Abstract
A facile method for the preparation of acid-resistant fabric with the fluoropolymer/SiO2 hybrid materials was presented. The polyester fabric was treated using a dip-dry technique and, subsequently, cured under high temperature. The results showed that the treated fabric displayed remarkable acid repellency with contact angles of 137.1°, 141.2°, and 139.5° for H2SO4 (80%), HCl (30%), and HNO3 (40%), respectively. The break force tests further confirmed the true potential of fluoropolymer/SiO2 hybrid materials to fabricate functional textile. We believe that this successful attempt offers an opportunity to fabricate acid-resistant fabric and advances the applications of protective clothing.
Introduction
Protective textiles, which are used to protect people from various bodily injuries, have attracted increasing attention due to their unique properties [1,2]. As one of the most important categories of protective textile, acid-resistant fabric could be used in areas ranging from petroleum, chemistry and metallurgy to electroplating. During the production, transportation, and application of strong acids, the workers must wear acid-resistant clothing to avoid corrosion and permeation. Over the past decades, the most effective acid-resistant textiles were coated with rubber and/or hydrocarbon resins, which could form a continuous film on the fabric surface and block the void of textile thus achieving an acid-resistant property [3]. However, it is uncomfortable to wear the resin-coated cloth during working due to the poor air permeability. Thanks to the efforts of various research groups, acid-proof fabrics were obtained by preparing intrinsic acid-resistant fibers. For example, Jin et al. [4] synthesized a polyurethane fiber by polyurethane urea and silane coupling agent. Polyurethane fiber could then be blended with cotton, rayon, and polyester. Yet, the single kind of polyurethane fiber limits its application. Developing chemical agents for the process of wet chemical coating was extensively used for the fabrication of functional textile [5]. Wet chemical coating method is popularly used because of the low cost, good homogeneity, and easy preparation for large-scale production. To this end, we prefer to develop wet chemical agents to prepare acid-proof fabrics. Most acidic solutions are of aqueous phase, so the acid-proof mechanism is similar to that of water repellent. Compared to superhydrophobic textiles [6–8] and other protective fabrics [9,10], only few studies are relative to the preparation of finishing agent for acid-resistant use [11,12]. And most of them mainly focused on the superhydrophobic property where the stability and durability was tested by checking the repellency of acidic aqueous solution [11]. Numerous researches have confirmed that the water-repellent or superhydrophobic surfaces could be achieved by the combination of the surface micro-nano structures and low surface energy materials [13,14]. In this work, we further extended this theory to the fabrication of acid-resistant fabric. Galbraikh [15] and several other authors have demonstrated that fluorochemicals could effectively provide resistant properties to corrosive solution and enable an acid-proof coating on fabric. However, the acid-proof permeability (i.e. the ability of prohibiting the acid to penetrate and destroy the fabric) could not reach the standard requirement (30 min, according to GB 24540-2009) owing to the defective and discontinuous film.
Recently, organic/inorganic nanocomposites have been widely applied in various functional coating applications including water-repellent surfaces, flame retardant, anti-ultraviolet, anti-reflection, etc. [16]. It was understandable that the superhydrophobic surfaces were achieved by the combination of low surface energy due to the organic polymer and rough surface structures caused by inorganic nanoparticles. Nanosilica have been widely used as inorganic components owing to its high strength, high corrosion resistance, and high roughness [17]. For example, Sun et al. [18] obtained excellent superhydrophobic surfaces by radical polymerization of vinyl-groups functioned silica nanoparticles with benzoyl peroxide. Xu et al. [19] obtained superhydrophobic, highly transparent, and stable organic/inorganic nanocoating by a simple sol-gel dip-coating method. Cui et al. [20] synthesized fluorinated-polyacrylate/SiO2 nanocomposite by seeded emulsion polymerization method. However, few studies aimed to apply organic/inorganic nanocomposites for the fabrication of acid-resistant surfaces. Zeng et al. [21] prepared superhydrophobic cotton fabrics with a very low contact angle hysteresis using fluorinated alkyl silane and silica nanoparticles. The coated fabric also showed good stability against long immersion times in organic solvents and acidic solutions. Wang et al. [22,23] developed robust super liquid-repellent fabrics using fluorinated polymers and demonstrated the durability of the coatings to withstand the attack of acid (pH=1). Yoo et al. [24] reported the solvent resistance of a polyester fabric coated with stacked polymer film through initiated chemical vapor deposition. But they did not study the properties of concentrated acid and did not report the mechanism of acid proofing. Acid-resistant fabrics hold more merits and properties against the corrosive acid than the water-repellent textile.
In this study, we first introduced the organic/inorganic hybrid materials to prepare acid-proof fabrics for the application of protective clothing. The fluoropolymer/SiO2 nanocomposites were coated onto the polyester (PET) fabrics by a simple dip-pad-cure process. Compared to the traditional preparation of acid-resistant fabrics, the obtained organic/inorganic surface showed excellent protective property toward concentrated H2SO4 due to the combination of a specific micro-nano rough surface and low surface free energy layer. This facile approach is expected to broaden their use for protective clothing.
Experimental section
Materials
Bleached twills woven polyester (PET) fabrics (150D (denier) / 144f (filament) × 100D/72f) were used as the substrates. The weight per unit area of the fabric was 128 g/cm2. H2SO4 (95–98%), HCl (36–38%), HNO3 (65–68%), methyl methacrylate (MMA), butyl acrylate (BA), 2-hydroxyethyl acrylate (HEA), ammonium persulfate (APS), anionic composite surfactant sodium dodecyl benzenesulfonate (SDBS) were the analytical reagents and purchased from Sinopharm Chemical Reagent Co. Ltd. (Shanghai, China). Nonionic surfactant aliphatic alcohol polyoxyethylene ether (AEO-9) and dodecafluoroheptyl methacrylate (DFMA) were obtained from Harbin Xeogia Fluorine-Silicon Chemical Co. Ltd. (Hangzhou, Heilongjiang, China).
Preparation of finishing agent for acid-resistant treatment
The main component of the finishing agent was fluoropolymer/SiO2, which was an organic/inorganic nanocomposite obtained and characterized using a protocol in our lab [25]. First, vinyl silica hydrosols were prepared by one-step water-based sol-gel method with the composite surfactant emulsification and used as seeds. Then, the fluorinated acrylic polymer/nanosilica was synthesized by seed semi-continuous emulsion polymerization. The acrylate monomers BA/MMA = 3/2 wt% and HEA (2 wt%, based on the total monomers) were pre-emulsified by AEO-9 and SDBS (AEO-9/SDBS = 2/1 wt%), and similarly for DFMA (20 wt%). The silica seeds (6 wt%) and a portion of the pre-emulsified monomers were introduced into a four-necked flask equipped with a refrigerating system. The mixture was stirred for 30 min and heated up to 60℃, then a portion of the initiator APS (0.8 wt%) was added dropwise. When the temperature reached 80℃, the residual monomers mixture and APS was introduced slowly. Simultaneously, the pre-emulsified DFMA monomer in second dropping funnel was fed into the flask. The reaction was continued at 80℃ for another 4 h. Then, the fluoropolymer/SiO2 nanocomposite was obtained.
Acid-resistant finish for PET fabrics
The acid-resistant fabrics were coated with the obtained fluoropolymer/SiO2 agent by dip-pad-cure, process. The PET fabrics were immersed in the fluoropolymer/SiO2 solution (50 g/L, pH was adjusted to 8–9 by ammonium hydroxide) for 10 min, and then squeezed using an automatic padder (Rapid Labortex Co. Lte., Taibei, Taiwan) with a nip pressure of 1 kg/cm2. This process was repeated twice. The fabrics were then dried at 100℃ for 150 s and cured at 170℃ for 120 s in the oven.
Characterization
Acid contact angle (CA) was determined according to ASTM standard 7334-08 via an OCA40 contact angle system (Dataphysics, Germany) using 10 µL of testing liquids (80% H2SO4, 30% HCl, 40% HNO3, according to GB 24540-2009) at room temperature. The testing was repeated 5 times for each sample then the average CA was obtained. The mechanical properties were determined by a universal testing machine (H10K-S, America Tiniius Olsen) in tensile mode according to ASTM standard 5035-06. After immersing in the acidic solution (80% H2SO4, 30% HCl, 40% HNO3, concentrations were determined according to GB 24540-2009) for 5 min and drying at 25℃, the samples were air-conditioned at 25 ± 2℃ and 65 ± 2% relative humidity for 24 h. Then the samples were stretched at a crosshead speed of 300 ± 10 mm/min to reach a constant strain rate. Five specimens were measured to obtain the average values.
The surface morphologies of the PET fabric before and after acid-proof treatment and acid immersion were examined by scanning electron microscopy (SEM, Hitachi TM-1000). Acid-proof permeability was measured by dropping 10 µL acid droplet (98% H2SO4, 36% HCl, 68% HNO3) with a micropipette injector onto the fabric surface to observe the damage severity over time. The photographs of the damaged fabric were taken by a digital camera (Canon SX700HS, 16.1 megapixel), which was available in our Lab.
Then the dissolution of a single yarn under H2SO4 (98%) was tested by an optical microscope (E400POL, Nikon). The CIE whiteness of fabric was measured according to GB/T 8424.2-2001 by a color measurement spectrophotometer (DATA COLOR machine, SF600 PLUS) with D65 light source and 10° visual angle. The air permeability was determined according to GB/T 5453-1997 by a textile testing equipment YG461G (Ningbo) at a pressure of 100 Pa.
Washing fastness of the coatings was tested according to the standard AATCC 61-2007 Laundering Testing. Specimens subjected to this test were similar to that produced by five typical hand launderings at a temperature of 40 ± 3℃. First, the fabric samples with dimensions 5 cm × 10 cm were prepared. Then the samples were washed in 200 mL 0.37% (percent detergent of total volume) detergent with 10 steel balls at 40 ± 3℃ for 45 min. After washing and drying, the contact angles were checked with H2SO4 (80%) after different washing cycles.
Results and discussion
In this study, the fluoropolymer/SiO2 nanocomposite emulsion was prepared by seeded semi-continuous emulsion polymerization by the monomers (MMA, BA, HEA), vinyl silica, and initiator APS. According to our previous research [25], a core (nanosilica)/ shell (fluoropolymer) structure was successfully obtained and the compatibility with organic materials was enhanced by many C=C active groups on the prepared vinyl silica. The C=C active groups could join the polymerization reaction with monomers thus leading to a chemical bond between SiO2 particles and fluoropolymer. The fluoropolymer/SiO2 nanocomposites were coated onto fabrics via dip-pad-cure method. Figure 1(a) shows the schematic illustration of the fabrication of acid-resistant fabric. The fluoropolymer/SiO2 nanocomposites were distributed on the surface during dip-pad process, and then the nanocomposites could gradually move closer and form an orderly arrangement with the evaporation of solvent. It has been demonstrated by many researchers that after the wet application of the fluorinated agents, an annealing or curing is necessary to obtain the repellence effect [26–28]. As a result, in our work a dense fluorinated layer was well formed on the fabric, together with a rough surface provided by inorganic nanosilica. Besides the intermolecular forces between the nanocomposites film and the polyester fabric, the polyester fibers could partially hydrolyze and form carboxylic groups under the weakly basic condition, then the carboxylic groups reacted with the SiO2 silanol to generate the chemical link [29]. Fluorine-containing groups have very low surface energy and self-aggregated property because of the extremely low polarization and high electronegativity of fluorine atoms. Herein, during the formation of the nanocomposite film, the fluoropolymer migrated to the surface preferentially. On the other hand, fluorinated molecules with high chemical stability can protect the carbon main chain as well as other molecules from the corrosion of concentrated acid.
Schematic illustration of (a) coating of organic/inorganic nanocomposites on PET fabric and (b) the contact between acid droplet and the micro-nano rough surface.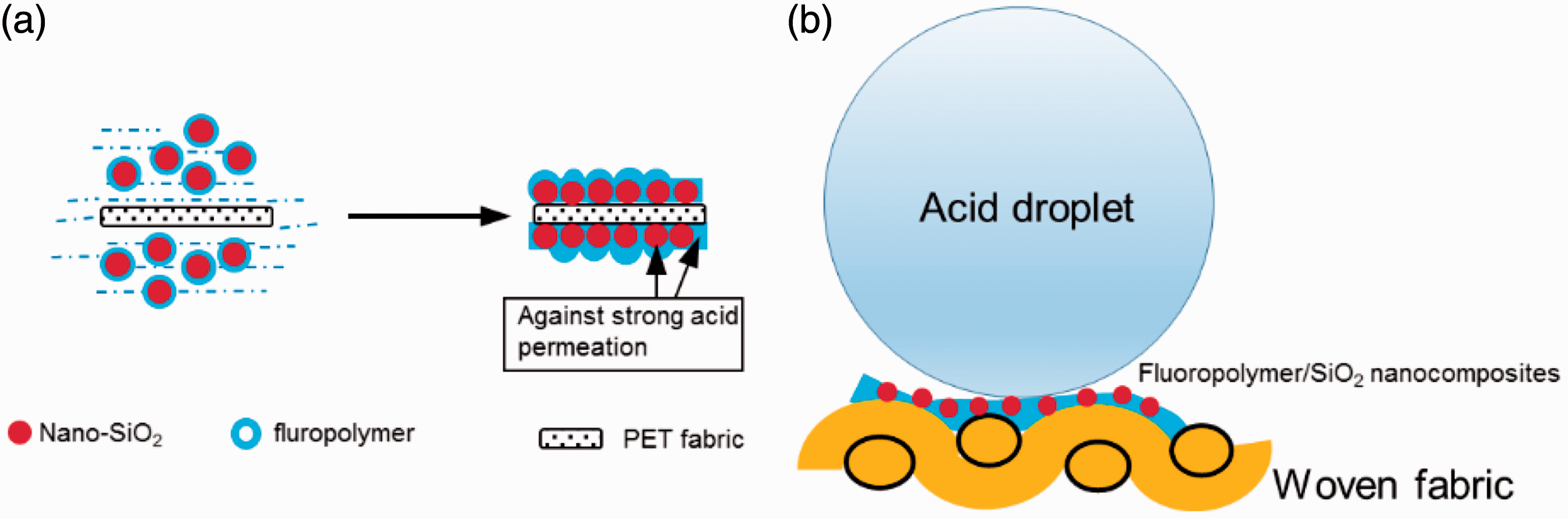
We evaluated the acid repellency by acid droplet contact angle. When the three types of acid drop (80% H2SO4, 30% HCl, 40%HNO3) were dripped on the untreated pristine PET fabric, it would be absorbed by the fabric and vanish rapidly. Figure 2(a) to (d) shows the images of water and acid droplets (10 µL) on the treated PET fabrics, which were coated by fluoropolymer/SiO2 nanocomposites. It displayed excellent superhydrophobicity with a water CA of 145.8° (Figure 2(a)); the acidic CA were 137.1°, 141.2°, and 139.5° for H2SO4 (80%), HCl (30%), HNO3 (40%), respectively, suggesting an outstanding acid repellency due to the combination of low surface energy and surface roughness. Compared with the water contact angle, the smaller contact angle of acid can be attributed to the lower surface tension of each acidic solution (water 72.8 mN/m, 80% H2SO4 66 mN/m, 40% HNO3 67 mN/m, 30% HCl 70 mN/m) [30–33]. Additionally, the slight reactivity of the acids toward the acid-proof surface could lead to the lowering of the contact angle. Obviously, the acid-resistant property of PET fabric was improved significantly after finishing with the organic/inorganic agent.
Contact angles of PET fabrics with acid-proof finishing: (a) water, (b) 80% H2SO4, (c) 30% HCl, and (d) 40% HNO3 acid droplet, respectively.
The excellent acid repellency of acid-proof treated fabric can be explained by Cassie-Baxter’ mode [34,35] as shown in Figure 1(b) that is similar to superhydrophobic surfaces. The coating substrate PET fabrics were woven by yarns that were produced by spinning of the microscale fibers. After incorporating with the nanoscale fluoropolymer/SiO2 nanocomposites, a micro-nano dual-size structure was generated, which is similar to the lotus leaf [36]. When a drop of corrosive acid was put onto the nanocomposites finished fabric, a large amount of air could be trapped due to the nanoscale silica stacking with large interfacial area, thus forming a liquid drop–air–solid interface. Therefore, the acid droplet can only sit on the top of the rough surface because of the large air pockets. Besides the water repellent theory, fluorinated polymer layer and silicon nanoparticles both hold high chemical stabilities that can further protect the fabric from strong acid penetration and provide the resistance against the acid liquid. The achievement of the acid-resistant effect of the fabric was attributed to the surface roughness, low surface energy, and chemical stability of the coatings.
Mechanical properties are important factors for evaluating a protective clothing fabric. Break force was measured to investigate the impact of the process on the physical characteristics. The break force of untreated and treated fabrics as well as the break force after immersion in different acids are presented in Figure 3. For pristine PET fabric, the break force of wrap and weft direction was 440 N/25 mm and 242 N/25 mm, respectively. After the treatment with the acid-proof agent, the break force of wrap and weft direction decreased to 430 N/25 mm and 231 N/25 mm, respectively. The minor decrease of break force could be attributed to the degradation of PET ester bond that occurred under finishing agent and high temperature curing. But the decrease of only 2–4% would not affect the wearability of a fabric. The acid resistance performance was further measured by immersing the fabrics in H2SO4 (80%), HCl (30%), HNO3 (40%) for 5 min. There was a noticeable decrease in the warp-wise break force of untreated fabrics from 440 N/25 mm to 393 N/25 mm (H2SO4), 416 N/25 mm (HCl), and 405 N/25 mm (HNO3), respectively. At the same time, the weft break force of untreated fabrics was obviously decreased from 242 N/25 mm to 206 N/25 mm (H2SO4), 230 N/25 mm (HCl), and 217 N/25 mm (HNO3), respectively. With the acid-resistant finishing, the break force of treated fabric was maintained well with a small decrease: the warp-wise break force slightly dropped from 432 N/25 mm to 422 N/25 mm (H2SO4), 426 N/25 mm (HCl), and 423 N/25 mm (HNO3), respectively, which confirmed that an excellent acid-resistant property was obtained. It can be concluded that the acid-resistant property against different acids was exhibited in the following order: H2SO4 (80%) < HNO3 (40%) < HCl (30%), which was consistent with the corrosivity order of these acids solution.
The break force (a) warp direction and (b) weft direction of untreated and acid-proof treated fabrics before and after immersion in 80% H2SO4, 30% HCl, 40% HNO3 for 5 min.
The SEM was used to observe the surface morphology of the PET samples. The original PET fabric has extremely smooth surface (Figure 4(a)). In contrast, PET fibers treated by fluoropolymer/SiO2 nanocomposites became rougher with uniform particles of around 500 nm due to the introduction of nanosilica, indicating that the organic/inorganic agent was indeed coated on the fiber surface resulting in a rough structure. From our previous study, the resultant fluoropolymer/SiO2 particles was about 120 nm [25] indicating that the nanocomposites were aggregated to about 500 nm during the drying and curing procedure. After immersion in H2SO4 (80%) for 60 min, it was found that the untreated fiber was extremely damaged, while the surface of treated fiber showed no significant change, as shown in Figure 4(c) and (d), in consistent with the results of break force test. The results suggest that the roughness of a surface and organic fluoropolymer plays a pivotal role in determining the acid wettability.
SEM images of fabrics before (a, b) and after (c, d) immersion in H2SO4 (80%) for 60 min. (a, c) untreated fabric, (b, d) acid-proof treated fabric, respectively. Insets in (a) and (b) are high magnification micrographs with a scale bar of 4 µm.
Then, the process of fabric damage was further investigated when a drop of H2SO4 (98%) was put on to the fabric. As shown in Figure 5, the droplet of acid spread on the untreated fabric immediately. As time passed, the fabric was damaged in circle with a diameter of 17 mm. Particularly, the center of the circle was leaked, which did not have any protective properties. In contrast, the fabric with nanocomposites agent coating exhibited an excellent acid-resistant property, the droplet maintained the spherical shape even after 60 min and the diameter of the damage area was only 5 mm. The color of H2SO4 (98%) droplet changed from transparent to white due to the oxidizing property of concentrated acid, but the fibers were not damaged. Figure 6 shows the images of untreated and acid-proof treated yarns, which were immersed in H2SO4 (98%) for different time periods. As shown in Figure 6(b), the pristine PET fibers were dissolved rapidly within 5 min of contact with H2SO4, and owing to the dehydration property of the acid, water of crystallization was removed and turned the fibers into carbon. After 30 min of contact with the concentrated acid, all the ester-bond were degraded and only liquid globules were left. In contrast, there was no change during the first 5 min when the acid-resistant fiber was immersed in H2SO4 (98%). After 10 min, a dark liquid (carbon) appeared under the fibers. Even after 30 min, only a small portion of fibers on the surface were dissolved, confirming the effective acid-resistant performance.
The fabric surface damages of pristine PET fabric and the acid-proof fabric during contact with a drop of H2SO4 (98%). The status of a single yarn from untreated fabric and acid-resistant fabric, after immersion in H2SO4 (98%) for (a) 0 min, (b) 5 min, (c) 10 min, and (d) 30 min, respectively.

Durability of the PET fabric with acid-proof treatment.

Conclusions
In summary, we first developed a simple and robust method for preparation of the acid-resistant fabric by coating fluoropolymer/SiO2 nanocomposites. The nanocomposites were applied to the PET fabrics by dip-pad-cure method, which is practical and convenient for large-scale production. The coated fabrics exhibited excellent acid-proof properties with a contact angle of 137.1°, 141.2°, and 139.5° for H2SO4 (80%), HCl (30%), HNO3 (40%), respectively, which can be attributed to the combination of a specific micro-nano rough surface and fluoropolymer layer, which were both stable to strong acid. The results of break force test and analysis of dissolubility of a single yarn further confirmed the true potential of fluoropolymer/SiO2 hybrid materials to generate acid-resistant fabrics with outstanding properties. We believe that the use of acid-resistant fabrics can be extended for a variety of applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Program for Specialized Research Fund for the Doctoral Program of Higher Education in China (No. 20130075130002), and the National Natural Science Foundation of China (No. 51303022). As a PhD student, H.W. was also partially supported by the China Scholarship Council.
