Abstract
In recent years, zinc oxide nano particles coating on textiles such as polyester is considered because of UV blocking and self-cleaning properties. Alkaline hydrolysis of polyester is a method in textile industry for surface treatment in large scale to enhance wettability. In the present work, polyester fabric was treated with sodium hydroxide, then was coated with ZnO nano particles, and also polyester fabric was treated with sodium hydroxide and ZnO nano particles at the same time. The bending length, water adsorption time, bactericidal properties, atomic absorption spectroscopy, and self-cleaning effect were measured according to the standard methods. Scanning electron microscopy and Fourier transform infrared spectroscopy analysis were used for the study of surface morphology and surface chemical bonding. The results demonstrated that increasing of zinc oxide nano particles concentration increased bending length, water adsorption time, antibacterial and self-cleaning effect. Comparing with pre-alkaline and simultaneous alkali treatment showed that pre-alkaline-treated fabric had more zinc oxide nano particles, therefore more self-cleaning and bactericidal effect. The scanning electron microscopy of alkaline-treated polyester fabric showed surface hydrolysis and nano-particles on the surface of polyester, and Fourier transform infrared spectroscopy spectroscopy indicated chemical bonding.
Introduction
Recently, zinc oxide has been found highly attractive because of its remarkable application potential in solar cells, sensors, displays, piezoelectric devices, electro-acoustic transducers, photo-diodes and UV light emitting devices, sun-screens, UV absorbers, anti-reflection coatings, photo-catalysis, and catalyst [1–13]. Zinc oxide is a modifier in textile industry [14] because of its use for the production of safety garments and all kinds of fabrics for the construction industry. It has a high UV blocking [15–18] and antibacterial properties [19]. Zinc oxide (ZnO) ultrafine particles are well known as UV blocking materials, which make it feasible to be widely used in polymers, fabrics, and cosmetic materials [16,18]. Zinc oxide has a band-gap at around 3.37 eV corresponding to 376 nm and thus it absorbs light that matches or exceeds this band gap energy [16–18]. UV-range of solar spectrum lies within this range and thus UV light gets absorbed by zinc oxide particles. The UV-blocking property of zinc oxide is only enhanced when it comes to nano-sized ultra-fine ZnO particles [18].
Zinc oxide has been studied as antimicrobial agents because of their photocatalytic activity under UV light [20–22]. Zinc oxide particles resist against microorganisms [20,23]. The inorganic oxides of CaO, MgO, and ZnO have antibacterial activity, which is attributed to the generation of reactive oxygen species on the surface of these oxides [20,24]. The antimicrobial mechanism of chemical agents is attributed to binding of the surface of the agent with a microorganism and the consequent metabolism of the agents inside the microorganism. Researchers have reported the antimicrobial activity of metal ions [25,26]. Many researchers have also attempted to correlate the biological activity of inorganic antibacterial agents with the size of the constituent particles [20,27,28].
In recent years, metal nano particles are used for microelectronics [29], photo catalysis [30], magnetic devices, and powder metallurgy [31]. Nano particles coating of textiles is considered because of UV blocking, antimicrobial and self-cleaning properties. It may affect the other fabric properties like dyeing, strength, bending length, air permeability, and friction. ZnO nanoparticles were used to improve antistatic properties of polyacrylonitrile. Jesionowski synthesized zinc oxide and deposited it on PES nonwoven [14]. Prasad synthesized and characterized the ZnO nanoparticles using solid-state reaction [32]. The ZnO nanoparticles applied onto cotton fabrics using acrylic binder. The UV-blocking property of these fabrics was evaluated in addition to their friction and mechanical properties. AbdElhady synthesized chitosan/ZnO nanoparticles using different concentrations of ZnO at different temperatures and applied it to cotton fabric for antibacterial and UV protection results [33]. Zinc oxide–soluble starch nanocomposites (nano-ZnO) synthesized using water as a solvent and soluble starch as a stabilizer was impregnated onto cotton fabrics to impart antibacterial and UV-protection functions [34]. Researchers also investigated antibacterial properties of textiles treated with zinc oxide nanoparticles [35–39]. Glass fiber–unsaturated polyester composites with different contents of ZnO were prepared by Peng et al. [40]. Their resistance after exposure to UV irradiation was evaluated. A sufficient content of ZnO in unsaturated polyester could significantly increase the resistance to simulation by exposure of the composites to Sun [40]. In the present study, zinc oxide colloidal solution was used for polyester fabric to enhance self cleaning and antibacterial effect of polyester fabric. Polyester fibers have hydrophobic properties, which need alkaline treatment to achieve hydrophilicity. Researchers worked on alkaline treatment of polyester fibers such as hydrolysis of PET treated with nano TiO2 and optimization of hot alkali treatment of polyester [41–46]. In the present work, polyester fabric was treated with sodium hydroxide, then was coated with ZnO nano particles and also treated with sodium hydroxide and ZnO nano particles at the same time to compare different treatments of ZnO nano particles on polyester fabric with alkali.
Materials and methods
Materials
The 100% gray plain weave polyester fabric with 25 warps and 20 wefts in cm was prepared from Baft Azadi Co (Iran). The chemicals used in this study were ZnO nano particles colloidal solution made by Sigma Aldrich (USA), sodium hydroxide from Romil (UK), Methylene blue (Merck, Germany), and Standard detergent without optical brightener (SDC, UK).
Methods
Alkaline hydrolysis and ZnO nano particles coating were conducted based on previous researches with some modifications. The plain weave polyester fabric was treated with sodium hydroxide in concentration of 1% (w/v %) at boiling temperature for 60 min with liquor ration of 50:1, then was neutralized with acetic acid, rinsed and dried [45,47,48]. Then ZnO nano particle colloidal solution was used on alkaline-treated and untreated polyester fabrics. The ZnO nano particles were coated at concentrations of 1, 2, 5, and 10% (% owf) at boiling temperature for 60 min [45]. On the other hand, polyester fabric was treated with ZnO nano particles and sodium hydroxide at the same time. The same concentrations of zinc oxide nano particles and sodium hydroxide also were used and the experiments were carried out at boiling temperature and 60 min to compare different treatments of ZnO nano particles with alkali (pre-alkali and simultaneous). Then, treated fabrics were dried and cured at temperatures of 200℃ for 3 min. The laundering test was carried out according to the AATCC 124-1996 standard test method to determine the durability of zinc oxide nano particles. The fabrics were washed for five cycles of washing at temperature of 47℃ for 30 min. After alkali treatment, the weight loss of alkali-treated polyester fabric was measured according equation (1):
The bending test was carried out according to the standard ASTM-D-1388 test method. A sample of specified width and length was placed on the top of the device and covered with a ruler. The fabric and the ruler were moved together until the sagging edge of the fabric touched the inclined plane. The length indicated on the ruler was recorded as the bending length. Water droplet adsorption time test was done according to the AATCC-39 1980 standard test method. A drop of water was poured from the burette to the surface of fabric. The time of disappearing water drop was measured.
The antibacterial test was done by AATCC standard method 100-1999 [49]. Two kinds of bacteria were used in this test (E. coli and S. aureus). The colonies were counted and bacteria reduction was calculated from equation (2):
The Zn ion content on polyester fabric was determined by atomic absorption spectroscopy. The polyester fabric was weighted and placed in furnace (Nabertherm, Germany) at temperature of 600℃ for 3 h [45,50], and then was diluted to a certain volume. The zinc ions were determined from the calibration curve of standard solutions and were reported in terms of gram per 100 g of fabric.
The ZnO nano particles treated polyester fabrics were dyed with methylene blue dye and were exposed to sunlight to determine self-cleaning effect. The color parameters of methylene blue dyed fabrics were obtained using reflectance spectrophotometer (D65/10°). Color difference of untreated and ZnO nano particles treated fabric were calculated according to equation (3):

Results and discussion
Bending length
Bending length and water droplet adsorption time results of ZnO nano particles treated polyester fabrics.

Water adsorption time
The time for water droplet adsorption on PET fabric is shown 0.2 s in Table 1. After alkaline treatment of polyester fabric, water droplet adsorption reached to 0.09 s. This is due to the physical or chemical changes on alkaline-treated polyester fiber, which may affect on water adsorption. Alkaline treatment of polyester fiber with sodium hydroxide acts on polymer structure of fiber surface and makes pores on the fiber surface to enhance the hydrophilicity of PET by forming roughness, so that increase in surface roughness promoted surface wetting further and water drop could penetrate into the pores [47,54–57]. In addition to physical changes, the chemical changes might be happen, the alkaline hydrolysis causes chain cleavages as OH− attacks ester-bonded carbonyl carbon resulting in hydroxyl (–OH) and –COO groups on the fiber surface [47,57–59].
By increasing of ZnO nano particles concentration, wetting time of a water drop into the fabric increased. This increase was shown for both of pre-alkaline and in the presence of alkaline-treated fabrics. Increasing the concentration of nano ZnO led to slight increase in surface roughness [53]. The present particles on fiber and fabric surface made a rough surface, which blocked the penetration of water drop into the fiber. The zinc oxide nano particles also penetrated into the pores, which made by alkaline treatment of polyester fiber. The other reason for increasing water adsorption time is, penetrating nano particles between the molecules, fibers, and yarns structure, which prevents of water penetrating by filling the free space between the molecules and fibers [52]. Adsorption of zinc oxide nano particles between the fibers lowered water adsorption through capillary action [54,60,61]. Pre-alkaline-treated polyester fabric showed higher water drop adsorption time. This higher adsorption time is attributed to the presence of higher zinc oxide nano particles on the fiber surface and between the fibers and molecules. Therefore, pre-alkaline-treated polyester fabric adsorbed more zinc oxide nano particles.
After repeated laundering, the wetting time decreased as compared with before laundering, because of the decrease of nano particles on the fiber surface. The nano particles were removed from the fabric surface by washing process. The removed particles were attributed to the physically adsorbed particles, which had no reaction with fiber.
Bactericidal properties
In the present work, the effect of ZnO nano particles treatment on polyester fabric with two types of microorganisms was investigated. In this regard, S. aureus and E. coli were chosen on the basis of being gram positive and gram negative. Figure 1 indicates bacteria reduction of ZnO nano particles treated polyester fabrics. By increasing the concentration of zinc oxide nano particles, the bacteria reduction increased to 100%. The antibacterial activity of the ZnO particles were studied by Zhang [19,36]. The active oxygen species generated by ZnO particles could be a mechanism for bactericidal effect. The presence of active oxygen species has been detected by Yamamoto [36,62]. The detailed mechanism for the activity of ZnO is still under debate. One possible explanation of the antibacterial effect of ZnO is based on the abrasive surface texture of ZnO. ZnO nanoparticles have been found to be abrasive due to surface defects [20,63]. Nano-sized and micron-sized ZnO suspensions are active in inhibiting the bacteria growth; the nano-sized ZnO suspension clearly has a much higher activity than the micron-sized ZnO suspension [19,36]. ZnO bulk treated fabrics show antibacterial activity but the activity in the ZnO nanoparticles treated fabrics is much higher [36].
Bacteria reduction percentage of ZnO nano particles treated polyester fabrics.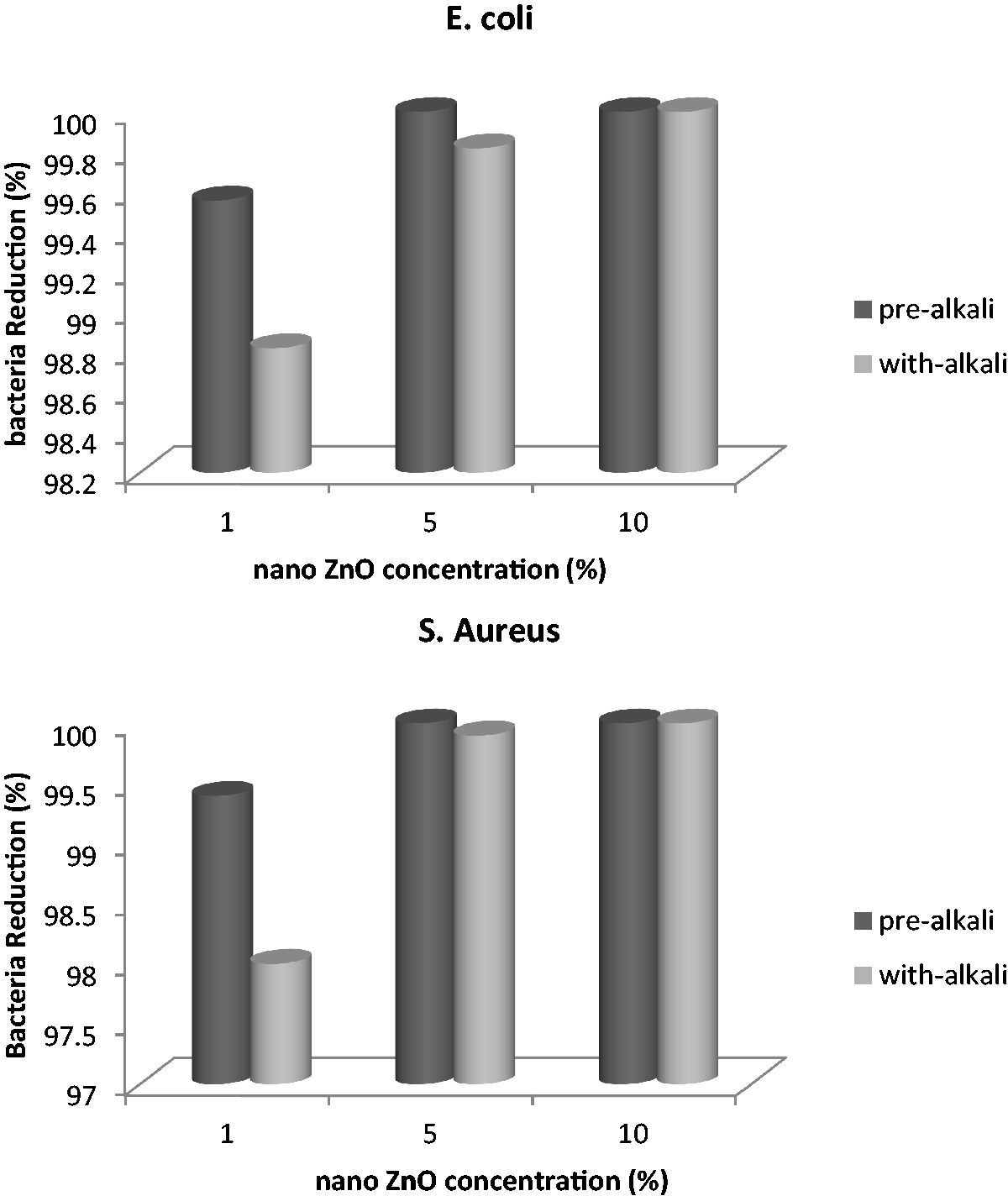
In comparison with pre-alkaline and with alkali treatment of zinc oxide nano particles treated polyester fabrics, pre-alkaline treatment of polyester fabric showed 99.56% of bacteria reduction at concentration of 1% of zinc oxide nano particles. ZnO nano particles coating in the presence of alkaline treatment of polyester fabric showed 98.82% of bacteria reduction. The higher bacteria reduction for pre-alkaline-treated fabric is attributed to the higher zinc oxide nano particles concentration. These results are agreement with the previous results of bending length and water droplet adsorption, which pre-alkaline-treated polyester fabric adsorbed higher ZnO nano particles. The probable reason that showed more ZnO concentration on pre-alkaline polyester fabric might be the presence of more pores on the surface of alkali-treated polyester fiber. In simultaneous treatment of alkali and ZnO nano particles on polyester, alkaline and zinc oxide nano particles affected on fiber surface at the same time. Therefore, there might be a reaction between alkali (sodium hydroxide) and ZnO nano particles, which could slow the reaction of alkali and ZnO to the fiber separately. Zinc oxide can react with NaOH to produce ZnOH+, Zn(OH)3−, and Zn(OH)4−2 at pH = 6–14. When the alkaline concentration is higher (pH > 12), NaZn(OH)3 or Na2Zn(OH)4 is produced. The probable reaction of ZnO nano particles in sodium hydroxide solution is shown as [64]:

Zinc ion content (atomic absorption spectroscopy)
For investigation of zinc oxide nano particles concentration on polyester fabrics, the atomic absorption analysis was used to determine zinc ions content on polyester fibers. Figure 2 shows the zinc ion content per 100 g of fabrics on pre-alkaline and in the presence of alkaline-treated polyester fabrics. It can be seen that the results were agreement with the previous results of bending length, water drop adsorption, and antibacterial test. Zinc ion content in pre-alkaline and 10% of zinc oxide nano particles treated was 0.2 g per 100 g of fabric, by simultaneous treatment of ZnO nano particles and sodium hydroxide it reduced to 0.1 g. Regarding the previous explanations, pre-alkaline treatment of polyester fibers had more zinc oxide nano particles. The alkaline treatment with sodium hydroxide caused some pores on the surface of polyester fiber, which zinc oxide nano particles could penetrate into the pores and fiber structure. The alkaline polyester fiber also had hydrophilic sites to adsorb zinc oxide nano particles (Schemes 1 and 2) [47]. Whereas alkaline and ZnO nano particles treatment of polyester fiber at the same time could affect less on the fiber surface, so that few amount of zinc oxide nano particles were adsorbed on the polyester surface. The probable reason for this low amount of ZnO nano particles might be the reaction between ZnO and sodium hydroxide (Scheme 3) [64]. Figure 2 also shows the zinc ions content after repeated laundering. Zinc ion content reduced after five cycles of washing. Zinc oxide nano particles were removed from the fabric surface by washing process. The removed particles are attributed to the physically adsorbed particles.
Zinc content per 100 g fabric. Alkaline treatment of polyester fiber [47]. ZnO nano particles treatment on pre-alkaline-treated polyester. Simultaneous treatment of polyester fiber in the presence of alkaline and ZnO nano particles.



Self-cleaning performance
The self-cleaning performance of hydrophilic coating is known as photo-catalysis and it chemically breaks down dirt when exposed to light. The hydrophilic coatings have parallels with photosynthesis, using sunlight to drive chemistry [65]. ZnO nano particles act as a photo-catalyst, and under light irradiation result in the electron-hole pair in which electron (e−) is reductive, and hole (h+) is oxidative [33,66]. Holes cause the oxidization of adsorbed organic molecules while electrons eventually combine with atmospheric oxygen to give the superoxide radical, which quickly attacks nearby organic molecules. The result is a cleaning of the surface by the conversion of organic molecules to carbon dioxide and water at ambient temperature [65]. The organic pollutants including aromatics, polymers, dyes, and surfactants can be broken down in this way. Photo-catalysis is usually tested by destructing of a pollutant. Methylene blue is a popular choice as it is easy to quantitatively measure by spectroscopy [65]. In the present work, methylene blue dye was used and colorant decomposition activity in the presence of zinc oxide nano particles was measured. Figure 3 shows the color difference of methyelene blue and ZnO nano particles treated polyester fabrics after 24 and 42 h of exposing sunlight. The higher color difference (ΔE) indicates higher methylene blue dye decomposition. By increasing zinc oxide nano particles concentration from 1 to 10%, color difference increased and showed higher dye decomposition after exposing sunlight. In comparison with pre-alkaline and at the presence of alkaline treatment of ZnO nano particles treated polyester, pre-alkaline-treated polyester fabric showed higher color difference, therefore higher dye decomposition and self-cleaning effect. This was because of higher amount of ZnO nano particles on pre-alkaline polyester, which was in line with finding of bending length, water adsorption, bactericidal properties, and atomic absorption analysis.
Color difference (ΔE) of ZnO nano particles treated polyester fabrics for self-cleaning effect.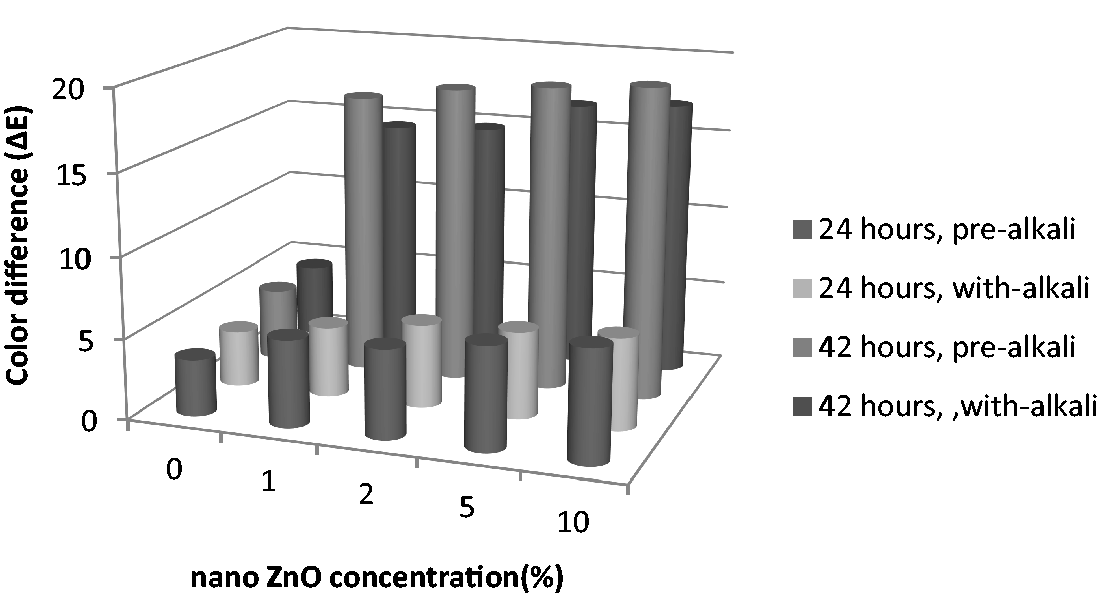
Scanning electron microscopy (SEM)
The treatment of polyester fabric with sodium hydroxide caused the weight loss of 5–6%. This was because of surface hydrolysis of polyester fiber. The weight reduction on polyester surface is attributed to chain cleavages as hydroxyl group attacks ester-bonded carbonyl groups [47]. Figure 4 shows the scanning electron micrographs of untreated polyester (a) alkaline polyester, (b) ZnO nano particles in the presence of alkali, (c) and pre-alkaline treatment (d). As shown in Figure 4(a), polyester fiber had a smooth surface, while the alkali treatment of polyester fabric in Figure 4(b) made some pores and scars on the surface of fibers. Surface etching of polyester by alkali treatment led to surface roughness [47]. The scars which were formed by alkali hydrolysis might be the sites for penetrating the nano particles into the fiber. Figure 4(d) demonstrates coating of zinc oxide nano particles on alkali polyester. Comparing with alkali-treated polyester, zinc oxide nano particles were observed on it. These results confirmed the previous results, which showed the higher penetration of zinc oxide nano particles on pre-alkaline-treated polyester fiber. The micrographs of pre-alkaline-treated polyester (Figure 4(d)) showed more ZnO nano particles as compared with the treatment of ZnO nano particles at the presence of alkali (Figure 4(c)).
SEM micrographs of (a) polyester, (b) alkaline polyester, (c) ZnO nano particles in the presence of alkali, and (d) ZnO nano particles on pre-alkaline treatment.
FTIR/ATR spectroscopy
Figure 5(a) shows the spectrum of untreated polyester fabric. According to the previous work of Natarajan and Jeyakodi Moses [41], the spectrum showed high peaks from 1700 cm−1 to 700 cm−1 according to the spectra of stretching vibration band of C = O at 1709 cm−1 and C–O–C stretching vibration band at 1087 cm−1 and 1237 cm−1, which were attributed to the ester groups of polyester [41,67]. Figure 5(b) shows the alkaline polyester fiber spectrum. Peak at the region of 2319 cm−1 confirmed the presence of carboxylic groups (–COOH) in alkaline-treated polyester. Figure 5(c) and (d) shows the spectra of pre-alkaline and in the presence of alkali treatment of zinc oxide nano particles on polyester fabrics. The band at 607 cm−1 was corresponded to the new bonds PET + ZnO nano particles. The presence of this band supported the ionic character of the new band formed due to the addition of ZnO nano particles to alkali hydrolyzed fabrics [50].
FTIR/ATR spectra of (a) polyester, (b) alkaline polyester, (c) ZnO nano particles on pre-alkaline treatment, and (d) ZnO nano particles in the presence of alkali.
Conclusion
In this research, polyester fabric was treated with sodium hydroxide, then was coated with ZnO nano particles, and also polyester fabric was treated with sodium hydroxide and ZnO nano particles at the same time. Alkaline hydrolysis of polyester fabric before treating ZnO nano particles and also in the presence of nano particles showed different amounts of zinc oxide nano particles in fabrics, therefore different properties. The properties such as bending length, water adsorption, antibacterial effect, and self-cleaning were investigated. The results of these properties demonstrated that increasing of zinc oxide nano particles concentration increased these properties. The antibacterial performance of pre-alkaline polyester fabric also showed higher bacteria reduction as compared with treatment in the presence of alkali. Self-cleaning effect of ZnO nano particles on polyester fabric was more significant when pre-alkaline treatment was used. Pre-alkaline-treated polyester fabric revealed higher water drop adsorption time and slightly increase in bending length. Results of atomic absorption spectroscopy confirmed that pre-alkaline-treated fabric had more zinc oxide nano particles. The SEM of alkaline-treated polyester fabric showed surface hydrolysis of polyester and scares on the fiber surface, and FTIR spectroscopy indicated chemical bonding. Self-cleaning and antibacterial effects of ZnO nano particles can be important in textile industries. Textile industries also need to reduce the processing time of finishing. This study reveals that although one step of chemical finishing is desirable for textile industries, two steps of alkaline and zinc oxide nano particles treatment on polyester fabric is significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


