Abstract
The presented work reported the growth of 3D-shaped TiO2 flower particles on the surface of polyester fabrics using two step approaches of sol–gel technology and hydrothermal method. The scanning electron microscopy, EDS analysis, Raman spectroscopy, and X-ray diffraction techniques were employed to study the effect of titanium isopropoxide (TTIP) concentration on the growth of flower-like TiO2 microstructures. Later, a layer of trimethoxy(octadecyl)silane was applied on TiO2-coated polyester fabrics to fabricate the self-cleaning textiles. The physical self-cleaning properties were examined based on superhydrophobicity and contact angle measurements, where maximum static contact angle of 160.1o and minimum roll off angle of 3° was found for 2 mL TTIP concentration. The degradation of methyl orange dyes under UV light irradiation was observed to confirm the photocatalytic chemical self-cleaning behavior, where the samples coated with 2 mL TTIP decolorized the dye solution in 150 min, whereas the samples coated with 1 mL and 1.5 mL TTIP took almost 300 and 210 min, respectively.
Introduction
Research related to self-cleaning surfaces has received considerable attention within the scientific community due to their potential applications in industry and daily life [1]. For example, it is useful for the textile products which are endangered by staining with heavy contaminants such as soot, oils, lubricants or those not capable of washing due to their size or water and/or detergent sensitivity (i.e. umbrellas, canopies, shade shelters, camps, curtains, blinds, roller blinds, or external sunshades) [2]. The self-cleaning textiles also hold great promise for military applications where there is a lack of time for laundering at severe conditions, and in business life when clothes get stained accidentally [3]. Currently, there are two main concepts used in developing self-cleaning textiles [4]. The first concept (physical self-cleaning) is based on the superhydrophobic approach where water droplets attain the spherical shape and then they roll off the surface carrying away the dirt particles [5]. The second concept (chemical self-cleaning) is based on the process of photocatalysis where the dirt/stain molecules break down to simpler species (such as CO2 and water) on exposure to light [6]. For physical self-cleaning, the surface roughness and low surface energy of materials are the two important factors to control the wettability of a surface and to obtain water contact angles greater than 150° [7]. As an inspiration from nature (e.g. lotus leaves, butterfly wings, and water strider legs), scientists have fabricated superhydrophobic textiles by creating surface roughness in combination with low surface energy materials such as organic silanes, fluorinated silanes, alkyl amines, and silicates [8–10]. For chemical self-cleaning, polycrystalline semiconductor oxides have been applied in the form of nano-coatings, leading to the successful development of a number of UV-active self-cleaning textiles [11]. In this regard, titanium dioxide (TiO2) has become the most popular high efficiency catalyst due to the advantages of its high refraction coefficient, absorption in the UV range, strong photocatalytic effect, biological inertness, and low cost [12,13]. It was reported that the particle size, crystal structure, and the morphology play important roles on photocatalytic activity of TiO2, which are mainly depended on synthetic method and reaction conditions including titanium salt, pH value, reaction temperature, time, additives, etc. [14].
Recently, numerous efforts have been made to simultaneously create superhydrophobic and photocatalytic surfaces which not only repel water but also decompose organic contaminates at the same time. However, the fabrication of superhydrophobic and photocatalytic surface is challenging because this surface either loses its superhydrophobicity upon irradiation with light or does not show a photocatalytic property [15]. Furthermore, the efficiency of TiO2 as a photocatalyst has been limited due to the recombination of photogenerated charge carriers, which is typically faster than the rate production of reactive oxidation species [6]. Apparently, many further developments were hampered by the unattainable robustness of hierarchical roughness and the instability of surface chemistry [7]. Thus, improving the charge separation efficiency needs a rational design of photocatalyst structure. To improve the photocatalytic activity, several methods are demonstrated, such as reducing the recombination of separated electron–hole pairs by blending various TiO2 phases, or differing the size of TiO2, depositing metal materials on a TiO2 surface to form a heterojunction at the interface, introducing electron–hole scavengers or trapping sites to inhibit the recombination, and doping specific metal or non-metal elements to extend the absorption regime to visible wavelengths [16]. To further improve their performance and the durability, a considerable amount of research is also focused on the controllable synthesis of TiO2 with aligned one/three-dimensional nanostructures such as nanorods, nanowires, nanoflowers, nanotubes because of their high specific surface area and favored charge-transfer rate [17,18]. Among them, the flower-like TiO2 hierarchical structure was found to demonstrate excellent photocatalytic properties and inhibit defects of micron and nanometer structures due to their more abundant porous structure, larger specific surface area, lower density, better surface permeability, greater light-harvesting capacity, good light absorption efficiency, and appropriate refractive index [19].
Despite the great progress in this field, the shape-controlled synthesis of TiO2 micrometer or nanometer structures remains a challenge. Currently, the methods used for the synthesis of flower-like TiO2 hierarchical structures include the hydrothermal, solvothermal, microemulsion, sol–gel, hydrolysis, and electrodeposition oxidation methods [18]. But these methods are impractical for large-scale production due to their high cost, occurrence of unexpected morphological changes, problems with heterogeneous impurities, and time consuming [20]. Here, the growth of 3D-shaped TiO2 flower particles on surface of polyester fabrics using two step approaches of sol–gel technology and hydrothermal method is reported. Compared to previous studies, it has number of advantages such as simple, inexpensive, high yield, and effective control over phase and morphology of TiO2 at low temperatures. The effect of titanium isopropoxide (TTIP) concentration on the growth of the flower-like TiO2 microstructures were investigated in detail using scanning electron microscopy (SEM), EDS analysis, Raman spectroscopy, and X-ray diffraction (XRD) techniques. Further, the layer of trimethoxy(octadecyl)silane (OTMS) was applied on TiO2-coated polyester fabrics to fabricate the self-cleaning textiles which are simultaneously superhydrophobic and photocatalytic. The non-fluorinated silanes were selected, as they are less hazardous to skin compared to fluorinated silanes. The physical self-cleaning properties were examined based on superhydrophobicity and contact angle measurements, whereas the degradation of methyl orange dyes under UV light irradiation was observed to confirm the photocatalytic chemical self-cleaning behavior.
Experimental methods
Materials
TTIP, titanium butoxide, hydrochloric acid, caustic soda, ethanol, acetic acid, and petroleum ether were purchased from Sigma Aldrich and used as received. A plain woven polyester fabric of warp count 98 dtex, weft count 185 dtex, ends/cm 52, picks/cm 27, and areal density 97 g/m2 was supplied by the local industry. Trimethoxy(octadecyl)silane (OTMS) was purchased from Merck.
Synthesis of flower-shaped TiO2 particles on polyester fabric
At the beginning, the surface of polyester fabric was cleaned by petroleum ether for 2 h at 60℃ in Soxhlet apparatus and later treated with 20 g/L caustic soda solution at 80℃ for 30 min. Then, the flower-shaped TiO2 particles were grown on the surface of polyester fibers using two step approaches of sol–gel technology and hydrothermal method. In the first step, a semitransparent TiO2 sol was formed by continuously stirring a mixed solution of titanium butoxide (6 mL), ethanol (60 mL), and acetic acid (2 mL) for 3 h at 60℃. The caustic soda-treated polyester fabric was immersed in the prepared TiO2 sol for 5 min, padded with a wet pickup of 70–80% and dried at 110℃. This process was repeated three times, and the samples were cured at 140℃. In the second step, hydrothermal method was used to grow the flower-shaped TiO2 particles on the already TiO2-seeded fabric. Here, different amounts of TTIP were added drop wise into the aqueous hydrochloric acid solutions (i.e. 10 mL HCl and 60 mL distilled water), and the mixtures were stirred for 10 min until the solutions turned clear. The resulting solution was further transferred into 150-mL Teflon-lined stainless-steel autoclave, and TiO2-seeded polyester fabric was placed in it. The autoclave was placed in an oven for 1 h at 125℃. Finally, the polyester fabric was removed from the autoclave, rinsed with deionized water and dried at room temperature. Figure 1 shows the schematic of deposition of TiO2 flowers onto polyester fabrics.
Schematic of deposition of TiO2 flower-shaped particles on polyester fabric.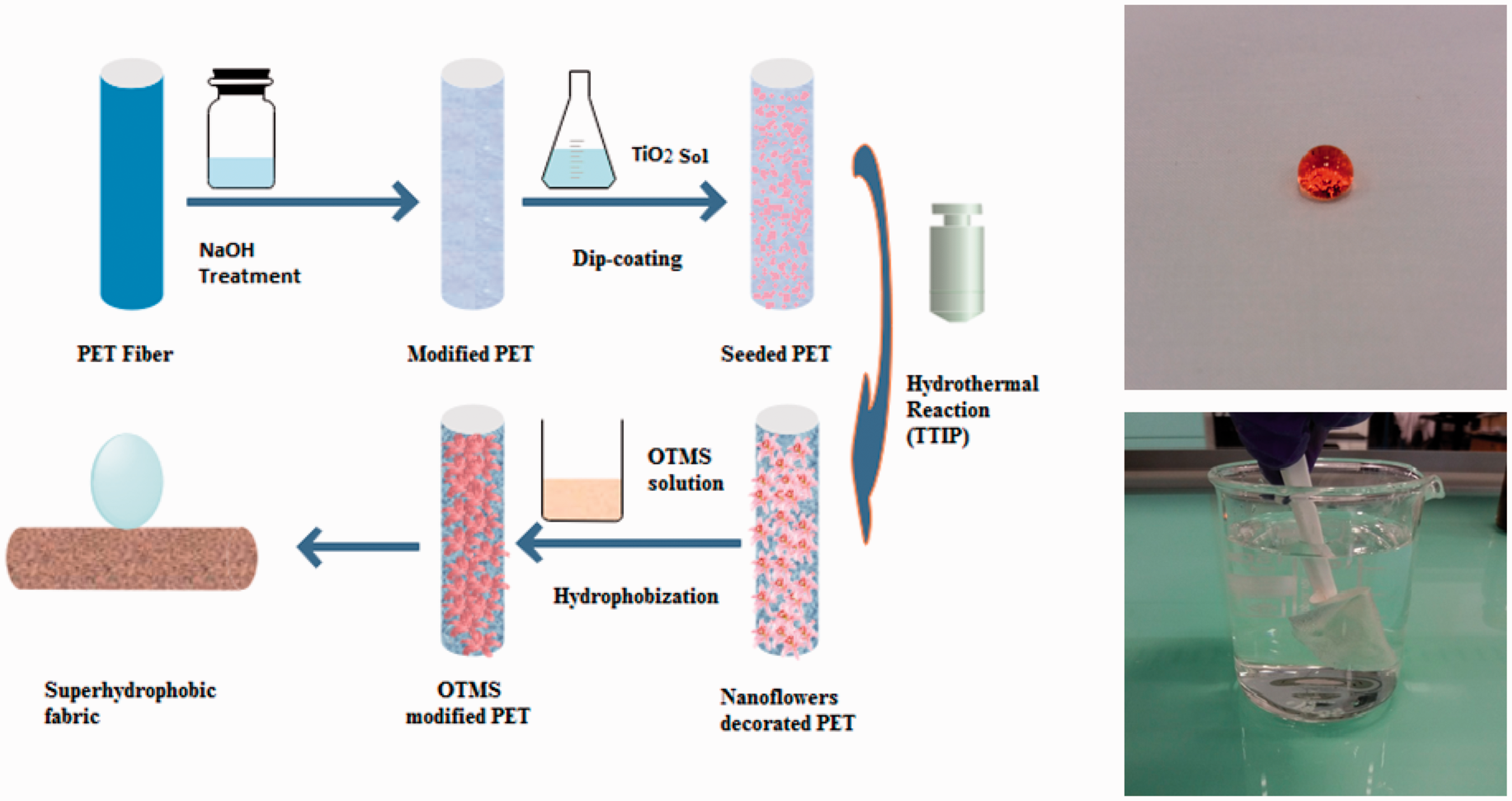
Characterization of surface structure of TiO2 nanoflower-coated polyester fabrics
The SEM of TS5130 Vega Tescan was employed to observe the surface microstructure of TiO2 flower-coated polyester fabrics at 30 kV acceleration voltages. The gold sputtering was performed to develop the conductive layer on surface of samples before SEM analysis. For better resolution, the surface structure of coated samples was also seen under the field emission SEM of Zeiss Ultra Plus at an accelerating voltage 2 kV equipped with an Energy Dispersive X-ray spectrometer Oxford X-max 20. For characterization of TiO2 phases, the Raman spectra were recorded by DXR Raman Microscope (Thermo Scientific, USA) with 532 nm laser, grating 900 lines/mm in spectral range of 3500–50 cm−1 and objective Olympus LMPlanFL 10 × and 50×. The 25 and 50 µm slit spectrograph aperture was used with 0.9 to 5.0 mW level of laser power, 1 s exposure time and 50 exposures. The crystalline structure of TiO2 was studied on XRD analysis of PAN analytical X’pert PRO equipment. The XRD analysis was performed with a diffractometer equipped with a conventional X-ray tube Cu kα1 radiation (λ = 1.5406 Å) power condition (40 KV/30 mA). The XRD pattern was measured in the 2θ range of 20–70 with the step size of 0.02.
Superhydrophobization of TiO2 nanoflower-coated polyester fabrics
The OTMS was applied on TiO2-coated polyester fabrics for fabrication of superhydrophobic surfaces by lowering the surface energy. The OTMS solution was prepared by addition of (8%, w/w) OTMS drop wise into ethanol and stirring it at room temperature for 45 min. The hydrolysis of OTMS was promoted by use of acetic acid and by adjusting pH values in 4–5 range. The polyester fabrics decorated with different concentrations of 3D-shaped TiO2 flowers were then immersed in hydrolyzed OTMS solution, dried in air, and cured at 120℃ for 1 h. One control sample was also prepared by just coating of OTMS alone and without any surface decoration of TiO2 nanoflowers. The schematic of fabrication of superhydrophobic surfaces can be seen as continued process to decoration of TiO2 flowers from Figure 1. Later, equation (1) was used to calculate the uptake (i.e. add on percentage) of TiO2/OTMS on polyester fabrics.
Furthermore, the comfort properties of coated fabrics were examined based on the measurements of stiffness from the TH-7 instrument. This instrument is similar to standard Kawabata KES-FB 2 device used for testing of low stress mechanical properties [21].
Characterization of superhydrophobicity of TiO2-OTMS-coated polyester fabrics
Contact angle measurement
The contact angle of TiO2-OTMS-coated polyester fabrics was measured by surface energy evaluation system of Advex Instruments. It is based on the goniometric method to calculate the contact angle between liquid and solid with 5 µL of water droplet. A total of six measurements were made on each sample, and the average contact angle was determined.
Roll off angle measurement
The sample was fixed on custom-made device, and 5 µL water droplet was dropped from a syringe on the fabric. The roll off angle was measured by inclining the plate slowly at an angle of 1° until the droplet started to move. On each sample, six measurements were made and mean value was taken.
Contact angle hysteresis
The advancing contact angle was measured when needle was brought in close proximity to the sample surface, and the deionized water was pumped until the drop reached a size of approximately 25 µL. Subsequently, the receding angle was measured when the liquid was pumped in at the same rate until the detached drop from the needle or all of the liquid returned to the syringe. Then, the contact angle hysteresis was estimated from the difference between the advancing and the receding contact angles. The test was repeated three times for each specimen at different locations.
Durability of contact angle
For the estimation of mechanical abrasion durability, TiO2-OTMS-coated polyester fabrics were dragged against the sand paper surface by putting 100 g load on top of the sample, and the change in contact angle after 20 abrasion cycles was examined [22–24]. Whereas for the washing durability, the change in contact angle of coated fabrics was measured after five laundering cycles according to ISO 105 C06 (B1M) standard (4 g L−1 detergent, 60℃ temperature and 45 min time). Further, the TiO2-OTMS-coated polyester fabrics were immersed into the acidic (pH=1) and alkaline (pH=13) solutions for the duration of 24 h and then the contact angle was measured to ensure their chemical durability. As exposure to sunlight during daily routine influences the properties of textile fibers, the durability of contact angles against UV exposure was studied by placing the coated samples under the ultraviolet lamp Philips TL 6 W/05CE UV tubes (315–400 nm) for 24 h. Lastly, the effect of jet of water when impacted on the fabric surface was also studied by observation of the bouncing of water droplets. A jet of water with speed of 3 m/s was impacted on coated fabrics by a syringe kept at 4 cm above the sample with an angle of 45° for 1 min, and the bouncing of water droplets was captured in the high-speed camera (Olympus, i-SPEED 3) at 5000 frame s−1.
Characterization of self-cleaning properties of TiO2-OTMS-coated polyester fabrics
Physical self-cleaning
Methyl orange dye (0.5 g) was used as a contaminant, and it was randomly sprinkled over the coated fabric surface. Then, the physical self-cleaning property was studied by rolling the water over the dye-contaminated surfaces. The directed movement of water droplets was observed in digital camera by introduction of water droplets at an angle of 45° on the coated fabric surface using micropipette.
Chemical self-cleaning
As the TiO2-OTMS-coated polyester fabrics were superhydrophobic in nature, they were not immersed directly in aqueous solution of methyl orange dye. The superhydrophobic fabrics were first soaked in acetone, and then immersed in dye solution for the chemical self-cleaning behavior using two different experiments (i.e. stain degradation and solution discoloration). For stain degradation, the dyed samples were irradiated under the UV light of Philips TL 6 W/05CE UV tubes (315–400 nm), and the stain degradation performance was evaluated as a function of exposure time to UV light. Later, the fabric surfaces discolored after the tests were scanned with 300 dpi, and the scanned images were analyzed to calculate the whiteness index by Image J software [25]. For solution discoloration experiments, the coated samples with diameter 2.8 cm were placed in a beaker having 15 mL of methyl orange dye solution. This beaker was then placed below the light source Philips TL 6 W/05CE UV tubes (315–400 nm) at a distance of 18 cm. To estimate the photocatalytic discoloration effect, an aliquot was removed from the solution after a pre-set time interval and its absorbance was measured at λmax (485 nm) using UV-Vis Spectrophotometer (UV-1600PC).
Results and discussion
Surface structure of polyester fabrics coated with TiO2 nanoflowers
SEM analysis
The polyester fiber surface has low surface free energy, low wettability, and poor adhesion due to the lack of polar groups (COOH and OH) on the main chains. In order to attach TiO2 nanoseeds on the polyester fabric surface, caustic soda treatment was carried out to generate the polar groups. Although the tensile strength of the polyester fabric decreased by 11% due to the cleavage of ester linkages and hydrolysis of polyester chains, it resulted in the formation of hydroxyl and carboxyl acid groups on the fiber surface [26]. From Figure 2(b), oligomers formed due to hydrolysis of polymer chains can be seen as small particles on the surface of the polyester fibers. The attachment of very fine TiO2 nanoseeds can be observed from the surface of polyester fibers as shown in Figure 2(c).
SEM micrographs of polyester fabric. (a) Before caustic soda, (b) after caustic soda and (c) after seeding.
The surface hierarchical structure is important to obtain the superhydrophobic property. The surface structure of coated fabrics was investigated at different TTIP concentrations (i.e. 0.5 L, 1 mL, 1.5 mL, and 2 mL) from SEM images as shown in Figure 3. It can be seen that the concentration of TTIP had a great effect on the growth and structure of TiO2 nanoflowers. At low concentration of TTIP (i.e. 0.5 mL), no TiO2 flower structures were found on the polyester fiber surface except some tiny crystal nuclei of TiO2 particles. However, the amount and the size of TiO2 flower structures greatly increased as the amount of TTIP increased from 1 mL to 1.5 mL (see Figure 3(a) and (b)). When the TTIP concentration was further increased to 2 mL (see Figure 3(c)), the flower-like structures started to aggregate sharply and a continuous thicker coating was constructed on the polyester fiber surface. A micro/nanoscale surface roughness mimicking the surface of lotus leaf was formed with each fiber of approximately 5–10 µm diameter, and the TiO2 particles of below 500 nm sizes. A good adhesion between the TiO2 particles and polyester fibers can be attributed to the presence of hydroxyl groups and formation of hydrogen bonds between them.
Effect of titanium isopropoxide concentration on growth of TiO2 flowers. (a) 1 mL of TTIP (b) 1.5 mL of TTIP (c) 2 mL of TTIP (d) 2 mL of TTIP with high resolution.
EDS analysis
It was carried out to know the chemical composition of TiO2-coated fabric. The EDS spectra in Figure 4 showed the presence of O, C, Ti, and Au as main elements on the surface of TiO2-coated fabric, whereas the pristine polyester surface mostly consisted of O and C elements. The relative atom ratio for O, C, and Ti was determined about 40.7%, 37.1%, and 22.1%, respectively. The presence of Au element was caused by sputtering of gold on TiO2-coated sample during SEM characterization.
EDS spectrum of flower-like TiO2 nanoparticles coated on polyester fabric.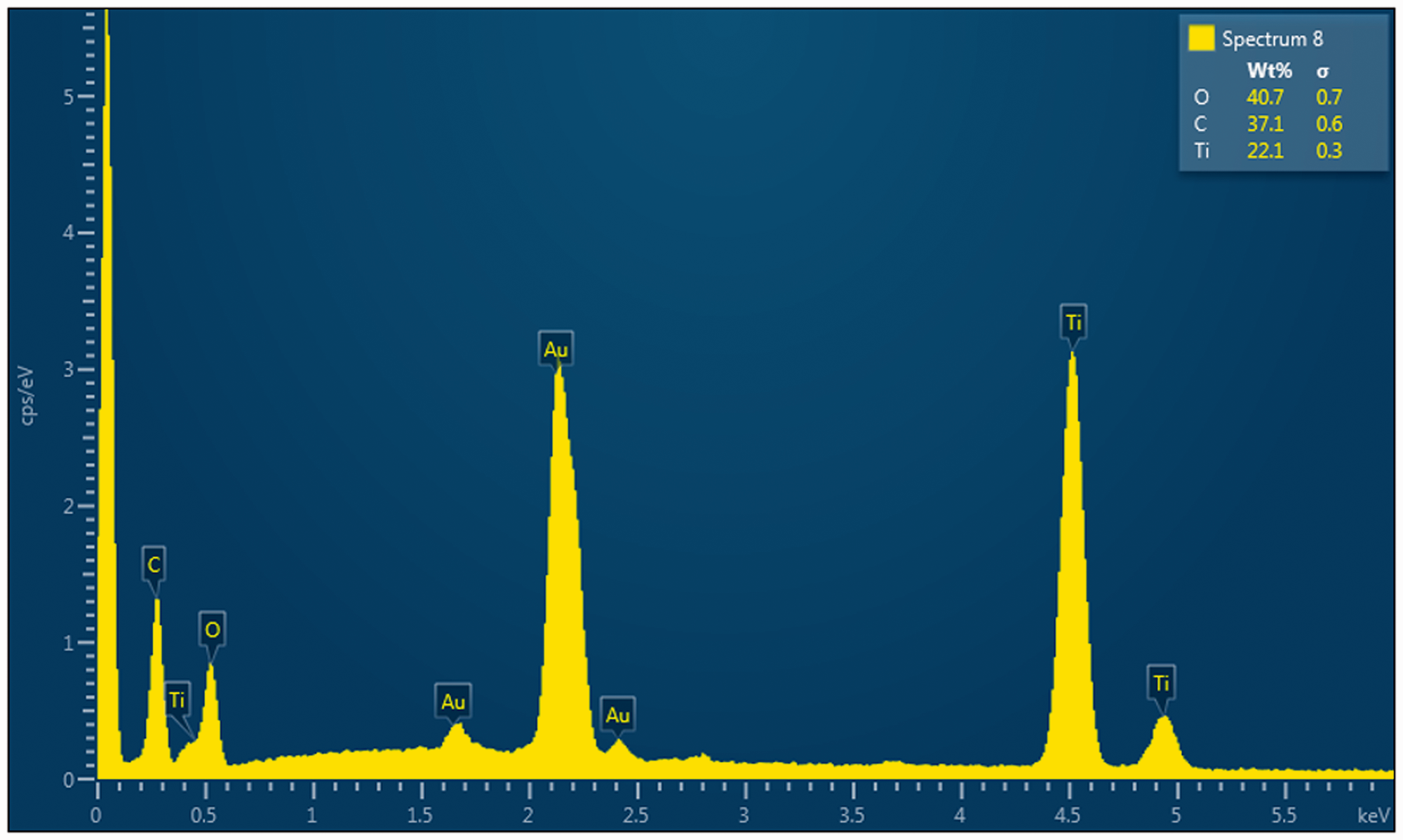
Raman spectroscopy
It was employed to investigate the crystal structure of TiO2 flower-shaped 3D structures coated on the polyester fabrics. The Raman spectra shown in Figure 5 displayed the vibration modes at about 231, 447, and 608 cm−1, which can be attributed to the second-order Raman scattering of tetragonal structure. The Raman peak observed at 146 cm−1 indicated the formation of the anatase TiO2 phase [27,28], whereas the two strong Raman peaks observed at 447 cm−1 and 608 cm−1 confirmed the formation of crystalline rutile phase [29–31]. The intensity and peak positions of observed Raman spectra are in good agreement with the previous studies reported in the literature.
Raman spectra of flower-like TiO2 nanoparticles coated on polyester fabric.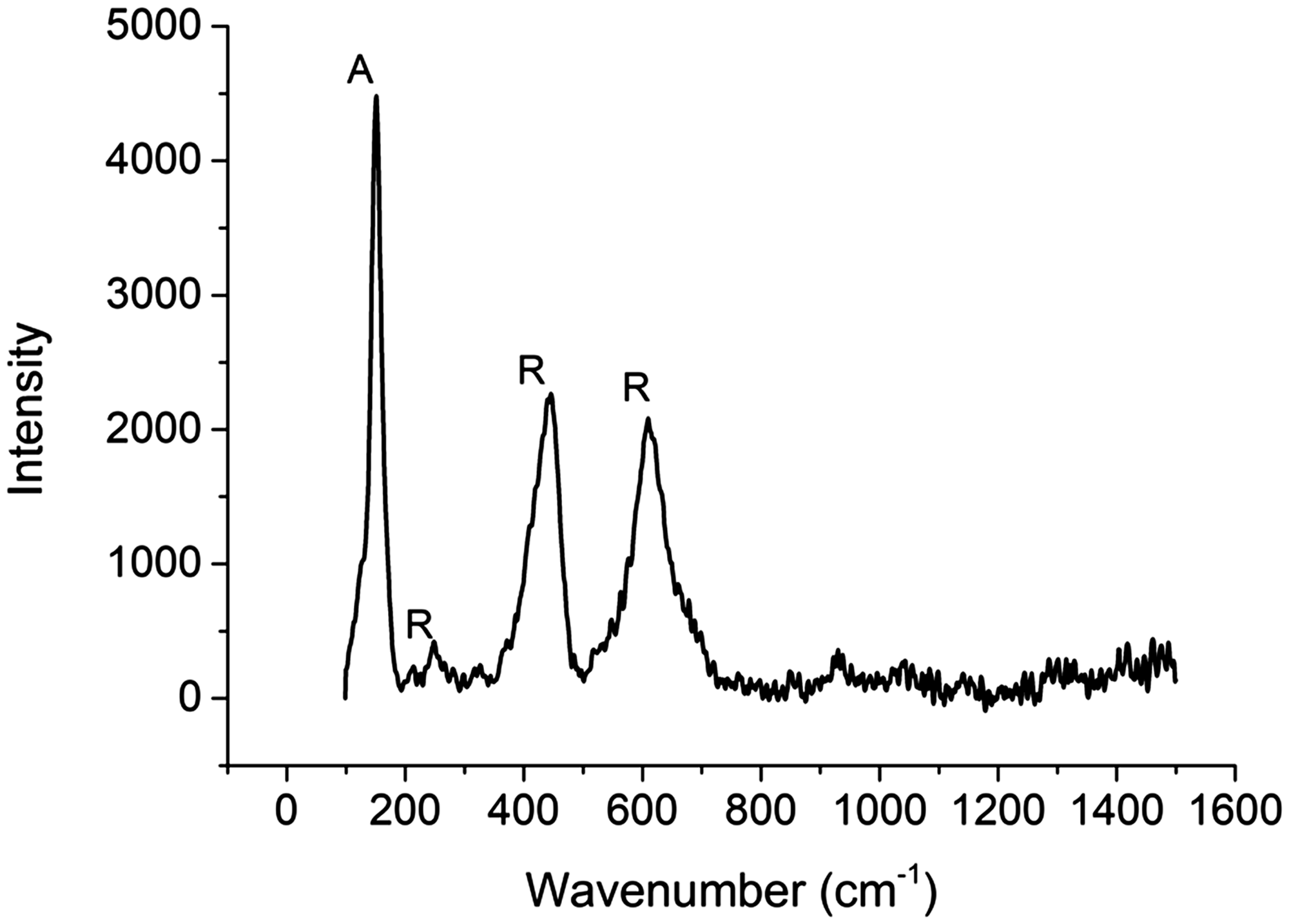
XRD analysis
It was carried out to determine the crystal phase of the complex TiO2 nanostructures coated onto the polyester fabrics. As discussed in previous section of Raman spectroscopy, the existence of both anatase and rutile TiO2 phases in the synthesized TiO2 flower structures was confirmed (see Figure 6) [30]. The Bragg peaks observed at 27.08, 36.02, 41.24, 54.26, and 62.89 (2θ) were due to (110), (101), (111), (211), and (002) planes of tetragonal rutile phase (JCPDS no. 76-0317). The peaks observed at 25.29 and 48.01 (2θ) were due to (101) and (200) planes of the anatase phase (JCPDS no 21-1272) [32,33]. Further, the shapes of the diffraction peaks suggested the formation of highly crystalline TiO2 structures.
XRD pattern of flower-like TiO2 nanoparticles coated on polyester fabric.
Physical properties of TiO2-OTMS-coated polyester fabrics
The add-on of coated fabrics at different TiO2-OTMS coatings were calculated, and it was found to increase in linear pattern with increase in TTIP concentration. The final dry add-on of the coated fabrics was measured as 1.8 % for 1 mL TTIP, 4.2 % for 1.5 mL TTIP, and 7.3 % for 2 mL TTIP concentrations. Subsequently, the hand (i.e. comfort) properties of the coated fabrics were assessed from the measurements of stiffness values. Stiffness is a tendency of the fabric to keep standing without support. It is a special property of the fabric for desirable draping and also influences the physical comfort of clothing [34–36]. It can be evaluated from bending properties of the fabric using bending length and flexural rigidity. Despite large increments in add-on of coated fabrics at higher TTIP concentration, the stiffness values were found to increase marginally. The stiffness of uncoated polyester fabric was 0.93 N m, whereas it was measured as 1.16, 1.32, and 1.42 N m for TiO2+OTMS-coated polyester fabrics at 1 mL, 1.5 mL, and 2 mL TTIP respectively. This indicated no loss in the draping behavior of TiO2-OTMS-coated polyester fabrics and therefore there was satisfactory physical comfort behavior.
Superhydrophobization of TiO2 nanoflower-coated polyester fabrics
The measurements of static water contact angle, roll-off angle, and contact angle hysteresis were made to understand the effect of surface decoration of TiO2 flowers on surface wettability of TiO2-OTMS-coated polyester fabrics (Figure 7). The water droplet depicted a spherical shape on the surface of TiO2-OTMS-coated textile. On the contrary, the surface of uncoated polyester fabric was partially wetted by the water droplet. Without any decoration of TiO2 flowers and with only coating of OTMS on polyester surface, the static water contact angle and roll off angle were measured around 129.8° and 15°, respectively. This indicated the hydrophobic behavior of polyester fabrics after OTMS coating alone. However, this surface was not suited for physical self-cleaning action, as the water droplets were difficult to roll off showing the strong adhesion between the water droplet and the coating surface. When polyester fabric surface was decorated with 3D-shaped TiO2 flowers and subsequent treatment of OTMS, the enhancement in contact angle and simultaneous reduction in roll off angle was found. The increase in contact angle and reduction in roll of angle was observed with the increase of TTIP concentration during the hydrothermal deposition of TiO2 flowers (Figure 7(a)). The maximum static contact angle of 160.1° and minimum roll off angle of 3° was found for 2 mL of TTIP concentration. Therefore, the improvement in water repellency from surface hydrophobicity to superhydrophobicity can be confirmed when polyester fabric surface is decorated with TiO2 nanoflowers before the OTMS coating. This phenomenon can be attributed to the formation of unique two-tier structural surface combined with the microscaled polyester fibers (5 µm) and the nanoscaled TiO2 flowers (500 nm).
Effect of titanium isopropoxide concentration on contact angle. (a) Static contact angle and (b) contact angle hysteresis.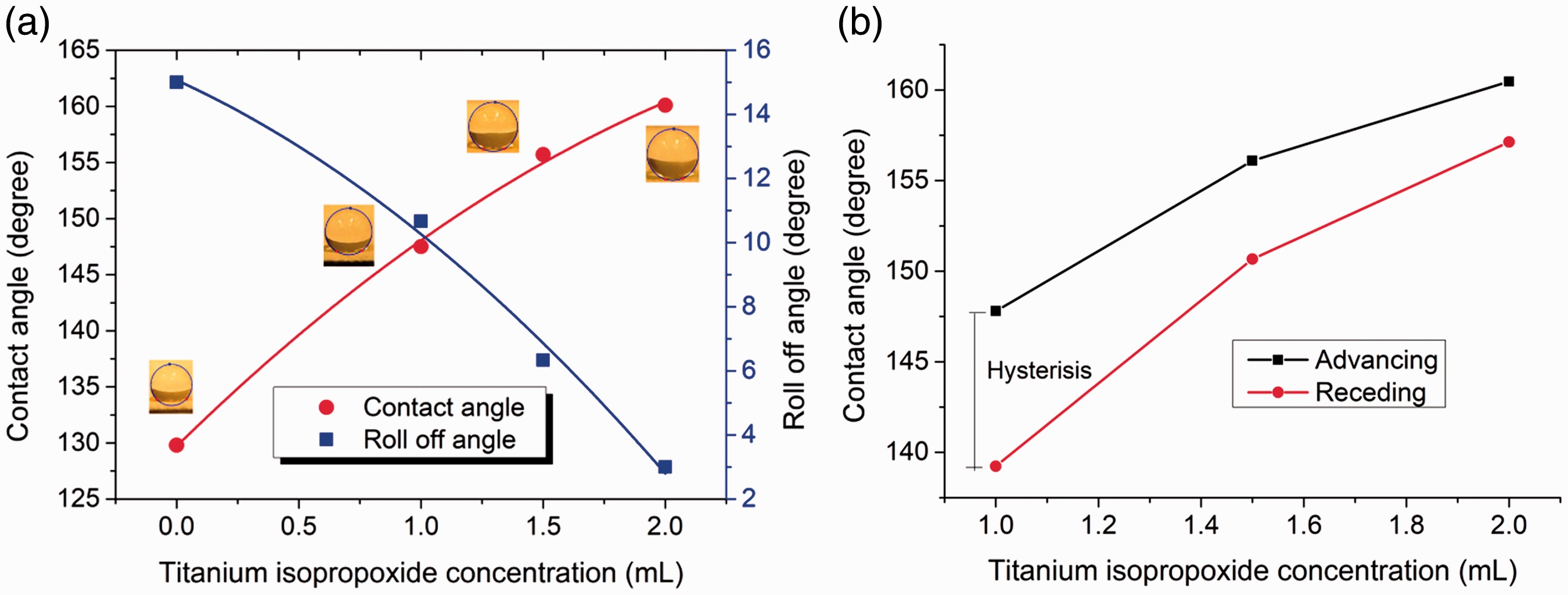
Due to the surface roughness and the heterogeneity of textile surfaces, the measured contact angle after the liquid drop advances is usually greater than the contact angle that results after the droplet recedes from a previously wetted surface [37,38]. Therefore, the measurement of static contact angle results can be misleading for the assessment of physical self-cleaning action. The contact angle hysteresis (i.e. difference between the advancing and receding contact angles) plays an important role in the sliding behavior of water droplets for self-cleaning effect [39,40]. Here, the advancing and receding angles of TiO2-OTMS-coated polyester fabrics were measured to examine the influence of TiO2 flower structures on the self-cleaning effect. From Figure 7(b), the contact angle hysteresis was found to reduce from 9° to 3° with increase of TTIP concentration from 1 mL to 2 mL, which indicated the formation of discontinuous, unstable, and contorted air/solid/liquid contact line due to the decoration of TiO2 flowers. For superhydrophobicity, the solid–liquid–air contact line can further become more discontinuous and a larger amount of air film can be trapped by depositing high density of TiO2 flowers using higher concentration of TTIP.
Based on previous studies, the wetting behavior of liquids can be explained either by Wenzel state or Cassie–Baxter state [41]. In Wenzel state, a liquid impregnates the rough surface to form a completely wetted contact with the surface, whereas the liquid does not completely fill up the rough surface due to air pockets in the Cassie–Baxter state. Therefore, the superhydrophobicity of the TiO2-OTMS-coated polyester fabrics can be believed in Cassie–Baxter state, where spherical water drop cannot penetrate entire fabric surface due to large amount of air trapped in the structure [38]. Further, the Cassie–Baxter state of TiO2-OTMS-coated polyester fabric was verified by immersing the coated fabrics into water. The trapped air between the water and the superhydrophobic fabric resulted in the formation of plastron layer and thus showed the silver mirror-like bright and reflective surface [42]. The Cassie–Baxter model given in equation (2) can be used to estimate the area fraction of a water droplet Analysis of superhydrophobicity of TiO2-OTMS-coated polyester fabrics. (a) Cassie–Baxter prediction, (b) work of adhesion and (c) force of attraction.
Furthermore, the Young–Duprè equation given in equation (3) was employed to describe the movement of liquid droplets on hydrophobic surfaces by calculation of work of adhesion
For the determination of the maximum force of attraction, the work of adhesion and the work of separation can be equated. Equations (4) and (5) describe the attractive force
Under equilibrium conditions
Therefore, the work of adhesion can be expressed as given in equations (6) and (7)
At the end, the force of attraction Fe was estimated by knowing the values of work of adhesion
Durability of superhydrophobic properties of coated polyester fabrics
The durability of TiO2-OTMS-coated polyester fabrics against mechanical abrasion, laundering, chemical, and UV action can be seen from the results of contact angle measurements shown in Figure 9(a). After 20 cycles of sand paper abrasion, the water contact angles were reduced for all the samples of different TiO2 depositions. However, the samples coated with TTIP concentrations of 1.5 and 2 mL continued to maintain their superhydrophobicity and presented the respective contact angles of 153° and 156° after mechanical abrasions. Likewise, the action of five laundering cycles resulted in the decrease of contact angles, but still higher than 150° for the samples coated with more than 1.5 mL of TTIP. The removal of only few particles for samples of 1.5 mL TTIP concentration due to shearing and friction forces indicated the robust and stable coating of TiO2 particles over polyester fabrics (see Figure 9) [5].
Durability of surface microstructure TiO2-OTMS-coated fabrics (a) against mechanical abrasion and (b) against laundering.
Further, all samples showed acceptable reductions in superhydrophobic properties after exposure to chemical environment. The reductions in contact angle after exposure to alkaline environment was found greater than the acidic environments due to the itching of polyester fiber surfaces in presence of alkali. As many types of materials lose their superhydrophobicity when exposed to ultraviolet irradiation, the ultraviolet durability of superhydrophobic TiO2-OTMS-coated polyester fabrics was also studied [43]. Compared to mechanical and chemical actions, the water contact angle of coated fabrics was not affected after exposure to ultraviolet rays and thus suitable for use under the solar radiations. Lastly, the effect of jet of water when impacted on coated fabric surface was examined to verify the durability of contact angles. The time-lapse photographs of water droplets bouncing on the fabric are shown in Figure 10(b), where water droplets completely left the fabric surface without wetting and thus confirmed their super hydrophobic behavior.
Durability of TiO2-OTMS-coated polyester fabrics. (a) Contact angle before and after exposure and (b) impact of water jet and bouncing of water droplets.
Physical self-cleaning properties of coated fabrics
Due to the superhydrophobic behavior and low roll off angles, the utility of TiO2-OTMS-coated polyester fabrics was studied for physical self-cleaning performance. The methyl orange dye was used as contaminant to demonstrate the self-cleaning action. When the water droplets were dropped on the contaminated fabric, it rolled over the surface and maintained the spherical shape. During the sliding process of water droplets, the dye particles were immediately picked up and taken away leaving behind a clear surface as seen in Figure 11 [44]. This behavior was attributed to the creation of lotus effect by high water surface tension and low surface energy of coated fabrics [45].
Physical self-cleaning property of TiO2-OTMS-coated polyester fabric.
Chemical self-cleaning properties of coated fabrics
The chemical self-cleaning properties of coated fabrics were examined based on two aspects (i.e. stain degradation and solution discoloration tests). For stain degradation performance, the methyl orange dye was applied as stain on the coated fabrics and their photo degradation behavior towards the dye was observed at different time intervals of exposure to the ultraviolet light. From Figure 12, the significant degradation of methyl orange dye was found for all the coated samples decorated with different concentrations of 3D-shaped TiO2 flowers. The reasons for low deviation under 30 min could be due to different locations of dyes and photocatalyst. When the dye stain was deposited on the coated fabric, the part of it which was on top of the TiO2 nanoflowers degraded quickly because it was completely exposed to UV light. On the other hand, the dye deposited on the sides of nanoflowers required more time to degrade. The stains disappeared mostly within 3 h, and the samples became approximately white after 4 h. Furthermore, the degradation rate was found to increase with increased density of TiO2 flowers coated onto fabric and also with increased time of UV exposure. On the other hand, the pristine polyester fabrics showed no change in stain reduction even after the exposure to UV light for more than 4 h.
Photocatalytic stain degradation of TiO2-OTMS-coated polyester fabric.
Later, the photocatalytic degradation rate of methyl orange dye was quantitatively estimated by measuring the whiteness index of samples on ImageJ software. The increase in measured color intensity from this software can be correlated to increase in the whiteness index (see Figure 13). Interestingly, the depth of stains was found much higher before the start of UV irradiation on samples decorated with TiO2 flowers than the sample coated with OTMS alone. This can be attributed to hydrophilic nature of TiO2 particles deposited onto the polyester fabrics. However, from the measurements of color intensity, the dye degradation of samples decorated with TiO2 flowers was found to increase after the UV irradiation, whereas the sample coated with OTMS alone showed a straight line and thus no degradation of dyes in absence of TiO2 particles. The photocatalytic activity of TiO2 coated textiles can be attributed to the decomposition of dyes by generation of highly oxidative radicals under the UV light [11].
Whiteness index of TiO2-OTMS-coated polyester fabric after photocatalytic degradation.
Another test of solution discoloration was also employed to characterize the chemical self-cleaning properties of coated fabrics. Here, the coated samples were added into the dye solution and then the dye concentration was measured by recording the UV-Vis spectra. Figure 14(a) shows the absorbance spectra of the dye solution at different UV illumination times. The straight line obtained for the pristine polyester fabric indicated the absence of any photocatalytic activity by polyester itself. The absorbance value of methyl orange dye in the solution was measured by observing the peak intensity at 485 nm. The decrease in absorbance value with the passage of time indicated the decrease in the concentration of dye due to its photocatalytic degradation. The samples coated with 2 mL of TTIP decolorized the dye solution in 150 min, whereas the samples coated with 1 mL and 1.5 mL of TTIP took almost 300 and 210 min, respectively. The higher rate of dye degradation in case of 2 mL of TTIP can be attributed to the decoration of high-density 3D-shaped TiO2 flowers on the fiber surface. Furthermore, the photocatalytic efficiency of the sample coated with 2 mL of TTIP was examined for repeated discolorations of dye solution. From Figure 14(b), no change in photocatalytic efficiency of treated fabric was found during the five successive discoloration cycles which confirmed its potential use for several times in the application.
Solution discoloration properties of TiO2-OTMS-coated polyester fabrics. (a) Effect of TTIP concentration and (b) effect of successive reuse cycles.
Mechanism of photocatalytic dye degradation
When TiO2 was illuminated by light energy higher than its band gaps, the electrons in TiO2 jumped from the valence band to the conduction band and then formed electron (e−) and electric hole (h+) pairs on the surface of the photocatalyst. The negative electrons and oxygen combined into O2−, whereas the positive electric holes and water generated hydroxyl radicals. When the organic compound falls on the surface of the photocatalyst, it can combine with O2− and OH−, respectively, and turn into carbon dioxide and water. In the present study, the degradation of dye was started with the azo bond cleavage by highly oxidizing hydroxyl radicals and was followed by the hydroxylation of the aromatic ring.
Conclusions
The concentration of TTIP was found to have a great effect on the growth and structure of TiO2 nanoflowers. The assembly of TiO2 particles in flower shape was not found at low concentration of TTIP. However, the amount and the size of TiO2 flower structures were greatly increased by further increase in TTIP concentration from 1 mL to 2.0 mL. The increase in contact angle and reduction in roll of angle was observed with the increase of TTIP concentration, where maximum static contact angle of 160.1° and minimum roll off angle of 3° was found for 2 mL of TTIP concentration. Furthermore, the formation of discontinuous, unstable, and contorted air/solid/liquid contact line was found, as the contact angle hysteresis reduced from 9° to 3° with increase in TTIP concentration. From the Cassie–Baxter state of superhydrophobicity, 58%, 78%, and 83% increase in unwetted fraction of the air pocket was calculated for 1 mL, 1.5 mL, and 2 mL TTIP concentrations, respectively. Moreover, the reduction in work of adhesion (11.4 to 4.34 mN/m) and force of attraction (10.27 to 3.9 10−8 dyne/cm2) indicated less force required for movements of water drops on the polyester fabric surface decorated with more number of TiO2 flowers. The surface of TiO2-OTMS-coated polyester fibers also showed the promising results to maintain the superhydrophobic durability against mechanical abrasion, laundering, chemical and UV action. When the coated fabrics were examined for physical self-cleaning, the methyl orange dye particles were immediately picked up and taken away by the rolling water droplets. The coated fabrics also exhibited the chemical self-cleaning action, as the stains of methyl orange dye disappeared mostly within 3 h and the samples became approximately white after 4 h.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the research project of Student grant competition of Technical University of Liberec no. 21312 granted by Ministry of Education, Youth and Sports of Czech Republic. This work was supported by the Ministry of Education, Youth and Sports of the Czech Republic and the European Union – European Structural and Investment Funds in the frames of Operational Programme Research, Development and Education – project Hybrid Materials for Hierarchical Structures (HyHi, Reg. No. CZ.02.1.01/0.0/0.0/16_019/0000843) and project Modular platform for autonomous chassis of specialized electric vehicles for freight and equipment transportation (Reg. No. CZ.02.1.01/0.0/0.0/16_025/0007293).








