Abstract
There is a growing interest in the development of natural fiber-reinforced composites, most likely due to their wide availability, low cost, environment friendliness, and sustainability. The market size for natural fiber-reinforced composites is projected to reach $5.83 billion by 2019, with a compound annual growth rate of 12.3%. The composite materials reinforced with wood, cotton, jute, flax or other natural fibers fall under this category. Meanwhile, some major factors limiting the large scale production of natural fiber composites include the tendency of natural fiber to absorb water, degradation by microorganisms and sunlight and ultimately low strength and service life. This paper has focused to review the different natural fiber treatments used to reduce the moisture absorption and fiber degradation. The effect of these treatments on the mechanical properties of these composites has also been summarized.
Introduction
The composite material is a combined assembly of two or more constituents, giving a synergizing effect in the resulting material. The matrix and reinforcement are two major components of the composite material. The matrix glues the reinforcement and provides desired shape and properties and may be either thermoplastic or thermoset in nature [1]. The thermoset polymeric resins harden or cure irreversibly when subjected to a specific curing temperature [2]. The reaction is exothermic and heat is produced as a result of polymer hardening [3]. Reinforcement is the bunch of fibers that carry the stress applied on composite and are largely responsible for mechanical behavior of the composite.
The glass fiber reinforced polymer composites (GFRPC) are the largest category of fiber-reinforced composites (FRP), as 87% of the 8.7 million ton global FRP market is based on E-glass composites [4]. Glass fiber has high density (2.50 g/cm3). It is obtained from non-renewable source [5] and is not bio-degradable. This has led to many problems including waste disposal and health concerns as glass fibers are suspected to cause skin irritations and lung cancer [6]. Every year heaps of waste are being added on the planet which is difficult to dump. There is also need to cut short the use of petroleum based materials and use environmental friendly sustainable materials.
All these things have impelled the researchers to focus on eco-friendly composite materials. There are different types of eco-composite materials, like biodegradable polymer based materials, natural fiber reinforced materials, etc. The natural fibers based eco-composite materials are more suitable for use [7] as compared to biodegradable polymeric materials due to two main reasons: cost and availability. The advantages of natural fibers over synthetic fibers include their low cost, low density, biodegradability, and recyclability [8, 9] as shown in Table 1, while Figure 1 presents the cost per unit weight of natural fibers [10].
Cost per weight comparison of natural fibers [10]. Comparison between natural fibers and synthetic fibers [92].

Natural fibers are abundant, based on renewable source, low cost, and have less density [11]. These properties reveal new horizon for researchers to replace the synthetic fibers reinforcements in composites [12]. Research in the field of natural fiber reinforced plastics has revealed that their mechanical properties are comparable to those of glass fiber-reinforced composites. Therefore the natural fiber reinforced plastics are mostly used to produce structural components of automotive industry [13] such as panels, doors, roofs and covers [7], furniture articles like office chairs, door panels, safety helmets, etc. [14]. The car interior and exterior parts have been manufactured by integrating natural fiber composites, serving a two-fold goal; to lower the overall weight of vehicle, thus increasing fuel efficiency and to increase sustainability of manufacturing process. Companies like Mercedes Benz, Toyota, and Daimler Chrysler are already using natural fiber composites [5]. The major advantages of using natural fibers in composites are the cost of materials, their sustainability, and low density as compared to glass fiber [14].
Classification of natural fibers
Natural fibers have good environmental impact as they solve the disposal problem, associated to polymeric composites. In short, the natural fibers are green substitute of synthetic fibers in composite applications. The natural fibers are classified on the basis of origin into plant, animal, and mineral. Plant based fibers are used extensively because of their renewability and availability [7]. Further subdivisions of plant fibers are: bast, leaf, seed, fruit, and wood, depending on the part of plant from which fibers are obtained [15]. Mostly used plant fibers are cotton, hemp, flax, kenaf, jute, bamboo, sisal, and coconut. Animal fibers are made up of proteins; for example silk, wool, hair, etc. Mineral fibers are not preferred in technical applications due to their carcinogenic properties [16].
Comparison between the properties of different reinforcements [7].

Structure of plant fiber
The plant fiber cell wall is divided into two sections as shown in Figure 2. First is the primary cell wall which contains irregular network of cellulose micro fibrils that are closely packed. The second wall is known as secondary wall, which is composed of three separated layers S1 (outer layer), S2 (middle layer), and S3 (inner layer). The S2 layer is responsible for mechanical properties of the fiber [22], which are dependent on the degree of polymerization, cellulose content, and microfibrillar angle. Fibers with higher degree of polymerization, higher cellulosic content, and a lower microfibrillar angle exhibit higher tensile strength and modulus [23].
Structure of the fiber cell wall.
Pectin is also polysaccharide which holds the fibers together, while lignin is a complex hydrocarbon polymer with both aromatic and aliphatic constituents. It absorbs less water, insoluble in most of the solvents and cannot be broken down into monomeric units. The glass transition temperature (Tg) of lignin is around 900℃ and melting temperature around 1700℃. It is considered to be thermoplastic polymer having amorphous nature. The lignin is not hydrolyzed by acids, but soluble in hot alkali, readily oxidized, and easily condensable with phenol [24,5]. The chemical structure of cellulose, hemicellulose, pectin, and lignin is shown in Figure 3 [25].
Structural representations of (a) cellulose, (b) hemicellulose, (c) pectin, and (d) lignin.
Extraction of natural fibers
Various methods are used for the extraction of natural fibers from the plant stems. Retting is the process that allows fibers to be separated from the woody core by controlled degradation of plant stem [26]. The retting of the straw is carried out with time by exposing it to moisture; sometimes mechanical decorticator is also used. The available techniques of retting are given in Figure 4. Most of the available methods of retting are based on the biological activity. In this process microorganism fungi and bacteria from the environment degrade the polysaccharides and hence separate the fiber bundles. The fiber extraction process has a major impact on final fiber quality and yield. To extract good quality cellulosic fibers from plants, microbial retting is one of the widely used techniques [27].
Retting techniques for fiber extraction.
Sain and Panthapulakkal used fungal retting for extraction of wheat straw fibers; the fungus was obtained from the bark of an elm tree. The enzymes produced by the fungus removed the pectinic glue holding the fiber bundles together and released cellulosic fibers from the stem. To demonstrate the effect of extraction process on the fiber quality, they also defibrillated the wheat straw mechanically (using laboratory-scale mechanical refiner) before and after fungal retting. They finally confirmed that extraction technique influences the chemical composition, structure, and properties of the fibers. The four broad categories of retting involve the chemical, biological, mechanical, and physical fiber separation process [28].
Biological retting
Biological retting is of two types: natural and artificial. The natural retting comprises field or dew retting and cold water retting. Field retting is the most commonly applied retting process in regions that have specific temperature and moisture ranges. The crops after being mowed are left on the fields until the microorganisms separate the fibers from the xylem and cortex. To prevent over-retting, the process needs to be stopped at the right time. In cold water retting, anaerobic bacteria is used that breakdowns the pectin of plant straw bundles submerged in huge water tanks, ponds or rivers, and vats. The process takes about 6–14 days and depends on the type of bacteria and temperature of the retting water. Environmental pollution is high due to unacceptable organic fermentation waste waters, even though the process produces high quality fibers. Artificial retting includes canal or warm-water retting to produce fibers of high quality in 4–5 days. The bast fibers are soaked in warm water, and separated after sufficient retting [29].
Mechanical extraction
A cost effective and simple method is to separate bast fiber from plant straw by mechanical means. The fibers produced by decortication are much coarser as compared to dew or water retting. It is also termed as the decortication technique, and is based on a simple mechanical process using toothed breaking rollers for the decortication of bast fibers conventionally. This technique is most suitable for retted fibers. It frees the fiber from stem in the form of strands by removing the bark and woody parts of the stem. When the stem comes in proximity of breaking roller, the rotating blades strike the stem and remove the green ribbon by breaking it into pieces [30]. The fiber produced by the decorticator requires additional cleaning to remove the shives. There are 20–25% impurities left with fiber after decortication, which are reduced to 4–5% after cleaning.
In advanced decortication technique, bast fibers are decorticated by impact stress and is suitable for both unretted and retted plants [31]. The impact stress is generated by thin hammers, which impact the surface of the stem with a very high speed of approximately 60 m/s. This thorough stress makes it possible to process the bast fibers without any retting and ensures a complete mechanical separation effect of fibers and shives. It differs from the conventional technology, where slow running crushing rollers are used, which require an intensive retting of straw stems before processing.
Physical retting
The physical retting involves ultrasound and steam explosion methods for the separation of fibers from plant straw. The stems are harvested and crushed; then immersed in hot water bath that contains small amounts of alkali and surfactants and subsequently exposed to high-intense ultrasound. This process separates the hurds (coarse parts of flax or hemp that adhere to fiber after it is separated) from the fiber. The steam explosion method shows another suitable alternative to the traditional field-retting procedure. Under high temperature and pressure, steam penetrates the spaces between fiber bundles in the bast. This sudden relaxation of the steam leads to an effective breaking up of binders, which subsequently results in an extensive decomposition into fine fibers [32].
Chemical and surfactant retting
This type of retting refers to the retting process in which the straws of fiber are submerged in heated tanks. These tanks contain aqueous solutions of sulfuric acid, sodium or potassium hydroxide, chlorinated lime, and soda ash to dissolve the pectin. The unwanted non-cellulosic components adhering to the fibers are removed by the use of surface active agents. High quality fibers are produced by chemical retting, but this adds costs to the final product [33].
Natural fiber composites
The natural fibers are considered as environment friendly reinforcement for composite materials, having potential of use in various applications. Different natural fibers (e.g. sisal, flax, jute, kenaf, coir, etc.), offer a number of advantages over synthetic fibers due to their renewable nature. Some of the unique properties of natural fibers include: low cost, biodegradability, recyclability, low density, good thermal properties, reduced tool wear, non-irritation to the skin, and enhanced energy recovery [34]. The acceptable mechanical properties of natural fiber composites such as elongation, ultimate breaking force, flexural properties, impact strength, acoustic absorption, suitability for processing, and crash behavior increases its demand for automobile components [35].
These natural fiber composites reinforce either thermoset or thermoplastic resins. The natural fiber-reinforced thermoplastic composites have been studied by a number of researchers for automotive applications. Among various thermoplastic polymers, polypropylene (PP) is perhaps one of the most widely used because of its distinct properties such as dimensional stability, high heat distortion temperature, flame resistance, and transparency. The thermoset resins reinforced with natural fiber have been prepared for structural applications. Phenolics, epoxy, unsaturated polyester (UP), and vinyl ester are the most widely used thermoset matrices for this purpose [19]. Most thermosetting composites are processed by simple processing techniques such as hand lay-up, spraying, resin transfer, compression, injection, and vacuum bag molding.
Flax, hemp, jute, sisal, and bamboo are the most popular reinforcement materials in bio-composites. Rice husk is used as filler for the composite materials [36,37]. The textile industry cotton waste is also used as reinforcing material for the composites, having tensile strength of 27 MPa [38]. The tensile strength and modulus of jute reinforced polyester composite are 60 MPa and 7 GPa, respectively [39], while reinforced polyester composites have tensile strength and modulus 249 ± 25 MPa and 23.3 ± 3.3 GPa, respectively [40]. Polymer matrix reinforced by woven fabric are most commonly in structural applications because it allows the control of fiber orientation and has good reproducibility and high productivity [41]. Hybrid composites are used to obtain the tailored properties, which is difficult to achieve with single fiber. The researchers reported a combination that natural/natural and natural/synthetic fiber-based hybrid composites have certain advantages over individual fiber-reinforced [42].
The incorporation of hydrophilic natural fibers in the polymer matrix results in a heterogeneous system, having inferior properties due to lack of adhesion between fibers and the matrix. Therefore, it is important to improve the fiber–matrix adhesion for the development of natural fiber-reinforced composites. The issues in the development of natural fiber-reinforced composites can be grouped into three categories namely thermal degradation, moisture absorption, and biodegradation.
Thermal degradation
The thermal degradation of natural fibers is a bad aspect in the development of natural fiber composites. The chances of thermal degradation increases with increase in the amount of natural fibers (fiber volume fraction) in polymer matrix composites [43]. It has been observed that in natural fibers, the thermal decomposition of about 60% mass fraction occurred within a temperature range of around 100℃ [44]. Flax fibers are thermally degraded at temperature around 200℃. It was investigated that initially degradation of flax fibers was not significant at 170℃ for 120 min, but the strength decreased at 210℃ by approximately 50% [12].
Moisture absorption
All the cellulosic fibers being hydrophilic in nature absorb moisture from environment until equilibrium is established. The moisture regain of natural fibers vary between 5% and 12%, as shown in Table 2. The moisture absorption causes dimensional variations in the fiber as well as composite material, thus affecting the interface and the mechanical properties of composite material. During composite fabrication, the moisture may lead to poor fiber–matrix interface. There will not be effective transfer of load due to poor interface and the composite material will deteriorate. Treatment of natural fibers with different chemicals [45] or grafting of vinyl monomers may help to reduce its moisture regain [46, 47].
Biodegradation and photo degradation
As discussed in the previous section, natural fibers are mostly composed of high molecular weight carbohydrate polymer, cellulose, or proteins. The microorganisms degrade lignocellulose present in the natural fibers. They can attack carbohydrate polymers in the cell wall, resulting in loss of strength. Lignocellulose also undergoes photochemical degradation when exposed to ultraviolet light. Resistance against biodegradation and UV radiation can be improved by bonding chemicals to cell wall polymers or by adding polymer to cell matrix.
Shaker et al. [48] investigated the bio-functionality of flax fiber-reinforced composites by using ZnO nanoparticles. The bioactivity was tested in terms of antibacterial activity (zone of inhibition). The ZnO nanoparticles have enhanced bioactivity against microorganisms [49]. The resulting composite material was bioactive even with lowest amount (0.02%) of ZnO nanoparticles. Galashina et al. [50] immobilized silver nanoparticles in flax fiber composites to impart protection against microorganisms. The silver nanoparticles are also antimicrobial [51] and the as-produced bio protected composites are supposed to have lower risk of fiber degradation and enhanced service life, by restricting the growth of bacteria.
Modification of natural fibers
Natural fibers being hydrophilic are incompatible with the hydrophobic polymer matrix. Fiber–matrix interface is affected by hydrophilic nature of cellulosic fibers [52]. In addition to this, waxy and pectin substances cap the reactive functional groups of the fiber and act as a hindrance to interlock with the matrix. Fiber surface needs to be modified with different chemical treatments to enhance the effectiveness of interfacial bonding [53]. The treatments performed for this purpose include grafting of monomers, bleaching, acetylation, etc. Grafting of fibers with monomers also helps to improve the thermal stability of the natural fibers. The compatibilizer or coupling agent can also be used for the effective stress transfer across the interface. Compatibilizer is a polymer with functional groups grafted onto the chain of the polymer. Coupling agents are tetra functional organometallic compounds based on titanium, silicon or zirconium and are commonly known as silane, titanate, and zirconate coupling agents [52].
More reactive groups are exposed on the fiber surface by chemical treatment, which in turn gives better interface and enhanced mechanical properties of the composite [54]. Some chemical surface modification techniques include treatment with sodium chlorite [55], metha acrylate [56], isocyanate [57], silane treatment [58], acetylation [59, 60], mercerization [61–64], etherification [65, 66], enzymatic treatment [67–71], peroxide treatments [62], benzoylation [61], dicumyl peroxide treatment [57], plasma treatment [72–75], ozone treatments [76,77], and grafting [78–80]. The oxidation of polyolefin [81,82] has also been reported to improve the incompatibility between the surfaces of natural fiber and polymer matrix.
The pre-treatment of natural fibers helps to chemically modify or clean the fiber surface [83]. The natural fibers have a hydroxyl group due to cellulose and lignin; therefore they are amenable to modification. This hydroxyl group is involved in hydrogen bonding within the cellulose, thereby reducing the activity towards the matrix. Therefore a number of treatments are performed to improve the natural fiber composite strength, ageing and fiber matrix adhesion [79]. A well-engineered interface not only improves the strength of composites significantly but also gives the structural stability [84–86]. The properties and performance of composite can be controlled by the fiber–matrix interfaces chemistry and character. Good interfacial properties are vital to confirm efficient load transmission from matrix to reinforcement which aids to reduce stress absorption and improves overall mechanical properties.
The strength of reinforced composite materials depends not only on the substrate but also on the interface strength. The composites having good interfacial properties can be exposed to diverse range of environmental conditionings. On the other hand, if composites have poor interface then environments degrade fiber/matrix interfacial bond and leads in the loss of microstructural integrity. Consequently, the creep rates of polymers are highly sensitive to temperature and moisture diffusion along the fiber direction than that transverse to the fiber direction. As a result the net age of composite is reduced [87,88]. Following are some important treatments performed for different types of natural fibers.
Sodium chlorite treatment
To improve the mechanical and thermal properties and to enhance the hydrophobicity, Arifuzzaman Khan et al. [55] chemically modified the okra bast fiber by NaClO2 treatment. The extent of modification reaction was evaluated by the Fourier transform infra-red (FTIR) spectroscopy. Scanning electron microscopy and the wide angle X-ray diffractometry were done to observe the morphology and crystalline index of jute fibers. They observed a noticeable variation due to chemical treatment in the fiber surface and improvement in its tensile properties (i.e. tensile strength, young’s modulus and extension at break) [89].
Zahran et al. developed a novel chemical formulation for bleaching flax fibers in one-step, based on activation of sodium chlorite by hexamethylene tetramine (HMTA) in the presence of a nonionic wetting agent. They suggested that at optimum formulation, HMTA activates decomposition of NaClO2 to mainly liberate nascent oxygen rather than chlorine dioxide [90].
Treatment with methacrylate
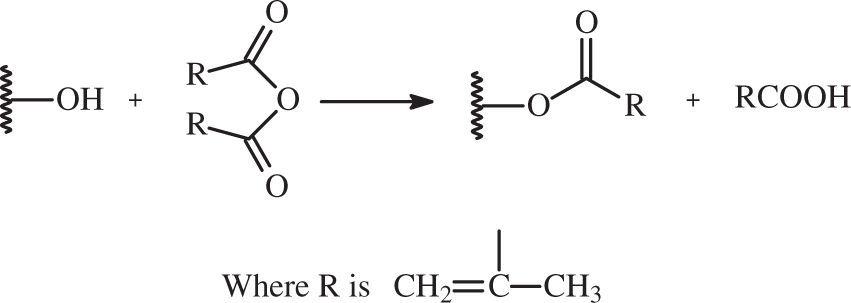
Canter et al. [56] performed the esterification of flax fibers to make them hydrophobic. They used 10 wt% methacrylate (MA) for 25 h at 50℃ and made flax/PP composite. They noted that the composites made by flax fibers showed good flexural and tensile strength. Kaith and Kalia [91] uses 20 wt% methyl methacrylate (MMA) and treated the flax fibers for 20 min then made the flax composite by using phenolic resin. They noticed less absorption of moisture in the treated flax fiber composites [59]. The treatment reaction is shown below [92]:
Silane treatment
Silanes act as coupling agents and to let glass fibers adhere to a polymeric matrix also stabilize the composite materials. Silane coupling agents reduce the hydroxyl groups of fibers and give better interface. Hydrolysable alkoxy group leads to the formation of silanols in the presence of moisture. Hydroxyl groups of the fibers react with silanol, forming stable covalent bonds to the cell wall that are chemisorbed on to the fiber surface. As a result silane restrains the swelling of the fibers by creating a cross-linked network due to covalent bonding. The reaction is as follows [93–95].

The flax fibers were made hydrophobic by silane treatment [58] and composites were fabricated using these treated and untreated flax fibers. UP was used as matrix and the interface of composite was studied.
The degree of crosslinking in the interface region can be improved by using the silane coupling agents. For the purpose of modifying the natural fiber matrix interface, silane coupling agents play a perfect role. Improvements in the interfacial adhesion and for getting better mechanical properties of fiber/polymer composites, proper treatment with silane is best by following the sol gel process. Actually silane is absorbed and condensed on the fiber surface. The hydrogen bonding is formed between the hydroxyl group of natural fibers and silane coupling agent. This link may be then converted to the covalent bonds by heating the treated fibers at high temperatures [96, 97]. Kalia et al. [23] investigated the effect of aminopropyl tri-ethoxy-silane (APS) to impart hydrophobicity in cellulosic fibers. Nevell et al. [24] observed improved mechanical properties and fungal resistance for UP and epoxy composites, after silane treatment.
Acetylation
A well-known esterification method is acetylation. Acetylation of natural fibers introduces plasticization to natural cellulosic fibers, mostly applied to stabilize the cell wall of wood cellulous against the moisture, environmental degradation and for improving the dimensional stability. When acetic anhydride is applied on lignocellulose material, it reacts with hydroxyl groups of cellulose and also prevents the diffusion of the reagents, as noticed by Yao et al. [44] who made flax/PP composite after treating the flax fiber. They have reported 18% increase in degree of acetylation along with significant increase in tensile and flexural strengths.
Acetylation reaction introduces an acetyl functional group (CH3COO–) into the fiber structure. The reaction involves the generation of acetic acid (CH3COOH) as by-product which must be removed from the lignocellulosic material before the fiber is used. Chemical modification with acetic anhydride (CH3–C(=O)–O–C(=O)–CH3) substitutes the polymer hydroxyl groups of the cell wall with acetyl groups, modifying the properties of these polymers so that they become hydrophobic [98]. The reaction of acetic anhydride with fiber is shown as:
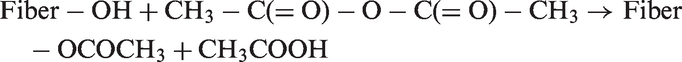

PP or polyethylene (PE) in a toluene or xylene solution is usually used for anhydride treatment. Fibers are impregnated in it to carry out the reaction with hydroxyl groups on fiber surface. Hughes et al. [99] noticed that propionic anhydride (PA) and methacrylic treatment on fiber surface showed better yield properties. Cantero et al. [56] reported that modification mechanisms are of two types: (a) introducing a reactive vinylic group by methacrylic anhydride (MA) and (b) coating of hydrocarbons on surface by PA. They made flax fiber composite with methacrylic polypropylene (MAPP) and checked the strength. The strength of unmodified flax fiber composite was less than MA treated composite.
Mercerization
It is a process in which natural fibers are treated with alkali which leads to fibrillation and causes the breakdown of bundles of fibers into smaller fibers. Thereby, it leads to develop a rough surface topography, that results in better fiber matrix interface adhesion and an increase in mechanical properties [61]. Moreover, fiber active sites increase by mercerization process and allow better fiber wetting. Chemical composition, degree of polymerization, and molecular orientation are also affected and give long lasting effect on mechanical properties. The alkali treatment also helps to improve the properties of cotton fiber composites [100].
By this treatment there is a removal of hydrogen bonding in the network structure. Reaction which takes place during this treatment is shown below.

Sreekala et al. [62] showed that a 10–30% solution of sodium hydroxide gave the best results when they treated flax fiber with 2.5%, 5%, 10%, 15%, 18%, 20%, 25%, and 30% NaOH. They observed that 5%, 18%, and 10% were the best concentration for mercerization. Ray et al. [101] also treated the jute fibers with 5% NaOH solution for 0, 2, 4, 6, and 8 h at 300℃. Then dried at room temperature for 48 h followed by oven drying at 100℃ for 6 h. Garcia-Jaldon et al. [102] found that 2% alkali solution at 200℃ for 90 s at 1.5 MPa is enough for defibrillation and degumming to individual fibers. Several workers performed mercerization and reported that mercerization leads to increase in the amount of amorphous cellulose and also removal of hydrogen bonding [63].
Jute fibers were washed using detergent (2 vol% in aqueous solution) then immersed in 5 wt% NaOH solution for 24 h at room temperature. Then again washed with distilled water to remove the excess NaOH and then dried at 70℃ for 24 h by using vacuum. Likewise banana fibers were also cleaned and refluxed in 0.25% solution of sodium hydroxide for 1 h and then washed in very dilute acid for removing non-reacting alkali. The fiber washing was carried out until they were alkali free, after that fibers were dried in an oven at 70℃ for 3 h [103,104].
The chemical composition of natural fiber is affected by mercerization process, as well as the degree of polymerization and molecular orientation of cellulose crystallites, while removing the lignin and hemicellulose [64]. This type of treatment also converts the crystalline form of cellulose I to cellulose II [105]. This transformation could be seen by FT Raman spectroscope by the intensity ratio of the stretching modes of symmetric (C–O–C) and asymmetric proposed by Bledzki et al. [64]. Wang et al. [106] checked the chemical properties of mercerized flax fiber composite. The treated flax fibers were used as reinforcement and polystyrene was used as matrix. They observed that mercerization of flax fiber improved the mechanical properties of polystyrene composites. In the same way Bledzki et al. [107] investigated the effect of acetylation on the flax/PP composites and noticed the increase in mechanical properties, as well as increase in the thermal stability of treated flax fiber is due to the compositional change by removal of lignin and hemicellulose.
Jabbar et al. studied the creep behavior of alkali treated woven jute/green epoxy composites incorporated with different loadings of chemically treated pulverized jute fibers (PJF) was presented at various environment temperatures [108]. Yan et al. [41] improved the mechanical properties of natural fiber composites by using the alkali treatment of flax fiber. They used 5% NaOH for 30 min, and then made the flax epoxy composite. They noticed a significant improvement in tensile strength (21.9%) and flexural strength (16%) along with some improvement in transverse strength. In the same way Van de Weyenberg et al. [105] used the same alkaline treatment technique for flax/epoxy composite but they used 4 wt% NaOH, 45 s treatment time and noticed favorable enhancement in mechanical properties. Manikandan Nair et al. [109] immersed dewatered sisal fiber in 5% and 10% NaOH solution for 1 h at 30℃. These alkaline treated fibers were then soaked in glacial acetic acid for 1 h at 30℃ followed by soaking in acetic anhydride containing one drop of concentrated H2SO4 for 5 min. There was significant improvement in fiber surface properties after the treatment [110].
Oladele et al. [111]. investigated the effect of chemical treatment on the constituents and tensile properties of sisal fiber. The leaves of sisal were cut and buried underground, then wetted with water regularly close to the stream in order to subject proper fermentation for about 15 days. These fermented leaves were completely washed and sun dried. The chemical and mechanical treatment was done over dried sisal fibers then fraction of their constituents and tensile properties were characterized. The results showed the removal of lignin and hemicelluloses, which is responsible for the bonding strength of composite produced from natural fibers. The results of the tensile test revealed that sample treated sequentially with KOH have best tensile properties. Figures 5–7 show the SEM images of untreated and 8% NaOH treated sisal, jute, and hemp fibers [112].
SEM images of sisal fiber (a) untreated and (b) 8% NaOH treated. SEM images of jute fiber (a) untreated and (b) 8% NaOH treated. SEM images of hemp fiber (a) untreated and (b) 8% NaOH treated.


The key issue in terms of overall performance is the interface between the reinforcing agent and the matrix. Coir fiber reinforced epoxy composites’ performance depends on alkali treatment and the length of fiber. Coir fibers were treated with sodium hydroxide for 10 days with 2%, 4%, 6%, 8%, and 10% concentration. The length of the fibers was about 10, 20, and 30 mm. It is observed that the mechanical properties of coir-based green composites, modulus of rupture and internal bond, increase as a result of chemical composition modification and surface modification [113]. The pretreated natural fiber based composite performed better in terms of mechanical properties as compared to the untreated natural fiber based composites [114, 115].
Etherification
Etherification of natural fiber composites makes it more useful and enhances certain properties [12]. Sodium hydroxide plays an important role in forming a charged intermediate species with the fiber, which allows the faster nucleophilic addition of epoxides, alkyl halides, benzyl chloride, acrylonitrile, and formaldehyde [12,65,66].

The cellulosic materials are reacted with acrylonitrile using 4% NaOH aqueous solution saturated by NaSCN as a swelling agent and catalyst at 40℃. The reaction of benzyl chloride with hydroxyl groups can produce etherified natural fiber [116]. Baiardo et al. [117] applied esterification and etherification reactions to flax fiber to modify its surface properties. The treated fiber showed unchanged structure and morphology, with desired chemical groups on its surface for reinforcing applications in polymer composites.
Enzymatic treatment
Enzymatic treatment is very useful and interesting step when enzymes are used in combination with chemical and mechanical methods for modification of materials. Enzymes are efficient catalysts and are highly specific in their work under mild and energy saving conditions. To further functionalize the lignocelluloses, oxidative enzymes such as peroxidases can be used [67]. Laccase do the oxidation of phenolic hydroxyls to phenolic radicals in the presence of oxygen [118]. It was further noticed that lignin contents of single cellulosic fibers were decreased from 35% to 24% by laccase treatment [68, 69]. Laccase when used with natural phenols such as acetosyringore,
Treatment with isocyanate
An isocyanate compound contains the isocyanate functional group (–N = C = O), which is highly susceptible to reaction with the hydroxyl groups of cellulose and lignin in fibers. Isocyanate is reported to work as a coupling agent used in fiber-reinforced composites. The reaction between fiber and isocyanate coupling agent is shown below:

Where, R may be any chemical groups (such as alkyl).
Dried fibers treated with alkali were placed in a round bottomed flask and soaked in an appropriate volume of carbon tetrachloride (CC14) and a little (1 ml) dibutyl tin dilaurate catalyst. After that, round bottomed flask was fitted with a pressure equalizing funnel containing the urethane derivative, then urethane derivative was added dropwise into the flask with constant stirring. This reaction was allowed to continue for one more hour after the addition of urethane. These urethane treated fibers were purified with acetone by refluxing for 8 h in a Soxhlet apparatus and repeated washing with distilled water, followed by drying in oven at 80℃ [57].
Peroxide treatments
Peroxide treatments have attracted the attractions of most of the researchers for treatment of cellulosic fibers, because peroxide treatment is very easy and also gives good mechanical properties. Organic peroxides are easily decomposed to free radicals, which further go to react with cellulose of fiber and also hydrogen group of matrix as proposed by Sreekala et al. [62]. The schematic of peroxide treatment is shown below:

During peroxide treatment, fibers are treated with 6% dicummyl peroxide or benzoyl peroxide in acetone solution for 30 min after alkali pretreatment of fibers. Flax fibers were treated with dicumyl peroxide from acetone solution. Fibers were soaked in solution at 70℃ for 30 min. Then fibers were washed with distilled water, and placed for 24 h in oven at 80℃, to improve the hydrophobic properties.
Benzoylation
Benzoylation is an important transformation in organic synthesis, and benzoyl chloride is most often used in fiber treatment. Benzoyl chloride includes benzoyl (C6H5C = O) which is attributed to decrease the hydrophilic nature of the treated fiber and improve interaction with the hydrophobic matrix. The reaction between the cellulosic hydroxyl group of the fiber and benzoyl chloride is shown below.

Joseph et al. [61] used sodium hydroxide and benzoyl chlorite (C6H5COCl) solution for treatment of surface of sisal fibers. The fibers were alkaline pre-treated to activate the hydroxyl group of lignin and cellulose of fibers. Then fibers were immersed in 10% NaOH and benzoyl chloride solution for 15 min. After that fibers were soaked in ethanol for 1 h to remove benzoyl chloride and fibers were then washed and dried at 80℃ for 24 h Surface modification and improvement in hydrophobicity was observed after the treatment.
Dicumyl peroxide treatment
Fibers pretreated with alkali (30 g) were soaked with 6% solution of dicumyl peroxide (DCP) in acetone for 30 min, followed by decantation of the solution and drying of the fibers. There was significant improvement in mechanical properties and decrease in moisture regain of the composites [57]. The SEM images of the untreated and treated fiber surfaces are given in Figure 8. The treated fiber surfaces underwent benzoylation, silane treatment, and dicumyl peroxide treatment [12].
SEM photographs of fiber surfaces after chemical treatment: (a) untreated, (b) silane treated, (c) benzoylated, and (d) dicumyl peroxide treated.
Sisal–PE composites, prepared with DCP, show reduced water absorption, although the mass% of the absorption has the same dependence on time as the untreated composites. Significantly lower mass% change values of DCP treated composites may be attributed to the improved interfacial bonding. Grafting reactions reduce the number of voids between sisal and the PE matrix, and partially prevent fibers getting into contact with water [119].
Plasma treatment
This is very effective treatment method to modify the surface of natural polymers without any change in their bulk properties. The plasma discharge can be generated by either cold plasma treatment or corona treatment. Both are the plasma treatment methods when ionized gas has an equivalent number of charged (positive and negative) molecules that react with the surface of the material present. Frequency of the electric discharge is the main distinguishing feature between the two categories of plasmas. Cold plasma of high-frequency can be produced by microwave energy, whereas alternating current of a lower frequency discharge at atmospheric pressure produces corona plasma. The type of ionized gas influenced the modification of the wood and synthetic polymer surfaces [7]. Maldas et al. [75] reported an avenue to activate a wood surface for getting better adhesion with polyolefin by exposure to plasmas. The plasma discharges were generated by either corona treatment or cold plasma treatment. Further, Podgorski and Roux [74] have recently studied the number of the polar component of surface energy of pine wood for plasma modification that include power, distance of samples to plasma source, treatment time, stability of plasma treatment, and type of gas. Corona discharge was used to treat the pulp sheets having moisture contents of up to 85%. The process was carried out in the presence of air and nitrogen atmospheres, and chemical modification of the sheets were evaluated by dye [73]. There were no evidence of an increase in carboxylic groups found on the treated paper surface but it was noticed that the quantity of aldehyde groups increased with increasing corona treatment. The authors were unable to know that either the increase in bonding strength was due to a reaction of hydroxyl groups with aldehyde groups or the degradation of hemicelluloses that adsorb between the surfaces of the paper sheets. Uehara and Sakata [72] noticed that air corona treatment caused a reduction in the molecular weight of cellulose and improvement in hydrophobic properties of cellulosic fibers.
Ozone treatments
The surface of cellulosic materials can be modified by exposure to ozone or oxygen-fluorine gas. Hedenberg and Gatenholm [76] exposed cellulose fibers and PE membranes and films to ozone gas. The adhesion properties of the composites were improved when low density poly-ethylene (LDPE) was treated with ozone treatment. These two mechanisms were proposed for the increase in bonding strength, which involved hydrogen bonding of LDPE that contained carbonyl groups as revealed by spectroscopy and covalent bonding initiated by the decomposition of hydro peroxides.
The ozone treatment affects the contact angles for different liquids with the surfaces and increases the total surface energies. More importantly, the oxidation procedure increases the polar component of the surface energy of LDPE. This increased polar character of LDPE will provide a significant better basis for interactions with the numerous hydroxyl groups on cellulose, which in turn reduces the hydrophilic character of the material.
Chtourou et al. [77] increased the strength properties of PE and pulp composites by exposing the PE to oxygen-fluorine gas. The authors found that by exposure, the specific (acid–base) interaction parameter increased, and the improvement of properties were attributed to hydrogen bonding between both fluorine and hydroxyl groups, carbonyl, and hydroxyl groups.
Grafting
Graft copolymerization of vinyl monomers onto wood and cellulose based materials was first reported in 1953. Since then grafting has received much attention for modification of cellulose. Meyer [79] studied the applications for wood graft copolymers. Hon [78] reviewed four challenges of cellulosic modification by graft copolymerization: limited cellulose involvement, homo polymerization of monomer, limited control of molecular weight, and poor replication of grafting yields. The efficiency of grafting monomers onto natural surfaces increases when the surfaces of fibers, wood and plant based materials have been activated. Hill et al. [120] noticed that wood modified by methyl acrylic anhydride allowed for the grafting of MMA and styrene. An adequate study by Hill and Cetin was made for using the method to protect wood against UV irradiation [121]. Grelier et al. [122] grafted with isocyanate, that have an UV absorbing chromophore to medium density fiber boards by microwave activation.
Oligo esterification method was used to activate pine wood blocks with maleic anhydride along with allylglycidyl ether, in order to graft hydride-terminated polydimethylsiloxane and bis (trimethylsiloxy) methylsilane [80]. Firstly, the absorbency of cellulose fibers was improved by activating the surface with an ethoxysilane and then by doing copolymerization of acrylic acid onto the surface, initiated by potassium persulfate [57, 123–134]. In comparison with the number of studies for graft copolymerization onto cellulose, research on the copolymerization onto more complex substances like wood and plant fiber is scarce. Presence of lignin and other extractives found in wood act as antioxidants through resonance stabilization of free radicals, thus inhibiting polymerization and grafting. Actually lignin inhibits the copolymerization process [124].
The type of catalyst and the nature of lignin has inhibiting ability in graft copolymerization [125]. However, lignin may become more modified than the holocellulose depending upon the reaction conditions [126] or may reduce homo-polymerization of monomer due to a chain transfer mechanism. Zheng et al. have described the participation of lignin in the graft copolymerization of polymethyl methacrylate onto bagasse pith initiated by Fe2+ and hydrogen peroxide [127] and potassium permanganate (KMNO4) [128]. Lignin was more susceptible to grafting and specifically, hydroxyl cyclohexadienyl radicals formed by lignin participated in the copolymerization.
Similar to the concept of bagasse treatment, same initiators were used in the graft copolymerization of stone ground wood pulp. It was noticed that phenolic hydroxy radicals produced by reaction with ferric ions initiated copolymerization [129]. The initial stage of delignification, in which loss of lignin occurred by 0.6% by the sodium chlorite method of pulping, increased the grafting efficiency, but delignification did not have the same effect. The authors also noticed, depending upon reaction conditions that the phenyl ring formed phenoxy radicals that either initiated copolymerization or decomposed the phenyl rings of lignin. Marchetti et al. reported the deposition of manganese dioxide (MnO2) on the wood cell wall when KMnO4 was used as an initiator, which contributed to the radical chain transfer onto wood [130]. Ghosh et al. noticed the effect of lignin on the copolymerization in partially delignified jute fibers. Only slightly improved initial delignification by sodium chlorite grafting, but in the presence of only 0.8% lignin, the grafting efficiency was doubled [131]. Bleached and unbleached sugarcane fibers were grafted with hydroxy phenyl benzotriazole UV absorber and nitroxide radical of piperidinyloxy followed by treatment with acetic anhydride [135].
Fluorocarbon treatment
Ali et al. [136] used fluorocarbons, hydrocarbons, and hybrid fluorocarbons for the treatment of jute fibers. These chemicals have a lower surface free energy. It is a known fact that lower the surface free energy of a material, lower will be the moisture regain. A significant difference in moisture regain values of treated and untreated reinforcement samples was observed at a concentration of 40 g/l. The composite made from treated reinforcement regained very low moisture content as well as exhibited improved mechanical properties (tensile and flexural strength). Being dual nature (hydrophilic and hydrophobic groups) of hybrid fluorocarbon, the treated jute fibers and corresponding composites showed better properties as compared to other two chemicals.
Comparison of results of the pre-treatments on natural fiber composites.

Conclusions
This article focused on the literature review about natural fiber-reinforced composites and the limitations evolving in the development and serviceability of these composites. These natural fiber-reinforced composites are a potential substitute of glass fiber-reinforced composites. In order to exploit the full potential of natural fibers, these limitations must be overcome. Firstly, an appropriate fiber surface treatment should be carried out. The paper also presents literature on the natural fiber treatments used by researchers to reduce the moisture regain of natural fiber composites and improve the compatibility between the fiber and the polymer matrix. A wide range of pretreatments are available, such as sodium chlorite, methacrylate, silane, peroxide, enzyme, plasma, ozone treatment, etc. Some researchers have also worked to impart functionality against microorganisms in these composite materials. Such composites not only help to maintain hygiene but also have enhanced service life. Hence natural fibers can be used effectively to reinforce the composite materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

