Abstract
The purpose of this research is to improve ignition properties and anti-dripping of polyester fabric by using adhesion promoter (AP). The ignition properties of the untreated and treated specimens with durable flame retardant coating and non-durable flame retardant coating were estimated by horizontal flame chamber (UL-94), single-flame source and limiting oxygen index (LOI). The chemical structures of the pre- and final composites have been determined by Fourier transform infrared spectra with attenuated total reflection analysis spectroscopy. The mechanical tests and thermal properties were applied to study their tensile strength and thermal behaviors. The results show that AP has improved the flame retardancy and dripping of PET fabric compared to blank. The char yield increased from 8% to 18%, LOI from 17.5% to 27.5%.
Introduction
Polyester/Poly(ethylene terephthalate) (PET) is a manufactured product; it contains ester groups by 85% of the total percentage of composites and terephthalic acid and a dihydric alcohol. It has been used widely for its many advantages such as: strength, stretch, wrinkle resistance, low cost, highly flammable, less wearing comfort, build-up of electrostatic charge, flexible, easy washing, easy recycling, and quick drying. It is used in fabricating plastic bottles, resistant to biological damage as mildew and mold [1–7]. The PET melts at 245–290℃ [8], so it is not safe during the combustion due to the dripping which is considered as the new source of ignition [9,10]. To overcome this it is important to improve the ignition and anti-dripping properties of PET. Adhesion promoter (AP) is used to improve the cohesion between components and material surface (textile and plastics) in the presence of solvent such as: methyl ethyl ketone, toluene, and butanol [11]. The coating thickness, coating method (spraying and dipping), and composition viscosity have the main effect on AP. Furthermore, APs such as aminosilane and epoxysilane have a significant effect on the synthesis of flame retardant coating [12]. AP can be used by two methods: immersing the specimen in AP then coating with different technique (spraying or dipping) or used as the main components during the stages of coating synthesis. Other kind of AP is chlorinated polyolefins, which are used in many industrial fields such as inks on polyolefin plastics and adhesion of paint to plastic surface in the automotive industry [13]. In this work, polyester was cured with AP as a new technique to modify mechanical, thermal, physical, and ignition properties. All specimens were subjected to thermal degradation using thermal gravimetric analysis (TGA) and differential scanning calorimeter (DSC) techniques. The values of the treated and untreated polyester specimens were obtained to inspect the effects of AP and flame retardant coatings on the thermal degradation of polyester. Further, the degree of flame retardancy was determined by limiting oxygen index (LOI) to get the information of degradation process and efficiency of flame retardant.
Materials and methods
Materials
Polyester fabric (100%) of plain 1/1 and 140 g/m2 (provided by the Misr Company for Spinning and Weaving, El-Mahala El-Kobra, Egypt) was used. Tetraethoxysilane (TEOS) (95%, Alfa Aesor), phosphoric acid (85%, Aldrich), acetic acid (98%, Aldrich), urea (99%, MP Biomedicals, Inc.), and ethanol were used as promoter. Monochloroacetic acid (99%, Aldrich), sodium carbonate (99.9%, Alpha Chemika), sodium hydroxide, and hydrogen peroxide (30%) were supplied from El-Gomhoria Company, Egypt.
Preparation of the specimens and coating
Pretreatment of polyester
The fabric was washed with distilled water at 80° C for 30 min and dried horizontally at ambient condition.
Adhesion promoter
The AP was prepared by mixing modified chlorinated polyolefin with a blend of toluene and xylene. The composition is mixed with slow speed and high torque mixer until the components are dispersed (supplied by the International Group for Modern Coatings, Egypt). Different coating techniques such as dipping, spraying, and brushing were used to coat the surface of the specimen with AP. After immersing the specimens with AP they are subjected to drying in an oven for 4 min at a temperature of 80℃ to remove the solvent by evaporation and the specimens are ready for adhesion with coating.
Non-durable flame retardant coating (NFRC)
NFRC was synthesized by mixing 2.5 ml of TEOS, 1.25 ml acetic acid (as a catalyst), and 1.25 ml acetyl acetone (as stabilizing agent to prevent rapid condensation) in a round flask (250 ml) with constant stirring for 10 min at ambient temperature. Through the mechanical stirring a mix of ethanol and distilled water was added slowly (to remove the silane layer as soon as formed) with continuous stirring for 60 min to complete hydrolysis and condensation reactions. Finally, a limited amount of phosphoric acid was added to the final polymer with continuous stirring for 15 min [14,15].
Durable flame retardant coating (DFRC)
DFRC was synthesized by mixing solution A with B. Firstly, solution A was synthesized by dissolving 0.01 mol of zinc acetate dehydrate in 50 ml of methanol with stirring for 90 min at 50℃; while dissolving 0.02 mol sodium hydroxide in 50 ml methanol in a round flask for 60 min at 50℃ with auto-stirring leads to synthesize solution B. Finally, ZnO nano-sol has been synthesized when solution B was added to solution A by wise constant stirring for 90 min at room temperature then heating for 90 min [16]. The specimens coated with DFRC were washed at 60℃ for 30 min in the presence of 2% NaHCO3 and 5% nonionic detergent, and then dried in an oven at 110℃ for 5 min to study the durability of the coating.
Application of AP, NFRC, and DFRC onto polyester
After pretreatment, the specimen PB was immersed in AP for 5 min, and then dried at 80℃ for 30 min, while specimens PC and PD can be formed when immersed in AP for 2 min then immersed in NFRC and DFRC for 5 min followed by drying in a furnace for 30 min at 60℃, respectively. Finally, PE and PF specimens were prepared by immersing PET specimen in NFRC and DFRC directly for 5 min then dried at the same condition.
Mechanical behavior
The maximum force (tensile strength) and elongation % at the break of the specimen were carried out according to the standard method, ISO 13934-1 [17], using Shimadzu, Universal Tester of (C.R.T)—type S-500 Japan.
Thermal gravimetric analysis
TGA is used to determine the mass weight of a specimen during thermal decomposition. It was performed by Thermal Analyzer (TGA-50 Shimadzu Instrument) at a heating rate of 10℃/min from ambient temperature to 750℃ under N2 gas at flow rate: 30 ml/min. The weight of tested specimen was ∼7 mg [18].
Differential scanning calorimetry
It is used to give information about thermal changes that do not involve a change in specimen weight, characterize the thermophysical properties of polymers such as: heat flow (ΔH) and melting temperature (Tm). It was carried out using a Shimadzu DSC-50 Instrument—Japan in nitrogen gas with flow rate of 30 ml/min and heating rate of 10℃/min. Scans are started from ambient temperature up to 650℃ [19].
FTIR study
FTIR chemical composition of the untreated and treated PET specimens was studied by Nicolet 380 Spectrometer Instrument—USA, with a wavelength range of 4000–400 cm−1, scan number 64 at 4 cm−1 resolution.
Flammability properties
Flame chamber
This test is achieved at ambient temperature of 25 ± 2℃ (ISO 3795) [20] to determine the burning rate (BR) of specimen mounted horizontally in an U-shaped holder. To study the burning behavior, a piece of cotton pad (weight: 70 g, size: 64 cm2 and thickness: 0.5 cm) was placed under the specimen to observe whether molten drips (with or without flame) could ignite and burn the cotton pad or not. The free end of the specimen was exposed to the ignition source with flame length 3.8 cm (1.9 cm inside the specimen and 1.9 free) for 15 s then the flame was removed. Five specimens were used for testing with dimension 36.5 x 10 cm2 (in this test 5 x 15 cm2).
BR was calculated from this formula:
Measurement of LOI
It can be defined as the lowest amount of oxygen in oxygen–nitrogen mixture required to support complete combustion of specimen. LOI was measured according to ISO 4589 standard by Rheometric Limiting Oxygen Index apparatus. The difference between the LOI of the treated and untreated specimens (ΔLOI) was calculated. The width and the length of the specimens were 5 cm and 15 cm, respectively, then clamped vertically in U holder and ignited with propane gas from the free side for 30 s in the mixture of N2/O2 gases. The average of four specimens was considered [22].
Single flame source test
Six test specimens (three lengthwise and three crosswise) with the dimensions of 25 x 9 cm2 are ignited with a height of 2 cm propane gas flame from the center of the free edge for 15 s and the total test duration is 20 s at an angle 45°. U-shaped holder is used to attach the specimen vertically and filter paper is placed below the specimen holder to observe the falling of flaming debris [23].
Results and discussion
Fourier transform infrared spectroscopy (FTIR)
Polyester fabric (untreated)
FTIR spectroscopy was used to characterize the presence of specific chemical groups in the materials. In case of blank specimen, the polyester fabric is composed of alcohol, ester, aromatic ring, anhydride, and heterocyclic aromatic rings. That was the reason, there was still alcohol and anhydride as a residual reactants left in the polyester [24]. The effect of AP and flame retardant coating (non-durable and durable) was studied by using FTIR-ATR spectroscopic technique on the range from 4000 to 400 cm−1 (see Figure 1). All polyester specimens exhibit broad bands near 3450–3465 cm−1 due to the OH-stretching vibration of free and hydrogen bonded groups [25,26]. The ester functional groups appeared at 1716 (symmetric stretching of carbonyl group), 1235, and 1093 cm−1, while the aromatic ring observed in 2960, 1546 (asymmetric stretching small band of the carboxylate group), 1505, 1011, and 725 cm−1. At 1985 cm−1, a small peak appeared in polyester due to anhydride groups. Bands assigned for an ethylene CH2 group of –O–(CH2CH2)–O– moiety is also observed at 1450–1465 cm−1 and 840–855 cm−1. The peak at 1410 cm−1 corresponded to C–C stretching vibration of the benzene ring which was a stable group.
FTIR-ATR spectra of polyester fabric (PA) untreated polyester; (PB) treated with adhesion promoter only; (PD) treated with DFRC after immersing in adhesion promoter then dried at 80℃ for 30 min; (PF) treated with DFRC directly then dried at 80℃ for 30 min.
Polyester fabric (treated)
By comparing PF specimen with PA, any new peaks can be found except a few changes in the peak position (intensity). It can be explained as no inter-chemical reaction or adhesion force formed between the surface of polyester fabric and the DFRC. The intensity is decreased in the case of PD specimen (1728 cm−1: 79.35%, 1393 cm−1: 93.76% and 1242 cm−1: 86.37%) compared to PB (1712 cm−1: 94.26%, 1450 cm−1: 96.30%, 1340 cm−1: 97.02% and 1241 cm−1: 91.60%). Asymmetrical C–O in the acetate group appeared in the range of 1240:1242 cm−1 (both of PB and PD specimens). In case of the spectra of PD specimen, the bands between 3500 and 3420 cm−1 have been assigned as the O–H stretching mode with the O–H bending modes observed in 1035 and 952 cm−1. Whereas, PB specimen displays two hydroxyl stretching bands at 3445 and 3342 cm−1, while O–H bending modes are found at 1015 and 870 cm−1 and the bands of the carbonate group are observed at 1509, 1091, and 869 cm-1. C–H broad in the benzene ring appeared in 2900:3000 cm−1. While, the most important vibration bands appeared in case of PB specimen at 1153 cm−1 due to formation of siloxane bond (Si–O–Si)ν [27] between AP and uncoated specimen (PA).
Mechanical properties
Effect of AP on the mechanical properties of treated and untreated polyester fabric.

Thermal measurements
TGA analysis
The associated TGA curve of untreated (PA) and treated specimens (PB–PF) was carried out in a nitrogen atmosphere from ambient temperature to 750℃ and is shown in Figure 2 and Table 2. Up to 350℃, the PA specimen is more stable than the others then starting to decompose. Above 490℃ charring reactions tend to be completed. It is observed that the percentage of char yield (20%) during thermal degradation of the PD specimen is related to the highest value in case of LOI test (27.6%).
TGA thermograms of untreated and treated specimens of polyester fabric. TGA data for degradation of the treated and untreated specimens.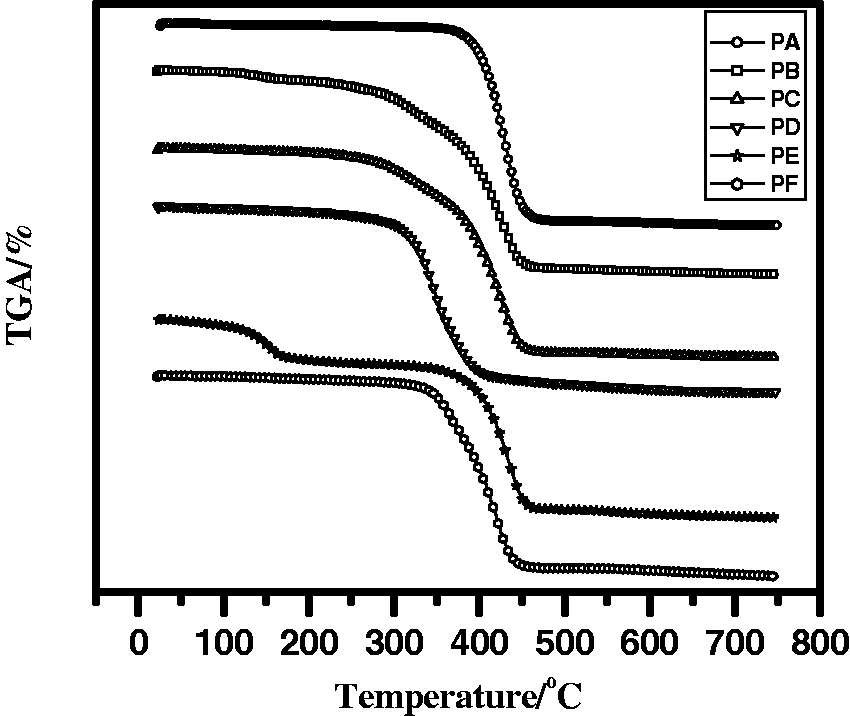
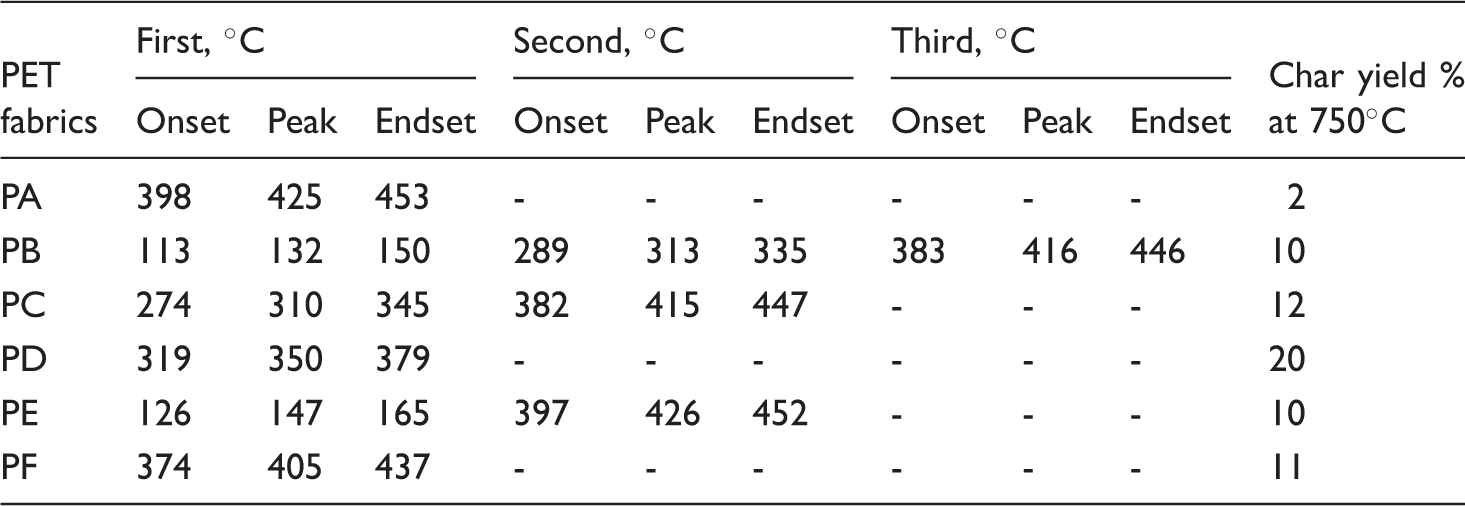
In the case of PE specimen, the degradation occurred in two steps. Firstly, the organic coating evaporated with smoking up to 185℃, polyester fabric degraded as mentioned in the case of untreated polyester fabric (char yield = 2%), whereas PF specimen decomposed in one step only (onset = 374℃, peak = 405℃, endset = 437℃ and char yield = 11%), due to the absence of AP which plays an important role to support the adhesion strength between the fabric and the coating. The polyester fabric immersed in AP (PB specimen) has different major stages of thermal degradation behaviors: the initial stage ends at 162℃ in which water is evaporated in the dehydration process [28]. From 163℃ to 338℃, the rate of decomposition became faster up to 470℃ (onset = 382.5℃, peak = 415.5℃, endset = 446.4℃ and char yield = 10%). Finally, PC specimen is thermally decomposed at two stages, up to 340℃ the first stage of decomposition occurred, followed by the second stage (peak = 413.6℃ and char yield = 28.4%).
DSC studies
DSC traces for PA and PB specimens are shown in Figure 3. In case of PA, it has a semi-crystalline structure and consists of a highly organized crystals and randomized amorphous regions with high molecular weight chains [29–31]. The specimen immersed in AP (PB) has a shorter chain length molecule which deposits easily on the surface of polyester, resulting in an internal plasticization of the fiber by reducing the dehydration temperature of the treated specimen than the blank from 29.6 oC to 26.6℃ [32–34]. The AP is playing an important role while increasing the value of the melting temperature (Tm) up to 267.5℃ and decreasing the value of the enthalpy (ΔH) less than −65.48 mJ compared to PA specimen (257.8℃ and −59.72 mJ, respectively). The decomposition is achieved in one and three steps in case of PA and PB specimens, respectively. Firstly, the peak appeared at 356.8℃, with heat flow at −662.10 mJ in the case of PA specimen, while at 367.7℃, a wide peak appeared with enthalpy −135.93 mJ. Second and third stages of decomposition appeared just in case of PB specimen. At the second stage, a sharp endothermic peak appeared at 400.5℃ with a heat flow at −120.32 mJ. Whereas, a wide peak at 497.78℃ with a heat flow + 738.23 mJ appeared in the final stage of decomposition.
DSC curves for untreated specimen; PA, and specimen immersed in adhesion promoter only then dried at 80℃ for 30 min; PB.
The thermal analysis of PA, PC, and PE specimens is shown in Figure 4. The heat flow for PC specimen is at −186.33 mJ with a sharp exothermic peak at about 262.9℃. The two peaks are appearing in the curve of PE specimen due to dehydration (sharp peak at 137.9℃) and evaporation of the solvent (wide peak at 177.3℃). The melting temperature of both PC and PE (262.9℃ and 263.9℃) slightly improved by 2% than the blank, so both do not melt during the ignition tests, but the area above the exothermic peak is broader than that of PC and PA specimens. A high difference was noticed at heat flow, since the PC specimen has the highest heat flow (−186.33 mJ) compared to the others (−147.33 mJ for PE and −59.72 mJ for PA). The peak appeared at 385.4℃ in case of PC specimen has moved out in both PA and PE specimens. Since PA has a wide exothermic peak at about 356.8℃ with heat flow at −662.10 mJ. While a wide endothermic peak appeared at 500.1℃ (ΔH = 702.82 mJ) and 474.6℃ (ΔH = 1120.0 mJ) in case of PC and PE specimens, respectively.
DSC curves for: PA; untreated specimen; treated specimen with NFRC after immersing in adhesion promoter, PC; and treated without immersing in adhesion promoter; PE.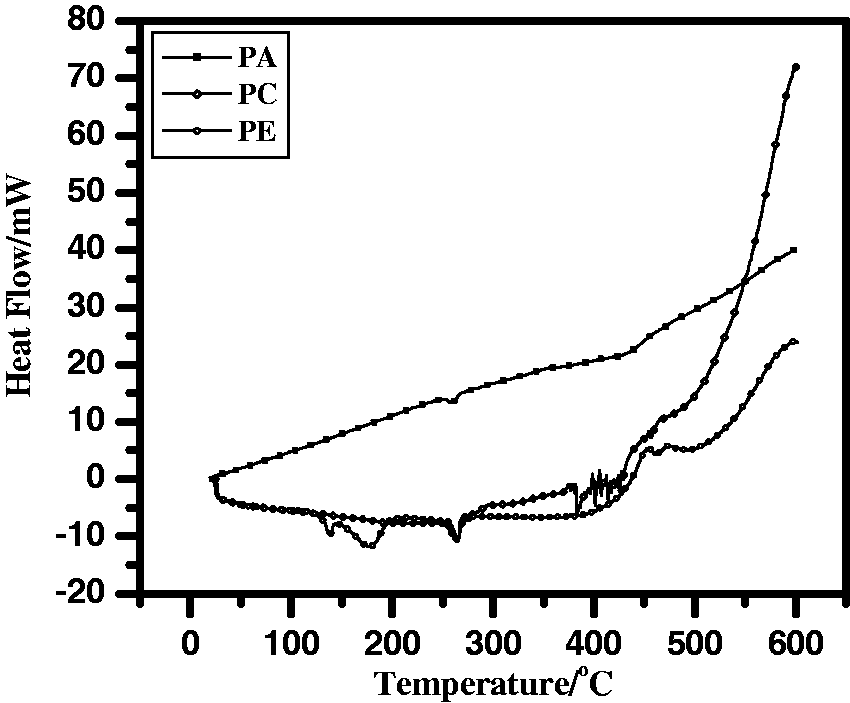
The melting temperature in Figure 5 has nearly the same value with high difference in enthalpy, since PF has a broad exothermic peak at around 253–273℃ with heat flow at −329.24 mJ. This is followed by a broad exothermic peak at about 397℃. The endothermic peak at around 430–505℃ has been found in both of PD and PF specimens but disappeared in the case of PA. The area under the endothermic peak at this range of 430–505℃ decreased in case of PD than PF and disappeared in the case of PA.
DSC curves for: untreated specimen; PA, treated specimen with DFRC after immersing in adhesion promoter; PD, and treated directly without immersing in adhesion promoter (PF).
Studying the burning behavior
Burning behavior of the untreated and treated PET fabric at room temperature.

Limiting oxygen index of the untreated and treated specimens.
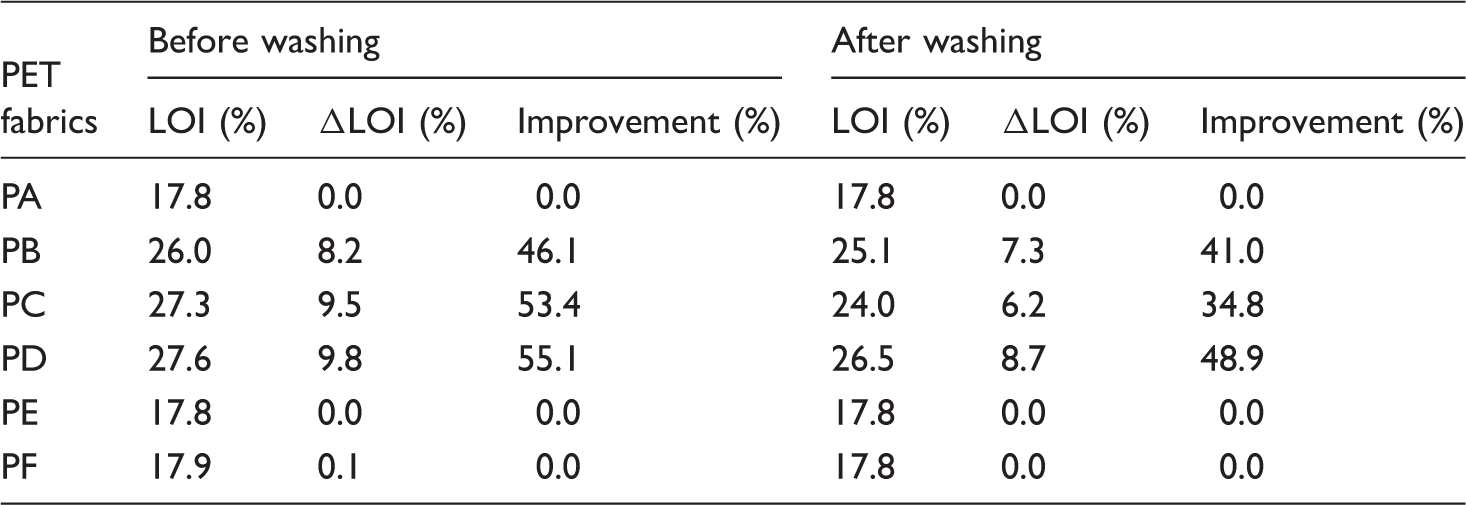
Table 4 illustrates that the LOI percentage of PB and PD specimens was not affected by the washing treatment due to the adhesive force of adhesion promoter. Figure 6 illustrates the ignited properties of the blank (PA) and treated specimens (PB–PF). Specimen PA was molten and many ignited droplets were dropped during flaming [14], while PB:PD specimens have improved in the ignition, dripping, and shrinking properties.
Studying the ignition properties of the untreated and treated specimens.
Single flame source
The flame application edge exposure to PET fabric and treated specimens.

Conclusions
It is important to protect polyester fabric from ignition with a new technique that does not consume time, fabrics, and money. So, thinking to apply AP technique was used as the answer for the previous inquirers. Depending on internal force formed between the surface of polyester fabric and the outer layer (DFRC or NFRC) this technique is applied to protect PET fabric from ignition, to improve the rate of burning, and increasing the LOI by ∼50% compared to PA, PE, and PF specimens. This indicated that AP has a direct effect to enhance the ignition characteristics of PET fabric. The untreated and treated specimens were exposed to thermal analysis test. The thermal decomposition of polyester fabric includes the oxidative decomposition with rapid mass loss, then dehydration and charring reactions tend to be completed. The ash residue % in the case of PD specimen at 750℃ supports the result of LOI. It can be explained as formation of isolated layers from ash residue prevents oxygen from reacting with PET specimen. The main role of adhesion promoter was observed in mechanical test. Specimen coated with DFRC as an outer layer recorded the lowest elongation % and the highest tensile force.
Footnotes
Acknowledgements
The author wishes to express his thanks to Prof Dr NA El-Zaher, Textile Metrology Lab, National Institute for Standards (NIS), Giza, Egypt and Dr Abd El-Aziz Z Gomaa (Consultant) for their help. Many thanks to International Group for Modern Coatings, Egypt for supplying me the Adhesion Promoter.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

