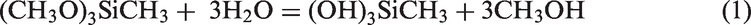Abstract
This research aimed to improve anti-dripping and ignitability properties of polyester fabric polyethylene terephthalate (PET) by using urea (CO(NH2)2), phosphoric acid (H3PO4), methyltrimethoxysilane (MTMS) via sol-gel technique. Methyl groups served as blocking groups preventing oxygen from penetrating into PET specimen, so the fire stopped. The interaction between the coating and irradiated surface of PET was detected by Fourier-transform infrared spectroscopy using UV/ozone for different time periods. The mechanical properties (tensile and elongation %) of the specimens were tested according to ISO 13934. The thermogravimetric analysis, derivative thermogravimetric analysis, and differential scanning calorimetry proved that the new coating has a direct efficiency to protect the PET fabric from ignition. Different standard test methods, ISO 4589, ASTM D635, and EN ISO 1716, were used to investigate the ignition properties of untreated and treated specimens. The results showed that the coated specimens with a urea solution only as a first layer, and then the sol solution as the second layer have the highest flame retardant effect compared to the other methods. This results in improving the dripping and ignition properties of a polyester fabric.
Introduction
Polyester fabric is a manufactured product, which is composed of at least 85 wt% of dihydric alcohol, terephthalic acid, and ester groups of the total percentage of composites [1]. Owing to itslow-cost, wrinkle-resistant, easy washing, quick drying, and high-strength properties, it is widely used in automotive industries, sportswear, home textiles, medical textiles, and packaging [2,3]. Polyethylene terephthalate (PET) has a melting point between 245℃ and 290℃ [4], so it is not safe during the combustion [5] due to drip, smoke, and shrink effects. Hence, it is important to improve the ignition properties of the PET fabrics in order to be used in many fields safely. The challenging issue with PET is its high rate of fire spread with melt-dripping [6,7]. Photo-induced grafting is one of the many methods used to improve the ignition properties of PET [8]. Urea has a direct reaction with phosphoric acid producing urea phosphate, which has amino and phosphorus groups. It is soluble in water forming urea and phosphoric acid [9].
The sol-gel process occurs in two stages: hydrolysis and condensation. Firstly, the sol is formed due to the presence of water and alcohol, which plays an important role in the exchange of the alkoxy groups with OH groups forming silanol groups that condense together to give siloxane bonds. The covalent bonds (siloxane bonds) between the treated surface and coating was formed by drying [10,11].
Hydrolysis reaction
Condensation reaction
In this study, PET fabric was irradiated with UV/O3 for 30 min before coating with a different solution prepared with a different method (PO2-5) to find the best one with high efficiency in protecting PET from ignition to be used in tents. Since urea played the main role as cross-linkage between the irradiated surface of PET and –OH groups at sol solution, all specimens were investigated by Fourier-transform infrared spectroscopy (FTIR), thermogravimetric analysis (TGA), differential scanning calorimetry (DSC), and mechanical (E% and T) tests to prove the changes due to the coating according to technique methods. The flame retardants were measured by a single flame source, flame chamber (UL-94), and limiting oxygen index (LOI). This research provides an effective process to prepare a flame retardant and hydrophobic coating to polyester fabric.
Materials and methods
Materials and chemicals
Mill bleached polyester fabric (PET) of weight 140 g/m2 plain 1/1 weave was kindly supplied by the Misr Company for Spinning and Weaving, El-Mahala El-Kobra, Egypt. Distilled H2O at 65℃ was used to wash PET fabric for 30 min, and then dried horizontally at ambient condition before testing [12].
Specimens irradiated with UV/ozone
The specimen were irradiated with a mercury lamp at low pressure, high intensity without an outer envelope (220 V, 200 W, made in Poland) hanging vertically in a closed cycle box. Finally, after irradiating the specimen for different times (0, 10, 20, 30, 40, and 50 min) the free radical was formed over the surface [13].
Silica sol synthesis
The sol was prepared by mixing 1.25 mL acetic acid (Tedia, 99%) as a catalyst with 5 mL methyltrimethoxysilane (Alfa Aesor, 95%), and then ethanol (Alfa Chemika, 95%) and distilled water were added during stirring for 10 min at ambient temperature. Ethanol has a direct effect in dissolving silane film as formed, so no layer was left on the surface. The sol was obtained after constant stirring for 4 h at room temperature [14,15].
Preparation of the untreated and treated specimens
PO1: The untreated specimen was irradiated by UV/ozone for 30 min to excite the surface of PET specimens [11];
PO2: Specimen was irradiated by UV/ozone for 30 min, immersed in sol solution only for 3 min, padded under pressure, 100%, and then dried at 110℃ for 10 min in an oven;
PO3: Specimen was irradiated by UV/ozone for 30 min, immersed in urea solution only with concentration 40%, dried at 80℃ for 5 min in an oven, then dipped in sol solution for 3 min, padded under pressure, 100%, and then dried at 110℃ for 10 min (Figure 1);
The scheme illustrates the role of urea as the cross-link between the irradiated surface of PET fabric and sol solution.
PO4: Specimen was irradiated by UV/ozone for 30 min, dipped in sol solution containing urea (0.5 mL of the urea solution with concentration 40%) and phosphoric acid (0.5 mL, 40%) for 3 min, padded under pressure, 100%, and then dried at 110℃ for 10 min (Figure 2); and
The scheme of the interaction between sol coating, urea, and H3PO4 on the surface of PET fabric.
PO5: Specimen was irradiated by UV/ozone for 30 min, dipped in sol solution containing a solution of urea (0.5 mL of the urea solution with concentration 40%) and phosphoric acid (0.5 mL, 5%) for 3 min, padded under pressure, 100%, and then dried at 110℃ for 10 min [16].
Fourier-transform infrared spectroscopy
IR spectra of the uncoated and coated specimens were recorded using Shimadzu’s TGA-50 Spectrometer (USA) instrument in the wave number range 4000–400 cm−1 with an average of 32 scans using a resolution of 4 cm−1 [17].
Mechanical properties
The tensile strength and elongation % of the PET fabric were tested by H5KT Atlas instrument (USA), according to the standard test method, ISO 13934 [18]. The tested specimens with dimensions 5 × 15 cm2 were used and the average of four specimens has been considered.
Wetting test
The wettability of PET fabric was evaluated by AATCC Test Method 39-1989 [19]. The specimen with dimensions 2.5 × 2.5cm2 was dropped into the beaker including water. The total time to complete specimen’s wetting was recorded. The average value of five determinations was taken as the result.
Contact angle (θ)
It is the angle between the specimen surface and the tangent drawn to the droplet at the point of contact with the specimen surface. A photographic method was used to measure the contact angle on polyester fabric. At contact angle measurement, a drop of deionized water or glycerol was placed on the outer surface of the fabric, photographed, and then measured [18]. In case of a hydrophobic specimen, a droplet of water or glycerol will be formed on the surface and vice versa in the hydrophilic specimen.
Thermal analysis
Thermogravimetric analysis and derivative thermogravimetric analysis
TGA is a thermal test for determining the stability, deterioration stages, and ash residue of the specimen by using thermal analyzer instrument (TGA-50 Shimadzu, Japan) [20,21]. Weighted polyester fabric (in milligram quantities) was put in empty crucible balanced with standard one, before exposing to a heated chamber at N2 gas (to remove the pyrolysis products). After that, the specimen was tested under normal atmospheric conditions with temperature ranging from 25℃ to 750℃ at a heating rate of 10℃/min with a flow rate of 30 mL/min [10]. The thermal analyzer records time, temperature, and weight loss during the test specimen. DTGA was used to follow the heat emission. Endothermic peaks with different lengths appeared in Figure 6 showed the first peak between 90 and 220℃ in the absence of oxygen due to the moisture. The second endothermic peak between 220 and 320℃ referred to the melting points. The maximum weight loss occurred between 320 and 460℃ and appeared as endothermic peaks with different width and lengths [22].
Differential scanning calorimeter
The change in enthalpy (melting temperature (Tm), a heat of melting, and glass transition (Tg)) for the specimen were determined by using a Shimadzu Analyzer DSC-50 (Japan). The test was carried out under the influence of nitrogen gas using specimens of weight ∼6.0 g, heating rate of 10℃/min, and flow rate of 30 mL/min. The test was carried out at temperature between 25℃ and 650℃ [23].
Ignition test
Flame chamber (UL-94) and limiting oxygen index
The horizontal burning rate (RB) of materials was determined by using the standard test method – ISO 3795 [24]. The PE fabric was cut in dimensions of 5 × 15cm2 with a thickness 0.2 cm and then held horizontally in a U-holder. The free edge of the tested specimen was exposed to the flame with length of 2 cm for 30 s at room temperature. The ignition source was removed and then the combustion behavior of the specimen was studied. The burning rate was calculated as follows
The minimum concentration of oxygen in a blend of nitrogen and oxygen required to keep specimen ignition was measured according to the standard test method [25]. The specimen with dimensions 5 × 15 cm2 was clamped in a U-type holder and made to stand vertically in a test column. The propane gas was used as a flame source for 30 s. The specimen in a mixture of nitrogen and oxygen was tested. The average of five specimens was recorded [26].
Oxygen bomb calorimeter
The heat released from the specimen combustion in pure oxygen can be measured directly, by using OBC according to standard test method EN ISO 1716 [27].
Results and discussion
Infrared spectroscopy
FTIR spectra of untreated and treated specimens of polyester fabric are shown in Figure 3. The major structure of the polyester fabric is alcohol, ester, anhydride, aromatic ring, and heterocyclic aromatic rings. The peaks of OH groups were observed at 3410–3431 cm−1. The untreated specimen had a sharp stretch peak, while a wide and short area under the broad peak had formed in the other specimen. All test specimens had the band in the range of 2904–2910 cm−1 corresponding to (C–H)ν groups as CH2 and CH3, but differed in length and width. The aromatic ring was observed at 2960–2975 cm−1, 1575–1585 cm−1, 1500–1510 cm−1, 1000–1020 cm−1, and 720–730 cm−1 [28]. The ester functional groups appeared in 1708–1720 cm−1, 1260–1280 cm−1, and 1090–1130 cm−1. For PO1 specimen, the CH2 group of the ethylene group appeared at 1454 cm−1, but semi-disappeared in the other tested specimen. The peak at 1410 cm−1 corresponded to C–C stretching vibration of the benzene ring, which was a stable group. For PO2 specimen, the peaks at 3516, 1653, and 905 cm−1 correspond to –OH bond [29].
FTIR spectrum of the polyester fabric untreated (PO1) and treated specimens (PO2-5) with UV/ozone before immersing in the sol-gel coating.
In all coated specimens, the methyl group (CH3–Si) appeared at 2918 and 1416 cm−1. Also, the band of silanol (Si–OH) appeared at 3500–3515, 1650–1650, and 900–905 cm−1. The bands at 1025–1017 cm−1, 890–895 cm−1, and 460–465 cm−1 are attributed to asymmetric, symmetric, and rocking vibrations of –Si–O–Si– bending, respectively [30]. A sharp peak of (P=O)ν at phosphoryl group appeared with different wave numbers in the range between 1220 and 1225 cm−1 but disappeared in the case of PO1 specimens. The peak of P–OH bond appeared at 780–785 cm−1 [31], while at the wave number 830 cm−1 the P–O–P peak appeared. The reaction between the free radical on the outer surface of the polyester fabric and urea solution resulted in the amide group (CONH)formation at 1540 cm−1 in the case of PO3 specimen only. For PO5 specimen, the peaks of C–H bond appeared at 2350 cm−1 and disappeared at the other.
Tensile and elongation properties
Mechanical properties of the PET fabric untreated and treated specimens.

Wetting and contact angle measurements
Wetting is the ability of liquids to keep in contact with solid surfaces. The overall wetting time of untreated specimen (PO1) is 4 s, but all treated specimens not wetted more than 30 min from the onset of the test. This proves that the hydrophobicity of the treated specimens was improved.
The contact angle is the measured angle between the liquid and the surface. When the liquid wets the specimen surface the hydrophilic surface has contact angle value <90°, whereas there is no wetting of the hydrophobic surface and the value of the contact angle is >90° and <180°. Figure 4 illustrates the contact angle measurement of polyester fabric of untreated and treated specimens. It is clear from the figure that the contact angle of the treated specimens is the obtuse angle (>90°), so PO2, PO3, and PO4 specimens were classified as hydrophobic surface and PO5 specimen as super hydrophobic, whereas PO1 as a hydrophilic surface.
Contact angle comparison between untreated and treated specimens.
Thermal analysis techniques
Thermogravimetric analysis
The weight change by heating the tested specimen has been studied by TGA. Figure 5 illustrates TGA thermograms for the untreated polyester specimen (PO1) and coated specimen (PO2-5). At first, the untreated specimen up to 340℃ is still stable and then starts to decompose. The major mass loss occurs between 340℃ and 495℃ due to the thermal degradation of the polymer backbone (ester bonds), and then the dehydration tends to be completed with char residue of 1.8%. In the case of the treated specimens, it is possible to detect the joint effect, including the sol solution, P, and N elements present in phosphoric acid and urea, respectively.
TGA curves of untreated and treated specimen with sol coating only after irradiated with UV/ozone for 30 min with nitrogen gas.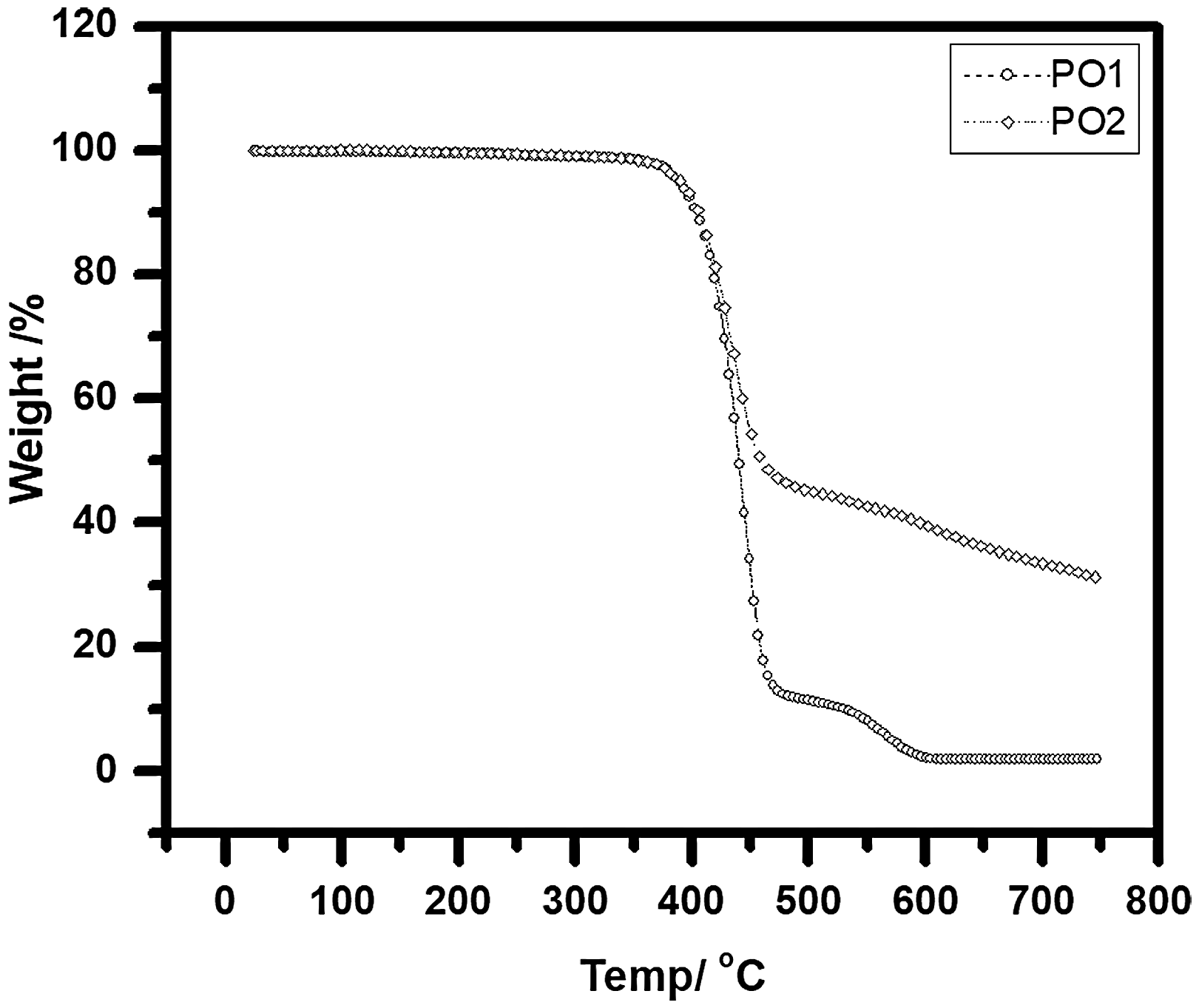
The PO2 specimen undergoes decay in two stages: first between 340℃ and 490℃, since the weight loss is higher and faster due to the change in the main series structure and bonds cracking, while the stage between 490℃ and 560℃ is the zone for complete dehydration and char formation. The ash residue % has been increased due to the presence of methyl groups, which played as a defender and protect the high percentage of the tested specimen from completely burning.
Figure 6 illustrates the degradation stages of the specimens coated with urea, phosphoric acid, and sol solution. PO3 specimen has decomposed differently from PO4 and PO5 due to the absence of phosphoric acid with the presence of urea, which plays the main role of cross-linking between the free radical on the specimen surface and siloxane groups in sol solution.
Derivative thermogravimetric analysis (DTGA) curves of untreated and treated specimens with urea and phosphoric acid.
The PO3 specimen has six stages of decomposition in comparison to PO4 and PO5, which have three analysis stages (Figure 7). Initially, the dehydration stage was achieved between 26℃ and 91℃, then crystal transformation (melting) was initiated in the range of 103–224℃ forming biuret acid, with the evolution of ammonium gas with weight loss of 30% [32]. The stage between 320℃ and 570℃ illustrates the change in physical properties of the PET by minimal loss of the fabric. Above 570℃, the dehydration and charring reaction tend to be completed. PO3 has the highest ash residue percentage (34.8%) compared to PO2 (28.7%), PO4 (32%), and PO5 (31.2%). This may be returned to the methyl groups (blocked area) at sol solution and hydrogen bonds (urea or urea–phosphate as cross-linkage) between sol solution and free radical on the surface of the irradiated specimens. Finally, both of PO4 and PO5 specimens have the same stages of dehydration and degradation except for the weight loss in the second stage between 95℃ and 190℃. The loss in weight is recorded to be 2% in the case of PO4, while PO5 has lost weight by 6%. This may owing to the different percentages of urea and phosphoric acid mixed with sol solution.
TGA curves of the treated specimen with sol solution, urea, and phosphoric acid after irradiated with UV/ozone for 30 min with nitrogen gas.
Differential scanning calorimeter
Figure 8 shows the DSC traces for untreated and treated polyester specimens. The glass transition (Tg) and heat fusion (ΔH) values have been tabulated in Table 2.
DSC curves for untreated specimen and treated specimens. Values of glass transition temperature and fusion heat for untreated and treated specimens. Tg: glass transition temperature; ΔH: heat of fusion.
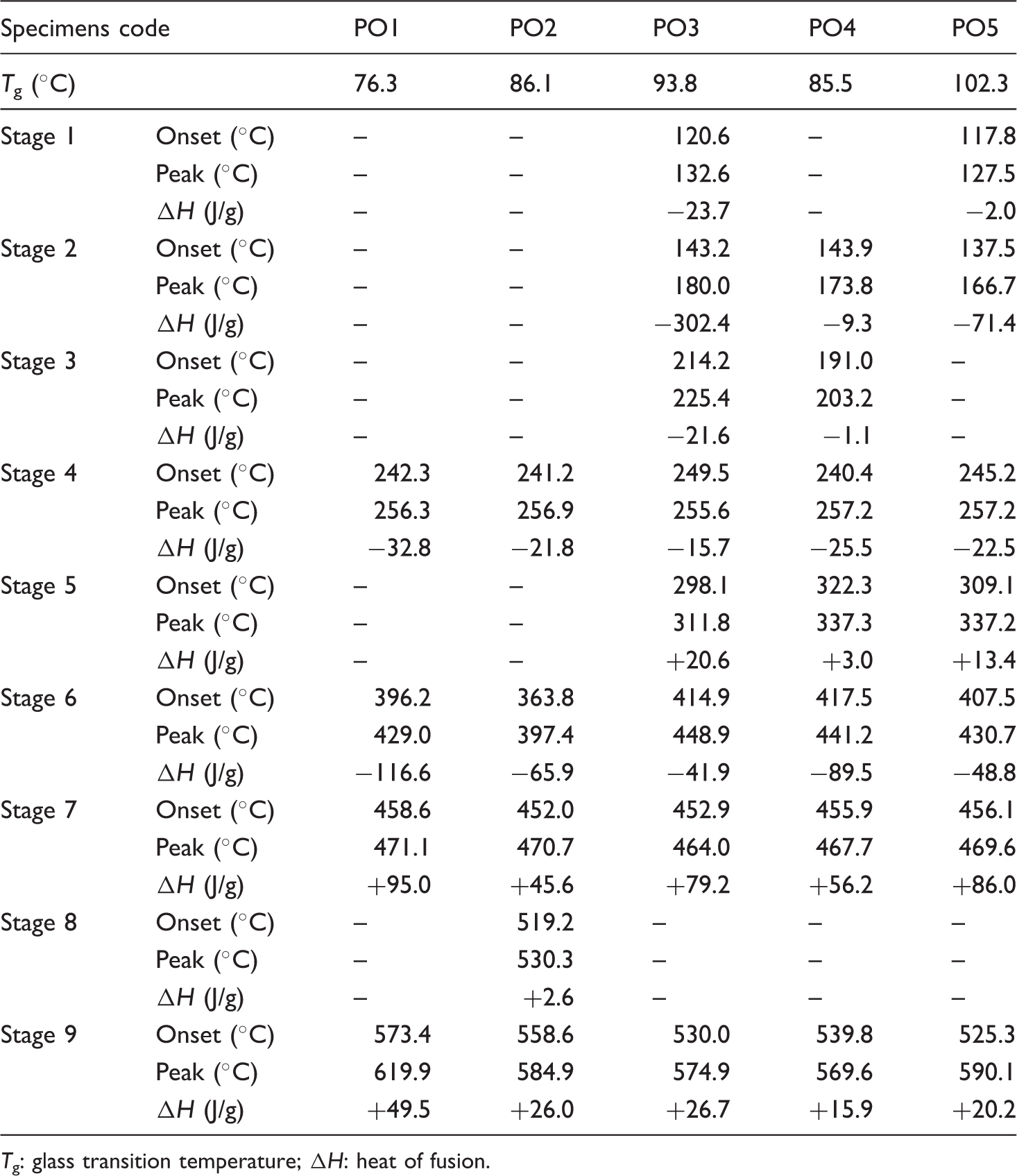
The tabulated data in Table 2 illustrate the main effect of the flame retardant coating. The Tg values increase in case of coated specimens compared to the blank one. This can be attributed to amorphousity, which decreased in PO3, PO4, and PO5 specimens, so both of them have the highest melting temperature.
Firstly, a sharp and long peak appeared in PO3 and decreased by 98% in PO5 and disappeared from a plot of PO4 (stage 1). In case of stage 2, there are melting point temperatures for PO3, PO4, and PO5 specimens only owing to the melting of the outer layer coating. These layers served as sacrificial layers in the protection of the PET from ignition. The effects of mixing urea and phosphoric acid by different percentages with sol solution can be seen in Table 2 (stages 2 and 5). A new peak appeared in 311.8℃, 337.3℃, and 337.2℃ for specimens PO3, PO4, and PO5 with heat fusion of +20.6, +3.0, and 13.4 J/g, respectively.
At stage 4, the onset temperature and peak results are slightly different. The onset temperature of PO3 specimen is the highest (249.5℃) followed by PO5 (245.2℃) while PO2 has the lowest onset temperature (241.2℃). The peak position of the tested specimens is nearly the same (255–258℃) but different in the width and area over the peak. The negative values of heat fusion for the specimens indicate that there are endothermic reactions occurring in this area. This peak is broad and long in case of PO1 and PO2 specimens, then changed to sharp and short in case of PO3, but returned to expand again with a sharp peak as found in both of PO4 and PO5 specimens. This variation in shape was attributed to the different degrees of crystallinity found in the specimens coated with sol solution, urea, and phosphoric acid.
Stage 6 has the melting point of the PET fabric for the tested specimens. The different values of the peak for specimens prove the effect of the presence of the new coatings. The PO3 specimen has the lowest heat fusion values (−41.9 J/g) compared to the other. The area over the peak is broad with a short length, and vice versa in case of PO1. At temperatures between 200℃ and 230℃ the peak length is the highest in the case of PO3 (−21.8 J/g), then decreased to disappear completely as in PO5 (stage 3). The irradiation of the specimen for 30 min before coating by sol-gel technique (PO2) has a new peak at 530℃ formed during the exothermic reaction with heat fusion of +2.6 J/g (stage 8). After melting stage of the PET fabric (360–418℃, stage 6), the exothermic peak was formed with different values due to the presence of urea solution before the sol solution coating or mixed with different percentages of phosphoric acid, which was used as a flame retardant coating (stage 7). All tested specimens completed the analysis test with exothermic peak (different fusion heat) and different peak temperatures, which changed between 569℃ and 620℃ (stage 9).
Studying the ignition behavior
Ignition behavior (UL/94) and oxygen index
Limiting oxygen index (LOI) and flame chamber of the specimens.

The LOI results for the PO3 specimens shows that immersed specimen fabric in urea solution (40%) before sol solution raises the LOI to 28.5%. The urea solution (primary layer) played a strong cross-linkage between free radical on the surface of the PET specimen and the second layer of the sol solution. These layers lead to prevention of the ignition and improved the melting characteristics (stop dripping). Mixing the sol solution with different percentages of urea and phosphoric acid reduced LOI of PO4 (21.0%) and PO5 (20.8%). This result agrees well the results of the flame chamber (UL/94).
Bomb calorimeter
The gross heat of untreated and treated specimens.


The cup after OBC test for untreated and treated specimens with different methods.
Conclusion
This work has shown that the addition of different amounts of urea and phosphoric acid to sol solution for synthesis sacrificial layers can be successfully protect polyester fabric from ignition by enhancing its flame retardancy. The interaction was illustrated by FTIR. This treatment leads to the improvement in the mechanical properties, wettability, and the contact angle. It was found that the flame retardancy and anti-dripping of PO3 polyester fabric perform greatly than the untreated and other treated specimens. Coated specimens (PO2-5) with different coatings prove that both of sol solution and urea as a cross-linkage have a main role in protecting the PET fabric surface from ignition and stop dripping and shrinking. The specimen (PO3) has the highest ash residue %, while the untreated specimen (PO1) has the lowest ash residue %. The ash residue percentages of all treated specimens have recorded an average between 28% and 35% compared to 0.5% in cases of the untreated specimen. The presence of sacrificial layer of sol coating only leads to the increase in the value of the oxygen index in case of the treated specimens where the PO3 specimen has recorded the highest LOI % and PO5 has a significant increase.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.