Abstract
The epoxidized styrene–butadiene–styrene nanofibers, which were prepared by epoxidation of the styrene–butadiene–styrene in toluene with peroxyformic acid generated in situ, were successfully embedded with nanometer titania dioxide by electrospinning. The optimum blended electrospinning parameters on preparing the nanometer titania dioxide/epoxidized styrene–butadiene–styrene fibers were obtained. The morphology and antiultraviolet property of nanometer titania dioxide/epoxidized styrene–butadiene–styrene composite fiber were examined using scanning electron microscope and an antiultraviolet transmission tester according to American Association of Textile Chemists and Colorists (AATCC183-2004) standard. The scanning electron microscope results showed that nanometer titania dioxide and the epoxy group could lead to the cementation of electrospinning fibers. The ultraviolet protection factor of rutile nanometer titania dioxide/epoxidized styrene–butadiene–styrene fiber membrane is the largest one of the four fiber membranes. The epoxidized styrene–butadiene–styrene nanofibers embedded with nanometer titania dioxide have great potential in the applications of antiultraviolet textiles.
Keywords
Introduction
In recent decades, polymeric nanofibers have been increasingly investigated due to its outstanding properties, including large specific surface area, great interconnectivity of pores, and high porosity with small pore size [1–5]. Different techniques have already been reported to produce polymeric nanofibers, such as phase separation, self-assembly method, and template polymerization, among which electrospinning is one of the most effective and high-potential methods to produce fibers ranging from the submicron level to several nanometers in diameter in a high voltage electrostatic field [6, 7]. Furthermore, the electrospun nanofibers have been used in various fields, such as tissue engineering scaffolds, filter media, semiconductive materials [8, 9].
Nowadays, with the aggravation of the fluorine contained pollution, the stratospheric ozone decreases and the ultraviolet (UV) radiation increases dramatically, which is harmful to humans [10, 11]. As a result, the attention of the hazards of UV radiation and the requirement of anti-UV textiles has been increased, respectively [12–14]. Therefore, increasing researches are focused on UV resistant fibers and UV absorbers [15, 16]. Zohdy et al. [12] investigated the UV-protective properties of cotton; PET fabrics; and their blend coated with ZnO, Alum, or Alum/ZnO by irradiation technique. Abundant literatures [17–19] are related to give the anti-UV property to commonly used textiles such as cotton, wool, PET, etc.
Styrene–butadiene–styrene (SBS) triblock copolymer, which has a two-phase morphology, possesses the maximum yield of the thermoplastic elastomer, due to the great elasticity and well wet-skid resistance [20]. However, owing to the lacking of functional groups and the sensitivity of the double bonds to light and heat, SBS is prone to hardening and deforming under UV irradiation [21, 22]. The modification of SBS is necessary to widen its applications. Among the SBS reported, few are concerning electrospinning with the modified ones. In previous experiment, due to less polarity, SBS was hard to dissolve in polar solvent at room temperature. So it was also difficult to prepare SBS nanofibers by electrospinning at room temperature. However, due to high polarity of epoxy group in epoxidized styrene–butadiene–styrene (ESBS), ESBS could dissolve in tetrahydrofuran (THF)/N-N dimethyl formamide (DMF) at room temperature, which made preparing ESBS nanofibers easy. Epoxidization may also increase the mechanical strength of prepared nanofibers by cementing them with each other.
The present work aims to prepare the ESBS nanoscale fibers with nanometer titania dioxide (nano-TiO2) embedded and discuss its anti-UV property and morphology. The double carbon bonds in the butadiene soft segment of the SBS were oxidized in toluene solution with peroxyformic acid generated in situ to yield the ESBS initially, and then the ESBS was characterized by infrared spectroscopy (IR) and the epoxy value was tested. Meanwhile, the nano-TiO2/ESBS composite fibers were prepared by blend electrospinning and observed by scanning electron microscope (SEM). The anti-UV property of different kinds of electrospun fiber membranes was investigated subsequently.
Materials and methods
Materials
All of the anatase and rutile nano-TiO2 and SBS were commercially available and used in form of powder. THF and DMF were purchased from National Chemical Co., Ltd, China. Formic acid (99%) and hydrogen peroxide (30%) were reagent grade for the epoxidation of SBS. All other reagents and solvents such as toluene, ethanol, hydrochloric acid, and acetone were commercially available and were of analytical grade.
Preparation of ESBS nanofibers
As shown in Figure 1, the 100 g/l SBS solution was prepared by dissolving the SBS powder into toluene by stirring. Then the temperature was raised to 70℃ and the formic acid (Vformic acid/VSBS = 1/40) was added. The H2O2 (Vhydrogen peroxide/VSBS = 3/40) was added into the mixture by pouring H2O2 aqueous solution slowly. The mixture was stirred at 70℃ for 2 h, and the production was agglomerated by ethanol after the solution got cold. Finally, the prepared product was washed to neutral (pH = 7.0 ± 0.5) by distilled water and dried at 60℃ in vacuum. The reaction equation is shown in Figure 1(b), and the formic acid was reacted with hydrogen peroxide under 70℃ as the step 1. Then the SBS was epoxidized by the peroxy acid which is prepared by step 1 at the same temperature as the step 2.
Schematic of ESBS nanofiber membrane preparation: (a) is the process of ESBS powder preparation, (b) is the reaction equations of epoxidation, (c) is the electrospinning process of ESBS nanofiber membrane.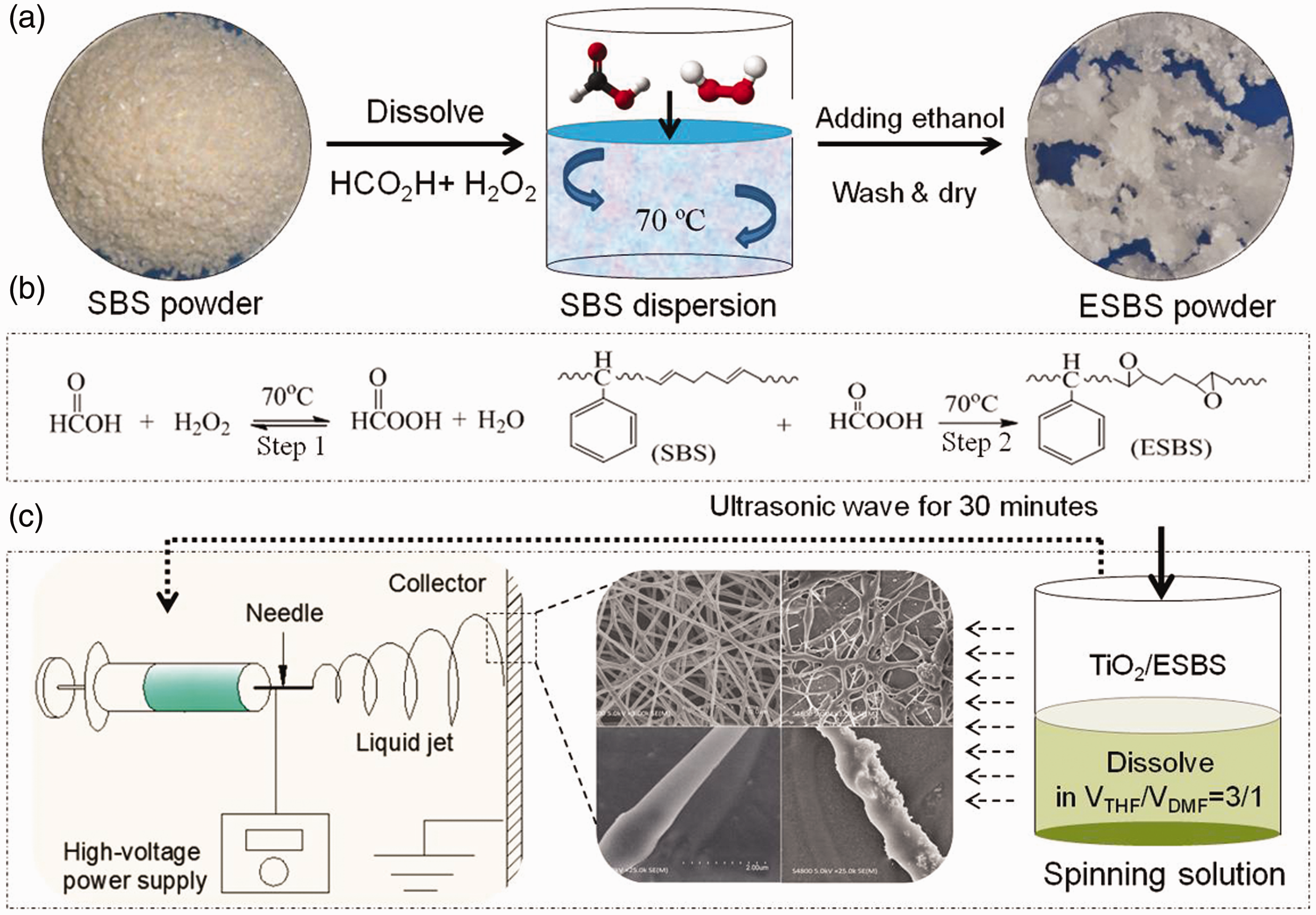
The two different crystalline structures of nano-TiO2 were dried at 70℃ and dispersed by ultrasonication with the same solvent (VTHF/VDMF = 3/1) for 30 min, respectively. All of the SBS spinning solution, ESBS solution, and nano-TiO2 dispersions with the same mass fraction of 12% were prepared by the same complex solvent (VTHF/VDMF = 3/1). The volume ratio of nano-TiO2 dispersion to ESBS solution was 1/4, and the two different kinds of nano-TiO2/ESBS spinning solutions were dispersed by ultrasonication at room temperature for 1 h. Then, the spinning solutions were put into a plastic syringe with a stainless steel needle 0.7 mm in inner diameter, and then the syringe was placed into the high voltage electrospinning machine (NEC-10, Japan). And all of the solutions were controlled at the same spinning condition. The distance between the needle tip and the aluminum-covered collector was 200 ± 10 mm, the applied voltage was 23 ± 1 kV, and the flow rate of the spinning solution was controlled at 0.4 ± 0.05 ml/h by a syringe pump (Figure 1(c)). After electrospinning for 8 h, the nanofiber membranes were taken off and dried in vacuum at room temperature for 5 h, respectively.
Characterization of ESBS nanofibers
Morphology
In order to compare the surface morphology of the ESBS and SBS, the samples were examined using a scanning electron microscopy (SEM) (Hitachi S-4800, Tokyo, Japan). All specimens were sprayed with gold prior to imaging and observed at a voltage of 5.0 kV.
Structural analysis
The infrared spectrums of SBS and ESBS were collected by Fourier transform infrared spectrometer (IR Prestige-21, Shimadzu, Japan). The epoxy value of the ESBS was tested by hydrochloric acid–acetone method [23]. Twenty-five milliliters mixed testing solvent of hydrochloric acid and acetone (1/80, v/v) was added into a solution of 1.0 g ESBS or SBS sample in 25.0 ml toluene and reacted at 20℃ for 2 h. Then three drops of phenolphthalein indicator and 0.1 mol/l potassium hydroxide–ethanol solution were dropped into the mixture to get the light pink color using base buret. The test and blank test were repeated for three times and the average epoxy value (E) was calculated by equation (1)

Anti-UV property test
The layer number (thickness) of electrospun fibers could affect UV protection factor (UPF) and UV transmission. In order to minimize the thickness effect, monolayer regions in each sample were marked through observing layer number of electrospun fibers from SEM. The anti-UV property was tested in marked monolayer regions by HB902 anti-UV transmission tester according to American Association of Textile Chemists and Colorists (AATCC183-2004) standard. For each sample, the identical experimentation was carried out five times.
Results and discussion
Morphology of ESBS nanofibers
The morphology and diameters of the ESBS electrospun fibers were analyzed before and after the nano-TiO2 embedding process. SEM in Figure 2 shows that the two different crystal structures of nano-TiO2 are combined with ESBS fibers. The ESBS nanofibers are entangled due to the hydrogen bonding between ESBS epoxy groups and hydroxyl groups in solution [24]. We can also observe that the ESBS nanofibers with nano-TiO2 are slightly more entangled than the ESBS pure nanofibers. The reason is that the addition of nano-TiO2, which can enhance the conductivity of spinning solution, may lead to the tendency of breakup of Taylor cone and promote the entanglement of fibers [25–27].
SEM images of electrospun fibers, (a) to (d) are SBS, ESBS, anatase-type TiO2/ESBS, and rutile-type TiO2/ESBS nanofibers, respectively.
Structural analysis
The infrared spectrums of SBS and ESBS nanofibers at the range of 500–2500 cm−1 are quite different as shown in Figure 3. The sharp peaks observed at 748, 962, and 908 cm−1 in curve A are attributed to the double bond of the cis-1,4-polybutadiene, trans-1,4-polybutadiene, and the 1,2-polybutadiene. The peaks observed at 1600, 1448, and 693 cm−1 in curve A are attributed to the styrene chain. However, the weakening of the peaks around 748, 962, and 908 cm−1 in curves B, C, D indicates that the double bond of the SBS has been damaged. Simultaneously, the appearing of the peaks at 1248 cm−1, 889 cm−1 and 854 cm−1 in curves B, C, D certify the existences of the epoxy group in the production. Furthermore, the broad peak of anatase-TiO2 is observed from 623 to 537 cm−1 in curve C and the broad peak of rutile TiO2 is observed from 630 to 550 cm−1 in curve D. It can be concluded that both the TiO2/ESBS nanofibers were successfully prepared, and the epoxy value of the ESBS electrospun fibers measured using the hydrochloric acid–acetone method is 13.6%.
FT-IR spectra of SBS and ESBS nanofibers, (a) to (d) are SBS, ESBS, anatase-type TiO2/ESBS, and rutile-type TiO2/ESBS nanofibers, respectively.
Anti-UV property
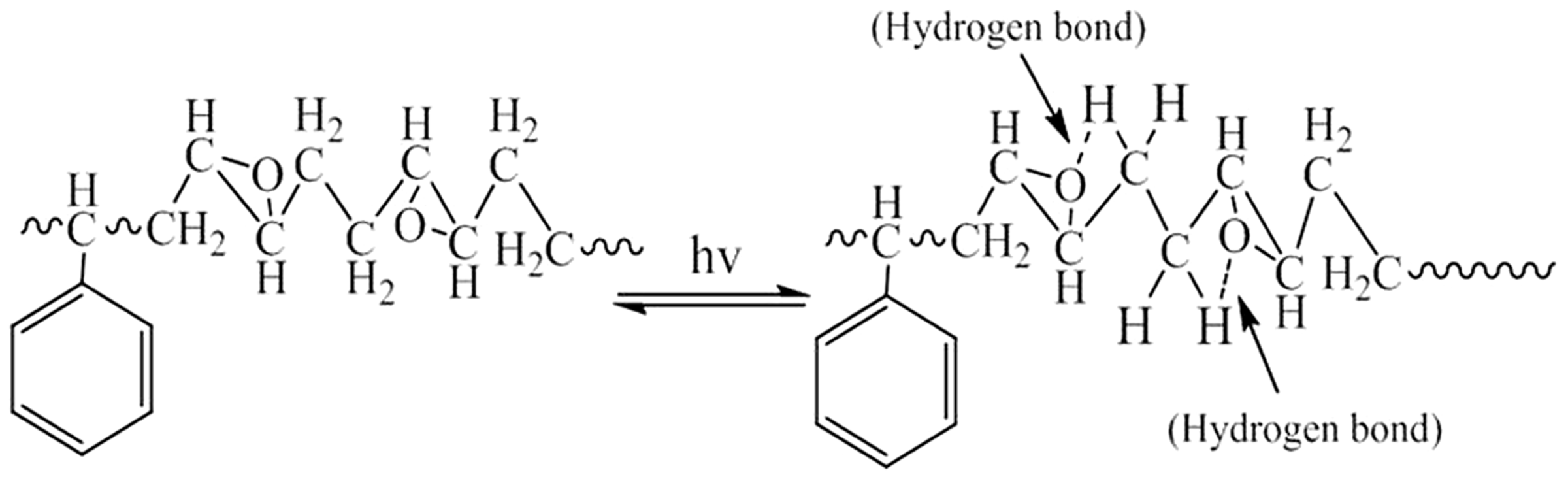
Owing to the conjugate system and the intramolecular hydrogen bond between the epoxide group and the hydrogen of ESBS, the average UPF (31.6) of it is bigger compared to SBS (Figure 4). As shown in Figure 5, the anti-UV mechanism describes that when exposing on the sunlight, ESBS absorbs the UV lay, and the epoxy group may combine with the hydrogen bond on the o-position carbon, and then the structure will restore while the energy is transformed and released [28]. The circular reaction may lead to the anti-UV property of ESBS repeatable and durable.
Comparison of antiultraviolet property of fiber membranes: (a) UPF and (b) UV transmission of SBS, ESBS, anatase-type TiO2/ESBS and rutile-type TiO2/ESBS nanofiber membranes. The antiultraviolet mechanism of ESBS nanofiber membrane. When exposing on the sunlight, the epoxy group may combine with the hydrogen bond on the o-position carbon, and then the structure will restore while the energy is transformed and released.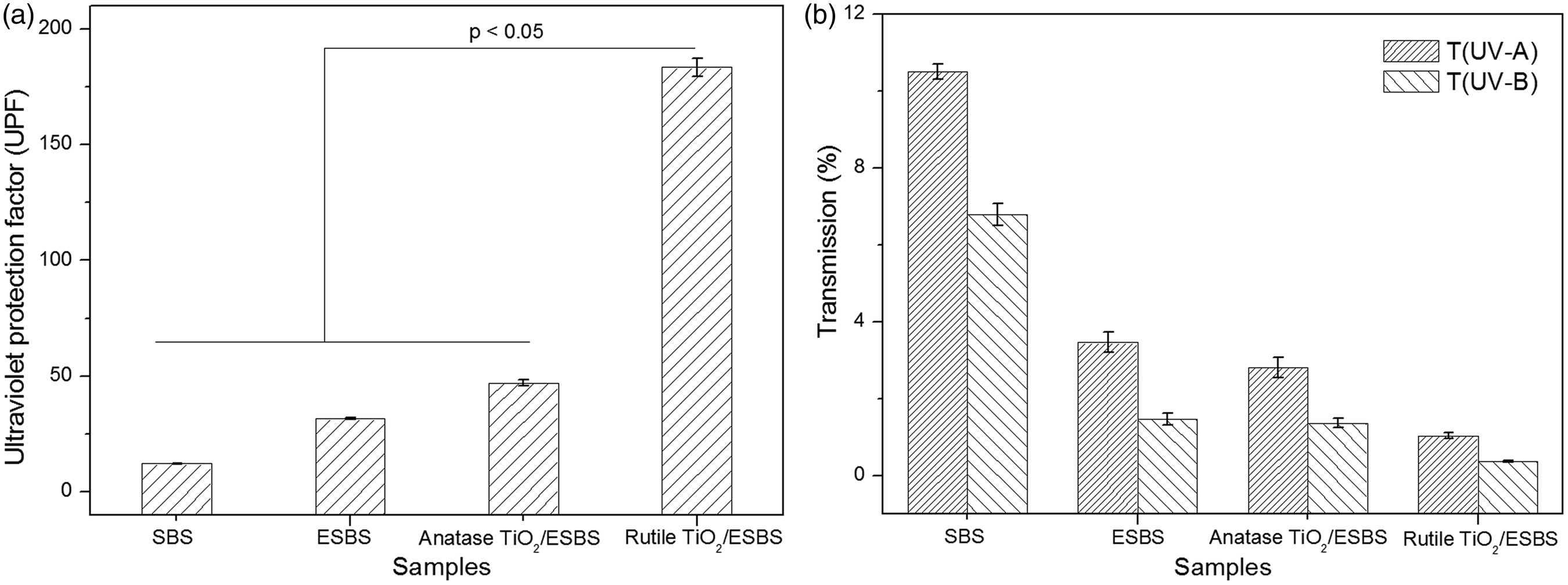
The nano-TiO2/ESBS fiber membranes show much better anti-UV property as data given in Figure 4. It can be explained that the anatase nano-TiO2/ESBS fiber membrane has double anti-UV effects: one is that the macromolecular structure of ESBS can absorb and scatter the ultraviolet ray; the other is that anatase nano-TiO2 can also absorb the ultraviolet ray with a match or exceed its band gap energy [28–30]. Meanwhile, the average UPF (183.4) of rutile nano-TiO2/ESBS fiber membrane shown in Figure 4 is the biggest one of the four fiber membranes and almost the four times of the UPF (47.1) of anatase nano-TiO2. It also can be found that the T (UV-A) and T (UV-B) of rutile nano-TiO2/ESBS reduce obviously, especially for the T (UV-B), which is less than 0.5%. It can be explained that as the higher of density and absorption limit and the narrower of the optical band gap, the light induced by electron/hole composite rate of rutile nano-TiO2 is much higher. At the same time, rutile nano-TiO2 possessing longer Ti–O bond length and weaker agglomeration tendency than anatase nano-TiO2 can be dispersed much more evenly, which may lead to the narrower distribution of particle size. All of above factors endow the rutile nano-TiO2 equip with lower refractive index and more suitable band gap energy corresponding to the wavelength of UV-A and UV-B. In summary, the anti-UV properties of different fiber membranes are decreasing in the sequence of rutile nano-TiO2/ESBS, anatase nano-TiO2/ESBS, ESBS, and SBS.
Conclusions
In this study, the ESBS nanofibers, which were prepared by epoxidation of the SBS in toluene with peroxyformic acid generated in situ, were successfully embedded with nano-TiO2 by electrospining. The optimum blended electrospining parameters on preparing the nano-TiO2/ESBS fibers were obtained. The results showed that nano-TiO2 and the epoxy group could lead to the cementation of electrospinning fibers. The UPF of rutile nano-TiO2/ESBS fiber membrane is the largest one of the four fiber membranes. In conclusion, the ESBS nanofibers embedded with nano-TiO2 have great potential in the applications of anti-UV textiles.
Footnotes
Authors’ contribution
Xuchen Tao and Chenggong Hu contributed to this manuscript equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by National Natural Science Foundation of China (21171003), Science Foundation of Education Department of Anhui Province (KJ2013B028, KJ2013A050), Textile Science and Technology Key Discipline in Anhui Polytechnic University, State Key Laboratory for Modification of Chemical Fibers and Polymer Materials, Donghua University with project code LK1421 and Natural Science Foundation of Jiangsu Province (BK20150372).
