Abstract
Carbon black has been replaced with silica as a reinforcing filler in tire tread compounds. This change has led to lower rolling resistance and improved hysteretic losses of so-called “green tires.” However, the dispersion of silica in the rubber matrix is an important issue due to the poor compatibility of hydrophilic silica with a hydrophobic rubber matrix. Recently, some rubbers with polar functional groups that can interact with silica have been studied to improve the interaction in silica-filled rubber composites. In this work, we fabricated the silica-filled rubber composites with solution styrene butadiene rubber (SSBR) and epoxidized natural rubber (ENR) and evaluated their properties in a silica-containing rubber formulation compared to conventional SBR and NR. The silica-embedded polar rubber matrix exhibits remarkable enhancement in the modulus, tensile strength, and abrasion properties due to an efficient dispersion of the silica and improvement of interfacial interactions with the rubber matrix. The polar rubber composite exhibits an enhanced dry and wet braking and improved rolling resistance due to the improved dispersion of the silica in the rubber matrix. These results show that rubber composites prepared with polar rubbers have great potential for tire engineering applications.
Keywords
Introduction
Carbon black (CB) has been most widely used as a reinforcing filler due to the high tensile abrasion properties that it provides among various fillers.1–6 However, in the tire industry, CB has been constrained by the lack of simultaneous improvements in rolling resistance and wet traction. In recent years, the improvements in the fuel efficiency of vehicles have become an important issue due to the depletion of fossil fuels and the environmental pollution caused by them. 7 From this point of view, silica has been used to replace carbon black as a reinforcing filler in tread compounds because of its low hysteresis loss and low rolling resistance to produce fuel saving tires.8,9 Furthermore, “green tires” made with silica fillers replacing carbon black have a greater wet grip and abrasion resistance.10–12 However, silica with silanol groups on its surface is highly polar and incompatible with non-polar rubbers such as styrene butadiene rubber, natural rubber and cis-butadiene rubber. Therefore, with a similar specific surface area, silica compared with carbon black provides a lower degree of performance improvement. Generally, to successfully use silica in rubber matrix for performance improvement, a silica coupling agent such as bis(triethoxysilypropyl)tetrasulfane (TESPT) is required to enhance the dispersion of silica in rubber matrix and to improve the rubber-filler interaction. As an alternative method to disperse silica in a rubber matrix, the sol-gel process has shown a good dispersion for silica filling in a rubber matrix. To obtain a preferable interfacial interaction between the silica and a rubber, 9 an in-situ synthesis method has also been widely used to reinforce rubber like NR latex, 13 NR, 14 SBR and NR which are grafted with methyl methacrylate 12 to increase the interaction between the hydroxyl of the in-situ silica and the rubber matrix. More recently, the microstructure of NR and SBR has been adjusted to improve the affinity between silica and rubber.15,16 Mark and Pan 17 suggested a new approach for preparing silica-filled rubber by generating the silica inside silicone rubber. Additionally, epoxidized natural rubber (ENR) with a higher polar epoxide functional groups interacts with the hydroxyl groups on the silica surfaces of silica. Cataldo 18 reported that the mechanical properties of silica-filled ENR were higher than those of silica-filled NR without a silane coupling agent due to the improved interaction between the ENR and silica surface via hydrogen bonds. 19 Furthermore, solution SBR (SSBR) modified at the end or the backbone of the rubber chain by functional groups was developed and commercialized. SSBR composites are very different compared to conventional emulsion SBR composite due to their rheology, mechanical and fatigue properties. 18 So far, few studies have compared the properties of ENR and SSNR composites to improve the affinity between silica and rubber.
In this study, silica-filled rubber composites with polar functionalized and non-polar functionalized SBR and NR were fabricated and characterized with the different silica contents. Generally, SSBR and ENR composites with hydrophilic functional end groups increase the modulus, tensile strength and abrasion properties compared to conventional SBR and NR composites. Furthermore, silica filled SSBR has more desirable properties than that of ENR with lower silica contents. The SSBR composites also have a lower rolling resistance and highly improved grip in both dry and wet conditions. This study shows the advantages and potential of silica-based fillers in tire engineering.
Materials and methods
Materials
The matrix material, an SBR (E-SBR 1500, Kumho Petrochem Co. Ltd., Korea), consisting of 23% styrene and 77% butadiene, was used. Natural rubber (RSS 3), solution styrene butadiene synthetic rubber (SSBR), and epoxidized natural rubbers with 35 mol% of epoxide solution styrene butadiene rubber with alkoxysilane-functional groups at two ends of macromolecular chains were prepared from Kumho Peterochem in Korea. Carbon black (N-330) was supplied by OCI Co. Ltd., Korea and the silica used in this study were supplied by Rhodia Silicas. N-tert-butylbenzothiazole sulfonamide (TBBS) and sulfur were obtained from Shangdong Shanxian Co. Ltd., China. Stearic acid (S/A) and ZnO were purchased from Sigma-Aldrich. Zinc ion processing aids were obtained from Struktol Co., USA, and bis(triethyoxysilylpropyl)tetrasulfide (TESPT) (Evonic Indusitrial AG, Si69) was applied to the mixed compounds as coupling agent.
Preparation of rubber composites
The formulations of the rubber composites are given in Table 1. The master batch is prepared by mixing the materials by using the laboratory mill with rolls dimensions at the speed of 4 rpm. And then, the diverse rubber latex (50 phr) was mixed with silica by vigorous stirring for 24 h and was added a sulfuric acid solution. The mixed rubber composites were washed with water until the pH reached 6–7. Also, the composites were dried for 24 h in an oven at 50°C. Subsequently, for master batches, SBR (50 phr) and silica (40, 60, 80 phr)/rubber latex emulsion (50 phr) were mixed at a rotor speed of 60 rpm in a Banbury mixer. The vulcanization agents and additives were mixed at the end of mixing to initiate the curing process at a rotor speed (60 rpm). The resulting materials were placed in an aluminum mold and cured at 160°C (T90) as determined by a rheometer under pressure.
Formation of rubber composites.

Characterization
Modulus and tensile tests were carried out on an Instron tensile machine (Instron Co., UK) at a crosshead speed of 300 mm/min. Tensile and modulus tests were carried out at a crosshead speed of 300 mm/min on an Instron tensile machine (Instron Co., UK) as ASTM 3039. The length and width of dumbbell-shaped samples were 100 mm and 5 mm, respectively. At least four tests were carried out for each type of sample. Fatigue test samples were carried out on a device (Wallace-Dematta Flexing Machine) and then calculate the number of cycles during crack growth of rubber composites until it is broken at 75°C. The abrasion testing is done with wallace test equipment, HZ 50 following test method as ASTM D2228. The device works with 250 Watts, where the sample is placed touching the lever, the lever spin 500 cycle for 5 min and then calculated the difference in weight before and after the test. Curing characteristics were measured for 30-min period at 160°C using a moving-die rheometer [DRM-100 (LP-171)]. Dynamic Mechanical Analyzer (DMA, A Metravib DMA+ 150) was used to complete temperature sweeps using two geometries, under deformation modes of shear and tension. The DMA was carried out from −60°C to 120°C at a heating rate of 3 K/min and a fixed frequency of 10 Hz in a mode of double cantilever deformation.
Results and discussion
Figure 1(a) schematically shows the reaction mechanism between ENR and the silica in sol-gel reaction processs.12–18 This reaction depends on the chemical bond between CH2–C–OH and Si–OH generated during the hot press process at 160°C for 20 min. This can again be attributed to the interaction between the hydroxyl and siloxane groups available on the silica and the epoxide groups of ENR through either hydrogen bonding or chemical bonding that occurs after a possible ring opening of the epoxy ring during mixing.20,21 Therefore, silica serves as a both self-vulcanizing agent and reinforcing agent in the ENR matrix. Generally, because the SSBR is synthesized by anionic polymerization with monomers of styrene and butadiene and an initiator dilithium, the –Si(OCH3)3 groups are grafted onto the two ends of the polymer macromolecular chains. After end-group functionalization, SSBR can react with silica following the hydrolysis reaction between the Si–(OCH3)3 groups of SSBR and the Si–OH groups of silica shown in Figure 1(b).
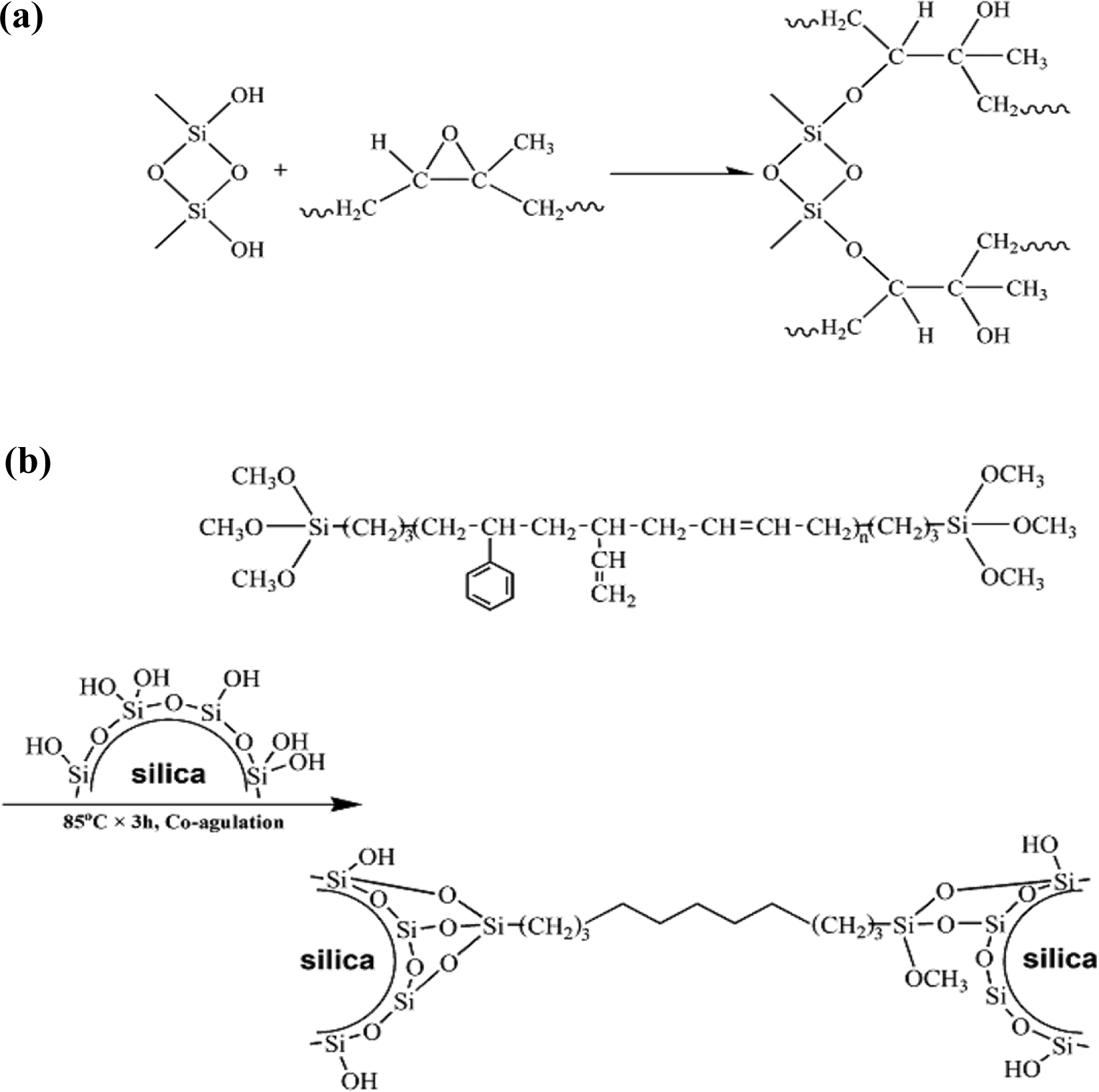
(a) Schematic diagram showing the overall reaction for preparing the ENR/silica composites. (b) Schematic diagram showing the overall reaction for preparing the SSBR/silica composites.
The influence of the silica contents on the characterization of the vulcanization process is presented in Figure 2. Figure 2(a) and (b) shows the effect of the silica contents on the vulcanization process of diverse rubber. Generally, natural rubber (NR) has a relative slow curing time compared with styrene butadiene rubber (SBR) due to its relatively low thermal conductivity.1–2 Moreover, it was observed that all the silica based rubber composites had a longer curing time (t40 and t90) for vulcanization with the increasing silica contents shown in Figure 2(a) and (b). Furthermore, in the case of the epoxidized NR, both curing times were decreased due to the higher polarities between the ENR and silica which less interfered with curatives leading to faster curing. 22 Even though part of the silanol groups are assumed to have interacted with the epoxide groups, the unreacted silanol groups can hydrogen bond with polar accelerators causing accelerator adsorption on the silica surface. Additionally, the curing time for SSBR is faster than that for SBR due to the high vinyl content in the rubber matrix. Furthermore, ENR and SSBR with the end-modified functional group can affect the curing time sensitivity with increasing silica content. The MH-ML (Δ torque) values of the rubber composites are shown in Figure 2(c) to (e). Generally, because the torque difference is directly proportional to the degree of crosslinking, it could be plausible that chemical crosslinks between the silica surface and the rubber chains formed during the mixing and curing.23–25 According to the increase in the silica contents, the ENR and SSBR composites with the hydrophilic functional end groups had increased delta torque values compared with NR and SBR, respectively, in Figure 2(c) to (e). Additionally, in Figure 2(e), the delta torque values of the SSBR composites and the crosslink density of the vulcanization meaningfully increased compared to those of the other rubber composites at silica 80 phr. This result indicates that the SSBR has a stronger crosslinking with the silica during the vulcanization compared to SBR.

(a) Curing properties (t40) of rubber composites with diverse silica contents. (b) Curing properties (t90) of rubber composites with diverse silica contents. (c) Torque of rubber composites with silica 40 phr. (d) Torque of rubber composites with silica 60 phr. (e) Torque of rubber composites with silica 80 phr.
The mechanical properties of all the rubber composites were examined along with the composites based on the different contents of silica shown in Figure 3. As more silica was dispersed into the rubber, the elasticity of the rubber decreased. Furthermore, the increment of the modulus with the increased silica loading may be due to the effect of strain increment, in other words, the silica is in the rigid phase which cannot be deformed. The modulus (300%) and tensile strength of the SBR composites with a high molecular weight were increased more than those of the NR composites shown in Figure 3(a) and (b). Moreover, compared to the NR composite, the modulus of the ENR composite with silica 80 phr is dramatically increased to 113 MPa shown in Figure 3(a). This is because the excellent dispersion of silica in the ENR matrix leading to a stronger rubber-filler hydrogen bond interaction although the rubber main matrix are different. Moreover, in Figure 3(a), the SSBR composite shows that the addition of silica 80 phr provided a significant enhancement in the modulus. The same behavior is observed for the tensile strength in Figure 3(b). As the concentration of the silica was increased, the tensile strength of all the rubber composites also increased. This result can be explain such that in the case of large quantities, silica will fill the spaces between the rubber chains, thus making a rigid structure with a better tensile strength. The tensile strength of the SSBR composite increased by as much as 22% compared to that of the SBR composite at silica 80 phr. While silica has poor compatibility with SBR due to its poor wettability, SSBR had a better adhesion and compatibility with silica because of the hydrophilic end functional group of the SSBR. Figure 3(c) shows a comparison of the elongation at the break of the rubber composites with different silica contents. The elongation property of the rubber composites decreased with the addition of large quantities of silica because when the rubber is highly contracted such that silica fills the spaces, it will restrict the movement of the chains and then decrease the elongation property. The elongation at the break of the SSBR composite (310%) shows it has decreased properties compared with those of the other rubber composites due to the enhanced interfacial adhesion and restricted motion of the rubber segmental chains. The fatigue properties of the rubber composites, shown in Figure 3(d), were measured after 2,000 and 10,000 cycles. Fatigue crack growth (dc/dn) was calculated with the following equation. 26

c; the crack length, n; cycles, G; the tear energy.

(a) Modulus of rubber composites with diverse silica contents. (b) Tensile strength of rubber composites with diverse silica contents. (c) Elongation at break of rubber composites with diverse silica contents. (d) Fatigue properties (fatigue crack growth) of rubber composites with diverse silica contents.
This equation means that fatigue crack growth is proportional to the tear energy, and equation (1) indicates that when the tearing energy is decreased, the fatigue life is increased. The SEM micrographs in Online Supplemental Figure S2 show that silica (20 phr) disperses without coagulation in the SSBR rubber matrix. As the amount of silica increases, agglomeration will take place, and an inhomogeneous distribution causes stress in the formulation, and the fatigue crack growth of the rubber composites increase according to the increase of the silica contents. The SSBR sample exhibited a remarkable increase in the crack length after 10,000 cycles, and the fatigue crack growth of the SSBR sample (3.89) was increased by over 87% compared to those of the SBR (2.39) and NR (2.08) at silica 80 phr. Also, NR, ENR, SBR and SSNR sample with silica 20 phr were subjected to TGA under a nitrogen atmosphere to examine their thermal properties, and the results were as exhibited in Online Supplemental Figure S1. The SSBR sample raised the thermal stability much more effectively than NR, ENR, and SBR samples, respectively. This was ascribed to the fact that the dispersion and interaction of SSBR matrix with the silica were better than NR and SBR in the corresponding composites.
In Figure 4, a comparison of the TEM images between NR and SSBR composites is shown. The silica fillers are shown to form globular aggregates of different sizes ranging from several microns to submicronic clusters in Figure 4. However, the large aggregates of silica are not observed and the silica are well distributed throughout SSBR matrix as smaller clusters of a micron or less in length. In Figure 4, TEM images provide direct evidence for evaluating the phase morphology and dispersion of silica in relation to the mechanical properties of rubber composites.

TEM images of (a) NR and (b) SSBR composites.
The dynamic mechanical properties (DMA) of all the rubber samples were also investigated to assess the elastic response. The glass transition temperatures (Tg) from −50 to −3°C are shown in Figure 5(a). Generally, the Tg of the SBR is higher than that of NR which means that the Tg of high styrene polymer composites is higher than that of high vinyl polymer composites. 27 Moreover, the increase in the silica contents shows no effect on the Tg; however, the ENR and SSBR composites have an enhanced Tg compared to the NR and SBR, respectively. This result supports that the functional groups of rubber improve the silica rubber interaction in the compound in accordance with the increased interaction between the rubber and silica. The addition of silica to the SSBR matrix increases the Tg from −14.8 to −12.8°C which is a higher value than that of the ENR composite from −23.8 to −21.8°C. This is attribute to the interaction between the functional group of the SSBR and the silica surface which enhances the interfacial adhesion and restricts the motion of the rubber segmental chains. The values of the tan δ (tan δ is defined as the ratio of the loss modulus to the storage modulus) for all the rubber composites versus the silica content at the temperature are shown in Figure 5(b) and (c). The dynamic mechanical properties are important because many engineered rubbers are frequently subjected to dynamic loading. The DMA test evaluates the resulting physical properties of the rubber and is often used to predict both the rolling resistance and the wet traction properties.23–25 Furthermore, rubber from the reactions of the active chain ends of the SBR and multifunctional groups with two or more hydrophilic groups are superior in terms of a lower rolling resistance and higher wet skid resistance. The tan δ of 0°C for the SSBR composite with silica 80 phr was about 81% higher than that of the SBR composite, shown in Figure 5(b), indicating that the pneumatic tire using SSBR has great potential for improving the wet grip tire performance. Additionally, in Figure 5(c), the 60°C tan δ of the SSBR composite was about 32% lower than that of the SBR composite. The tendency for the 0°C tan δ and 60°C tan δ of ENR with silica contents is also similar. Generally, the lowering of the tan δ at 60°C correlates to an improved rolling resistance. Furthermore, the SSBR composite by the reaction of the active chain ends of the SBR and the multifunctional groups is superior in terms of a lower rolling resistance and a higher wet grip due to the increase in the affinity (strength of the chemical bonds) for silica with its higher polarity. 28

(a) Glass transition temperature of rubber composites with diverse silica contents. (b) 0°C tan δ of rubber composites with diverse silica contents. (c) 60°C tan δ of rubber composites with diverse silica contents.
Volume loss caused by the abrasion of the rubber composites with the loading of silica is shown in Figure 6. The abrasion resistance of the rubber composites was measured in terms of the relative volume loss. A higher volume loss shows a lower abrasion resistance and vice versa. The abrasion properties increased up to an 80 phr loading of silica, and all three wear (Akron, William, Lambourn) properties are similar shown in Figure 6(a) to (c). This result is due to the interaction between the rubber matrix and the silica which restricts wear on the rubber during abrasion. At a higher loading of silica, the filler-filler interactions are increased the filler-rubber interactions resulting in a decreased rate in the abrasion loss values. Additionally, for the silica based rubber composites using ENR and SSBR, the wear resistance was improved due to their stronger interfacial adhesion between the rubber functional group and the surface of the silica compared to that of the NR and SBR composites. Lower abrasion loss values were obtained with the SSBR composites containing 80 phr silica compared to those of the other rubber composites. The silica-embedded SSBR composite exhibits a remarkable enhancement in the modulus, tensile strength, and abrasion properties which is consistent with the efficient dispersion of the silica and improved interfacial interactions with the rubber matrix. Therefore, we can confirm that our new and fascinating approach has great potential in commercial polymer engineering.
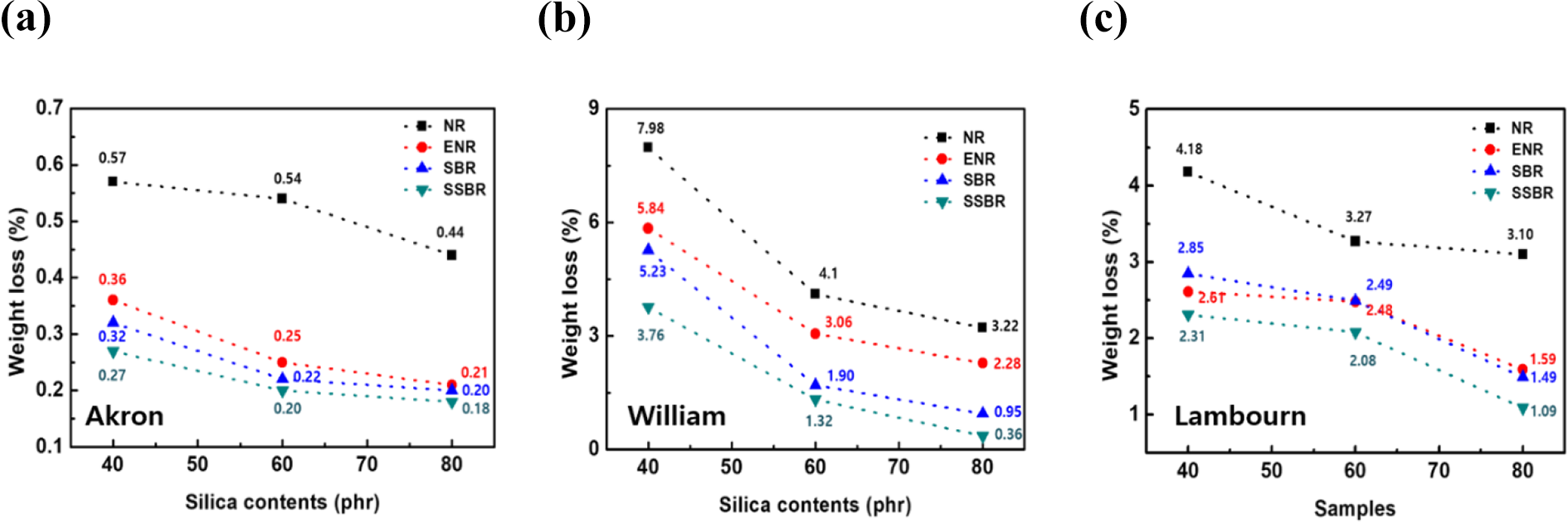
(a) Akron abrasion properties of rubber composites with diverse silica contents. (b) William abrasion properties of rubber composites with diverse silica contents. (c) Lambourn abrasion properties of rubber composites with diverse silica contents.
Conclusions
This study demonstrated the effect of the rubber type on the properties of composites containing different silica contents. The functionalization of the end and backbone of the rubber facilitates the better dispersion of silica with high solubility and stability which prevent from stacking and aggregating between fillers. The SSBR composite had a significantly improved modulus, tensile strength and abrasion performance due to the resulting homogeneous dispersion of the silica in the rubber matrix. Furthermore, the viscoelastic properties, such as the change in Tg and tan δ at 0 and 60°C, improved with the silica contents which indicate that the compatibility between the rubber and silica is a critical factor in determining the viscoelastic properties. Moreover, among the types of rubber, ENR and SSBR showed high crack growth lengths which in agreement with their mechanical properties. These results may provide new opportunities in the preparation of rubber composites with high performance.
Supplemental material
supplemental_material_(1) - Study on silica-based rubber composites with epoxidized natural rubber and solution styrene butadiene rubber
supplemental_material_(1) for Study on silica-based rubber composites with epoxidized natural rubber and solution styrene butadiene rubber by Sung Ho Song in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2019R1A6A1A03032988; NRF-2017R1C1B507647614; NRF-2020R1I1A307162811).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
