Abstract
Layer-by-layer self-assembly technology was introduced with adsorption of oppositely charged polycations and polyanions. The composite thin film consisting of quaternized chitosan derivative/poly(2-acrylamide-2-methylpropane sulfonic acid sodium salt) was created on the surface of cotton fabrics by using this method without much damage for breaking strength of swatches. The oxidative chlorine of five bilayers deposited on the swatches could reach 0.25%. The treated cotton fabrics exposure to dilute household bleach possessed excellent antimicrobial property, which could inactivate 100% of Staphylococcus aureus and Escherichia coli O157:H7 bacteria with a contact time of 1 min. The chemical surface modification, surface morphology, washing stability, storage stability, and mechanical property of the modified swatches were characterized and confirmed by Fourier transform infrared spectroscopy, scanning electron microscopy, atomic force microscopy, stability measurement, and tensile strength test, respectively.
Introduction
Chitosan is a natural cationic polysaccharide produced by deacetylation of chitin, the second most abundant natural polymer in the world [1]. It is reported that a substantial amount of work has been studied on chitosan, which show promising applications due to its biocompatibility, biodegradability, nontoxicity, and antimicrobial property [2, 3]. Textile materials including cotton [4–6], silk [7], wool [8], polyester [9], and linen [10] have been modified with chitosan or its derivatives, and desirable antibacterial activity has been reported. The modification of chitosan mostly can be carried out in the C2-amine groups and C5-oxhydryl groups of chitosan due to their active reaction [11–13]. Different other groups can be introduced into repeating units of chitosan and provide chitosan with properties and functionalities for various applications.
Layer-by-layer (LbL) self-assembly technique introduced by Decher et al. [14] has been attracting more and more attention due to its potential applications. This unique method involves the sequential adsorption of oppositely charged polyanions and polycations [15] and creates composite thin films on the surfaces of substrates by dipping the substrates into the solutions with oppositely charges. LbL technique can select suitable polyelectrolyte solutions to create a broad range of layer components according to different and versatile applications. Moreover, the roughness and thickness of the multilayered films can be controlled via the number of deposition layers and the types of polyelectrolytes [16]. It is noted that the substrates used to support and deposit multilayer possess various shapes and sizes, and the multilayered films can be applied to the wide range of substrates [17–19]. On the other hand, LbL technique is one of simple and environmentally friendly processes in coatings and uses water as solvent at ambient temperature instead of organic solvent at high temperature [15, 20]. This cost-effective method not only saves energy but also does not have adverse effect on environment.
Cerkez et al. synthesized poly(2,2,6,6-tetramethyl-4-piperidyl methacrylate-co-acrylic acid potassium salt) and poly(2,2,6,6-tetramethyl-4-piperidyl methacrylate-co-trimethyl-2-methacryloxy-ethylammonium chloride, and first developed antimicrobial function of N-halamine biocidal coatings utilizing the LbL assembly technique [21]. Liu et al. synthesized a cationic homopolymer poly((3-acrylamidopropyl) trimethylammonium chloride) and an anionic homopolymer poly(2-acrylamido-2-methylpropane sulfonic acid sodium salt), and coated them onto cotton fabrics via the LbL deposition technique [22]. Shirvan et al. used chitosan and pentasodium tripolyphosphate to assemble the cationized woven cotton fabrics via the LbL technique [15]. Although chitosan possesses biological property, the antibacterial activity was not as good as N-halamines. The LbL deposition technique can broaden the use of N-halamine biocides combined with antimicrobial property of chitosan. Some N-halamine chitosan derivatives have been also synthesized for use in antibacterial treatment [23]. If they were used to treat cotton fabrics by the LbL deposition technique, procedure of ionizing the polymers is essential.
In our previous work, a novel chitosan derivative was synthesized successfully and coated onto cotton fabrics using chemical cross-linking, which provided cotton fabrics with stable antimicrobial activity under the conditions of different pH [24]. However, the chemical cross-linking caused certain degree of damage to the tensile strength of the treated swatches, and the cotton fabrics after treatment became stiffer. In this study, the N-halamine chitosan derivative was further quaternized with glycidyl trimethyl ammonium chloride (GTMAC). It is feasible to use quaternized chitosan derivative positively charged as the cationic polyelectrolyte to treat cotton fabrics by LbL technology. LbL technique avoided the using of chemical cross-linking and its negative effect on cotton fabrics. In addition, PAMPAS was used as the anionic polyelectrolyte for LbL coating. It is reported that chlorinated PAMPAS is nonirritant to the rabbits tested [22]. The result of the skin stimulation test indicates that PAMPAS-Cl is also a safe antibacterial agent which can be used for antibacterial coating on textiles or other materials. So it would not cause negative effect to the quaternized chitosan derivative as polyelectrolyte.
Experimental
Materials
Chitosan (deacetylated degree 90.32%, molecular weight 50 kDa) was purchased from Zhejiang Aoxing Biochemical Co., Ltd, China. Household bleach (the active chlorine content was 5%) was supplied from Sinopharm Chemical Reagent Co., Ltd, Shanghai. 2-Acrylamide-2-methylpropane sulfonic acid (AMPA) was purchased from J&K Chemicals, Shanghai, China. All reagents were used as received without further purification. Bleached cotton fabrics (133 × 72/40S × 40S) was purchased by Zhejiang Guandong Textile Dyeing and Garment Co., Ltd. The bacteria involved were Staphylococcus aureus (ATCC 6538) and Escherichia coli O157:H7 (ATCC 43895) (American Type Culture Collection, Rockville, MD). The Trypticase soy agar was from Difco Laboratories, Detroit, MI.
Instruments
The FT-IR spectra of the N-halamine chitosan derivative-treated cotton fabrics were recorded by a Nicolet Nexus 470 spectrometer in the optical range of 400–4000 cm−1 by averaging 32 scans at a resolution of 4 cm−1. All swatches were prepared as potassium bromide pellets. The 13C NMR spectrum of N-halamine chitosan derivative was recorded on a Bruker AV-300 spectrometer. The SEM images were obtained by SU-1510 field-emission scanning electron microscope (Hitachi, Tokyo, Japan).
Synthesis of chitosan derivatives (Q-CS-HDH)
The CS-HDH was prepared as early work [24]. Then 6.02 g of CS-HDH (0.02 mol) and 15.16 g of GTMAC (0.1 mol) were distracted in 200 mL of isopropanol. Nitrogen was bubbled through the solution for 15 min and then the mixture was stirred at 70℃ for 30 h [13]. Then the solid was filtered, washed with acetone and ethanol solution, filtered and dried under vacuum yielding 7.34 g of Q-CS-HDH as a buff powder (Scheme 1). 13C NMR (D2O and CF3COOD): δ 98.76 (OCHO of CS), 63.05 (CHNH of CS), 70.70 (CHOH of CS), 78.51 (CH(CH)2 of CS), 75.81 (CHO of CS), 61.29 (CH2OH of CS) 55.39 (CH2NH), 178.07 (CON, CONH), 63.05 (C(CH3)2), 20.48 ((CH3)2), 56.96 (OCH2), 69.02 (CHOH), 70.24 (CH2N+(CH3)3), 55.39 ((CH3)3 N+R). FTIR (KBr) (cm−1): 3447 (νO-H), 1767 and 1700 (νC=O), 1473 (δC-H of (CH3)3N+R).
Preparation of PAMPAS
A total of 4.14 g of AMPA (0.02 mol) was dissolved in 100 ml deionized water containing a small amount of sodium persulfate (0.04 g, 1%) as initiator [22]. The solution was adjusted to neutral with 10% sodium hydroxide. The mixture was stirred and reacted at 65℃ for 3 h. Then water was removed at reduced pressure. The obtained product was dried in a vacuum oven at 45℃ for 2 days (Scheme 2). FTIR (cm−1): 1658 (νC=O), 1549 (νN-H), 1129 and 1045 (νO=S=O), 625 (νS-O).
Functionalization of the cotton fabrics
Q-CS-HDH was dissolved in 2% acetic acid solution at a concentration of 2 wt% as polycationic solution. PAMPAS was dissolved in deionized water at a concentration of 2 wt%, and the solution was adjusted to neutral with 10% sodium hydroxide as polyanionic solution. Cotton fabrics were first immersed into deionized water for 15 min. Then the cotton fabrics were immersed into a solution of Q-CS-HDH for 5 min and washed with deionized water three times to remove excess polycation. The coated cotton fabrics were dried at 80℃ for 5 min. Subsequently, the coated cotton fabrics were immersed into a solution of PAMPAS for 5 min at room temperature and washed again to remove excess polyanion. The coated cotton fabrics were dried at 80℃ for 5 min. Each time the swatches were immersed into the opposite charged polyelectrolyte solution and then washed and dried. This process was repeated five times (Figure 1). The cotton swatch-assembled five layers were used for all the tests.
The process in the formation of Q-CS-HDH/PAMPAS multilayers via LbL technology.
Chlorinated procedure of the swatches
The swatches chlorination were carried out with household bleach which was diluted to the concentration of 0.5% by distilled water and adjusted to pH 7 with 1 N H2SO4. The chlorine content loaded onto the swatches was evaluated by the iodometric/thiosulfate titration procedure [25].
Antimicrobial testing
AATCC Test Method 100-1999 was employed for the antimicrobial efficacy testing. The pH 7 phosphate buffer solution (100 mM) contained the test microorganisms which were gram-positive S. aureus and gram-negative E. coli O157:H7. An aliquot of (25 μl) bacterial suspension was added to the interlayer center of two 2.54 cm × 2.54 cm swatches using the method of “sandwich.” After contact times of 1, 5, 10, and 30 min, the swatches were quenched with 5 ml of 0.02 N sodium thiosulfate solutions to remove any remaining oxidative chlorine which could result in extended disinfection. Serial dilutions of the quenched swatches were made and plated on Trypticase agar, and the agar plates were incubated for 24 h at 37℃. Antimicrobial efficacy was determined and evaluated by the colony counts [26].
Washing testing
AATCC Test Method 61-1996 was used to evaluate the durability of the LbL assembly cotton fabrics and the stability of chlorine on the swatches after repeated standard washings. Stainless steel canisters containing 0.15% AATCC detergent in 150 ml of water and 50 stainless steel balls were fixed in a Launder-Ometer (Darong Textile Instrument Co., Ltd, Zhejiang, China) and rotated at 42 r/min and 49℃ for 45 min. The swatches were rinsed several times with distilled water and then dried after washing test. The Cl+% loadings of swatches before and after washing were measured by the iodometric/thiosulfate titration.
Storage stability testing
The storage stability of the covalently bound chlorines in the Q-CS-HDH on the surface of swatches was evaluated by determining the remaining chlorine on the swatches after different storage times. The swatches were tested periodically over 5, 10, 15, 20, 25, and 30 days at room temperature in dark environment. After the storage time, the swatches were taken out and the active chlorine contents were tested by the iodometric/thiosulfate titration procedure as mentioned above.
Mechanical properties measurement
Tensile strength of swatches was evaluated by GB/T3923-1998 standard. The swatches were prepared and measured under the same condition, and the data were acquired by averaging five experimental values.
Results and discussion
Characterization of surface modification of cotton fabrics via the LbL technique
The FTIR spectra of the uncoated and LbL-coated cotton fabrics are shown in Figure 2. Compared with the uncoated cotton, the FTIR spectra of treated cotton fabrics with five layers through LbL technique present a new absorption peak at 1551 cm−1, which is attributed to the amide II region of Q-CS-HDH and PAMPAS [22]. The peak at 1713 cm−1 of the swatches was assigned to the existence of carboxyl stretching mode of hydantoin ring. Additionally, the intensity of bands at 1551 and 1713 cm−1 increased with the growth of LbL deposition number. The results suggest that a thin layer deposition was formed upon each dipping process by LbL technique successfully.
FTIR spectra of (a) cotton fabrics, (b) treated cotton fabrics with three layer depositions and (c) treated cotton fabrics with five layer depositions.
The surface morphology is one of significant parameters for coating fibers, which can provide further information and more detail of coated fibers’ surfaces. SEM and AFM observation of cotton fabrics and treated cotton fabrics are carried out and shown in Figure 3. It was observed that the pristine fibers exhibited a relative smooth 2D surface. As shown in Figure 3(b), there were a large amount of substance aggregated on the surface of cotton fibers using LbL technology. The AFM images clearly illustrated the results. The pristine fibers (c) were in a flat form of the surface morphology, but instead were observed after coating that the surface morphology of the multilayer-assembled fibers became uneven and rougher, which was coincident with the visibility on the SEM images. The roughness of five-layer films on the surface of cotton fibers measured by AFM was 21 nm. The thickness of five-layer films obtained by AFM was 195 nm. The values for film thickness were related to the layer numbers, and thickness increased with increasing layer numbers of assembly.
SEM images of (a) cotton fabrics × 5000 and (b) coated cotton fabrics × 5000 and AFM images of (c) cotton fabrics and (d) coated cotton fabrics.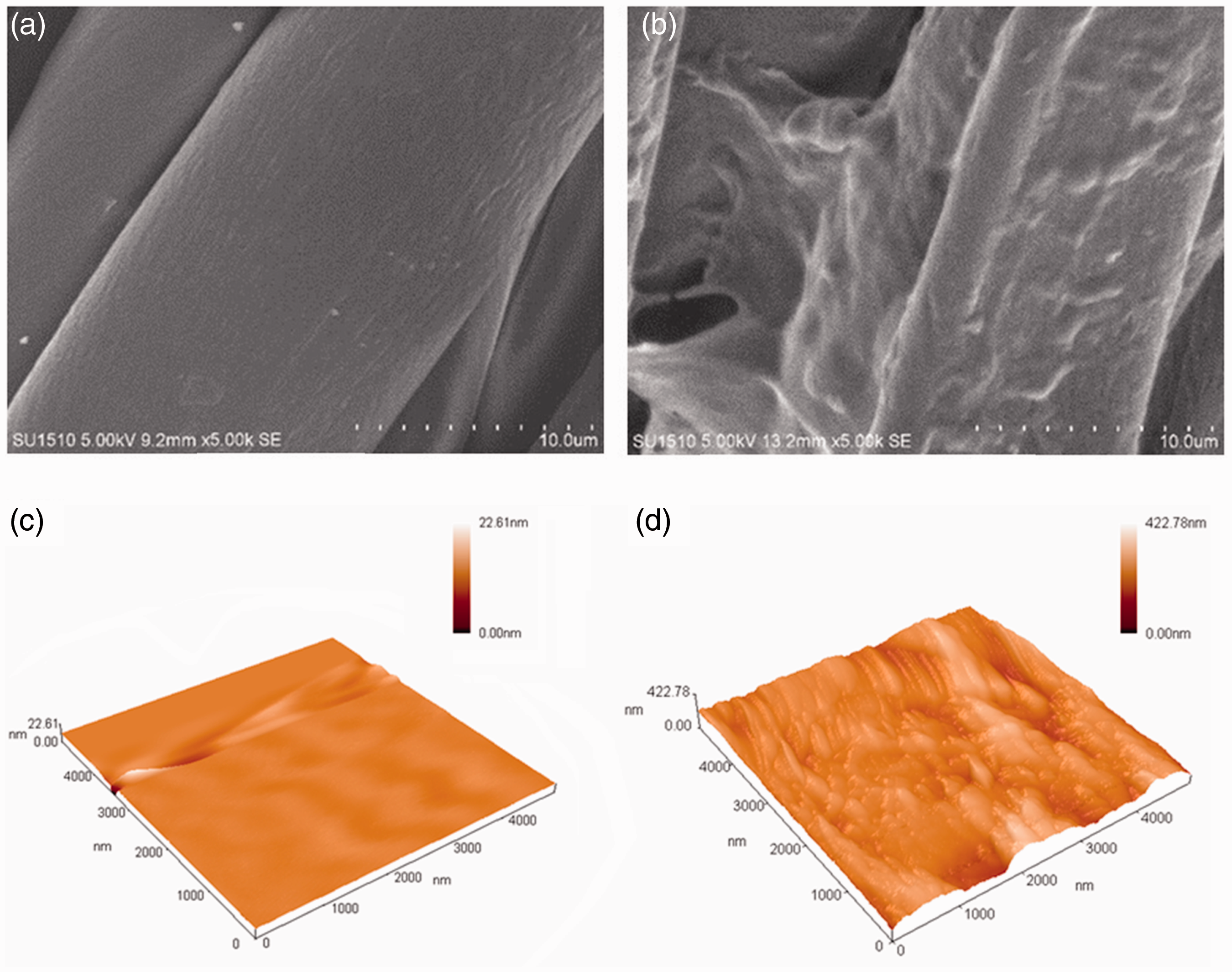
Antimicrobial property
The results of tests that swatches were challenged with S. aureus and E. coli O157:H7 bacteria are presented in Figure 4. Although the original cotton fabrics performed a small degree inactive property from the antimicrobial data, actually the fabrics did not kill the bacteria. The adhesion of bacteria to fabrics may make for the reduction of the bacteria to a certain extent [25, 27]. The cotton fabric treated by Q-CS-HDH and unchlorated cotton fabrics treated by LbL technology led to a little better antimicrobial property than the original cotton fabrics, and the results may be attributed to the quaternized groups. Quaternized chitosan derivative shows polycationic, porous, and absorbent properties, and bind microorganisms to their cell membrane and disrupt the structure resulting in the breakdown of the cell [28]. But the antimicrobial effect was not strong enough and the swatches did not kill all the bacteria which were inoculated to swatches. The cotton fabrics treated by Q-CS-HDH can completely inactivate the inoculated S. aureus and E. coli within contact time of 1 min by the chlorinated swatches with chlorine loadings of 0.25%. N-halamine groups of modified chitosan played a critical role in the excellent antibacterial activity, and the active chlorine can kill bacteria quickly and effectively.
Antibacterial property against S. aureus and E. coli O157:H7 (Inoculum population of S. aureus was 1.10 × 106 CFU/swatch, and inoculum population of E. coli O157:H7 was 1.87 × 106 CFU/swatch. Cotton-0.5 was treated by Q-CS-HDH one time. Coated cotton was treated by Q-CS-HDH/PAMPA using LbL technology and the thick multilayered films were five deposited layers.)
Washing stability
Washing stability of Q-CS-HDH/PAMPAS LbL-coated cotton fabrics. a

Conditions: A: chlorinated before washing; B: chlorinated before washing and rechlorinated after washing; C: unchlorinated before washing but chlorinated after washing.
The error in the measured Cl+% values was ± 0.01.
Storage stability
The storage stability of the covalently bound chlorines in the oppositely charged polyanions and polycations on the surface of cotton fabrics is shown in Figure 5. The chlorine content of swatches decreased gradually with the increase of storage time. The chlorine content reduces from 0.32 to 0.15% after one month of storage. The remaining chlorines on the swatches are sufficient to perform good antimicrobial activity [30]. Due to the hydrophilic nature of chitosan which absorb moistures from the surrounding environment, the absorbed water might promote the dissociation of N-halamines and lead to the decrease of chlorine content [31].
Storage stability of the chlorine on the treated cotton fabrics.
Mechanical property
The results of Q-CS-HDH/PAMPAS LbL-coated cotton fabrics surfaces as the outside layer are presented in Figure 6. It was noted that the tensile strength of the assembled fabrics gradually and slowly reduced with the increasing of surface layers compared with the pristine cotton fabrics. The self-assembly and chlorination process only caused a small degree of tensile strength loss. It indicated that the application of LbL technology in the treatment of cotton fabrics played only a little influence on the tensile strength for cotton fabrics. LbL self-assembly involves the sequential adsorption of oppositely charged polyanions and polycations, and creates a multilayer film on the surface of cotton fabrics. The method did not break the hydrogen bonds between cellulose intermoleculars which happens to chemical cross-linking [32]. The cotton fabrics treated by Q-CS-HDH via LbL technology can keep their original tensile strength and also be equipped with excellent antibacterial property.
The tensile strengths of (C-0) uncoated and (C-1, C-2, and C-3 are expressed as means + five layers, eight layers, and 10 layers, respectively) LbL-coated cotton fabrics. The schematic description of synthesis of Q-CS-HDH [13]. The schematic description of synthesis of PAMPAS [22].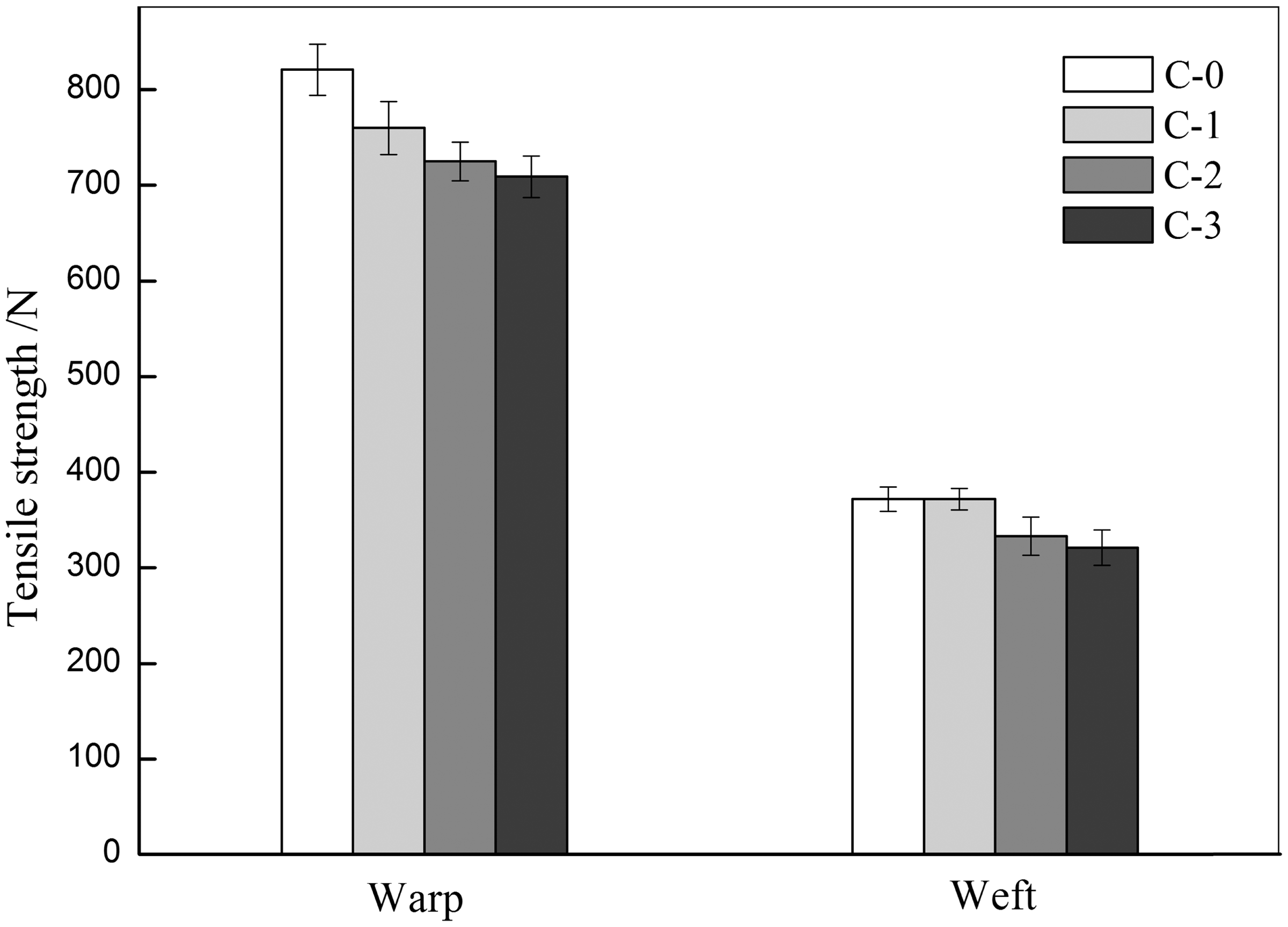


Conclusion
Q-CS-HDH/PAMPA films coated on cotton fabrics using LbL technology were successfully produced. The thick multilayered films via five deposited layers with polyelectrolytes on the coated swatches surfaces can be observed by SEM and AFM micrographs analysis. The coated cotton fabrics possess potent antimicrobial effects exposure to dilute household bleach. The chlorinated coated swatches can inactivate 100% of S. aureus and E. coli O157:H7 bacteria with a contact time of 1 min, respectively. It can be observed that the amount of chlorine remaining on the coated cotton was 0.07% after 25 washing cycles, which was still sufficient for an effective biocidal activity. The durability of LbL films in the air has revealed a reasonable result during the storage. The oxidative chlorine on the swatches reduced about 50% after one month of storage, but the remaining was still effective in inactivating bacteria, and most of the lost chlorine could be regenerated after rechlorination. LbL technology was introduced with adsorption of oppositely charged polyelectrolytes without much damage for tensile strength of swatches.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial support was provided by the Project for Jiangsu Scientific and Technological Innovation Team, the Graduate Student Innovation Plan of Jiangsu Province of China (KYLX-1141), the research fund from the Science and Technology Department of Jiangsu Province of China (BY2014023-09), the National Thousand Young Talents Program, and the Scientific Research Foundation for Returned Overseas Chinese Scholars, Ministry of Education, China.
