Abstract
Duck and chicken feather fibers are waste products of the poultry industry, creating a serious solid waste problem around the world. Previous works showed that feather fibers can be reused to adsorb heavy metal ions from water. But the raw feather fiber only exhibits moderate heavy metal ions adsorption capacity, not cost-effective to be reused as adsorbent in a large scale. To improve the adsorption capacity of feather fibers, sodium pyrosulfite (Na2S2O5) was used in this paper to modify the feather fiber in order to improve its Pb2+ adsorption capacity. Scanning electron microscopy and Fourier transform infrared spectroscopy quantified chemical and structural changes of Na2S2O5-modified feather fibers. In addition, Na2S2O5-modified feather fibers were processed into feather/polypropylene melt-blown filter cartridges and their dynamic Pb2+ adsorption properties were investigated by using the test equipment set up in our lab. Finally, the desorption effects of NaOH and sodium sulfide nonahydrate (Na2S·9H2O) aqueous solutions on the feather fiber adsorbed with Pb2+ were studied. It is found that Pb2+ adsorption capacity of the feather fiber increased after being modified by Na2S2O5. Modified feather/polypropylene filter cartridge exhibited higher Pb2+ adsorption capacity than feather/polypropylene filter cartridge and pure polypropylene filter cartridge in the whole dynamic adsorption process.
Introduction
The wastewater generated from many industries, such as tanning, battery, glassware, ceramics, electroplating, mining, paints and photographic industry, contains a number of heavy metal ions like chromium, lead, cadmium, etc. which have significant toxic effects and these pollutants requires removal before discharge into the main stream [1]. Nowadays people have realized that heavy metal ions remaining in the drinking water are harmful and toxic to human health. Long-term drinking of Pb2+ ion-containing water, even at very low concentrations, could lead to a broad spectrum of health problems, including nausea, convulsions, coma, renal failure, cancer and subtle effects on metabolism and intelligence [2].
Different approaches to remove Pb2+ ions from wastewaters, including chemical precipitation, ion exchange, reverse osmosis and adsorption, have been reported. Among them, adsorption is one of the most important methods because of its easy operation and versatility [3]. Various adsorbents have been explored to remove heavy metal ions from wastewaters. Biosorption material, a kind of low-cost adsorbent with excellent sorption capacity, is gaining more and more attention. Peat, sugar cane bagasse, eucalyptus bark and hen feather have been implemented for the removal of specific metals from wastewaters [4,5]. However, the feasibility of biosorption versus traditional methods has been questioned on the basis of a series of limitations: the relatively low metal uptake, the high brittleness of the biomass and the inherent poor reproducibility. Compared to other biomass sorbents, the feather fiber comprises excellent mechanical properties, resists hydrolytic degradation and is durable. It is said that the feather fiber is constituted with four parts from outside to inward, which are named as epicuticle film, cuticle layer, skin layer and cortex, of which the epicuticle film is a layer of biologic cell membrane with thickness approximately 0.02–0.10 µm. The biologic cell membrane is mainly made of sterol and bimolecular layers of triphosphate ester, and these chemicals are responsible for the excellent water repellant properties of feather fibers [6]. But as a biomass sorbent, the hydrophobic properties of the feather fiber are a serious drawback in the processing of aqueous metal ion solutions. The raw feather fiber only exhibits moderate heavy metal ions absorption capacity, not cost-effective to be reused as adsorbent in a large scale.
Recently, much research has focused on the sorption properties of feather fibers as sorbents for the removal of heavy metals, phenol and organic dyes from their aqueous solutions [7–11]. Banat et al. compared the sorption capacities of some keratin-based compounds, including the feather fiber, for the removal of Zn2+ ions from aqueous solutions in a batch configuration and using different conditions of pH and temperature [12]. The research on Zn2+ adsorption properties of feather fiber before and chemical treatment showed that the feather fiber treated with alkaline aqueous solutions absorbed more metal ions than those treated with an anionic surfactant and untreated feather fiber [13]. Many research works have been carried by chemical grafting, alkaline treatment, powdering, dissolution, etc. to improve the adsorption capacity of the feather fiber [14]. Keratin mainly comprises amino and carboxyl groups, has a good biocompatibility, nontoxic and skin-friendly properties present in wool and feather fiber. These fibers are generally used to extract keratin. Reduction is the main method of dissolving wool, where the disulfide bonds of keratin are cleaved [15]. Sodium pyrosulfite (Na2S2O5) aqueous solution, one of the important reducing reagents, is widely used to dissolve wool or remove the scale of wool. Wang and Liu [16] inferred that it can be used to modify the feather fiber for improving its Pb2+ adsorption capacity.
In another study, Kar and Misra reported the removal of Zn2+ using the feather fiber in a multicomponent aqueous solution of Cu2+, Pb2+, Zn2+, Cd2+ and Ni2+ at different pH values [17]. Most of the above-mentioned studies on the removal of Zn2+ ions using the feather fiber have been focused on sorption experiments using batch configurations. Aguayo-Villarreal et al. first studied the batch and column Zn2+ removal from aqueous solution using chicken feather fibers as sorbents [18]. Duck feather was processed into a kind of composite made from nonwoven fabric with bicomponent fiber, which showed good mechanical properties and absorption capacities of Cu2+ and Cr6+ bicomponent aqueous solution [14]. As we know, melt-blown filter cartridge is widely used nowadays to clean water, filtering out insoluble impurities. It is significant to endow it with heavy metal ions adsorption property at the same time. In this paper, feather/polypropylene (PP) melt-blown filter cartridges were prepared and their dynamic Pb2+ adsorption properties were investigated as well.
In addition, recovering the metal biosorbed or reusing the biomass is one of the essential issues to scale up biosorption technology. This work will examine the effects of sodium hydroxide (NaOH) and sodium sulfide nonahydrate (Na2S·9H2O) as eluents to regenerate the feather fiber adsorbed with Pb2+.
Experimental
Materials
The duck feather fiber without scapus was supplied by Yangzhou Ganquan Down Products Factory. PP with melt flow index 35 was purchased from Sinopec Shanghai Petrochemical Company Ltd. Lead nitrate (Pb(NO3)2) was used to prepare Pb2+ solution. NaOH and Na2S·9H2O were used as desorption reagents. Na2S2O5 and urea were used to modify feather fibers. Nitric acid (HNO3) was used in the inductively coupled plasma atomic emission spectrometry (ICP-AES) measurement. Ammonia aqueous solution (NH3·H2O) was used to adjust the pH value of solutions. All chemicals are all analytical grade.
Modification of the feather fiber
A total of 1, 3, 5, 10 and 15 g Na2S2O5 were added into 200 ml de-ionized water under 65℃, respectively. Twenty grams of urea was added to every solution as well. Ten grams of feather fiber was put into the above solutions, respectively. The solutions were stirred for 5 min and then were allowed to stand for 2 h. Finally, the solutions were filtered and feather fibers were washed thoroughly with de-ionized water and air-dried at room temperature for further use.
Preparation of feather/PP melt-blown filter cartridges
Feather/PP melt-blown filter cartridges were made by fixing an extra feather fiber feeding unit on a traditional melt-blown nonwoven production machine, as shown in Figure 1. The feather fiber was blown out by a fan uniformly from the unit and mixed with PP melt-blown nonwoven fibers spun from the spinneret of the melt-blown equipment and reached a pivot with a core, which rotated and moved back and forth to form the cylindrical shape of a filter cartridge. In this paper, three kinds of filter cartridges were made, i.e. feather/PP melt-blown filter cartridge, modified feather/PP melt-blown filter cartridge and pure PP melt-blown filter cartridge. Their structural properties were listed in Table 1. Photo of the feather /PP melt-blown filter cartridge was shown in Figure 2. Feather/PP and modified feather/PP melt-blown filter cartridges are constituted with multilayers. The inner layer is composed of pure PP melt-blown nonwoven fibers, the middle layer is composed of the feather fiber and PP melt-blown nonwoven fiber in a ratio listed in Table 1 and the outer layer is composed of pure PP melt-blown nonwoven fibers to endow the whole filter cartridge with high physical and mechanical properties. The inside diameter of the cartridge is 30 mm and the length of the cartridge is 250 mm.
Schematic diagram of the production process of feather/PP melt-blown filter cartridge. Properties of feather/PP melt-blown filter cartridges. Content of feather fibers is the ratio of the weight of feather fibers to the weight of the filter cartridge expressed as a fraction of 100. Photo of the feather/PP melt-blown filter cartridge.


Adsorption experiment of the feather fiber
Two grams of duck feather fiber was immersed in 250 ml Pb(NO3)2 aqueous solution containing 100 mg/l Pb2+ under 25℃. The solution was stirred for 5 min and then was allowed to stand for 1 h. Finally, the solution was filtered and the residual Pb2+ concentration of the filtrate was measured by using ICP-AES. The Pb2+ adsorption capacity of the feather fiber is calculated according to equation (1)

Dynamic Pb2+ adsorption measurement of filter cartridges
Most studies on the removal of heavy metal ions using the feather fiber have been focused on sorption experiments using batch configurations. In this paper, melt-blown filter cartridge was prepared and dynamic Pb2+ adsorption test equipment was set up by us. The melt-blown filter cartridge was fixed by hot melt adhesive on both ends with two end caps, one of which has hole in the center as shown in Figure 3(b). The end of the cartridge with hole was inserted into the support post of the filter cartridge vessel, as shown in Figure 3(c).
Photos of installation of the melt-blown filter cartridge. (a) Filter cartridge, (b) filter cartridge with caps on both ends and (c) filter cartridge vessel.
Dynamic Pb2+ adsorption equipment was set up in our lab as shown in Figure 4, which is constituted with pressure gauge, flow meter, filter cartridge and pump. Pb(NO3)2 aqueous solution with concentration of about 0.3 mol/l was sucked from the reservoir by the pump. It flowed through the valve, pressure gauge 1 and flow meter 1, and then went into the filter cartridge from the outer layer into the inner layer and then flowed out from the hole under the filter cartridge. The solution went through pressure gauge 2 and flow meter 2 and went back to the reservoir, and then repeated in this way. A small amount of the solution was taken from the reservoir every 30 min and Pb2+ concentration was measured by using ICP-AES.
Schematic figure of the dynamic adsorption testing equipment.
Desorption experiment
Duck feather fibers adsorbed with Pb2+ from the above Pb2+ adsorption experiment were washed thoroughly with de-ionized water and air-dried at room temperature. Then they were immersed in NaOH and Na2S·9H2O aqueous solutions with different concentration under 40℃ for 1 h, respectively. Finally, the solutions were filtered and Pb2+ concentration of the residual solutions was measured by using ICP-AES. The desorption capacity of the feather fiber under different solutions is calculated according to equation (2)

Duck feather fiber after NaOH desorption experiment was used to absorb Pb2+ according to the above procedure of adsorption experiment. Its Pb2+ absorption capacity was measured by the same method. The adsorption and desorption experiment was repeated for 12 times.
Measurement
ICP-AES
ICP-AES was used to measure Pb2+ concentration in the solution. Five per cent HNO3 was used as the media.
Scanning electronic microscopy (SEM)
Surface of feather fibers before and after Na2S2O5 modification was observed by using Japan JSM-5600LV SEM. The surface of samples was spurred with gold and observed under 10 kV.
Fourier transform infrared (FTIR) spectroscopy analysis
Duck feather fiber was mixed and grounded with KBr and made into a pellet. Then it was tested with Nicollet NEXUS670 infrared-Raman spectroscopy tester. The wavenumber was 500–4000 cm−1, the resolution was 8 cm−1 and the scan number was 64.
Results and discussion
Influences of Na2S2O5 treatment on Pb2+ adsorption of the feather fiber
Urea is a kind of protein denaturant. It is often used together with Na2S2O5 to modify protein [19]. In this paper, the feather fiber was treated by Na2S2O5 under different concentrations, while the concentration of urea remained unchanged. The Pb2+ adsorption capacity of modified feather fibers was shown in Figure 5.
Influences of Na2S2O5 treatment on Pb2+ adsorption of the feather fiber.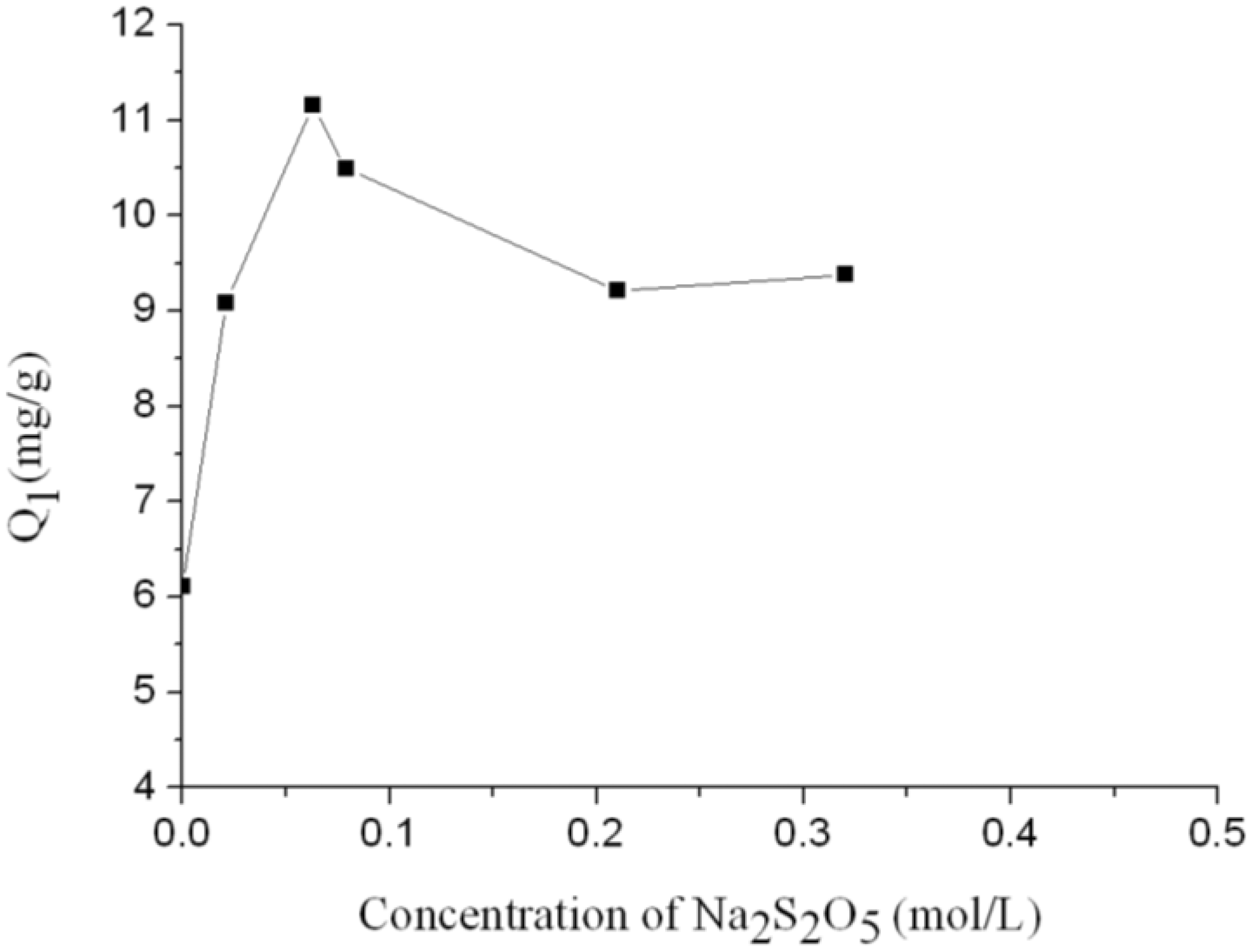
It can be seen from Figure 5 that Pb2+ adsorption capacity of the feather fiber increased with increase in the concentration of Na2S2O5 aqueous solution and reached the highest value when the concentration of Na2S2O5 aqueous solution was 0.079 mol/l. The Pb2+ adsorption capacity of raw feather fiber was 6.11 mg/g, while the highest Pb2+ adsorption capacity of modified feather fiber was 11.16 mg/g, over 80% higher than that of the raw feather fiber. Then the Pb2+ adsorption capacity of the feather fiber decreased a little and kept unchanged with the increase of Na2S2O5 aqueous solution concentration.
Structural changes of the feather fiber before and after Na2S2O5 modification
In order to uncover the reason why Pb2+ adsorption capacity of the feather fiber was improved after Na2S2O5 modification, surface structure and secondary morphology of the feather fiber before and after Na2S2O5 modification were investigated by SEM and FTIR and the results were shown in Figures 6 to 8, respectively.
SEM picture of the raw feather fiber. (a) Sub-branch of the feather fiber, (b) fibril of the sub-branch. SEM picture of the feather fiber after Na2S2O5 modification. (a) Sub-branch of the feather fiber, (b) fibril of the sub-branch. FTIR spectra of the feather fiber before and after Na2S2O5 modification. A – raw feather fiber; B – feather fiber after Na2S2O5modification.


It can be seen from Figure 6 that the sub-branch of the feather fiber carried a number of fibrils, which showed knars or pinaculums around the surface. Rough grooves appeared on the surface of the sub-branches of the feather fiber. After Na2S2O5 modification, the surface of sub-branches of the feather fiber became smooth and the feather fiber became slimmer. It is hypothesized that keratin fragments scaled off from the feather fiber surface layer by layer when it was treated by Na2S2O5 aqueous solution, which was also observed by Sun when the feather fiber was treated by NaOH solution [20].
Difference in peak intensity of α and β structures of modified feather fibers.

Y1630 and Y1540 represent the infrared transmittance of the bands at 1630 and 1540 cm−1, respectively.
It can be found from Table 2 that the difference in the band intensity of the two characteristic bands became more noticeable for the feather fiber after Na2S2O5 modification. In other words, it seems that more α-helix structure appeared after the feather fiber was modified by Na2S2O5. Compared to β-sheet, α-helix is more loose structure of the feather proteins, in which more hydrogen and/or disulfide bonds of feather keratin were broken during Na2S2O5 modification. Hence more adsorptive sites are available for Pb2+ and the adsorption capacity of the feather fiber was improved after Na2S2O5 treatment.
In addition, it is reported that the bands at 1079 and 1117 cm−1 arise from specific stretching and bending vibrations of S–O bond of cysteine, resulting from the broken disulfide bond of cysteine [22]. However, the spectra of feather fibers with and without Na2S2O5 modification were identical in the frequency range, suggesting that Na2S2O5 modification did not change the chemical structure of feather fibers to a detectable extent by FTIR spectroscopy. Perhaps Na2S2O5 modification only changed hydrogen bonds of feather keratin. More hydrogen bonds of feather keratin were broken with the increase in the concentration of Na2S2O5 aqueous solution. However, only a fraction of hydrogen bonds of feather keratin can be broken by Na2S2O5 aqueous solutions because of the constraint of disulfide bond. Hence it can be found from Figure 5 that Pb2+ adsorption capacity of the feather fiber increased with increase in the concentration of Na2S2O5 aqueous solution and reached the highest value when the concentration of Na2S2O5 aqueous solution was 0.079 mol/l.
Desorption effects of Na2S·9H2O and NaOH solutions on the feather fiber
Desorption properties of NaOH and Na2S·9H2O solutions on feather fibers.

It can be seen from Table 3 that Na2S·9H2O showed quite low Pb2+ desorption capacity and its desorption effect did not change obviously with the increase of the concentration of desorbent solutions. Only about 1.3% Pb2+ of the feather fiber was desorbed by Na2S·9H2O. The desorption capacity of NaOH is 24–31% of the adsorption capacity of raw feather fibers, which is 6.11 mg/g as shown in Figure 5. It is hypothesized that Pb(OH)2 and PbS would form, when the Pb2 + adsorbed-feather fiber was immersed in NaOH and Na2S·9H2O aqueous solution, respectively. The solubility product constant of Pb(OH)2 is 1.43 × 10−20, which is much higher than that of PbS (3.4 × 10−28). Hence more PbS deposit should be formed. However, it was found that the feather fiber changed color obviously and became nearly broken up when the concentration of Na2S·9H2O aqueous solution was as high as 0.03 mol/l, suggesting that Na2S·9H2O preferentially involved in the reaction with keratin of the feather fiber rather than with Pb2+ ion. Hence NaOH showed better desorption effect than Na2S·9H2O. In addition, the desorption capacity of NaOH did not change obviously when its concentration was increased to 0.05 mol/l. It is reported that complex ions, such as [Pb(OH)4]2−, Pb4(OH)44+, Pb3(OH)42+ and Pb6O(OH)64+ will be formed when the pH value is higher than 9. Pb(OH)2 deposit will be re-dissolved correspondingly.
In order to further investigate the desorption effect of NaOH, duck feather fibers were treated by adsorption and desorption experiment for 12 times. The Pb2+ absorption capacity of feather fibers after each desorption cycle was shown in Figure 9.
Repeated Pb2+ absorption capacity of feather fibers.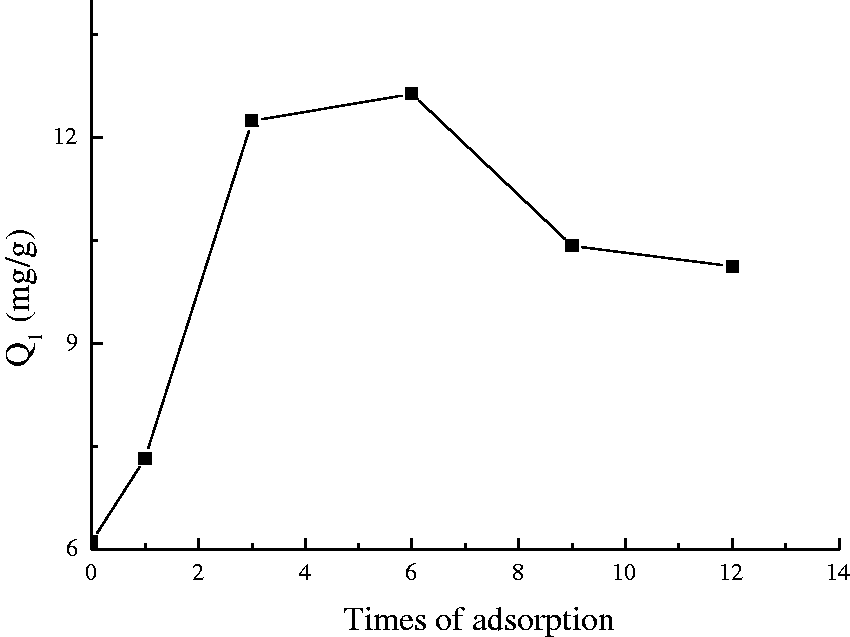
It can be found from Figure 9 that Pb2+ absorption capacity of the NaOH-treated feather fibers increased with the increase of the desorption cycles, suggesting that NaOH is an available desorbent reagent to reuse feather fibers as an Pb2+ absorbent.
Dynamic Pb2+adsorption properties of feather/PP melt-blown filter cartridges
In this paper, the feather fibers with and without Na2S2O5 modification were made into feather/PP and modified feather/PP melt-blown filter cartridge according to the patent titled a kind of feather/PP composite melt-blown filter cartridge and its process [27]. It is found that the feather fiber could be easily bonded to each other by PP melt-blown nonwoven fibers and the filter cartridges had good physical and mechanical properties. In addition, the dynamic Pb2+ adsorption properties of the three kinds of filter cartridges as listed in Table 1 were measured by using the test equipment set up in our lab as shown in Figure 10.
Dynamic adsorption properties of three kinds of filter cartridges.
It is found from Figure 10 that modified feather/PP filter cartridge had higher Pb2+ adsorption capacity than feather/PP filter cartridge and pure PP filter cartridge in the whole dynamic adsorption process measured in this paper. In addition, the Pb2+ adsorption capacity of pure PP filter cartridge was a little lower than that of feather/PP filter cartridge. As shown above, the Pb2+ uptake capacity of raw feather fibers is about 6.11 mg/g. It is reported that the Pb2+ adsorption capacity of PP strip is about 0.1 mg/g [28], which is much lower than that of feather fibers. It is believed that PP fibers of these two kinds of filter cartridges have higher specific surface area, resulting in higher Pb2+ uptake capacity than that of PP strip. When modified feather/PP filter cartridge was used as the filter cartridge, Pb2+ concentration in the solution decreased faster in the first hour in comparison with the other two filter cartridges, suggesting that modified feather/PP filter cartridge had faster Pb2+ adsorption speed in the initial dynamic Pb2+ adsorption process. In the third hour of the dynamic adsorption process, the residual Pb2+ in the solution was about 0.09 mol/l. Based on the assumption that melt-blown filter cartridge is widely used as liquid filtration material, modified feather/PP melt-blown filter cartridge is a promising material, exhibiting filtration and Pb2+ adsorption functions at the same time.
Conclusion
In this paper, Na2S2O5 was first used to modify the feather fiber in order to improve its Pb2+ adsorption capacity. SEM and FTIR were used to investigate the structural changes of the feather fiber before and after modification. In addition, the desorption effects of NaOH and Na2S·9H2O aqueous solutions on the feather fiber were studied. Finally, feather fibers were processed into feather/PP melt-blown filter cartridges and their dynamic Pb2+ adsorption properties were investigated by using the test equipment set up in our lab. First, it is concluded that Pb2+ adsorption capacity of the feather fiber increases after being modified by Na2S2O5. The highest Pb2+ adsorption capacity of the modified feather fiber is over 80% higher than that of the raw feather fiber. Second, NaOH is an effective desorbent agent for the Pb2+ loaded feather fibers; Third, Na2S·9H2O did not show expected Pb2+ desorption effect. It involved in the reaction with feather keratin rather than with Pb2+ during the desorption experiment. Finally, modified feather/PP filter cartridge has higher Pb2+ adsorption capacity than feather/PP filter cartridge and pure PP filter cartridge in the whole dynamic adsorption process.
Na2S2O5-treated feather/PP melt-blown filter cartridge is a kind of promising material for wastewater effluent treatment. For example, it can be used to remove Pb2+ existing in the wastewater yielded in the course of mechanical crushing and hydraulic sorting for waste lead acid batteries recycling. Compared to other processes, the filter cartridge can filter the insoluble particles in the wastewaters while keeping the Pb2+ adsorption property. In addition, feather fibers are waste products of the poultry industry. Na2S2O5-treated feather/PP melt-blown filter cartridge can be prepared easily by modified commercial melt-blown equipment. But more work should be done to test the long-term filtration performance and the desorption properties of the Na2S2O5-treated feather/PP melt-blown filter cartridge prepared in this paper.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
The authors gratefully acknowledge the financial support from the Shanghai Municipal Natural Science Foundation (11ZR1400900) and Shanghai University Students’ Innovative Entrepreneurship Training Project ‘research and development of waste feather fiber melt-blown filter cartridge’.
