Abstract
Permeability improvement of coal seams by hydraulic fracturing can promote desorption of coalbed methane. However, the water-block effect will restrain desorption of coalbed methane. Based on the essence of the physical adsorption of methane, competitive adsorption, substitution desorption, the capillary effect, and the Jamin effect are analyzed to study the effect of the critical moisture content and liquid water on methane adsorption and desorption. Simultaneously, the effects of the fluid pressure, fracturing fluid, fracturing time, pore pressure, and other variables on methane adsorption and desorption characteristics are analyzed. Then, problems that require further study are described.
Keywords
Introduction
Hydraulic fracturing technology currently plays an important role in the exploitation of coalbed methane (CBM). Hydraulic fracturing in coal-rock mass can transform a reservoir with low permeability, low reservoir pressure, and a low gas saturation level, thereby improving the production of CBM wells. Moreover, hydraulic fracturing can also be conducted to control coal mine gas and prevent coal–gas outbursts (Li et al., 2015a, 2015b; Yan et al., 2015; Yuan et al., 2015a, 2015b).
In hydraulic fracturing, the original joints, beddings, cleavages, and micro-fractures in the coal-rock mass are connected with the propagating hydraulic fractures, resulting in a fracture network. Within the fracture network, coal bed permeability is improved and desorption and migration of CBM are accelerated (Huang et al., 2012; Xu et al., 2011). Meanwhile, the flowing fracturing fluid in the pores and fractures will generate water pressure, resulting in gas displacement and competitive adsorption (Gensterblum et al., 2014; Huang et al., 2016; Ottiger et al., 2008). However, the fracturing fluid cannot be completely recycled due to the fluid loss caused by the original pores and fractures in the coal-rock mass. Additionally, the fracturing fluid will be imbibed into the pores and fractures under the effect of capillary pressure. As a result, the gas migration passages are blocked (which is called the water-block effect) and gas desorption is restrained (Bahrami et al., 2012; Kamath and Laroche, 2003).
The fracturing process will increase the water content of coal and change the saturation of coal gas and water. A critical moisture content of coal exists, under which gas adsorption is inversely proportional to the water content (Day et al., 2008; Joubert et al., 1973; Krooss et al., 2002; Zhang et al., 2014a; Zhao et al., 2011). In hydraulic fracturing, the higher the fracture pressure, the smaller the pore-size range (Gensterblum et al., 2013; Le et al., 2012; Qin and Zhang, 2000). The water-blocking mechanism, which is caused by residual fracturing fluid around the fracturing well or borehole, and the methods to eliminate water blocking have long been debated issues (Ahmadi et al., 2011; Bazin et al., 2013; Ni et al., 2016). It is widely recognized by researchers that the water-blocking effect can be relieved or eliminated by optimizing fracturing fluids with, for example, an alcohol and acid mixture, perfluorooctane sulfonamide surfactant solution, clean fracturing liquid, or similar (Chen et al., 2009; Fink, 2013; Khair et al., 2011).
Recent research and development on the effects of hydraulic fracturing on the adsorption and desorption of CBM are summarized in this paper. It is expected that this study will provide instructions for the comprehensive control of gas in high gas mines and coal–gas outburst mines, field tests of hydraulic fracturing, and optimization of CBM extraction by hydraulic fracturing.
Coal structure and the essence of CBM adsorption
Coal structure
Coal seams are double-porosity media that consist of a matrix pore system and a fracture system. The distribution of matrix pores is uneven, and the connecting type of various pores is different. Two kinds of pore space exist in coal, i.e., the effective pore space and isolated pore space. The former can act as a passage through which fluid can flow in coal, while the latter is completely closed and is referred to as closed pores (or dead pores; Rouquerol et al., 1999). Furthermore, effective pores include open pores and half-closed pores, and half-closed pores consist of cross-linked pores and blind pores.
CBM storage is closely related to the pore size. In the field of CBM exploitation and coal mine gas control, the decimal classification method of pore size, proposed by BHODB, has been widely used. Based on the above method, pore sizes are divided into micro-pores (<10 nm in diameter), transitional pores (10–100 nm in diameter), meso-pores (100–1000 nm in diameter), and macro-pores (>1000 nm in diameter), and the surface of the first three kinds of pores are the main places where gas is adsorbed (BHODB, 1996).
Methane adsorption theory and relevant models
The gas-retention capacity of a prepared coal sample can be estimated using methods that directly measure the volume of gas released from a coal sample into a sealed desorption canister or by indirect methods based on adsorption/desorption isotherms obtained in the laboratory (Diamond and Schatzel, 1998; Mazzotti et al., 2009). To model gas adsorption behavior, a large collection of adsorption models has been proposed by previous researchers to model adsorption isotherms, such as Henry’s model, Langmuir’s model, Brunauer emmett teller (BET) model, and the pore-filling model (Mukherjee and Misra, 2018; Tang et al., 2017b; Wang and Tang, 2018).
Langmuir model
On the basis of the monolayer adsorption assumption, the Langmuir model is the most widely used model because of its simplicity, effectiveness, and the reasonable explanation of its parameters (Tang et al., 2016). The Langmuir’s model can be shown in the following form:
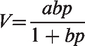
BET model
The BET model accounts for multilayer adsorbed molecules of subcritical methane in the surface of the adsorbent. The model is developed for interpreting type II isotherms and the reversible part of type IV isotherms. The BET model has the following form:

Pore-filling model
The pore-filling model is based on the Polanyi adsorption potential theory, which assumes that the attraction of the gas to the surface is largely due to van der Waals forces, which can be determined by the position of the gas particle from the surface (Dubinin, 1960). This model has been widely applied to characterize micropore-rich materials such as activated carbon. The form of the Pore-filling model is as follows:

where
Essence of CBM adsorption
Adsorption can be divided into physical and chemical adsorption. CBM adsorption is a CBM gas–solid physical process in which coal is the adsorbent and gas is the adsorbate.
From the perspective of molecular thermodynamics and surface chemical physics, methane adsorption results from the mutual attraction between the coal surface and gas molecules. Gas molecules stay on the surface of coal due to the Van der Waals forces between the gas and coal molecules. The larger the Van der Waals force, the more gas molecules are on the coal surface (Mosher et al., 2013; Zhang and Sang, 2009).
If the coal matrix is considered to be an organic solid consisting of carbon atoms, the inner carbon atoms exist in a force-balanced situation due to the surrounding carbon atoms. However, for surface atoms, at least one side of the atom is vacant. As a result, the atomic force is not in balance. Under the attraction of the inner atomic force, surface atoms tend to move toward the interior of the coal matrix. As a result, extra energies due to the surface atoms are generated, called surface energies (Wu, 1994; Yang and Xing, 2010; Zhang et al., 2014b), as illustrated in Figure 1. Therefore, from the energetic perspective, the essence of CBM adsorption is caused by the excessive free energy of the coal surface, which attracts CBM molecules to the surface. Afterwards, the free energy of the coal surface is decreased.

Schematic diagram of the distribution of the coal matrix excessive free surface energy. (a) CBM molecule; (b) surface carbon atoms; and (c) inner carbon atoms.
The states of CBM storage are determined by the physical adsorption essence of coal and gas. More than 80–90% of the total gas content in a coal seam is adsorbed gas, which exists within the coal matrix pores and fractures, and the remainder is distributed in the coal matrix in free and dissolved forms (Zhou and Lin, 1999).
Improvement of coal seam permeability by hydraulic fracturing
Development mechanisms of the hydraulic fracturing network
During hydraulic fracturing, the stress states around the fracturing well or borehole are initially changed by the transmitted water pressure. Then, hydraulic fractures initiate from the surface of the borehole or fracturing well and propagate outward. Then, new propagation of hydraulic fractures is controlled by the water pressure within the fractures. Meanwhile, due to the propagation of the main hydraulic fracture, the pressurized water in the fractures will seep into the coal body, forming seepage water pressure. Then, the seepage water will flow into the joint and fracture system in the coal seam and result in the propagation of the original joints and fractures. Generally, the propagation direction of the original joint and fractures is perpendicular to the minimum principal stress orientation. As a result, a certain angle exists between the expanding branch fracture (called a wing branch fracture) and the original fissure. After the above process, a fracture network containing the original joints and fractures, wing branch fractures, and main hydraulic fracture is formed in the coal body (Huang, 2012).
Permeability improvement of coal seam structural transformation
In hydraulic fracturing, the tensile stress generated by the high hydrodynamics force will overcome the resistance of the coal seam to fracture. As a result, a macro-fracture will extend fully in the space, forming the main seepage passage, and the micro-fractures will also extend farther due to the tensile stress. The connectivity of fractures and connectivity between fractures and pores are enhanced during hydraulic fracturing. As a result, an inter-connected fracture network is spatially formed in the coal seam, by which the gas flow passages are connected and the permeability of the coal seam is improved. Meanwhile, the fractures and fissures are supported by a proppant (e.g. fine sand) with regular roundness and compressive strength, and the newly formed fractures cannot close and the opening of hydraulic fractures is retained. As a result, gas can flow throughout the fracture network, and the flow conductivity of the coal seam is correspondingly improved (Keshavarz et al., 2015; Lee et al., 2016).
Processes of water drainage, pressure decline, and methane desorption
In the water drainage and pressure decline processes, the water pressure in macro-fractures drops the fastest, followed by that in micro-fractures and macro-pores, while that in micro-pores drops the slowest. When the pressure in the coal seam is below a critical desorption pressure, methane initially desorbs in macro-fractures, followed by micro-fractures, macro-pores, and micro-pores. The desorption-diffusion-flow model of gas is as follows: pores in the coal matrix → micro-fractures in the coal matrix → opening original fracture network → main hydraulic fractures → fracturing well or borehole (Huang et al., 2016).
Effect of water on CBM adsorption and desorption
Effect of the moisture content on CBM adsorption and desorption
Moisture adsorption behavior in coal
The presence of water in coal can adversely influence the sorption behavior of other molecules (Allardice and Evans, 1971; Tang et al., 2017a; 2017b; Yu et al., 2013). It has been reported that water clusters formed at or across the opening of a pore can effectively block the entire pore space, thereby hindering the access of other adsorbing molecules to pores. Apart from blocking pores, water leads to reduced CH4 uptake due to partial competition for sorption sites, a decrease in gas diffusivity, and induced swelling. The combined effect of these processes leads to a decrease in CH4 uptake and slowdown of the CH4 sorption kinetics (Krooss et al., 2002; Pan et al., 2010).
The isotherms of moisture adsorption fit the Dent model. The Dent model enables us to distinguish water adsorbed at primary and secondary sites. For a low relative pressure, adsorption at primary sites dominates, while with increasing pressure, adsorption at secondary sites gains in significance. The percentage of secondary site adsorption depends on the concentration of oxygen-containing functional groups because a higher concentration of functional groups supports water molecule bridging, which is shown in Figure 2 (Nishino, 2001; Švábová et al., 2011; Youssef, 1974).

(a) Molecular configuration of a model of disordered porous carbon obtained by Radiative Monte Carlo (RMC). (b) The same model as in a panel after hydrogen and hydroxyl groups are added (this work). The sticks are C−C, C−H, and C−O bonds. Red and white spheres in the functionalized sample are the oxygen and hydrogen atoms, respectively.
Critical moisture content
The concept of the critical moisture content was first proposed by Joubert et al. (1973). The authors also stated that the critical water content is equal to the equilibrium moisture content, which includes water in macro-pores and physically adsorbed in small pores (Joubert et al., 1973). Extensive adsorption studies show that the moisture content will decrease the gas adsorption capacity (Busch and Gensterblum, 2011; Crosdale et al., 2008; Gensterblum et al., 2013; Krooss et al., 2002). Dry coal samples are initially used to measure the gas adsorption capacity. Then, they are gradually replaced with wet coal samples or coal samples with equilibrium water content, which are closer to the actual conditions within the coal seam because the water content of an in-situ coal seam is greater than or equal to the equilibrium water content.
Micromechanics of the effect of the moisture content on CBM adsorption and desorption
It is revealed that water may significantly reduce methane sorption in nanopores. In small pores (1 nm), water and methane are adsorbed in the same layer. In large pores (>2 nm), water molecules adsorb on the first layer, and methane forms a weak second layer of adsorption (Jin and Firoozabadi, 2014).
The effects of water on the adsorption and diffusion of methane in nanopores is studied by ex-situ and in-situ nuclear magnetic resonance. The presence of water suppresses methane adsorption, but this suppression effect is stronger in pores with a diameter of 0.5–3 nm than those with a diameter of 6 nm. The self-diffusion coefficients of methane within pores with a diameter of 0.5–3 nm are not significantly influenced by the presence of water. However, in the pores whose size is 6 nm, the diffusion coefficient of methane increases as the amount of water adsorption increases, peaks, and then decreases to a constant value with further water adsorption (Hu et al., 2018).
The interactions of CH4 and H2O with coal surfaces were studied and characterized by their adsorption energy according to the density functional theory. It was demonstrated that the interaction between coal and CH4 was weak physical adsorption, but that the interaction between coal and H2O consisted of physical and chemical adsorption. The adsorption energy between coal and H2O is larger than that between coal and CH4 for all ranks at coals, suggesting that the adsorption priority of H2O is higher than that of CH4 (Zhou et al., 2016).
Based on the molecular structure model of coal (Chen et al., 2000), the adsorption configurations of the coal structure with H2O (Figure 3a) and CH4 (Figure 3b) were established by Zhang and Sang (2013) using Chem3D software. They also calculated the interaction potential energy of the adsorption configurations of graphite-like coal with CH4 and H2O, respectively, using the Quantum chemistry AB initio calculation Gussian 03 software (Chen et al., 2000; Zhang and Sang, 2013). The calculated results are shown in Table 1.

Configuration of the coal structure model and methane and water molecules. (a) Graphite-like structure of coal and methane molecule and (b) graphite-like structure of coal and water molecule. Grey sphere represents carbon atom, white sphere represents hydrogen atom, and red sphere represents oxygen atom.
Adsorption energy and corresponding adsorption distance between CH4 and H2O molecules.

Source: reproduced with permission from Zhang and Sang, 2013.
From the calculated results of the CH4 and H2O molecules, it is concluded that the adsorption energy between coal and water is larger than that between coal and methane. If water molecules exist in coal, they will be preferentially attracted to the coal surface. However, the active adsorption sites of the coal surface are limited. The adsorption site is occupied by a water molecule, and it cannot be occupied by methane, resulting in a decrease of the methane adsorption capacity of coal.
Effect of liquid water on CBM adsorption and desorption
Competitive adsorption and substitution desorption
Based on the essence of physical adsorption, the adsorption of methane is caused by the excessive free surface energy of coal. When methane molecules are adsorbed onto the coal surface, the excessive free surface energy of coal will decrease. The adsorption and desorption of methane molecules in coal are in a dynamic balance. Because the adsorption sites of the coal surface are limited, water molecules will compete with methane molecules for adsorption sites (Zhang et al., 2014). However, the adsorption ability of the coal surface for water molecules is greater than that for methane molecules. As a result, some of the adsorbed methane molecules will be replaced by water molecules and become free. The displacement of adsorbed methane by water and the acceleration of methane desorption have been widely confirmed by researchers (Chen, 2013; Chen and Cheng, 2014; Gensterblum et al., 2014). Figure 4 shows the displacement process of gas molecules by water molecules.

Drawing of a methane molecule substituted by a water molecule.
A coalbed may be wet for a long time after completion of the water flow and water–gas two-phase flow stages. In this period, water flows out as moisture vapor, but the water in the matrix does not change as much. The moisture loss is mainly from the water film in the fracture network. It is also observed that this moisture loss has a profound impact on the storage and transport of CBM. This moisture loss in coal seams can significantly improve coal permeability and thus enhance methane production (Teng et al., 2016).
Fractures are the main pathways of fluid accumulation and migration and they exert some control over the stability of the rock mass. The aperture, contact area ratio, and stress during permeability and fracture development have a strong influence on the permeability and seepage paths, which in turn, control the effective radius by absolute water injection. All of these factors should be considered when studying the structural characteristics of rock masses (Chen et al., 2017).
Effect of capillary pressure
When two kinds of unmixed fluids flow in a capillary pipe, one fluid usually scatters in the other fluid with a plunger-like shape, resulting in many meniscus interfaces in the two-phase fluid flow region. The forces at the two sides of the interface are discontinuous, and their difference is called the capillary pressure and represented by 
In general, coal matrix pores can be considered to be capillary pipes. Water will be imbibed into available pores under the capillary imbibition effect. The capillary imbibition process of semi-closed pores is shown in Figure 5, in which the solid arrow represents the direction of capillary pressure and the dotted arrow represents the motion of liquid water. If the pore gas pressure is not larger than the capillary pressure, the CBM will be enclosed in pores. In this case, the capillary pressure resists gas desorption and migration, which is one form of the water-block effect. The above process has been verified by laboratory experiments, which are shown in Figure 6.

Capillary imbibition process in a semi-closed pore.

Stimulation results of the capillary imbibition process in a semi-closed pore from the laboratory experiment. (a) water, (b) coal seam gas, and (c) matrix.
Figure 7 shows two kinds of capillary imbibition processes of open pores with at least two outlets. Figure 7(a) shows capillary imbibition taking place only at one outlet, and Figure 7(b) shows capillary imbibition taking place at both outlets.

Capillary imbibition process of open pores with two outlets. (a) Capillary imbibition at one outlet and (b) capillary imbibition at both outlets.
For the displacement of gas by water in open pores, Newton’s second law holds,

When
The wetting properties of porous media can drastically vary depending on the type of media and pore fluids. It is found that increasing the substrate’s affinity to the invading fluid results in a more efficient displacement of the defending fluid up to a critical wetting transition, beyond which the trend is reversed (Zhao et al., 2016).
Jamin effect
Bubbles in water or water drops in gas will always try to maintain a spherical shape due to surface tension. If the diameter of a bubble or water drop flowing through a pore is larger than the pore throat diameter, the pore will be blocked. The shape of bubbles and water drops must be lengthened to pass through the pore throat. To change the shape, some energy must be consumed, resulting in a decrease in the velocity of the movement of bubbles or water drops and an increase in the resistance to be overcome. The above phenomenon is called the Jamin effect. The Jamin effect is a kind of block effect, also called the gas block effect or liquid block effect. Figure 8 shows the Jamin effect of a bubble in water.

Jamin effect of a bubble in water.
The bubble is blocked at the pore throat. To pass through the pore throat, the resistance force, 

The calculation formula for the resistance of the Jamin effect can be obtained by subtracting equation (4) from equation (3) (Guo and Zhang, 2011):
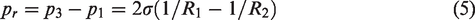
The bubble can only pass through the narrow pore throat when the difference of the internal and external pore pressures is greater than
Studies show that the Jamin effect is affected by the coal seam permeability, the pore-to-throat ratio, the water invasion depth, the water saturation, the interface tension, and other factors. The lower the coal seam permeability, the larger the pore-to-throat ratio. Correspondingly, the Jamin effect is more notable. The greater the water liquid invasion depth, the larger the water saturation. Accordingly, the liquid resistance is stronger. With the increase of the gas–liquid interfacial tension, the Jamin effect increases (Tian et al., 2015). Based on the above analysis, hydraulic fracturing can be conducted to improve the permeability of coal seams, and acidification fracturing can be adopted to increase the pore throat diameter. Additionally, residual water saturation can be decreased by increasing the water discharge amount, and the gas–water interfacial tension can be decreased by adding a surfactant to the fracturing fluid. Meanwhile, a wettability reversal agent can invert the coal seam wettability and turn the liquid or gas resistance into power to drive gas (Fan et al., 1996, 2006; Wang et al., 2008).
Effect of hydraulic fracturing on CBM adsorption and desorption
Methane driven phenomenon caused by hydraulic fracturing
It has been shown that during hydraulic fracturing in a high gas coal seam, the methane concentration significantly increases in the air flow in the ventilation roadway, indicating that the methane in the coal seam is driven out. This phenomenon is called the methane driven phenomenon (or effect) of hydraulic fracturing. The presence of the methane driven phenomenon of hydraulic fracturing was also confirmed by later experiments (Chen, 2015).
In the process of hydraulic fracturing, high-pressure water will flow within the channel system, which consists of cleats, micro-fractures, and pores in the coal seam. When the water pressure in the cleats and pores increases, the original mechanical balance of the channel system is disturbed, and stress redistribution occurs in the coal seam. At the forefront of water seepage, pore water will overcome the resistance of the channel to move forward, resulting in compression of the methane within a certain range of the water seepage forefront. As a result, the methane pressure increases. The pressure difference caused by the increase in the local methane pressure can lead to the migration of methane, which is proposed to be the methane driven phenomenon of hydraulic fracturing. Due to the methane driven phenomenon of hydraulic fracturing, three elliptical regions, i.e., the wet region, gas-driven region, and stress disturbance region, are formed in front of the developing face (Huang et al., 2016).
Dual effects of hydraulic fracturing on CBM desorption
Hydraulic fracturing can simultaneously promote and inhibit the desorption of methane. Therefore, hydraulic fracturing has dual effects on methane desorption. From the perspective of time, the process of hydraulic fracturing can be divided into two stages, i.e., during and after hydraulic fracturing. During hydraulic fracturing, the high water pressure will drive the free methane in pores and fractures. Due to the existence of competitive adsorption and substitution desorption between water and methane, displacement of methane by water will occur, which has been verified by experiments (Chen, 2013; Chen and Cheng, 2014; Zhang, 2011). In this process, hydraulic fracturing has a stimulative effect, causing the adsorbed methane to desorb into free methane.
The structure of coal can be transformed by hydraulic fracturing, and thus, the permeability of a coal seam can be improved. As a result, the adsorbed methane will desorb and become free methane and then flow along the fracture network and be extracted. During hydraulic fracturing, increases and decrease of the pore gas pressure will occur, resulting in a change of the pressure of the original methane. If the pore gas pressure decreases to below the critical desorption pressure, some of the adsorbed methane will desorb and become free methane. Conversely, if the pore gas pressure increases, some of the free methane will become adsorbed methane.
When hydraulic fracturing is carried out, free methane in pores and fractures is driven by high-pressure water. Meanwhile, water can substitute the adsorbed methane to some extent because of the competitive desorption and substitution desorption phenomena between water and methane, which have been proved by many laboratory experiments (Chen, 2013; Chen and Cheng, 2014; Zhang, 2011). The above process can be regarded to be a direct effect of enhancing the methane desorption ratio by hydraulic fracturing.
After hydraulic fracturing, the liquid water content in the coal seam increases, and the migration passages of methane are occupied and blocked by water. As a result, the permeability of the coal seam decreases, and the desorption of methane is inhibited. Meanwhile, during hydraulic fracturing, pressurized water enters into pores in the coal seam. After hydraulic fracturing, the borehole water pressure sharply decreases, and the pore water pressure decreases as well. As a result, the Jamin effect and the capillary imbibition effect of pore water will occur and the desorption of methane in pores will be restrained.
Effect factors of hydraulic fracturing on CBM adsorption and desorption
The effects of hydraulic fracturing on CBM adsorption and desorption are mainly affected by the water injection pressure, fracturing fluid, fracturing time, pore pressure, and other factors.
Water injection pressure
The minimum pore diameter that water can pass through under different water injection pressures is called the critical pore scale (Zhao, 2013). It is assumed that no gas pressure exists in the coal samples. Before and after water injection with different water pressures, the water content and the distribution law of pore diameters are different. Consequently, taking mercury injection experiments into consideration, the calculated critical pore sizes of coal samples are shown in Table 2.
Critical pore scale and corresponding desorption rate under different water pressures.

Source: reproduced with permission from Zhao, 2013.
Table 2 shows that the higher the water injection pressure, the smaller the minimum pore diameter that water can enter, and the restraining effect of water on methane desorption is correspondingly high because during hydraulic fracturing, pressurized water enters coal matrix pores and interacts with free and adsorbed methane. With high pressure water removed, the smaller the pore diameter, the greater the Jamin effect and capillary inhabitation and the stronger the methane closure effect caused by pore water. Therefore, the higher the water pressure, the more obvious the inhibitory effect of methane desorption and the lower the methane desorption ratio.
Type of fracturing fluid
Currently, there are many kinds of coal seam fracturing fluids, such as clean water fracturing fluid, guar gum fracturing fluid, clean fracturing fluid, and so on. In general, the pore canal can be simplified as a capillary. When fracturing fluid contacts methane in the capillary, the magnitude of the capillary force between the two fluids can be calculated by the formula:

From the above formula, it is observed that the capillary force decreases with the reduction of the fracturing fluid surface tension. The surface tension of clean water fracturing fluid is 73 mN/m. The surface tension of a clean fracturing fluid, which consists of a Viscoelastic surfactant (VES) surfactant, Synthesis (SYN) micelle accelerating agent, and Potassium chloride (KCL) swelling agent, is 35 mN/m. The surface tension of a clean fracturing fluid with an additive of penetrant JFC and concentration of 1% is 39 mN/m (Lu et al., 2015a, 2015b, 2015c; Wang et al., 2015). In practice, the clean water fracturing fluid is usually replaced by a clean water fracturing fluid containing a viscoelasticity surfactant fracturing fluid, nanoparticle–surfactant-stabilized foam fracturing fluid, and clean fracturing fluid containing a micro-foamed fracturing fluid stabilized by silica nanoparticles. By performing this replacement, the surface tension of the fracturing fluid can be decreased, and the restraining effect of high pressure water on methane desorption can be relieved or eliminated (Cai et al., 2016; Lu, et al., 2015a; 2015b; 2015c; Lv et al., 2015; You et al., 2015).
Fracturing time
The longer the fracturing time, the more fracturing fluid there is in the coal seam. The substitution of water for adsorbed methane does not begin until completion of hydraulic fracturing. In fact, this substitution begins immediately when high-pressure water contacts with coal. In other words, during the injection of high-pressure water, some of the adsorbed methane is replaced by water (Chen, 2013; Wang et al., 2015).
For a dry coal seam, the longer the high-pressure water injection time, the higher the water saturation degree. The curve of typical gas–water relative permeability is shown in Figure 9. It can be observed that the restraining effect of water on methane desorption becomes increasingly obvious with the increase of the water saturation degree.

Typical gas and water relative permeability curves indicating how the near wellbore permeability is reduced due to water invasion.
The inhibition effect of water on methane desorption can be relieved by reducing the filtration rate and increasing the discharge capacity of high-pressure water. During the process of huffing–puffing hydraulic fracturing, the fracturing fluid is repeatedly injected into the coal seam and then rapidly discharged with coal cinders. In this way, the coal seam permeability can be increased, and the process of pressure-declining and desorption of CBM and the displacement of methane by water can be promoted (Xin et al., 2014).
Pore pressure
During hydraulic fracturing, increases and decreases of water pressure will occur, resulting in changes of the pore pressure in the coal seam. On the basis that the adsorption and desorption of methane is totally reversible, the following experiments are performed to study the relationships among the pore pressure, methane desorption amount, and desorption time. First, adsorption balances of coal samples under different pore pressures are achieved. Then, methane desorption experiments are carried out. The experimental results are shown in Figure 10.

Diagram showing the relationships among pore water and the amount and time of methane desorption.
Figure 10 shows that the methane desorption amount increases with the increasing pore pressure. The methane desorption amount under a pore pressure of 1 MPa and 1.5 MPa increases by 78.5% and 107%, respectively, more than that under a pore pressure of 0.5 MPa. The desorption amount of methane under a pore pressure of 1.5 MPa is 1.16 times that under a pore pressure of 1 MPa.
Problems that require further study
Change of pore characteristics and the distribution law of methane in coal
A system is formed by the combination of CBM and the coal structure. The adsorption properties of methane are determined by the characteristics of pores in coal. Methane in pores and fractures can be driven, substituted, or blocked by high-pressure water, resulting in changes to the pore pressure and matrix stress of coal. Additionally, the characteristics of pores and the distribution of methane in coal are changed. Experiments show that, after injection of water, the volume, surface area, and average diameter of pores as well as the porosity of coal all increases to different extents (Xiao, 2010; Lu, 2015a, 2015b, 2015c). As a consequence, the change of the characteristics of pores in coal and the distribution law of methane after hydraulic fracturing need to be further studied.
Redistribution of the pore pressure in a coal seam
During the hydraulic fracturing process, methane is compressed within a certain range in front of the tip of the water seepage. As a result, the pore pressure along the migration direction of pore water changes dynamically. Both CBM adsorption and desorption processes and the coal matrix stress are influenced by the pore pressure. The dynamic changes of the pore pressure and coal matrix stress during the hydraulic fracturing process need to be further studied. The hydraulic fracturing process is a pressure-increasing process, but a pressure-reduced zone and pressure-increased zone exist at the same time. That is, in some places, the pressure is reduced, but in other places, the pressure is increased (Huang et al., 2016). When the hydraulic fracturing process is over, the local area pore pressure cannot be relieved, resulting in a change of the pore pressure. Therefore, in-depth studies are required to study the pore pressure redistribution laws in the coal seam after hydraulic fracturing.
Visualization of the process of methane displaced by water
In the current study, because water and oil are both liquid, oil displaced by water can be visualized. Because methane is gas, it is difficult to visualize the process of methane displaced by water. Although visualization of the process of methane displaced by water has been studied with the help of numerical simulation, many problems inconsistent with the practical condition exist. The process of methane displacement by water should be observed under a scanning tunneling microscope. Considering the pressure and temperature, it is currently difficult visualize water-displacing-methane process. However, this process may be able to be visualized in the future with further developments of scanning microscopy.
Evaluation of the effects of hydraulic fracturing on methane adsorption and desorption
At present, many studies have been carried out focusing on the impact of hydraulic fracturing on methane adsorption and desorption. Nevertheless, the effects of influence factors are studied independently. Only one factor is analyzed and summarized independently. Evaluations of the combined effect of more than one factor and the combined effect with factual geological conditions are insufficient or even non-existent.
The characteristics of CBM adsorption and desorption are influenced by the intrinsic properties of the coal seam, methane, and various geological factors. The permeability of a coal seam can be improved, and the methane pressure can be decreased by hydraulic fracturing in the coal seam. Then, methane desorption can be promoted. In the gas driven process, with the free methane in the passages driven out, the balance between free and adsorbed methane is also disturbed. Liquid water can replace adsorbed methane, but the capillary imbibition effect and Jamin effect will lead to water blocking. As a result, the seepage of high-pressure water has dual functions of promoting and restraining methane desorption. One or more indexes or an index system are needed to evaluate the combined effect of the two functions quantifiably or semi-quantifiably. In evaluating the two functions, the characteristics of the coal seam, the geological factors, the permeability improvement effect of hydraulic fracturing, the gas driving effect, the substitution effect of methane by water, and the water-block effect should be taken into consideration. The evaluation should be performed based on an overall analysis.
Footnotes
Authors' Note
Weiyong Lu is also affiliated with School of Mines, Luliang University, Shanxi, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by the National Natural Science Foundation of China (No. 51704285 and 51774272), Technical Innovation Team for Fault Diagnosis and Health Management of Coal Mine Machinery and Equipment (TD201812), research start-up funding of doctors in Luliang University (1901010145), Scientific and Technological Innovation Programs of Higher Education Institutions in Shanxi (STIP) and Transformation of Scientific and Technological Achievements Programs of Higher Education Institutions in Shanxi (TSTAP),which is gratefully acknowledged.
