Abstract
In this study, bisphenol-A-based acrylated epoxy oligomers were prepared and utilized to improve the adhesion strength of polyester cords onto rubber. The structure of the oligomers was characterized by Fourier transform infrared spectroscopy and 1H-NMR spectroscopy. Ultraviolet-curable adhesive formulations were prepared by using acrylated epoxy oligomers and applied onto the polyester cord fabric by a dip-coating method and irradiated. Ultraviolet-cured coatings were characterized by thermal and scanning electron microscope analysis, contact angle measurements. In the second stage of the experiment, ultraviolet-cured polyester cords were adhered onto rubber under heat and pressure. The prepared adhesive formulation was expected to improve the adhesion strength. The adhesion strength of the coated material was evaluated by using peel test as a function of the carboxyl/epoxide ratio. The adhesion strength of 18.0 N/cm was obtained when the carboxyl/epoxide ratio was set as 1. It was observed that peel strength, contact angle, and surface energy values of acrylated epoxies strongly depend on the acrylic acid content of the oligomer.
Introduction
Epoxy resins have found widespread applications in automotive, space, aviation, and OLED technologies, etc. They can be used as a matrix resin in preparation of composite materials due to its unusual chemical and mechanical behavior over the wide temperature ranges. Generally, epoxy resins are cured by the reaction of the epoxy group with amines, anhydrides, and carboxylic acids to form linear, branched, or cross-linked structures depending on the functionality [1–4]. The preparation of epoxy-based ultraviolet (UV) sensitive vinyl-esters is a direct one-step reaction of acrylic or methacrylic acid (AA or MAA) with epoxy groups. Acrylated epoxy oligomers having vinyl ester groups with carbon–carbon double bonds at the end of the epoxy resin, are generally used in industrial applications because of excellent adhesion and non-yellowing properties, flexibility, hardness, and chemical resistance. The epoxy part gives toughness to the cured films, whilst carbon–carbon and ether bonds give wettability and flexibility to the structure. The reaction between epoxy and an acid produces hydroxyl groups, thereby introducing polarity which can improve the wettability of the UV-curable coating material to help to adhere difficult surfaces [1,5–7]. Multifunctional acrylated oligomers are generally used in order to increase the crosslinking density, tear resistance, and the surface hardness of the UV-curable material [8].
UV-curable coatings are favored because of low-energy consumption with high productivity and can be applied onto the surfaces in a wide temperature range that is important for heat-sensitive materials. It is also environmentally friendly due to non-solvent dash-free process, having high scratch resistance, high optical clarity, etc. The drawbacks of UV curing are as follows: difficult to homogeneous curing on complex shapes, having poor weatherability due to continuous absorption of UV rays, and showing high curing shrinkage. UV-curing techniques have found wide spread applications in automotive, aerospace, printing, wood, dentistry, biomedical industries, etc. In that mentioned fields it can be used as lacquers, varnishes, printing inks, adhesives, and so on [2,9–12]. In order to increase the polymerization rate, during the UV-curing process of epoxyacrylates, the elimination of air and any impurities is the most important parameter to stop undesired radical formations. A large number of studies are focused on to overcome adhesion problems mainly concentrated on low conversion of double bonds and wide differences in surface energies between the coating material and cord surface [13].
In literature, various studies have dealt with the adhesion property between cord/rubber by using cobalt boron acylate [14], hydrated silica-resorcinol-hexamethoxymethylmelamine [15], and resorcinol-formaldehyde-latex [16] as adhesion promoters. However, these additives present toxicity, health and environment-related problems, and undesired coating properties. Very few studies have been reported on the synthesis of dual-curable epoxyacrylate [5,7,10,17,18] and there was no research considering their application between textile cords and rubber surfaces as an adhesive. Therefore, development of formaldehyde-free adhesive formulations for cord/rubber application seemed as an innovative approach for tire industry. This study was aimed to synthesize a formaldehyde-free dual-curable oligomer containing both epoxide and unsaturated groups in the same molecule allowing to be cured by both UV light and thermal energy. For this purpose diglycidyl ether of bisphenol-A (DGEBA)-based epoxy resin was acrylated by AA at various concentrations. These oligomers were included in adhesive formulations and applied onto the cord surface by dip-coating. By using UV light, the unsaturated groups of the oligomer were cross-linked onto the polyester structure. In the second stage thermal curing of the epoxide groups was carried out by using heat and pressure to adhere the treated cord surface to the rubber surface. The effect of carboxyl/epoxide ratio on the thermal characteristics and adhesion properties of coatings was investigated.
Experimental
Materials
Epoxy resin (EPIKOTE Resin 162) based upon DGEBA with an epoxy equivalent weight (EEW) of 167–171 g equiv.−1, acrylic acid (AA), triphenyl phosphine (TPP), hydroquinone (HQ), potassium hydroxide (KOH), phenol phthalein, ethanol, crystal violet, tetraethyl ammonium bromide, perchloric acid, acetic acid, methyl ethyl ketone (MEK), were all purchased from Sigma-Aldrich and used as received. Polyester-based cord fabrics (360 g/m2, 12 warp/cm, 5 weft/cm, 360 fibers/warp, 360 fibers/weft) were obtained from Izomas Co. Dipropylene glycol diacrylate (DPGDA) was obtained from Allnex Corporation. 1-hydroxycyclohexyl phenyl ketone (irgacure 184) was supplied by Ciba specialty chemicals.
Synthesis of acrylated epoxy oligomer
The compositions of the synthesized EA oligomers.

EA: epoxy acrylate; DGEBA: diglycidyl ether of bisphenol-A; AA: acrylic acid.
Preparation of dual-cured polyester cord/rubber composites
For dip-coating process, epoxy acrylate (EA) (about 15% of the total weight of the fabric layers) and photoinitiator (3 wt%) were dissolved in MEK in order to lower the viscosity of the coating material and to give a better wetting property to the polyester cord. Polyester cord fabrics were cut in 13 × 16 cm dimensions as six layers that are required to make the peel test according to the American Society for Testing and Materials (ASTM) standard [20]. Then polyester cords were dipped into the solution for 3 min and then the excess of the solution was removed from the fabrics by placing the fabric between polyethylene sheets and using a squeezing roller in 500 g weight in order to spread the adhesive solution homogeneously on both sides of the polyester cord. Fabrics were dried in an oven for 10 min for evaporation of the MEK. Fabric layers were individually exposed to UV light by using OSRAM Ultra-Vitalux 300 W UV lamp as a light source for 2 min from both sides of the fabric. After dip-coating process with various EA-based adhesive formulations an average 6.5% weight increment was detected.
After UV curing, the weight increment of the fabrics was measured and thermal curing of the epoxide groups between the cord fabric and styrene butadiene rubber (SBR, butadiene/styrene 75/25%) was achieved in a two-stage oven by using a catalyst based on organometallic compounds. The first part of oven is 60 m in length and 170℃, whereas the second part is 12 m in length and 220℃ temperature. During the thermal curing, cord fabrics are extended about 2%, exposed to about 300–400 kg/m loading on average depending on the thickness of the layers and pass through the oven with 13 m/min speed. The main thermal-curing process was carried out in the second stage at the higher temperature.
Characterizations
FTIR spectra were recorded on a Perkin Elmer Spectrum 100 FTIR spectrophotometer equipped with an ATR sampling accessory. Crosslinking density of EA100-coated UV-cured polyester fabric sample was determined by Soxhlet extraction for 4 h using acetone as a solvent. EEW means that the weight of resin in grams that contains a 1 g equivalent of an epoxy group [1] and was calculated by using a specifically designed formula that was given in a previous study [21]. The acid value means the potassium hydroxide amount in milligram that is needed to neutralize 1 g of epoxyacrylate oligomer. Acid value of the oligomers was measured according to the ASTM D974 standard [22]. 1H-NMR characterization was carried out on Agilent VNMRS at 500 MHz, by using deuterated chloroform (CDCl3) as a solvent and tetramethylsilane (TMS) as an internal standard. Thermogravimetric analysis (TGA) was performed with a TA TGA Q50 instrument under nitrogen atmosphere at a heating rate of 20℃/min. Nitrogen flow rate was 90 mL/min. Thermal analysis was performed by differential scanning calorimetry (DSC) TA DSC Q10 at a heating rate of 20℃/min, from −80℃ to 350℃. Scanning electron microscopy (SEM; JEOL Ltd, JSM-5910LV) was used for topological characterization. Water contact angles and surface energy were measured by using a Gardco PGX + goniometer equipped with a camera. Measurement was applied on EA-coated UV-cured polyester fabrics at room temperature. The volume of droplets was controlled to be about 3 µL. In order to increase the reliability of the test, each sample was measured three times and the average of these results was taken. The adhesion strength of the cured samples was evaluated by peel test after application on the cord/rubber surfaces according to the ASTM standard [20]. The sample of EA100 has been determined as the optimum adhesive formulation considering the peel test result; thus, the further characterization methods of 1H-NMR, DSC-TGA and SEM was only applied on EA100 sample.
Results and discussion
FTIR spectroscopy
FTIR spectroscopy was employed to observe the formation of ester bonds during the synthesis of EA. As clearly seen in Figure 1, the appearance of the characteristic ester carbonyl peak at 1720 cm−1 and a broad peak at about 3460 cm−1, which is assigned to secondary hydroxyl groups as well as a decrease in the characteristic absorption peak of an epoxide ring at 1230 cm−1 and 880 cm−1 indicates the opening of epoxy ring. It was observed that increase in the carboxyl/epoxide ratio increases with the extent of esterification reaction.
FTIR spectra of pure epoxy and EA prepared with different equivalent ratios of carboxyl/epoxide.
Figure 2 shows the FTIR spectra of pure polyester fabric, and EA100-coated fabric samples before and after UV curing. Accordingly, after the dip-coating process, acrylate functionality was observed at 1636 cm−1 associated with the resin [23]. After UV curing, the unsaturated acrylate peak almost disappeared due to the double bond cleavage caused by UV light; thus, photo polymerization process was completed by exposing each side of the fabric surfaces to UV light for 2 min. When the polymerization proceed, movements of monomeric and polymeric radicals become difficult due to the increased viscosity and crosslinking density resulting some of unsaturated acrylate double bonds remain as it is. This may also cause tackiness on the surface. Acetone extraction method was performed to obtain information on the degree of crosslinking density qualitatively by measuring the weight loss of EA100-coated UV-cured fabric. Accordingly, after acetone extraction for 4 h, a 0.02% weight loss was observed. This result proves that 2 min UV exposure time is adequate for curing of the EA-based coating materials.
FTIR spectra of pure polyester fabric, EA100-coated polyester fabric before and after UV curing.
Determination of EEW and acid value of the oligomer
Characteristics of EA samples.

EA: epoxy acrylate; DGEBA: diglycidyl ether of bisphenol-A; AA: acrylic acid; EEW: epoxy equivalent weight.
Completion of the esterification reaction was followed by checking the acid values as shown in Table 2. Accordingly, acid values of EA25 and EA50 indicate that almost all acidic groups reacted with epoxy groups. Whilst in EA75 and EA100 formulations, AA is in excess compared to the molar ratio of DGEBA. In the case of EA100 formulation, the initial carboxyl and epoxide groups are in stoichiometric amounts. It is obvious that AA had not reacted completely with the DGEBA in EA100 system. The acid value and EEW values are 43.4 mg KOH/g and 1040.4 g equiv.−1, respectively. At the end of the reaction, the carboxyl and epoxide concentrations decrease due to the EA formation; thus, the highest acid value and EEW were obtained in the case of EA100.
1H-NMR spectroscopy
Figure 3 shows the 1H-NMR spectra of EA100 oligomer in CDCl3. Accordingly, the peaks at 5.9–6.5 ppm prove the existence of acrylic group (CH2 = CH–). The peaks at 3.9–4.3 ppm illustrate –CH2 protons attached to the ester group oxygen atoms. The methylene and methine protons in the oxirane ring can be seen at 2.6–2.8 and 3.3 ppm, respectively. The aromatic protons of phenyl rings were appeared at 6.7–7.2 ppm. The peak at 1.65 ppm is due to the –CH3 protons in the cyclic ring [24,25].
1H-NMR spectra of EA100 oligomer.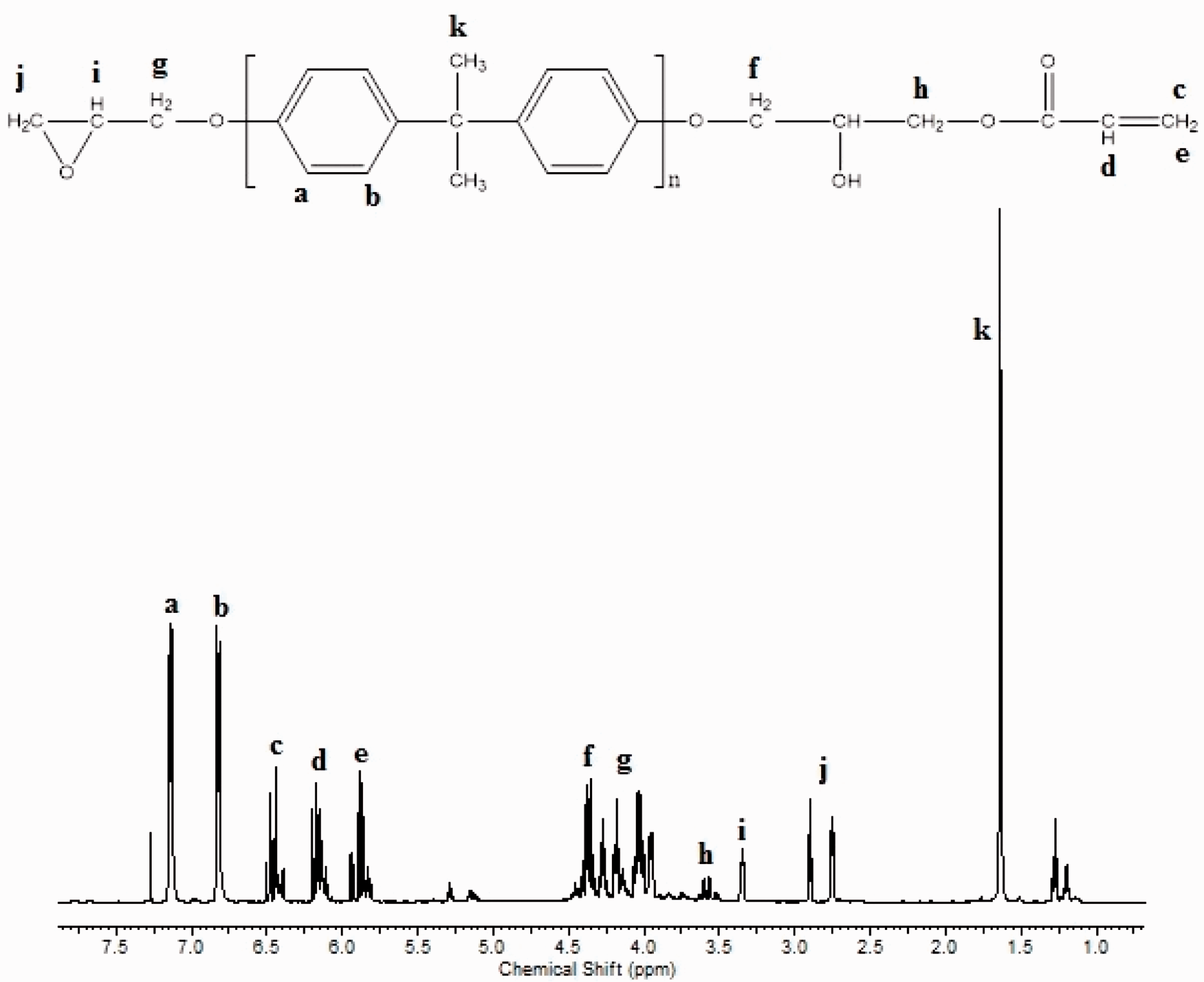
TGA and DSC analysis
The TGA thermograms of polyester and EA100-coated polyester fabrics are shown in Figure 4. Accordingly, a one-step decomposition was observed. There was no decomposition till 400℃ in polyester cord structure. Initial and rapid decomposition was observed at about 450℃, while in the range of 500–550℃ TGA curve showed a horizontal line. There was no weight loss above 700℃ and the char residue was about 15%. Because of the pyrolysis under nitrogen gas, carbonization occurred; thus, char residue was mainly formed by that carbonization step. Thermal stability increased after UV coating because of several reasons such as the aromatic structure of EA100, increment of carbon amount, and processing in the inert atmosphere.
TGA curves of the polyester and EA100-coated polyester fabric samples.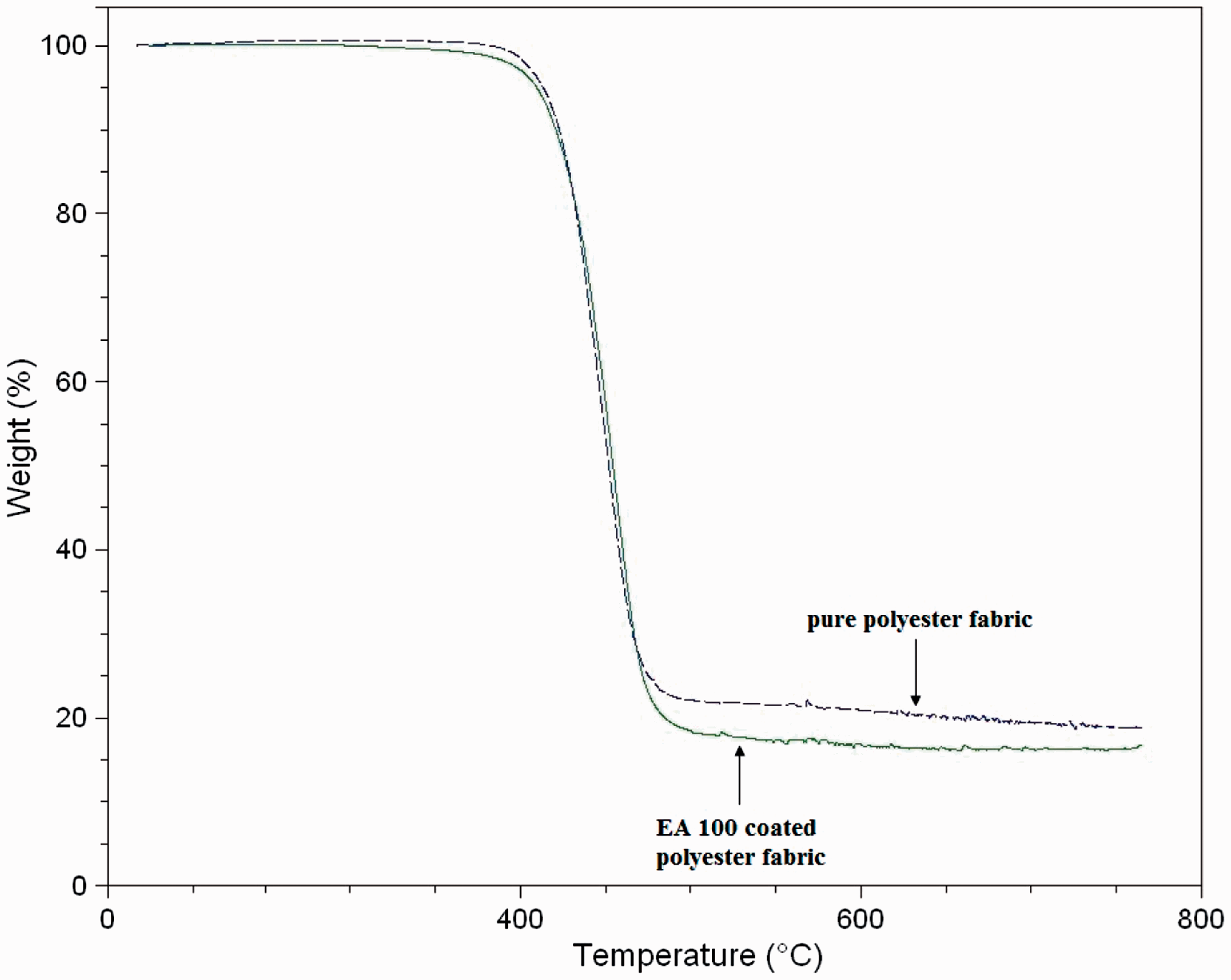
Thermogravimetric data of the polyester and EA100-coated polyester fabric samples.
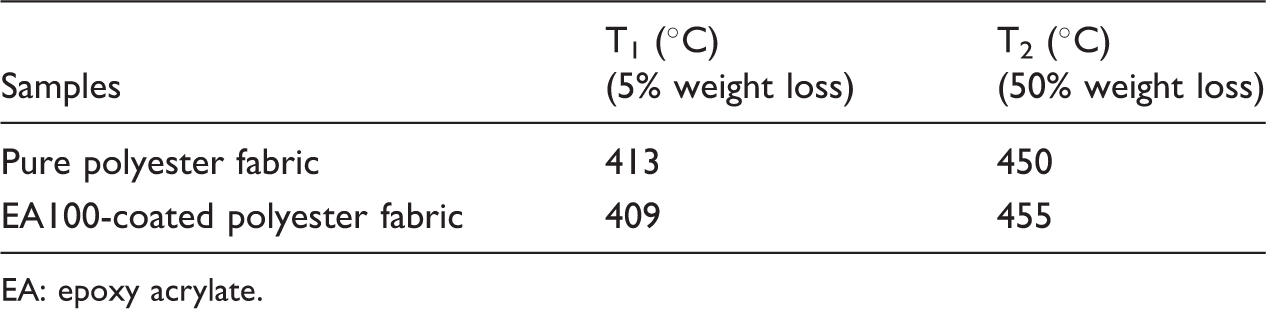
EA: epoxy acrylate.
Figure 5 shows the DSC thermograms of the polyester fabric and EA100-coated polyester fabrics, respectively. According to the DSC thermogram, a sharp peak was observed at about 260℃ which belongs to typical polyester crystals melting temperature. After coating with EA100, the amorf structure of EA is partially diffused onto the polyester cord surface and mixed with the crystalline structure of the polyester. Thus, melting temperature of the EA100-coated polyester fabric shifted to 250℃.
DSC curves for polyester fabric and EA100-coated polyester fabric samples.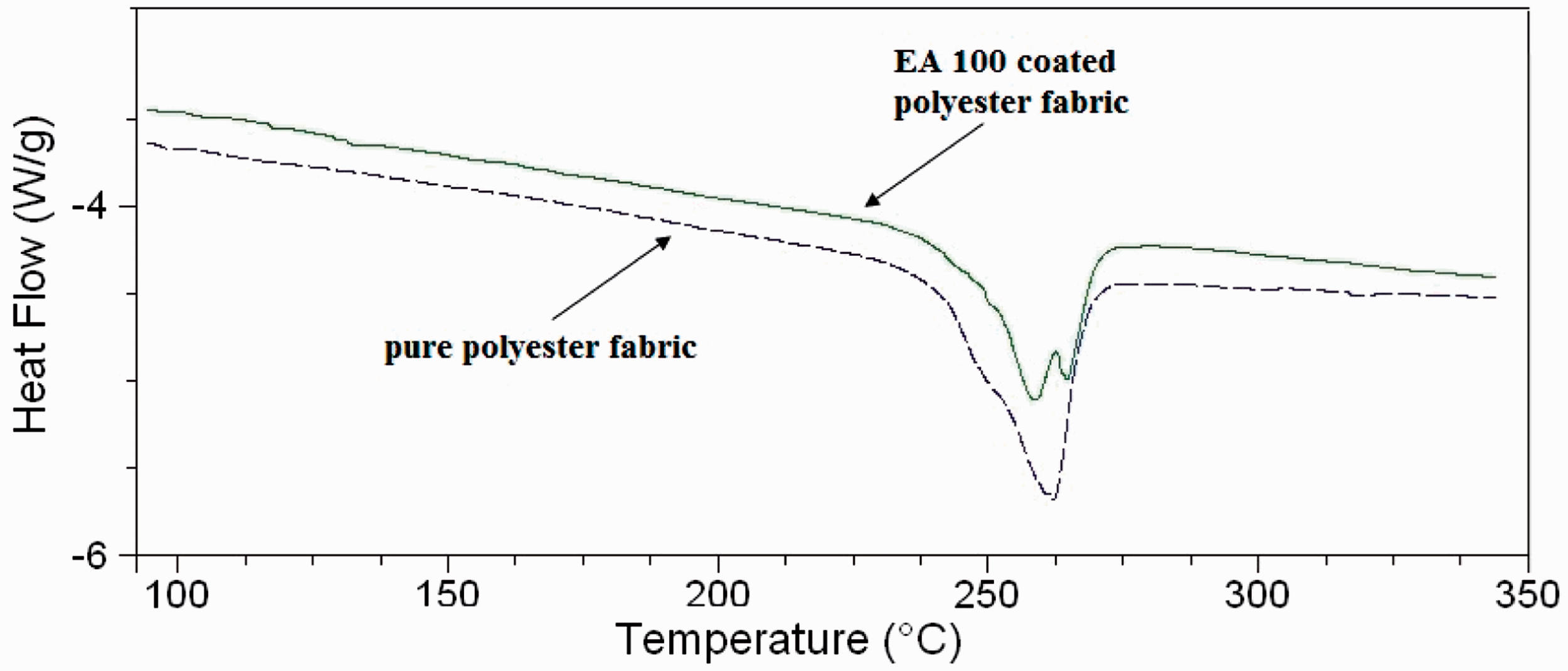
SEM analysis
Figure 6 shows the SEM images of polyester fiber and EA100-coated polyester fiber in various magnifications. According to the images, after coating, a very thin coating layer (about 0.5 µ) was obtained on single fiber surface without any agglomeration. As can be seen in Figure 6(d) and (f), after coating with EA100 the fiber surface gained a smoother structure. This can be attributed to the formation of well-dispersed EA layer on fabric surface and complete coating of each fiber within the fabric layer.
SEM images of polyester fiber and EA100-coated polyester fiber samples in various magnifications.
Contact angle and surface energy measurement
Contact angle and surface energy values of EA-coated UV-cured polyester fabrics.

EA: epoxy acrylate; UV: ultraviolet.

Contact angles of water droplets on EA-coated UV-cured polyester fabrics.
Peel test
Peel test results of polyester fabric for each adhesive formulation type.

EA: epoxy acrylate.
Conclusions
In the present study, formaldehyde-free oligomers were synthesized and utilized as an adhesive for cord/rubber applications. For this purpose, EA oligomers have been synthesized by addition esterification reaction of carboxyl and epoxide groups in various molar ratios and then characterized. These oligomers were included in the adhesive formulations together with other ingredients such as reactive diluents and photo initiator to bind cord and rubber surfaces. FTIR and 1H-NMR analysis proved the formation of epoxyacrylate oligomer. The completion of photopolymerization was proven by FTIR analysis by observing the disappearance of acrylate absorption peak after UV curing. In EA100 formulation, due to the EA formation, the carboxyl and epoxide concentrations decrease, resulting to give the highest acid value and EEW. EA coating layer caused a 6.5% weight increment on average for each polyester cord sample. In EA-coated UV-cured polyester fabric samples, thermal stability increased because of the increment in carbon amount. The EA layer provided more amorphous structure; thus, lower melting temperature values were observed compared to the polyester cord fabric. SEM images showed that EA-coated fiber surface gained a smoother structure without any agglomeration. Contact angle measurement of the UV-cured polyester cord surface proved that AA increment in oligomer caused a more hydrophobic behavior because hydroxyl groups of the acid form hydrogen bonding with the oxygen atom of the polyester cord.
The EA-coated UV-cured polyester cords were thermally cured in an oven under pressure to provide adhesion on rubber surfaces. After dual-curing process, peel test was applied to evaluate the adhesion property of the oligomer. Peel testing results showed that the best peel strength with 18.0 N/cm has been obtained by using the EA100 adhesive formulation with the highest acrylate functionality allowing more connection point between the double bonds of SBR during the thermal-curing process. Further studies are now in progress to improve adhesion strength between the cord and rubber surfaces by using adhesion promoters.
Synthesis of EA oligomer.
Footnotes
Acknowledgement
The authors are pleased to acknowledge Ms. Zuhal Nart from Izomas Co. for her assistance in peel testing and polyester cord fabric supplement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Istanbul Technical University Research Fund is gratefully acknowledged for financial support.
