Abstract
Phytochemicals have been used over the centuries in order to cure various diseases and still do to this day. Phytochemicals are herbal extracts that are also named herbal remedies. Curcumin (CUR), which is a natural polyphenolic compound derived from the roots of the plant Curcuma Longa, has many therapeutic properties. CUR has poor water solubility and instability that has confined its further applications, thus there is need for a carrier to deliver the drug consistently. In this research, the feasibility of using polycaprolactone/gelatin nanofibers as carriers for CUR is examined. Nanofibers are obtained by electrospinning method. The morphology of nanofibers was observed by scanning electron microscopy. Fourier Transform Infrared Spectroscopy and differential scanning calorimetry is used to study the thermal behavior of the nanofibers. Antibacterial tests were conducted against methicillin-resistant staphylococcus aureus (MRSA) and extended spectrum β lactamase (ESBL). These pathogens are very dangerous and versatile pathogens emerging rapidly causing nosocomial infections in hospitals. The nanofibers were 99.9% antibacterial against MRSA and 82.56% against ESBL. The results showed that these nanofibers have potent antibacterial activity against both Gram positive and Gram negative bacteria. As a result, these nanofibers are very promising materials for antibacterial applications.
Introduction
Over the years, there has been a growing interest in herbal and natural substances. Herbal remedies are natural products that are extracted from plants. Till this day, majority of people in developing countries depend on traditional medicine and traditional practitioners due to historical and cultural reasons. In the traditional Chinese medicine, mineral and animal materials and botanical sources were used [1–3]. For many years, due to the lack of scientific knowledge and complications in preparation, the advancement of herbal medicines was not a matter of consideration. It was in the early 19th century that chemical analysis became feasible and convenient. With the help of modern phytopharmaceuticals, novel procedures such as nanoparticles, microemulsions, matrix systems, solid dispersions, liposomes, etc are in hand [4,5]. By designing novel drug delivery systems for herbal constituents, they can be delivered in a sustained manner with the benefit of reduction in administration repetition and increasing the bioavailability.
Liu et al. [6] developed uniform, spherical, and smooth triptolide-loaded poly(
Curcumin (1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione) is a natural, low molecular weight, polyphenolic compound derived from the roots of the plant Curcuma Longa (Figure 1). This natural compound is called turmeric in English, haldi in Hindi, and ukon in Japanese. It has been used in Asian medicine and as spice and traditional medicine in south Asia for centuries. The chemical structure of CUR comprises two phenolic groups and one active methylene group, which can be applied as potential sites for chemical variations and covalent linkage with biomolecules. The molecular weight of CUR is 368, and it possesses a melting point of 183℃. Maximum light absorption of CUR, using ultraviolet spectrophotometry, appears at 420 nm [9,10].
Chemical structure of curcumin.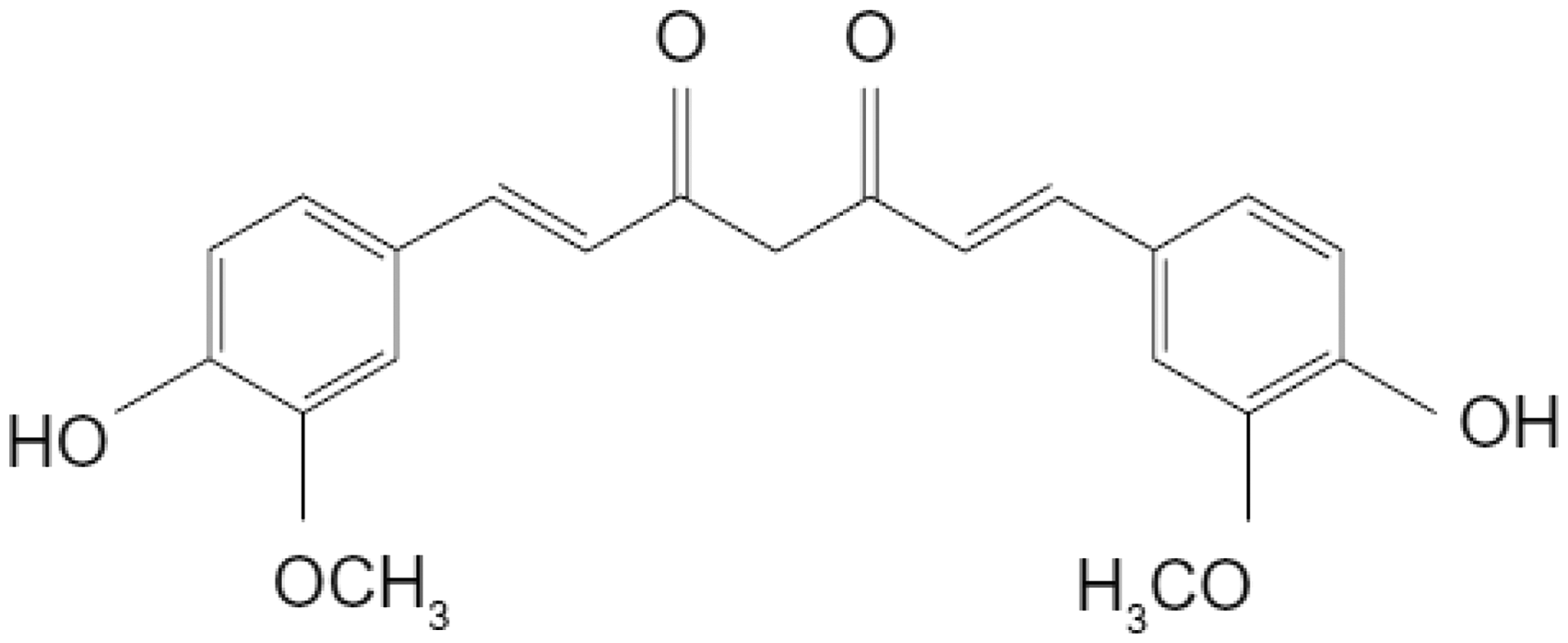
Various natural polymers such as collagen, chondroitin sulfate, chitin, chitosan, fibronectin, and laminin have been used in biological and biomedical applications [11, 12]. In a novel approach by Ghadi et al., nano-magnetic chitosan nanoparticles were obtained using Fe3O4 as the core and chitosan as the shell. These nanoparticles have potential applications for nanomedicine, biomedical engineering, industrial, and pharmaceutical field [13]. In a recent investigation, Cardoso et al. synthesized silver nanoparticles stabilized with type I collagen to build a nanomaterial with enhanced biological properties. Antibacterial activity in vitro and cell viability assays were also analyzed. Their research concluded that these particles showed antimicrobial activity against both Staphylococcus aureus and Escherichia coli and showed no toxicity to the cells at the examined concentrations [14].
Additionally, gelatin (Gel) is a natural polymer extracted from the collagen of bones and animal skin. Depending on the pretreatments on collagen, there are two types of Gel: type A and type B. If collagen undergoes acidic pretreatments, Gel type A is extracted and if alkaline pretreatments are processed, type B is obtained. This natural polypeptide, unlike synthetic polymers that are mainly nonionic, is a polyelectrolyte with many ionizable groups due to its carboxyl and amide functional groups [15]. Gelatin’s biocompatible and biodegradable properties are similar to collagen, but pure collagen is much more expensive than gelatin. This natural polymer also prevents fluid loss, which is an important factor in wound-healing applications [16]. Therefore, because of its many merits, gelatin is used extensively in biomedical applications. Nevertheless, in spite of many merits natural polymers possess, they do not show good mechanical properties. On the other hand, there are many biocompatible and biodegradable synthetic polymers with better mechanical properties [17]. Hence blending natural and synthetic polymers are often used to overcome this problem.
Many synthetic polymers such as poly(caprolactone) (PCL), PLA, poly(glycolic acid), poly(lactic-co-glycolic acid) have been used with natural polymers to produce polymer blends with proper mechanical and biological applications such as in skin, cartilage, bone, tendons, vessels, nerves, bladder, and liver tissue engineering [11]. Recently Munj et al. have discussed the suitability of a ternary polymethylmethacrylate–PCL–gelatin blend in the preparation of electrospun scaffolds for biomedical applications. Careful control over blend composition and infusion conditions develops the needed flexibility in order to produce biocompatible electrospun scaffolds for a variety of biomedical applications [18].
Lately, attention to applying herbs to polymers in order to increase their biological properties has increased. There have been extensive studies over the application of CUR for medicinal purposes. Over the past three decades, studies have proven CUR’s biological abilities. This natural substance has a broad range of therapeutic properties, which include antioxidant, anti-inflammatory, antimicrobial, and anticarcinogenic [19, 20]. It also can be used as an antimicrobial agent for textile materials. For example, the mixture of CUR with aloe vera and chitosan has shown that it can be used as a potential suppressor for microbial growth in cotton, wool, and rabbit hair determined by the exhaustion method. Also the combination of CUR with different antibacterial agents and hydrogel silver nanoparticles is used for wound dressing applications [21]. Brahatheeswaran et al. [22] achieved zein–CUR fibrous scaffolds with antioxidant properties that showed promotion of cell attachment.
In 2014, Malathi et al. electrospun CUR-loaded poly (lactic-co-glycolic) acid nanofiber for the treatment of carcinoma. Their studies showed that CUR was released in a sustained manner. The cell viability assay indicated that these nanofibers can be applied in skin cancer treatments [23]. CUR has also been electrospun with cellulose acetate, PLA, chitosan–Gel, and PCL [16, 19, 24, 25].
Electrospinning is a unique, simple, and efficient technique to produce electrospun fibrous structures with diameters in the range of nanometers to micrometers depending on the polymer and different electrospinning parameters implied. The morphology of electrospun fibers is influenced by various parameters, which are mainly categorized as follows: polymer solution parameters, processing conditions (applied voltage, effect of collector, and temperature), and ambient conditions [26]. Choktaweesap et al. studied the effect of various solvents such as glacial acetic acid, 2,2,2-trifluoroethanol, dimethyl sulfoxide, ethylene glycol, and formamide and their effects on morphology and fiber diameters. They concluded that only acetic acid and 2,2,2-trifluoroethanol were proper solvents [27]. Also Haung et al. mentioned that fluorinated alcohols such as hexafluoro isopropanol are good solvents for polypeptide biopolymers [17]. Song et al. used a water-based cosolvent approach that comprised of ethyl acetate and acetic acid in water. They found that the optimum composition ratio of the cosolvent is 2:3 (ethyl acetate:acetic acid). The maximum Gel concentration that could be dissolved in this condition was 11 wt% and the diameter of the nanofibers was found to be in the range of 47–145 nm [28].
To the best of our knowledge, no studies have been reported on CUR-loaded electrospun Gel/PCL nanofibers. In this paper, we investigate the synergistic effect of these polymers and the effect of adding CUR in electrospinning and biological properties. Effect of various electrospinning parameters on the morphology of fibers was investigated. The parameters investigated in this study are the polymer solution, distance between needle, and collector and the applied voltage.
Experimental
Materials
Gelatin for microbiology was obtained from Merck-Germany. CUR extracted from turmeric was obtained from Shahid Beheshti University. PCL and acetic acid were also purchased from Merck-Germany and used without further purification. Phosphate-buffered saline (PBS) 1× was prepared in the laboratory.
Fabrication of Gel/PCL-loaded CUR nanofibers
Gel powder (15% w/v) and 15% w/v PCL were separately dissolved in acetic acid (90% v/v). The solutions were stirred for 12 h at room temperature. PCL was added in a dropwise manner to Gel solution at a ratio of 1:1 w/w. After acquiring an even solution, 2% w/w CUR was added to the solution and was stirred for another 30 min before electrospinning.
For the electrospinning process, a 20-ml syringe with a blunt 19 gauge needle was used. An aluminum foil was used as a collector. Different electrospinning parameters were implied which are as follows: tip-to-collector distance = 10 cm, 18 cm, 21 cm; voltage = 10 kV, 15 kV, 20 kV; and the mass flow rate was adjusted on 1 ml/h, 1.5 ml/h, and 2 ml/h.
Characterization of nanofibers
The morphology and diameter of the nanofibers were analyzed by scanning electron microscopy (SEM) (JEOL, 6400). Samples were sputter coated with gold at an accelerating voltage of 15 kV. The diameter of the nanofibers was measured using microstructure measurement software. Differential scanning calorimetry (DSC) measurements were conducted under nitrogen atmosphere (Mettler–Toledo DSC 821e, Switzerland). The samples were heated from −100 to 300℃ at a rate of 10℃/min. The chemical structures of nanofibers and curcumin powder were characterized with Fourier Transform Infrared Spectroscopy (Thermo Nicolet FTIR, Nexus 670). In order to determine the wettability of nanofibers, the contact angle of nanofibers was measured by a video contact angle system (Sony colour video camera, Japan). Five samples were used for each test and the average value was reported. The droplet was set at 0.5 μl.
Biodegradability
PCL/Gel/CUR nanofibers were placed in 24-well plate containing 1 ml of a PBS (pH 7.4) in each well and were incubated in vitro at 37℃ for 15 and 30 days. After this period, SEM pictures were used to observe any change in the morphology of nanofibers.
Antibacterial activity
In vitro antibacterial activity of nanofibers was tested against methicillin-resistant staphylococcus aureus (MRSA) and extended spectrum β lactamase (ESBL), respectively, Gram positive and Gram negative bacteria by broth dilution method. Various dilutions were prepared in nutrient broth. The media was then inoculated with freshly prepared bacterial suspension. The inoculated tubes are incubated at 37℃ for 24 h. After incubation, the cultures were diluted several times. An amount of the diluted culture was then plated on Mueller Hinton Agar plates and incubated at 37℃ for 24 h. A blank sample, not treated with CUR, was used as control. After incubation, the number of colonies grown in each plate was counted. All the steps were carried out in aseptic conditions. The antibacterial activity was then calculated using the following formula:
Electrospinning was conducted under various conditions. The effect of electrospinning parameters on the nanofibers diameter was studied.
Statistical analysis
All data presented are expressed as mean standard deviation (SD). Statistical analysis was carried out using single-factor analysis of variance. A value of p ≤ 0.05 was considered statistically significant.
Results and discussion
Effect of applied voltage
Nanofibers prepared from the polymer blend solutions under different applied voltage changed from 10 kV to 20 kV. The mean nanofibers diameters were 123 nm, 127 nm, and 3.3 µm for 10 kV, 15 kV, and 20 kV, respectively.
With increasing the voltage, it was expected to obtain fibers with lower diameters due to the electrostatic repulsive forces on the jet and as a result, the jet is stretched and a narrow diameter is concluded [29]. Other researchers claimed that there is more polymer ejection when higher voltages are implemented and this assists the formation of a larger diameter fiber [29, 30]. In our study, with the increasing of applied voltage, for all the samples, increase of diameter was observed. When the applied voltage was 15 kV, uniform fibers with smooth surface were obtained. However, gelatin is a polycathionic natural polymer and acetic acid also encompasses acidic groups. The upshot of these ion groups with the implementation of a high voltage results an increase in diameter of nanofibers. When the voltage increased to 20 kV, due to high repulsion charges on the surface of the jet and electrical field, there was a uniformity in the distribution of charge on individual fiber strands. This resulted in production of nanofibers with uneven and much thicker diameter. Therefore, 20 kV was not a suitable voltage to obtain smooth and uniform nanofibers.
Figure 2 shows the SEM images of PCL/Gel/CUR nanofibers with 10 kV, 15 kV, and 20 kV applied voltages.
SEM images showing effect of voltage on morphology and diameter of nanofibers, (a) 10 kV, (b) 15 kV, (c) 20 kV.
Tip to collector distance
Electrospinning was carried out by varying different parameters (Table 1). Three different distances were applied: 10 cm, 18 cm, and 21 cm. The mean diameter of nanofibers produced at constant flow rate was 0.5 ml/h and applied voltage of 15 kV, but various distances showed an increasing trend from 118 nm ± 22% to 95 nm ± 20% for 10 cm and 18 cm. However, when the distance increased to 21 cm, the mean diameter of nanofibers decreased to 119 nm. Doshi and Reneker [31] and Megelski et al. [32] also had similar observations for other polymer systems. This is due to the fact that when the distance is increased from 10 cm to 18 cm, the solvent in the jet has more time to evaporate before it reaches the collector, therefore the diameter is reduced. When the distance is 21 cm, due to the reduction of electrostatic force on the ejected jet or in other words, the charge per unit distance, the jet, and the resulting nanofibers are not stretched enough due to the whipping of the nanofibers strand. One may conclude that there is a limit for increasing distance for reducing the nanofiber diameter. The main point may not be the total applied voltage per unit length of the distance between the nozzle and the collector. The change is observed in Figure 3.
Change in diameter of nanofibers with the change in distance.
Rate of electrospinning
Electrospinning parameters used in this study.

Effect of mass flow rate on the mean diameters of nanofibers produced with 15 kV and 15 cm.

Thermal analysis
The DSC thermograms of raw CUR powder, Gel, PCL, and PCL/Gel/CUR nanofibers are shown in Figure 4. An endothermic peak for gelatin nanofibers appeared at 100℃. This peak is due to the loss of water from gelatin nanofibers. Raw CUR powder shows a significant peak at 167℃ corresponding to the melting temperature of CUR. A sharp endothermic peak is observed at 60℃ that shows the melting point of PCL. In the PCL/Gel/CUR mixture, the melting points of PCL and CUR have respectively reduced to 51℃ and 133℃. The broad peak 90–100℃ indicates the loss of water in Gel.
DSC thermograms of CUR powder, Gel nanofibers, PCL nanofibers, and PCL/Gel/CUR.
FTIR
FTIR was used to investigate interactions between functional groups of Gel and CUR. Proteins consist of amino acids, which have nine characteristic FTIR absorption bands [33]. Figure 5 represents the infrared spectra. The protein bands appeared at approximately 1652 cm−1 due to C = O stretching vibrations, N–H bending at 1540 cm−1, N–H stretching at 3323 cm−1.
FTIR spectrum, (a) gel nanofibers, PCL nanofibers, CUR powder and (b) PCL/Gel//CUR nanofibers.
The amid I band is attributed to a random coil and α-Helix conformation of Gel [34]. For CUR, the characteristic bands appeared at 3400–3505 cm−1 due to phenolic O–H stretching, stretching vibrations at 1602 cm−1, carbonyl group C = O at 1627 cm−1 [22], C = C vibrations at 1511 cm−1, C–O–C stretching at 1154, 2945, 2867, 1294, 1240, 1731, and 1173 cm−1 are the characteristic peaks for PCL which are respectively: CH2 asymmetric stretching, CH2 symmetric stretch, carbonyl stretching, C–O–C asymmetric stretching, C–O and C–C stretching, symmetric C–O–C stretching. The characteristic peaks in PCL/Gel/CUR nanofibers were observed as follows: CH2 asymmetric stretch at 2929 cm−1, 2865 cm−1 CH2 symmetric stretch were both reduced. But C–O–C asymmetric stretching at 1240 cm−1 did not change. According to previous studies [35], PCL and Gel are chemically bonded. This leads to the orientation of NH2 and COOH functional groups to the surface of nanofibers. Bands at 1651 cm−1 and 1538 cm−1 show amid I and amid II bands of Gel which both reduced. 500–1085 cm−1 indicates CUR.
Contact angle
Contact angle measurement of nanofibrous samples.

Note: PCL, poly(caprolactone); CUR, curcumin.
Evaluation of biodegradability
The morphological changes in PCL/Gel/CUR nanofibers after 15- and 30-day period are shown in Figure 6. Slight degradation is observed after 15-day period, but nanofibers are more swollen after 30 days. PCL has a slow degrading rate [36] and this is favorable in wounds, like diabetic wounds, that have a long healing process. With increasing CUR in the PCL/Gel solution from 1 to 6%, the biodegradability of produced nanofibers did not change significantly. But from SEM images, it appears that the presence of CUR somehow delayed the degradation of nanofibers.
SEM images of nanofibers showing biodegradability after a 15-day and 30-day period.
Antibacterial activity
Antibacterial activity against MRSA and ESBL, control, and samples containing CUR (average CFU/ml).

Note: MRSA, methicillin-resistant staphylococcus aureus; ESBL, extended spectrum β lactamase; CUR, curcumin.
Conclusions
In this paper, CUR was used as herbal drug and was loaded on PCL/Gel/CUR nanofibers. The feasibility of electrospinning nanofibers in acetic acid was investigated. Addition of CUR, applied voltage, and distance were the different parameters applied to the system. The increase of voltage had a major effect on the diameter of nanofibers. As the voltage increased, the diameter drastically increased. This can be helpful in order to easily obtain different diameters proportionate to the application. Contact angle measurements showed that the addition of CUR decreases contact angle very much and creates a hydrophile surface. After a 15 - and 30-day period of incubation in PBS, with increasing CUR in the PCL/Gel solution from 1 to 6%, the biodegradability of produced nanofibers did not change significantly. But from SEM images, it appears that the presence of CUR somehow delayed the degradation of nanofibers. This is beneficial in wounds that have a long healing process. Evaluation of the antibacterial tests against MRSA and ESBL, which are both very dangerous pathogens that are difficult to cure, revealed that these nanofibers have great antibacterial properties.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

