Abstract
Today, with substantial global demand from patients suffering from wounds and burns, the wound care sector has gained great deal of interest in medical industry. Herein, coaxial electrospun poly(caprolactone)/poly(vinyl alcohol) core–shell nanofibers incorporated with Thyme extract in the core structure were prepared using coaxial electrospinning, and the effect of various operational parameters such as polymer concentration, applied voltage, flow rate, distance, and Thyme concentration on nanofiber diameter were studied. Physical and mechanical properties of the nanofibers were determined by analytical techniques. The results revealed that desired nanofibers with uniform surface morphology and acceptable tensile strength could be obtained at applied voltage of 15 kV, needle tip-target of 13 cm, core flow rate of 0.2 mL/h, and shell flow rate of l mL/h. Moreover, MTT assay shows that the nanofibers are highly biocompatible regardless of Thyme encapsulation. Antibacterial activities of the prepared core–shell nanofibers were also examined against two bacteria—gram-positive Staphylococcus and gram-negative Escherichia. Encapsulation of 5% (w/v) Thyme extract concentration in the core–shell poly(caprolactone)/poly(vinyl alcohol) nanofibers led to high antibacterial activity of the produced nanofibers.
Introduction
Textile materials are used in a wide range of applications such as traditional application, structural reinforcements, filtration, membranes, and medical application. Nanofibrous textile materials because of their special characteristics have found potential applications in a wide range of medical applications including tissue engineering, drug delivery systems, and woundcare. Considering the rise in the number of accidents worldwide, the demand for wound dressing has remarkably increased. Numerous therapeutic approaches such as autografting, using skin replacement products like various hydrogels, foams, and injectable scaffolds have been applied for treatment of either complicated or flesh wounds [1,2]. Most surgical techniques have not been efficient because of limitations such as lack of available donor sites and structural integrity and need for multiple surguries. Recently, nanofibers have been emerged as a potential alternative to be widely used in wound care and facilitate tissue cell activities for efficient treatment of partial and full thickness wounds [3]. The reason behind the increase in demand for nanofiber materials is their fascinating characteristics such as superior mechanical properties, high surface area, high mass-to-volume ratio, tunable surface functionalities, and tiny interfibrous pore size with high porosity compared to other forms of the products [4,5]. In general, a desirable nanofiber wound dressing should be biocompatible, nonallergenic, and cost effective. However, it should prevent infection, remove blood and exudates, and have suitable mechanical and physical properties depending on the wound type [3,6]. Several studies have examined the encapsulation of antimicrobial agents and growth factors in nanofiberous wound dressings to hamper infections [7].
One of the promising and cost-effective technologies to produce sub-micrometer fibers is by forcing a polymer melt or solution with an electric field through a spinneret called as electrospinning [8–10]. Polymer solution properties, electrospinning setup, and environmental properties are variables, which can influence the formation of superfine fibers with diameters ranging from a few micrometers down to tens of nanometers and their morphology [11]. Type of collector used and the speed of rotating collector can result in aligned or randomly oriented nanofibers [12].
Electrospun nanofibers have been shown to be used as scaffolds due to their similarities with the extracellular matrix (ECM) in terms of chemical and physical structure, resulting in enhancement of cell attachment, proliferation, and differentiation [13]. Moreover, nanofibers created by electrospinning can be designed to serve as delivery vehicles by encapsulating drugs into them. Certain biological compounds released by nanofiber scaffolds are antibiotics, proteins, and growth factors [14]. The success in design of these nanofiber scaffolds can be obtained either via direct mixing of drugs into the polymer solution prior to electrospinning [15], or through the exerting of core–shell electrospinning known as coaxial electrospinning, which is an elaborated method of generating nanofibers with an organized core–shell structure [16,17].
Coaxial electrospinning is an attractive process which contains a concentric spinneret that can accommodate two different solutions and result in nanofiber structures with high quality and improved functionality [14]. During the coaxial electrospinning, the core solution, which most commonly consists of diverse bioactive substances including antibiotics, drugs, DNA, and proteins, is injected through the inner needle, whereas the shell solution composed of a spinnable polymeric material is injected through the outer needle, and under the effect of electric field at the tip of capillary, the liquid jet can be transformed into the conical shape [18].
Different biocompatible and biodegradable polymers have been electrospun into nanofibers and have indicated high potential for being used as effective wound dressing patches. Poly(ɛ-caprolactone) (PCL) as an aliphatic polyester and poly(vinyl alcohol) (PVA) are biocompatible, nontoxic, and high potential biomaterials commonly used as a controlled drug releasing vehicle and scaffold due to their good tissue compatibility and excellent electrospinng ability [19]. PCL as a semicrystalline polyester with high compatibility with various types of polymers and low melting point is easily processable and have good mechanical properties [20]. However, its hydrophobic property, which can affect the cell attachment and its degradation, limits its application as a scaffold in tissue engineering. The hydrophobicity of PCL can be tailored by blending it with an appropriate hydrophilic polymer [21,22].
Suganya et al. [19] fabricated PCL/PVP nanofibers containing herbal drug and evaluated its bactericidal property. They showed that the herbal drug encapsulated in nanofibers have antibacterial properties and can be implicated in drug delivery applications and used as wound dressings to protect wounds against common infections. Zhao et al. [23] prepared biodegradable core–shell fibrous scaffold with PCL comprising the core structure and gelatin surrounding the surface of the PCL fibers. The resulting core–shell fibrous scaffolds showed higher mechanical properties compared to single component scaffold, and they were capable of stimulating cell attachment and proliferation. PVA is a hydrophilic polymer with hydroxyl groups on its molecular chains, which enable it to be modified to attach growth factors and other biomolecules [24]. Song et al. [25] fabricated electrospun PVA/Col/HA nanofibers and indicated that the impregnation of hydroxyapatite and collagen in PVA matrix can remarkably enhance the nanofiber stability and mechanical properties as well as cell adhesion and proliferation. Yao et al. [26] and Koski et al. [21] studied the application of electrospun PVA nanofiber scaffolds as ultrafine separation filters and biodegradable mats. Ranjbar et al. [22] produced nanofibers from PVA/gum tragacanth; they showed that fabricated nanofibers are good candidates for application in wound healing. Song et al. [14] fabricated electrospun PCL/PVA core–shell nanofiber, incorporated with drugs, and found that nanofibers with core–shell structure have more controllable release in comparison with the blended ones.
Active components such as antifungal, growth factors, antibiotics, and other therapeutic compounds can be easily encapsulated into the multifunctional bioactive nanofibrous dressings via a co-spinning process [27]. These achievements could be gained by adding drug or other therapeutic compounds into the prepared polymer solution either before electrospinning or via application of coaxial electrospinning [14].
It is demonstrated that essential oils extracted from plants, seeds, and leaves are volatile in nature and have antifungal, antibacterial, and antioxidant properties because of the presence of biologically active compounds including Thymol and carvacrol [28]. Thyme is an aromatic plant, which is rich in Thymol and carvacrol. Figure 1 represents the chemical structure of Thymol, which is the main constituent of Thyme. The aqueous extract of Thyme leave consists of polyphenolic compounds whose major components are Thymol [29]. Not only do these compounds possess antimicrobial activity against a wide range of gram-positive or gram-negative bacteria, but also they are the main phenolic components, which give Thyme high antioxidant capacity [30,31]. It is also evident that Thymol can be applied as anti-inflammatory agent, which is crucial for efficient wound healing [32].
Chemical structure of Thymol.
Blending PCL with PVA results in a suitable biomaterial with appropriate hydrophilicity, flexibility, and sufficient mechanical strength forces imparted by cells during tissue treatment. Many studies have been done on core–shell PVA/PCL electrospun nanofibers but limited studies have considered the combination of thyme and these nanofibers as antibacterial agent for drug delivery and wound dressing. In this study, a new core–shell nanofibers based on PCL and PVA containing herbal antibacterial have been developed. PVA and Thyme extraction were combined and formed the core of nanofibers and PCL-surrounded surface of PVA fibers via applying coaxial electrospinning. The effects of electrospinning parameters (e.g. flow rate, collector distance, and applied voltage) and Thyme concentration on nanofibers’ morphology were investigated, and the optimum formulation was designed. Finally, properties of the prepared core–shell fibrous mats such as chemical properties, hydrophilicity, tensile strength, antibacterial properties, and biocompatibility were investigated.
Experimental
Materials
Poly(ɛ-caprolactone) with the average molecular weight of 80,000 Da was purchased from Sigma Company. PVA powder with molecular weight of 72,000 Da, chloroform, and dimethylformamide (DMF) were supplied from Merk Company (Germany). For antibacterial purpose, ethanolic extract of Thyme was prepared according to the method previously reported [33]. Briefly, 200 g of plant was added to 1 L of absolute ethanol followed by sealing the container with foil paper and keeping at room temperature for an overnight. Afterwards, the contents were filtered and concentrated by evaporating the solvent at 40℃ for 24 h. At the final step, the extract powder was weighed and conserved in sterile vessels.
Methods
Coaxial electrospinning of PCL/PVA nanofibers loaded with Thyme
Coaxial electrospinning set up and Thyme concentration of designed nanofibers.

Characterization
Fourier transform infrared spectroscopy analysis
Chemical characteristics of the prepared core–shell nanofibrous structures were determined using NEXUS 670 (Nicolet Company) Fourier transform infrared spectrometer. KBr pellets combined with the samples were supplied and the spectrums of samples were recorded in the range of 500–4000 cm−1.
Field-emission scanning electron microscopy
The coaxial electrospun nanofibers were firstly sputtered with thin layer of gold before SEM observation. The morphology of the nanofibers was observed utilizing field-emission scanning electron microscopy (FE-SEM) (Hitachi, Model S- 4160) at an accelerating voltage of 30 kV, and the average fiber diameter (AFD) was estimated using Microstructure Measurement Software.
Transmission electron microscopy
Transmission electron microscopy (TEM) model CM30 was used to accurately recognize the core–shell structure of coaxial nanofibers. The specimens were covered with ultrathin carbon layers and observed under an accelerating voltage of 300 kV. The average fiber diameter and distribution were determined by choosing 100 fibers from SEM images and using Measurement software.
Contact angle measurement
A sessile drop method was adopted in which deionized water was automatically dropped in the form of droplets on the surface of the fibrous specimens until equilibrium was applied to examine the effects of addition of PVA and Thyme on the hydrophilicity of the coaxial electrospun nanofibers at ambient temperature. The drop shapes on the nanofibers surfaces were imaged by Video contact analyzer, Model SSC-DC318P color, and contact angles were evaluated using Image J Software. These test was repeated for three samples with the same formulation.
Tensile strength evaluation
As an ideal wound dressing should be durable enough for handling and application. Tensile behavior of the designed nanofibers was measured using a tensile testing machine model Instron 5566 (England). The test was performed on rectangular webs with equal dimensions (30 × 5 mm2). Firstly, the thickness of the prepared samples was recorded. Then three samples of each composition were drawn until their break point with the tensile rate of 10 mm/min, and the average values were reported.
Cytotoxicity assessment
The biocompatibility of core–shell nanofibers was analyzed using sample extraction [33]. Briefly, the specimens (3 ± 0.5 cm2) were sterilized using UV rays and incubated in 1 mL culture medium for 7 days at 37℃ in a 5% CO2 atmosphere. Pure culture medium maintained in similar condition was chosen as a control. After the incubation period, the mediums were collected for use in viability assays. Rabbit fibroblast cell lines L929 were supplied by National Cell Bank of Pasteur Institute of Iran, and 1 × 104 cells/well were cultured in 96-well microtiterplate in Dulbecco’s Modified Eagles’ Medium (DMEM, GIBCO, Scotland)/Ham’s F12 (GIBCO, Scotland) supplemented with 10% fetal bovine serum (FBS, Nanobioarray, Iran) and left overnight. Subsequently, the culture medium in each well was replaced with 90 µL of the sample extracts plus 10 µL FBS. After 24 h, the mediums were aspirated and to quantify metabolically active cells, 100 µL of 0.5 mg/mL MTT (3-(4, 5-dimethyl-2-thiazolyl)-2, 5-diphenyl-2 htetrazolium bromide) (Sigma USA) was added into each well, followed by incubation for 4 h at 37℃. Finally, the purple formazan crystals were dissolved by 100 µL of isopropanol (Sigma USA) per well, and optical density (OD) of the colored solutions was measured at 540 nm using ELISA reader (STAT FAX 2100, USA). Three samples from each formulation were tested for reporting best results of cytotoxicity assesment. The cell viability (%) was measured from the following equation:

Antibacterial activity evaluation
Quantitative evaluation of the degree of the antibacterial activity of electrospun nanofibers was examined according to the AATCC method against two bacteria namely Staphylococcus, (gram-positive) and
Statistical analysis
All quantitative results were reported as mean ± standard deviation, and each experiment was performed in triplicates. One-way analysis of variance (ANOVA) was applied to compare various groups and
Result and discussions
Effects of process and material parameters
Applied voltage
During coaxial electrospinning process, the jet stability and fiber morphology remarkably depend on the applied electrical voltage. Figure 2 shows the SEM images of coaxial electrospun nanofibers fabricated from 11% PCL and PVA containing 5% Thyme under various applied voltages of 8, 13, and 15 kV with constant core and shell flow rate of 0.2 and 1 mL/h, respectively, and tip-to-target distance of 13 cm. As illustrated in the figure, an increase in the applied voltage from 8 to 15 kV causes decrease in the average fiber diameter from 750 ± 86 to 201 ± 22 nm. Hence, the voltage of 15 kV in which the formation of thinner fibers and more stable compound Taylor cone was occurred, found to be the optimum amount and used in subsequent stages. The results are consistent with those obtained in similar investigations. It is reported that the enhancement of the applied voltage decreases fiber diameter owing to the increased electrostatic force on solution jet leading to the narrower fiber formation [8]. Moghe et al. [34] also showed that in coaxial electrospinning, there is a small range of applied voltage in which increase in strength of the electric field causes decrease in both the inner and outer diameters of the electrospun structures. However, above the critical amount, increase in the voltage of the applied electric field to exceed that required for the given material, results in separation of jets from the shell and core and no stable Taylor cone and core–shell fibers formation is occurred.
FE-SEM image of PCL/PVA/Thyme core–shell nanofibers under different voltages: (a) sample 1 (8 kV), (b) sample 2 (13 kV), (c) sample 3 (15 kV).
Distance between needle tip and collector
Another parameter affecting the fiber size is the distance between tip and the collector. As demonstrated in Figure 3, diameter of samples 4, 5, and 6, decreases on increasing the distance between tip of the needle and collector (Table 1). It could be due to the increase in the remained time for the solvent evaporation of the created jet [35,36]. It has been reported that decrease in tip–collector distance reduces the time for the solvent evaporation and enhances the strength of electric field, resulting in acceleration of created jet towards the collector. As shown in Figure 3, further reducing the distance in sample 4 (7 cm) led to the formation of beads on fibrous structures, which can be due to the assembling of the excessive solvent as a result of the decrease in residual time for solvent evaporation [36]. In this study, the distance of 13 cm (sample 6) was selected as an optimized distance based on better evaporation of solvent through wider area between the needle and collector.
FE-SEM image of PCL/PVA/Thyme core–shell nanofibers with different distances between needle tip and collector: (a) sample 4 (7 cm), (b) sample 5 (10 cm), (c) Sample 6 (13 cm).
Core flow rate
Many studies have shown that flow rate can directly affect the dimension of core–shell fiber in coaxial electrospinning [34]. It has been reported that in constant operating conditions and shell flow rate, there is a range of core flow rate in which sustained compound Taylor cone and core–shell construct can be obtained. As illustrated in Figure 4(a) and (b) for respectively samples 7 and 8 and also as presented in Table 1, increasing the core flow rate from 0.2 to 0.5 mL/h resulted in increase in average fiber diameter of the composite nanofibers from 215 ± 37 nm to 310 ± 54 nm, size distribution, as well as formation of beads on nanofibers’ structure. In a similar investigation, Zhao et al. [37] indicated that if core flow rate is higher than the optimum value, lack of uniformity in size distribution can be observed due to the removal of excess solution from the needle tip. Moreover, in extremely high core flow rates, the inner cone loses its characteristic shape due to the lack of the viscous drag exerted by the shell solution to uniformly encapsulate fast moving core solution [38,39]. As a result, the core flow rate should generally be lower than that of the shell. In this investigation, the core flow rate of 0.2 mL/h was chosen as an ideal amount because of achieving sustained fibrous structure with high uniformity in size distribution.
FE-SEM image of PCL/PVA/Thyme core–shell nanofibers with different core flow rates: (a) sample 7 (0.5 mL/h), (b) sample 8 (0.2 mL/h).
Thyme concentration
To study the influence of Thyme concentration on nanofibers’ morphology, samples 9, 10, and 11 were selected for comparison, and it was found that increase in the Thyme content in the core solution containing 5% (w/v) PVA from 0 to 10 % increased the average fiber diameter from 235 ± 44 nm to 746 ± 75 nm as well as increase in amount of entanglements (Figure 5). The obtained results were consistent with those reported by Sedri et al. in a similar study in which they evaluated the effects of Thyme concentration as herbal medicine on morphology of nanofibers based on chitosan/polyethylene oxide [40]. Increase in the amount of Thyme leads to increase in viscosity of the polymer solution, resulting in decrease in processability of the solution as well as enhancement of entanglements. In the present work, core solution containing 5% (w/v) Thyme was selected as an optimum solution due to the resulting uniform nanofibrous structure with small amount of entanglements.
FE-SEM image of PCL/PVA/Thyme core–shell nanofibers with different Thyme concentrations: (a) sample 9 (0% (w/v)), (b) sample 10 (5% (w/v)), (c) sample 11 (10% (w/v)).
Transmission electron microscopy
As demonstrated in Figure 6, the formation of distinguishable core–shell nanofiber structure was observed by TEM. A sharp boundary between the shell and core fibers was observed by TEM in Figure 7.
Representative TEM images of PCL The FTIR spectra of pure PCL, Thyme, and their core–shell nanofibrous constructs with incorporation of 0 and 5% (w/v) Thyme in the core containing 5% (w/v) PVA.

FTIR spectroscopic analysis
Figure 7 presents the FTIR spectrum of pure PCL, Thyme, and their core–shell nanofibrous constructs with incorporation of 0 and 5% (w/v) Thyme in the core containing 5% (w/v) PVA. The characteristic bands in FTIR spectra of electrospun PCL nanofibers appeared at 1234, 1730, 1274 cm−1 for C–O, C=O, and C–H stretching, respectively; 1431 cm−1 for C–H bending and 2852 cm−1 for symmetric CH2 stretching. As shown in Figure 7, the FTIR spectrum of Thyme has a strong absorption at 3400 cm−1 corresponding to O–H (phenol) stretching. The absorptions appeared at 2920, 1615, 1516, and 1046 cm−1 are related to C–H band stretching, C–C deformation mode band, aromatic C–C stretching, and C–O alcohol stretching, respectively. Moreover, the appeared peaks at 1377 and 1257 cm−1 correspond to aryl O–H [41].
PCL/PVA core–shell electrospun nanofibers showed all characteristic broad absorbing of PCL and some prominent peaks of PVA. It can be due to the encapsulation of PVA into PCL, which masked some functional groups of PVA and prevent appearance of all distinguishing bands of PVA. Moreover, FTIR spectrum of PCL/PVA/Thyme core–shell nanofibers was similar to that of PCL/PVA, and certain characteristic bands of Thyme and PVA were not evident. This phenomenon can be for the incorporation of PVA containing Thyme into the shell layer. In addition, overlapping of the bands corresponding to O–H groups in PVA and Thyme avoids them to be distinguishable.
Contact angle
Water contact angle was measured in order to evaluate effects of PVA and Thyme on electrospun nanofibers’ hydrophilicity. The water contact angle of PCL was 110 ± 5.3° and that of PCL/PVA coaxial electrospun nanofibers was 83 ± 4.03°. The results showed that incorporation of PVA into PCL nanofibers reduced hydrophobicity of the final core–shell structure. In general, it has been reported that the presence of hydroxyl groups (O–H) and hydrogen bands in PVA makes it highly hydrophilic, so incorporation of PVA in to the hydrophobic polymer such as PCL can result in increase in the hydrophilicity of the final structure [24,42]. The obtained results were consistent with similar research conducted by Kim et al. in which they demonstrated that the hydrophilicity of PCL mats was significantly improved by double-spinneret electrospinning. Moreover, Neethu et al. showed that addition of PVA to PCL in semi-IPN compositions remarkably increased the hydrophilicity of the prepared constructs. In addition, incorporation of Thyme into the core structure containing PVA (PCL/PVA/Thyme) contributed to higher reduction in the water contact angle values (76 ± 3.3°) than that of PCL/PVA coaxial electrospun nanofibers. These results could be mainly due to the presence of polar and phenolic components such as Thymol and carvacrol in Thyme extract that make it water soluble and hydrophilic plant.
Tensile strength
Tensile strength, elongation, and Young’s modulus were measured and calculated to investigate the influences of PVA and Thyme on the coaxial nanofibrous structures. It was expected that incorporation of PVA into the core structure of designed coaxial nanofibers led to increase in the tensile strength of the prepared structures due to the high mechanical properties of PVA in comparison with that of PCL. It could be postulated that encapsulation of PVA into PCL reinforced the resulting core–shell nanofibers and under mechanical forces, high portion of stress applied will be endured by polymer part with higher strength (PVA). Hence, as illustrated in Figure 8, for a constant deformation value, higher forces should be exerted on the core–shell structures, and break occurred at higher tensile strain compared with that of pure PCL nanofibers. The values obtained are in accordance with those found by other researchers [43,44]. Moreover, the results showed that incorporation of PVA into PCL caused final core–shell nanofibers to be more fragile than pure PCL, and elongation at break was lower than that of PCL. On the other hand, as represented in Figure 8 and Table 2, encapsulation of 5% (w/v) Thyme into the designed core–shell nanofibers caused reduction in tensile strength compared to PCL/PVA core–shell nanofibers without Thyme. Similar to the tensile strength, Young’s modulus and elongation at break also decreased. It has been reported that addition of Thyme increases pore sizes and porosity of the designed nanofibers. Hence, dense constructs are stiffer and have higher mechanical properties than porous structures [6]. These results confirm outcomes of the report by Altiok et al. and Cagri et al. who had concluded earlier that similar to additives other than crosslink agents, addition of Thyme lowers tensile strength value of nanofibers [6,45]. Moreover, dressing must be able to withstand the load applied by cells and may be strong enough to resist the forces of the body movement or outer environment. The strength of fabricated nanofiber is 1.08 MPa and this data show that this mat is very strong to withstand the applied forces, on the other hand the reported strain for this scaffold is 143%, so this mat can elongate enough. In other papers [6], data for strength and strain are more or less than these data and there is no specified data for this application.
Typical tensile stress–strain curves of pure PCL and PC/PVA, PCL/PVA/Thyme nanofibers membranes. Mechanical properties of nanofibers.

Cytotoxicity assay
MTT assay in which metabolically active cells react with a tetrozolium salt to produce a soluble formazan dye was applied in order to examine the biocompatibility of the designed electrospun nanofibers. Fibroblast cells are frequently used in cytotoxicity test. Experiments were performed at least three times. As illustrated in Figure 9, all specimens showed cell viability of more than 90% indicating no serious cytotoxic effects on cells exposed to 7 days nanofibers’ extracts. Moreover, it can be observed that there is no significant difference between coaxial electrospun nanofibrous structures containing Thyme (PCL/PVA/Thyme) and PCL/PVA core–shell nanofibers. The obtained results showed that the designed core–shell nanofibers consisting of Thyme in the core structure with no toxicity are proper to select for other biological investigation like in vivo investigations.
MTT assay of the cell viability after the cell was treated with extracts of electrospun PCL, PCL/PVA/Thyme nanofiber membranes.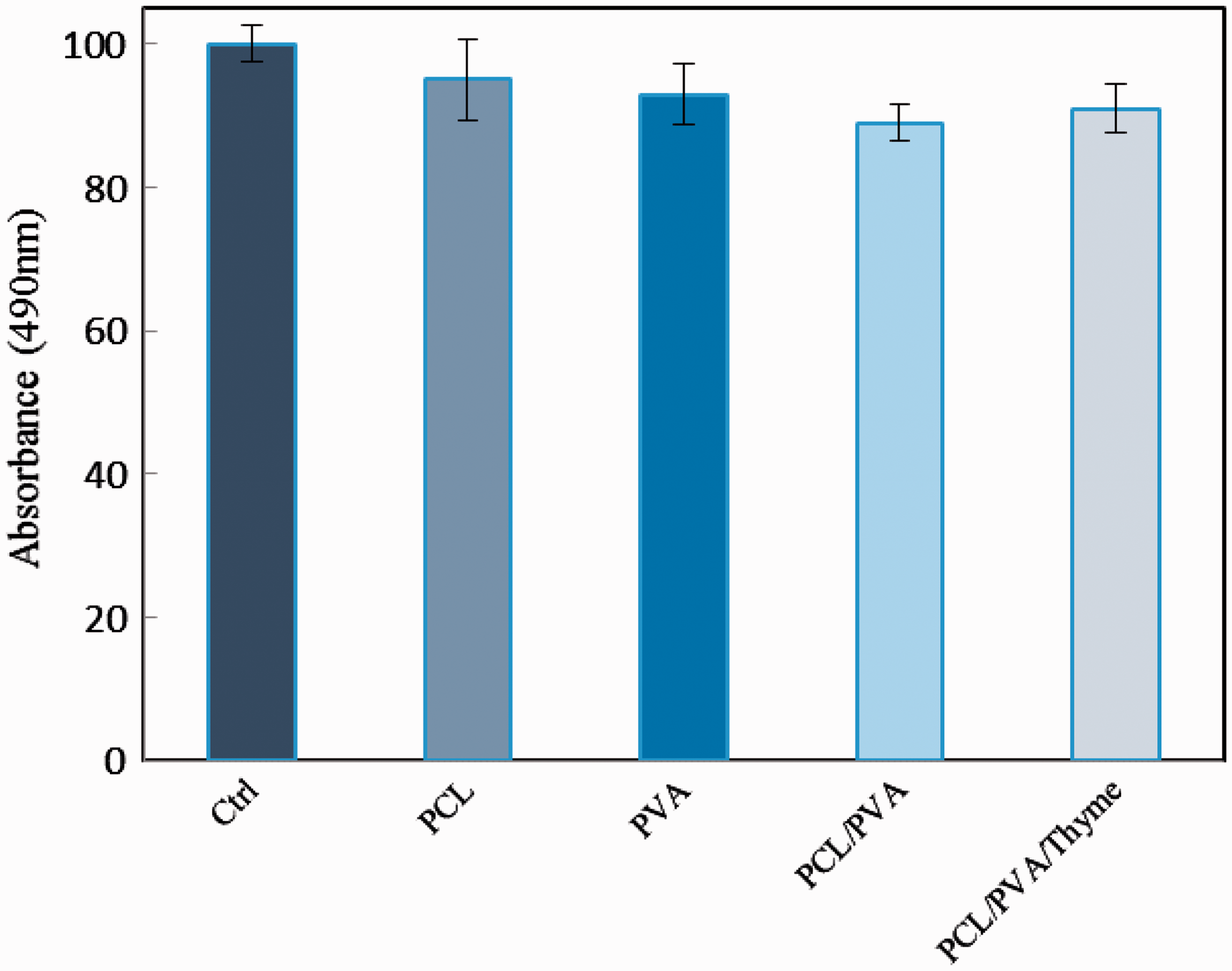
Antibacterial evaluation
The results of anti-microbial tests are shown in Figures 10 and 11. In this test, PCL/PVA core–shell nanofiber were considered as the control sample. According to the results, it is deduced that designed nanowebs, which are treated with Thymes, have an antibacterial activity. As shown in the figures, Thyme inhibits the growth of bacteria in all three concentrations of bacteria in the plates, so the antibacterial activity is more than 99%. Studies indicate that phenolic compounds in Thyme structure are responsible for antibacterial activities [46]. These compounds are Thymol and carvacrol where the antibacterial efficacy of carvacrol is explained by Didry et al. [47]. Hosseini et al. [48] also showed that the Thyme essential oil exhibited the highest antimicrobial efficacy against the tested gram-positive and gram-negative bacteria.
Antibacterial activity against Antibacterial activity against 

This indicates that even the presence of Thyme in the core is able to contribute for antibacterial properties for the nanofibers structure.
Conclusion
In the present work, Thyme-incorporated PCL/PVA core–shell nanofibers were prepared by coaxial electrospinning technique under optimum operating conditions. The effect of Thyme extract addition into the core structure of PCL/PVA coaxial eletrospun nanofibers was investigated for the physical, mechanical, and antibacterial properties of the designed nanofibers. Hydrophilicity assessment indicated reduction in contact angle as a result of incorporation of PVA and Thyme in the core structure, and it was mainly due to the presence of phenolic and polar groups in the back bone of PVA and components of Thyme extract. Moreover, tensile test showed that addition of PVA with higher tensile strength than PCL in the core structure increased the mechanical properties of the core–shell nanofibers; however, encapsulation of Thyme in the core caused decrease in the tensile strength due to the high porosity and pore size compared to PCL/PVA, but it was still stiffer than constituents of nanofibers. The produced electrospun constructs were also biocompatible and incorporation of Thyme into the nanofibrous structures introduced higher antibacterial activity compared to that of PVA/PCL nanofibers. As these attributes are the crucial properties for ideal wound-healing materials, Thyme extract encapsulated nanofibers fabricated by our system could be used as a potential wound-healing material as well as promising materials for treatment of surfaces that contain pathogenic microorganisms.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
