Abstract
Polyethylene terephthalate multifilament fabrics used as filtration and operating room textiles possess through-thickness pore channels at the yarn intersections (meso-pores). These pore channels pose a risk for the penetration of contaminated fluids and particles. The size of pore channels may be reduced by high-density weaving. However, this leads to reduced drapability and thus to degraded application properties of the fabric. To satisfy the requirements without impeding the physiological properties of the textile, fluid- and particle-tight fabrics are developed. This was realized by partial immobilization of functionalized micro particles into the meso-pores. A reduction of the pore size without complete pore-closure is achieved by establishing a net-like particle structure in the meso-pores. To match the requirements of intensive use, permanent particle-bonding to the fiber surface is necessary. This can be achieved by suitable polyethylene terephthalate fabric surface-modification, dependent on the particle functionalization. The investigations have shown that functionalized particles establish a very good inter particle bonding as well as to the fiber surface. An increased permanent bonding can be realized by a modification of the fabric surface which is tuned to the functionalization of the particle.
Keywords
Introduction
The use of functionalized micro- and nano-particles may be considered as an important area in the field of thin-layer technology. Due to the large bandwidth of modification techniques and particle systems it is possible to generate particles with novel functionalities. By using functionalized micro- and nano-particles as coating substrates, these functionalities may be transferred onto textiles. Hence, it is possible to create superhydrophobic surfaces [1] or antibacterial properties by the immobilization of silver nano-particles [2], respectively. The cores of these novelties are smallest particles which may be applied on textile materials by thin-layer technologies (sol-gel process, plasma, spraying, etc.). The low particle diameter enables the creation of layer-thicknesses in the micro-/nanometer range. Compared to fully coated materials, thin-layer techniques ensure the preservation of textile-physiological properties like drapability or breathability. Thus, those techniques provide good preconditions to improve the barrier function of polyethylene terephthalate (PET) fabrics without reducing their wearing comfort.
PET fabrics, which could be used as operating room (OR), clean room, or protective textiles are expected to exhibit a barrier against particle-loaded fluids which serve as carrier media for microorganisms [3]. The barrier properties of the fabric are defined by the construction-dependent pore morphology (meso- and micro-pores) [4] as well as the surface properties. At the same time, high wearing comfort is necessary for the use as safety garments. Therefore, it is very challenging to meet those opposing requirements (barrier and comfort) in one textile structure. Through partial finishing in the yarn intersections it is possible to improve the barrier effect in these critical areas. The textile-physiological properties are secured through the retention of the micro-pores (pores between the single filaments). Compared to nano-scaled particles which are mainly deposited on the fiber surface, micro particles are more likely to be placed between the single filaments, which positively support partial coating in the yarn interstice [5]. Combined with an application technique based on vacuum-filtration which allows partial and aimed particle application in the meso-pores, porous structures composed of micro particles have successfully been created in the meso-pores without pore closure [6]. The effectiveness of the finishing was evaluated by extensive physiological and physical characterization of the woven fabrics before and after particle application, supplemented by a comparative study of selected textile-physiological fabric, properties (resistance to water vapor permeability, air permeability and bending stiffness) with highly dense barrier fabrics in [6]. Although reduced air permeability and increased bending stiffness was observed, the textile-physiological properties were still superior to those of standard high-density woven fabrics, thus ensuring high wearing comfort. The resistance to water vapor permeability stayed below the guidance value of
Furthermore, plasma surface treatment is a widely used technique for textile finishing [8,9]. The choice of different reactive gases makes it possible to create many reactive groups at the surface, which enable a functionalization of the textile surface towards improved wetting behavior. Another technique to activate textile fabrics is alkali treatment. This is a wet chemical pre-treatment in which the textile is treated by alkaline solution. Alkaline hydrolysis causes splitting of the ester groups present in PET. This results in elevated surface energy levels and thus in more likeliness to interact with other compounds [10]. Initiators functioning as adhesion agent are an additional method to support particle bonding to the fabric surface. Especially for the usage as OR-garments a solid and permanent particle-bonding is of major importance. Particles which are released from the fiber-surface and penetrate through the textile are potential carrier media for viruses and bacteria. This additional transfer potential must be avoided.
Until now there are no processes available where a partial and targeted functionalization of PET-fabrics with micro particles can be realized in the yarn intersections. The permanent binding of the particles is essential. Therefore, the current research focuses on the stability of hydrophobic properties after washing treatment of fabrics functionalized with nano-particles [11,12]. However, particle release is not considered.
Therefore, the goal of this study is the realization of PET-surfaces with permanently bonded functionalized particles. For this purpose, different particle systems are chosen. It is shown that the pre-treatment of the fabric depends on the functional groups present on the particles. The washing process is one of the strongest mechanical loads in the lifecycle of apparel fabrics. The obtained results are expected to be the basis of a novel particle coating technique for improved barrier functions of textiles which withstands the washing process.
Materials and methods
Materials
N,N′,N′,N″,N″-pentamethyldiethylenetriamine (PMDTA, Aldrich, 99%), ethyl α-bromoisobutyrate (EBiB, Aldrich, 98%), 3-aminopropyltriethoxysilane (APS, ABCR, 97%), α-bromoisobutyric acid (BiBA, Aldrich, 98%), α-bromoisobutyrylbromide (Aldrich, 98%) anhydrous dichloromethane (Fluka), triethylamine (Fluka), L-ascorbic acid (Sigma, ≥99%), copper(II) bromide (Aldrich, 99.999%), 1-ethyl-3-[3-(dimethylamino)propyl] carbodiimide hydrochloride (EDC, Pierce), tetraethyl orthosilicate (TEOS, Fluka, ≥99%), ammonia solution (Acros, 28–30% solution), ethanol abs. (VWR, 99.9%), anisol (Fluka, >99%), tin(II) 2-ethylhexanoate (Aldrich, ∼95%), styrene (Sigma, 99%), tert butylacrylate, glycidyl methacrylate (Polymer Source and Fluka) were used for the investigation of this study. Tert-butyl acrylate (tBA) was distilled under nitrogen steam prior to polymerization.
For the investigations, three particle systems are considered in more detail. The basic system is formed by untreated silica particles. In this particle system there are 1 µm sized particles, which are prepared by the Stöber method and can form a nearly ideal monodisperse particle size distribution in the selection of suitable process parameters. They also show an ideal spherical morphology over the whole size range [13,14]. Because of their good mechanical and chemical resistance, these particles are favorable for the synthesis with numerous functionalities and thus for the development of new particle systems with tunable properties. For this reason they are used as the core for two further particle systems and thus form the basis for the “core-shell” particles. The core particles are functionalized with two types of polymers, in order to provide new surface properties. The first particle system is functionalized with a shell of poly acrylic acid (PAA). The second particle system is covered by a shell of poly (glycidyl methacrylate) (PGMA). A textile fabric made from poly (ethylene terephthalate) PET microfilament yarn is used as substrate for the particle application.
Poly(ethylene terephthalate) fabric
Parameters of PET fabric.
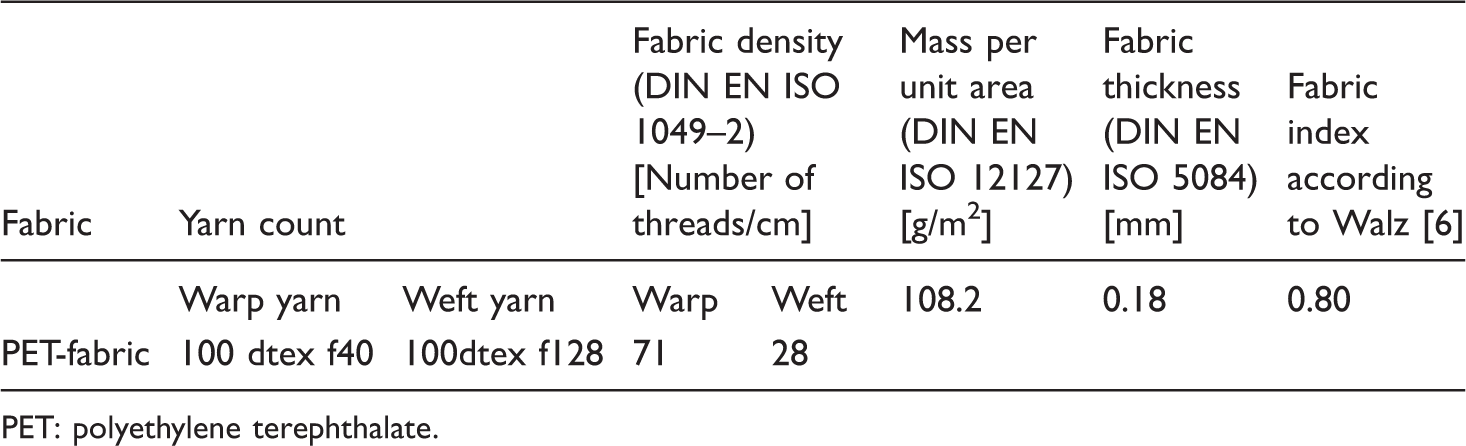
PET: polyethylene terephthalate.
Methods
Synthesis and modification of monodisperse SiO2 particles
The micrometer-sized silica particles were prepared using a multi-step hydrolysis-condensation procedure of Tetraethylorthosilicate (TEOS) in ammonia hydroxideethanol solution based on Stöber approach described in [13,14]. Briefly, TEOS was poured via sequential addition into a mixture of ethanol and ammonia solution in a polypropylene bottle using a syringe. Silica particles generated within one-step synthesis were used as seeds for the next synthesis step. Each reaction was carried out stirring at room temperature overnight (starting after the last addition of TEOS) at 600 r/min stirring speed. The final dispersion was separated by centrifugation to yield monodisperse 1 µm large silica spheres. Purified particles were dried at reduced pressure and 100℃ using a vacuum oven.
Synthesis of fully covered polymeric modified core-shell particles
In this step, dry spherical silica particles were added to the ethanol solution of 3-aminopropyltriethoxysilane (APS) and stirred. After this, particles were centrifuged and washed several times. Finally, APS-modified silica particles were dried. APS-modified particles were dispersed in anhydrous dichloromethane. Then, α-bromoisobutyryl bromide and triethylamine were added to the suspension. Particles were centrifuged and washed several times. Initiator-modified particles were dispersed in N,N-dimethylformamide (DMF). Then, respective monomer, copper(II) bromide, N,N′,N′,N″,N″- pentamethyldiethylenetriamine were added. After addition of tin(II) 2-ethylhexanoate, the flask was heated up to 70℃. The particles were centrifuged and washed several times with DMF and ethanol after termination of polymerization [15]. Previous experiments demonstrated that grafting density in this case is typically 0.1–0.3 chains/nm2. Shells of PGMA and polyacrylic acid (PAA) on the silica particles were synthesized. PGMA brushes were obtained directly by polymerization of respective monomers (Figure 1). The PAA brushes were obtained in two steps. In the first step, poly(tert butylacrylate) (PTBA) brushes were prepared by polymerization of tert butylacrylate. In the second step, PTBA brushes were hydrolyzed in PAA using methanesulfonic acid. Details of the synthesis of fully covered core-shell particles are given in [16,17,18].
Scheme of the synthesis of core-shell particles by “grafting from” approach.
Pretreatment and modification of textile fabric
Particle systems and their properties.

PAA possesses reactive carboxyl groups which permanently bond to epoxy or amino groups. Therefore, two options are checked for the generation of reactive groups on the polyester surface.
The first option is a two-step pre-treatment of the fabric with the adhesives calcium chloride (CaCl2) and dissolved PGMA. The clean and dry fabric is immersed in a 2% solution of CaCl2 and distilled water for 30 min. Thereafter, the fabric is soaked for another 30 min with a solution of toluene and PGMA. After a short drying time for about 15 min in the oven at 70℃, the functionalized PAA particles are applied. The epoxy groups on the textile, which are created by the pre-treatment, support the chemical bonding with the carboxyl groups of the PAA particles. In the next step, binding of the particles is completed by a thermal treatment at 120℃ for 60 min.
The second option for the fabric modification is a wet chemical amino-functionalization with a fleet of 25% synthetic amine tetraethylenepentamine and 0.02 g of nonionic surfactant, at pH 11. After a 15 min dip treatment in the solution, in order to finish the amino-functionalization, the textile was padded on a laboratory padder Type HVF (Mathis AG, Switzerland) at a pneumatic roller pressure of 3 × 105 Pa and with a web speed of 3 m/min [19]. The functionalized samples were subsequently dried in an oven at 70℃. The detection of functional amino groups on the fabric was done with Ninhydrin. Ninhydrin is used in the textile chemistry for the detection of primary amino groups, in particular amino acid on a fiber, and thus for the identification of protein fibers or the protein on the fiber. The presence of amino groups is indicated by a purple-red discoloration on the fabric.
The particles coated with PGMA exhibit reactive epoxy groups in their shell. An alkaline treatment of the fabric results in an ester cleavage and thereby to a formation of functional carboxyl groups on the surface. This results in a chemical interaction between both reactive groups which has a positive effect on the particle bonding. For this effect, the textile is boiled in a 30% solution of sodium hydroxide and afterwards washed with distilled water and dried at room temperature. The hydrolysis process leads to etching of the fiber surface and at the same time a formation of small craters, thus resulting in a roughened fiber surface. The roughening contributes to an increase in surface energy and supports the bonding of the functionalized particles on the fabric. Another option to generate functional groups on the fabric surface constitutes a plasma treatment via atmospheric pressure plasma (system “AS Coating Star”, Fa. Ahlbrandt System GmbH, Germany) with reactive gases like oxygen and argon. The reactive gases cause an increase in polarity and therefore in the reactivity of textile. The application of the functional groups on the fiber surface effects a stable connection between fiber and functionalized particles. An additional roughening of the fiber surface caused by the plasma treatment supports the formation of a chemical interaction which supports the adhesion of the particles. Because the plasma treatment is not long-term stable a quick riser test is done directly after the treatment. A strip of plasma-treated fabric is dipped for about four min vertically in a test solution of ink and water. The height of rise of the test solution in the textile compared to an untreated blank denotes the increase of hydrophilicity, which is a characteristic for increasing the surface energy.
The SiO2 particles are non-functionalized bare particles. These native particles contain silanol (Si-OH) groups, which can be used for further modification as well as for particle binding. The Si–OH groups are the only possibility to achieve chemical interaction between the particle and the fabric surface. The pre-treatment of the PET textile is carried out analogous to the methods for PAA and PGMA particles. The purpose is to demonstrate the necessity of particle functionalization.
Particle immobilisation and post-treatment
After cleaning and modification process of the textile, the particles are applied by dipping the fabric in the particle solution with a particle concentration of 5 wt%. The hydrophilic SiO2 and PAA-modified particles are dispersed in distilled water; for the hydrophobic PGMA-system a suspension with toluene is prepared. The binding of the PAA-modified particles on the fiber surface and their deposition on the fabric depend on the pH-value (Table 2). An increase of pH leads to changed surface charges and swelling of the PAA shell which may result in different particle-bonding performance. The pH-value is regulated to pH 2 and pH 7 by addition of hydrochloric acid [20]. After drying of the treated polyester fabric at room temperature, the chemical attachment of the particles takes place in the laboratory dryer LTE 76206 (Mathis AG, Switzerland). The functionalized polymer shells of the core-shell particles are thermally cross-linkable, which means that the thermal energy leads to inter-particle bonding and particle-fabric bonding, respectively. The thermal treatment takes 60 min, on the basis of preliminary experiments. The treatment temperature for PAA particles is 120℃, for the PGMA particles 170℃.
Testing
Thermogravimetric analysis
Thermogravimetric analysis (TGA) measurements were conducted on a TGA Q 5000IR device (TA Instruments Co., USA). The chosen measurement conditions were a temperature span up to 800℃ and measurement under air. TGA results were used for the quantification of the thickness of the polymer shell. Thickness of the grafted polymer layer was estimated based on TGA results using equation (1)
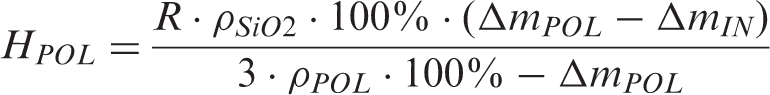
Scanning electron microscopy
The morphology of the core-shell particles and the coated fabrics was investigated using SEM technique. The SEM images were acquired on a NEON 40 EsB Crossbeam scanning electron microscope (Carl Zeiss NTS GmbH, Germany), operating at 3 kV in the secondary electron (SE) mode. In order to enhance electron density contrast, samples were coated with platinum (3.5 nm) using a Leica EM SCD500 sputter coater.
Transmission electron microscopy and cryogenic transmission electron microscopy
TEM images and cryogenic transmission electron microscopy (cryo-TEM) images were taken with a Libra 120 cryo-TEM (Carl Zeiss NTS GmbH, Germany), equipped with a Lanthanhexaborid (LaB6) source. The acceleration voltage was 120 kV and the energy filter with an energy window of 15 eV was used.
Characterization of particles
The size and pH-dependent electrokinetic measurements (via electrophoresis) of the particles in dispersion were carried out with a Zetasizer Nano ZS (Malvern Instruments Ltd., UK) and an MPT-2 autotitrator. For all measurements, the particles were suspended in a solution of 10−3 M KCl in water (10 mg/24 ml or 0.42 mg/ml). The pH of the prepared dispersion was controlled by adding 0.1 M KOH or HCl aqueous solutions. Three measurements were recorded for each sample at each pH value.
Characterization of particle application
The main aim of the particle treatment is a permanent binding of the micro particles on the fabric surface as well as an examination of the change of the fabric surface properties (water-repellent properties). Especially for surgical clothes and of course all clothing textiles, the washing process implies very high exposures of mechanical stress. Therefore, an investigation of particle binding after the washing process is obvious. For a closer study of the durability of the particle treatment, the finished textiles are washed in soapsuds at 40℃ for a total of 360 min. The analysis of the results from the washing test takes place via SEM (DSM 982 Gemini, Zeiss Germany) as well as a count of the particles in 10 ml of the used soapsuds (Abakus® mobil fluid).
The hydrophobic PGMA-modified particles additionally enable the generation of water-repellent surfaces on the textile fabric. To examine this effect, measurements of contact angle are performed by the sessile drop method by using the DSA 100 (Fa. Krüss, Germany).
Results and discussion
Characterization of polymers and functional core-shell particles
The different steps of the chemical modification were studied and characterized using complementary analytical tools such as TGA, SEM, TEM, cryo-TEM, and electrokinetic (electrophoresis).
Thermogravimetric analysis
TGA results were used for the quantification of the thickness of the polymer shell on the particle surface. It was found that PTBA shell thickness is around 30 nm. After the hydrolysis of PTBA into PAA, the shell thickness decreases down to 15 nm (Figure 2). The samples KK315 to KK316 and KK005 represent the phases of functionalization process.
Results of TGA analysis for native SiO2 as well as PAA modified core-shell particles (examples from several parallel syntheses).
Evaluation of surface morphology and TEM analysis
The SEM images show that the particles cohere to each other (Figure 3). That is the indication of the soft and thermal cross linkable polymer shell, which acts like a composite system. The thickness of the polymer shell was estimated from TEM images and is about 20 nm that correlates with TGA results.
Representative TEM (left and right) as well as SEM (middle) images of PAA-modified core-shell particle.
Swelling and pH responsive properties of functional core-shell particles
The formation of surface charges during the contact of the polyanion brushes' shells with aqueous solutions and the changes of these charges as a function of the pH of the aqueous solution were studied by electro kinetic potential measurements (Figure 4). The dependence of zeta potential on pH has a step-like character that is intrinsic to behavior of surfaces where surface charges are produced by dissociating groups. The particles with PGMA shell as well as the native SiO2 particles demonstrate a clear switching of charge: the native SiO2 particles are positively charged at low pH and negatively charged at high pH (Figure 4) and same at PGMA particles. At pH 2 the PAA particles are uncharged, otherwise they are negatively charged at high pH. These results are important for the binding properties.
Dependence of electrokinetic potential values (zeta-potential measured by electrophoresis) of SiO2 –native, SiO2-PAA and SiO2-PGMA.
The size of the particles (proved by DLS as hydrodynamic diameter and by cryo-TEM) with polymer shell also strongly depends on pH value. In PAA-coated core shell particles the PAA shell is swollen and the hydrodynamic diameter of the PAA-modified particles is larger at high pH values (pH 7 and pH 10) as seen in Figure 5. That is due to electrostatic repulsion of negatively charged carboxyl groups (Figure 6). The opposite scenario is observed at low pH (pH 2): the hydrodynamic diameter is smaller because the PAA is collapsed (Figure 6). The density of the shell gradually decreases with the distance from the particle surface. The shell-thickness depends on the pH value. The PAA shell is thin and dense at low pH when the polymer is almost uncharged. This behavior is important and will be used for the following binding properties between the particles and substrates. Similar pH value responsive behavior of PAA-coated particles was also observed with cryo-TEM investigations (Figure 5).
PH responsive behavior of PAA-modified core-shell particles vs. pH proved by cryo-TEM: PAA shell in collapsed state (left) and PAA shell strongly swollen (right). Changes in hydrodynamic particle diameter of PAA modified core-shell particle compared to native SiO2 particle.

Particle deposition on fabric surface
PAA core-shell particles
Based on the results of pH-dependent behavior of the PAA-modified particles, the subsequent experiments towards their depositions on textile fabrics were performed at pH 2 [19]. The SEM analysis shows a reasonable good particle immobilization onto the fiber surface and “networking”/binding between them at pH 2. At low pH values (pH = 2) PAA-modified particles are almost uncharged (Figure 4) and the PAA chains are collapsed (Figures 5 and 6). This leads to higher particle hydrophobicity and higher adhesion to the fiber surface, as was shown in our own work on direct force measurements [19].
For a robust surface modification of the PET-textiles with particles two approaches have been proven. On the one hand the above-described pretreatment by a two-step treatment bath in dissolved CaCl2 (first treatment bath) and PGMA (second treatment bath) and on the other hand a wet chemical amino treatment. The above-mentioned tests and results [19] have shown that the pH has an influence on the reactivity and adhesiveness of the particle shell. The immobilization of PAA particles is therefore carried out at low pH (pH 2, Figure 7). The results for two approaches for the surface modification based on the particle immobilization onto textile fibers and the subsequent thermal treatment are shown in representative SEM images (Figures 8 and 9). A good binding between the particles is proved for both variants. However, the binding points of the amino-treated fabric between particle and fabric are much smaller and less pronounced compared to the two-step treatment (Figures 8 and 9). After the first examination of the particle binding by the washing permanence test this first optical impression was confirmed. The particle binding on the amino-treated fabric was not stable enough, which resulted in a nearly complete particle removal during the washing permanence test.
SEM images of PAA particles on PET fiber surface, pH = 2. PAA-particle bonding after pre-treatment with CaCl2 and PGMA. PAA-particle bonding after wet chemical amino-pre-treatment.


Treatment with PGMA particles
For the treatment with the PGMA particles, the polyester fabric was prepared by means of a pre-described sequence analogous to Synytska et al. [5]. For a permanent binding of the particles on the fiber surface, two possibilities of fabric modification were examined. (1) Alkali treatment and (2) corona treatment with the reactive gases oxygen and argon. A very good binding behavior of the particles to the PET fiber could be observed for both pretreatments, wherein in the alkali-treated fabric occurs an increased deposition of particles on the fiber surface. A steady, almost fully covered thin particle layer is formed on the PET fabric (Figure 10). Due to the hydrophobic properties of the particles, the flat deposit favors the formation of a water-repellent fabric surface. In spite of roughened fiber surfaces, particle deposition was observed mainly between individual fibers when plasma treated fabrics were used (Figure 11). Therefore, for the purpose of hydrophobizing of the fabric, alkali treatment is preferable. The results of the washing permanence test also favor the alkali treatment. At the plasma samples an almost complete cleaning of the PGMA particles could be observed after an evaluation of the wash resistance. The particles on the alkalized sample, however, remained preserved.
PGMA-particle bonding on fiber surface after alkali treatment. PGMA-particle bonding on fiber surface after plasma treatment.

Comparison between particle systems
For comparison of three examined particle-modified systems, (SiO2, PAA, PGMA) a direct comparison of the results was carried out. Therefore, three variants from the preliminary experiments were selected. For the PAA-modified particles, the variant of the fabric pre-treatment by means of the two-stage bath of PGMA and CACl2 and a particle application at pH 2 has proven to be useful. The bonding-quality of the PGMA-modified particles on the textile surface was closer examined on the alkali treated fabric. The fabric pre-treatment to support the binding of non-functionalized SiO2 particles was carried out analogous to the pre-treatment of PAA- and PGMA-modified particle systems (CaCl2 + PGMA as well as alkali treatment).
As can be seen in Figure 12(a) to (c), the three particle systems already show differences in the morphology on the fabric surface. The non-functionalized SiO2 particles are deposited primarily in the space between filaments (Figure 12(a)). A similar behavior is observed with the PAA-modified particles, however, due to the pH value of the amplified particle suspension they form agglomerates on the fabric surface, where the particles build dense networks (Figure 12(b)). However, the PGMA-modified particles are deposited almost completely flat in a steady layer on the fabric surface (Figure 12(c)). This is particularly advantageous for the generation of hydrophobic surfaces. Analysis of the particle bonding on the fiber surface shows a similar behavior with PAA-modified, as well as PGMA-modified particles. Both systems form a binding with each other as well as to the fiber surface. In contrast only a low particle to particle binding of SiO2 particles can be recognized as well as to the fiber surface. One reason for this is the missing organic polymer-shell which is soft and supports the physical binding after the thermal treatment. It is only possible to use the low chemical adhesion caused by the OH-groups of the silinol-groups which is not enough for a permanent binding. The PAA- and PGMA-modified core-shell particle systems possess better binding behavior. At low pH values (pH = 2), as discussed above, PAA is uncharged, collapsed and shows good binding properties. PGMA is also well known as a system with high binding performance. The binding behavior of the functionalized hybrid particles (PAA and PGMA) results from three binding possibilities. At first there is a chemical reaction caused by the functional groups on the fabric surface as well as on the particle shell. This will be supported by physical interaction between polymer chains of particle and fabric which form a connection between each other. The combination of this chemical and physical interaction is essential for a permanent particle binding.
Representative SEM images for particle systems SiO2 (a), PAA (b), PGMA (c) in comparison of surface morphology, topography and particle bonding.
Particle adhering after washing test
The stability of the particle binding is examined by a washing treatment. The optical results (Figure 13) after the washing treatment confirm the supposition about the binding behavior of the particle from the results of the SEM images (Figure 12). The un-functionalized SiO2 system shows a very low washing permanence (Figure 13(a)), despite the same pre-treatment of PET fabric was used for all particle systems. Only some sparse particle agglomerates remain on the fabric. The low particle bonding is caused by the inorganic and hard particle which does not offer the possibility to form a permanent physical binding between the particles as well as the particle and the PET-fiber. The low chemical interaction of OH groups is not enough to generate a permanent binding (Figure 15).
Representative SEM images of particle treated textile fibers after five washings. Chemical and physical interactions between PGAM or PAA particles and fabric. Chemical interaction between SiO2 particles and fabric.


In contrast, the PAA-modified particles as well as the PGMA-modified particles show good washing permanence. This demonstrates the need for a functionalization of both particles and fibers in order to generate reactive groups on the surfaces. Compared to the unwashed fabric sample with PAA- and PGMA- particle system, only a low removal of the particles from the fabric surface is recognizable (Figures 12(b) and (c) and 13(b) and (c)).
The reason for these results is that the textile fabrics were preliminarily treated with sodium hydroxide for PGMA particles and a solution of CaCl2 and PGMA for the binding of PAA-modified particles, which led to the surface functionalization and forming either reactive carboxyl or epoxy functional groups.
Then PGMA- and PAA-modified particles are deposited and chemically bound to the modified textile surface due to reaction between carboxylic and epoxy groups (Figure 14). The already mentioned physical interaction between the polymer brushes on the particle surface and fabric surface supports the bonding positively after thermal treatment (Figure 14). In contrast to the high adhesion behavior of PGMA and PAA shell, the chemical adhesion of silinol groups (native SiO2 particle) is very low and not resistant against the washing process (Figure 15).
Both types of particle coatings remain stable on the textile surface after several washing cycles as shown in the Figure 16. The analysis of the washing lyes by the counting of the particle located in the lye before and after the examination of the washing permanence with PAA and PGMA particles equipped fabric, shows at both tests only low divergences in comparison to the unused raw lye (Figure 16). The particle diameter of functionalizes particle is about 1.0 µm. For the evaluation of particle concentration in the washing lye, a particle size area of 1.0 to 1.4 µm is examined. Particles >1.4 µm can be assumed to be dirt/detergent residues and thus as a foreign body. Considering the results of washing solution of fabrics treated with PAA particles, the increase of the particle concentration is about 16% compared to the concentration of particles in the raw liquor (Figure 16). However, the washing lye of the fabrics treated with PGMA particles shows a lower increase of the particle concentration. The increase is only about 4% compared to the unused wash solution. These results demonstrate the stable and solid connection between the functionalized particles and the modified fabric surface.
Numbers of particles in the washing liquor compared to a raw washing liquor. Water contact angle of textile with PGMA particle treatment and the analysis as a function of time.

Water repellent properties
On the untreated PET-fabric as well as the fabric with SiO2- and PAA-particle treatment, measurements of water contact angle are not possible. After the deposition of the droplet, the test liquid rapidly penetrates into the textile immediately. By the application of PGMA particles, it is succeeded to generate water repellent surfaces on the fabric with a water contact angle of
Conclusion
The contents of this research consist in analyzing the possibilities for modification of plain weave PET fabric to generate reactive groups for a permanent binding of functionalized monodisperse core-shell particles on the fiber surface. The permanence of this connection was evaluated by the simulation of usual terms of utilization based on washing tests. The investigations form the basis for a partial and targeted treatment of the permeable meso-pores of PET-fabrics with functionalized micro particles. An aim is the specific and gradual reduction of the size of this permeable pore structure to increase the barrier effect of the fabrics under preservation of the physiological properties [6]. For the investigations three different particle systems were selected. Un-functionalized SiO2-core particles, particles with a hydrophilic PAA shell as well as particles with a hydrophobic PGMA shell. The investigations have shown that core-shell particles functionalized with PAA and PGMA establish a very good inter particle bonding as well as to the fiber surface. This can be realized by a modification of the fabric surface which is tuned to the functionalization of the particle. A very good adhesion of the PAA particle was achieved through the treatment of the PET fabric with adhesion promoters PGMA and CaCl2. An alkaline treatment of the PET fabrics was used for a durable binding of the particles functionalized with PGMA. The binding of both particle-systems on the fabric surface was analyzed and evaluated by a 360 min long washing test and the subsequent analysis of the wash liquors. An increase of the particle concentration of only 4% for the PGMA particles and 16% of PAA as compared to particles of the raw liquor was determined after the washing test. A permanent binding of the un-functionalized particles could not be achieved, thus proving the need of the particle functionalization. Due to the hydrophobicity of the PGMA particles a water-repellent fabric surface having a contact angle
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by “Deutsche Forschungsgemeinschaft” (German Research Foundation, DFG).
