Abstract
Nanotechnology provides the ability to engineer the properties of materials. Nano zinc oxide having particle size ∼50 nm was used with different concentrations to coat the following textiles fabrics: cotton, polyester and blend cotton/ polyester (65/35) using the pad-dry-cure method. The main advantages of these treated samples were their light weight, increasing surface area per unit volume with increasing treatment concentration. The treated samples were characterized through the following measurements: wide angle X-ray diffraction, mechanical tests (tensile strength and elongation percentage) and thermal properties, i.e. diffraction scanning calorimeter. The results indicated that the behavior of the change in crystal parameters for treated cotton samples greatly differed as compared for polyester. The highest intensity and d-spacing in case of treated cotton were observed at zinc oxide concentration 0.25% followed by a decrease. The polyester samples showed gradual increase in intensity by increasing the zinc oxide concentration, while the hybrid structure blend samples followed the mixture behavior rule. The order of improvement in crystalline parameters for the three treated samples was cotton > blend > polyester. Also, the order of improvement in mechanical properties was cotton > polyester > blend; these results may be due to the uniform distribution of ZnO nano-particles on the fabric surface thus the elasticity of the coated fabric increases.
Introduction
The development of new clothing products based on the immobilization of nanoparticles on textile fibers has recently received growing interest from both the academic and industrial sectors [1,2]. Today, a wide range of nanoparticles and nanostructures can be immobilized on fibers, which brings new properties to the final clothing product. Lately, much interest has been given to the development of clean textiles by means of surface modification of the fibers with either anti-stain or self-cleaning coatings. Self-cleaning surfaces can be designed through the use, for example titanium dioxide nanoparticles which can act as photo-catalyst under UV light [3]. Numerous methods can be used for the surface modification of fibers with nanoparticles, including blending of the nanoparticles in the polymer matrix before spinning or chemical grafting of the desired functional groups onto the fibers [4]. Among the properties imparted to textiles using nanoparticles are water repellence [5], anti-bacteria [6–9], UV-protection and flame retardant [7,9,10].
Flame-retardant finishes of textiles include a variety of chemical compounds and polymers that are appropriate of each fiber type, fiber blend, fabric weight and construction [11]. The flammability characteristics of commercially available fiber vary widely, cellulosic burn readily with glow followed by the formation of char, while wool supports combustion only with difficulty. Synthetic fibers may melt slowly without burning (nylon 6 and 66), burn and melt readily (polyester and acrylic) [12]. The thermal processes and combustion of organic polymers, whether in fiber film or in some other forms, occur in progressive and definable sequence in which thermal decomposition precedes ignition and combustion of the material. After combustion, the polymer may burst into flame, melt, shrink, char or thermally degrade without flame [13,14]. The role of flame retardants is to inhibit the formation of combustible products and/or to alter the normal distribution of decomposition products emanating from the original material [15]. Various finishing techniques have been used to impart flame resistance to textiles. Among them are coating, graft or homopolymerization, cross-linking, covalent bond formation and so on [16,17]. Fiber blends are more difficult to flame proof than fabrics composed of only a single fiber type. Change in flame retarding steadily may be antagonistic, synergistic or neutral for a particular flame retardant system applied to a fiber blend relative to either fiber type alone [18,19].
The principle aim of this research work was to investigate the effect of treating different textile fabrics with nano zinc oxide (ZnO) particles. Thus, cotton, polyester and blend (cotton/polyester) were treated with different concentrations of nano ZnO, wide angle X-ray diffraction (XRD), tensile strength and elongation percent and diffraction scanning calorimetry (DSC) were used to evaluate the coated samples.
Materials and method
Fabrics
Pure cotton fabric (100%) with weight 190.64 gm/m2, thickness 0.43 mm, number of yarns/cm in warp direction is 23 and in weft direction is 23. Pure polyester fabric (100%) with weight 99.56 g/m2, thickness 0.32 mm, number of yarns/cm in warp direction is 22 and in weft direction is 16. Blend cotton/polyester (65:35) fabric (100%) with weight 98.45 gm/m2, thickness 0.29 mm, number of yarns/cm in warp direction is 25, and in weft direction is 22. These fabrics were purified by scouring with 2 g/L of non-ionic detergent using a liquor ratio 1:50 at temperature 60 ℃ for 15 min.
Chemicals
Nano ZnO with particle size ∼ 50 nm having molecular weight 81.39 (Aldrich). Acrylic binder (Acrylate Base SME-2) pH ∼ 6. Propanol was used as dispersion medium. These materials were used as received.
Treatment methods
All the samples under test were conditioned at ambient conditions. Four different concentrations of nano ZnO were prepared (0.25%, 0.5 %, 1%, 2%). For each concentration, ZnO and propanol is added to 100 ml deionized water together with acrylic binder in a ratio 1:1:0.5. Strips of samples were immersed and soaked in the prepared solutions with liquor ratio 1:100 for 10 min under magnetic stirring followed by padding to ensure complete pick up. The padded samples were dried at 100 ℃ for 15 min, then thermo fixed at 150℃ for 2 min. Finally, the treated samples were washed with 2 gm/L sodium lauryl sulphate for 5 min, then rinsed in distilled water and air dried.
Instrumental techniques
X-ray diffraction (XRD)
XRD measurements of all the treated samples were carried out using a Scintag Irc (USA) X-ray Diffractometer of CuKa-radiation was used. It operates at 45 kV and 40 mA at wavelength 154.6 cm−1. The diffractograms were recorded with a diffraction angle 2 Θ ranging from 0 to 60° with a scan rate 2°/min. The crystalline index (CrI) was calculated for the different samples using the following equation [20]
Mechanical measurements
Tensile strength and elongation percentage at break for all samples under test were measured and evaluated using a Shimadzu Universal Tester of (C.R.T.) type S-500, Japan. The measurements were carried out according to standard method for tensile properties of textile (ASTM D 3822-07).
Differential scanning calorimetry (DSC)
Experiments were performed with DSC-50 Shimadzu (Kyoto, Japan). Each sample was heated and cooled at a scanning rate of 10℃ /min, under nitrogen atmosphere to prevent oxidation. A test sample of ∼ 5 mg was placed in aluminum capsule and heated from 30–200℃ for each run. The melting temperature (Tm) and enthalpy (ΔHm) were determined.
Results and discussion
X-ray diffraction (XRD)
The most common method to obtain structural characteristic changes is XRD, as it gives information about crystal parameters namely inter-planar space (d) and crystal size. Figure 1 (a) to(c) shows diffraction pattern of cotton, polyester and blend fabrics coated with difference concentrations of ZnO nanoparticles. The peak area crystallinity was measured in the angle range 18°: 36°; it can be seen that, there is a change in peak height and width.
(a–c) XRD spectra diagram of the three treated samples using different concentration of ZnO nano particles.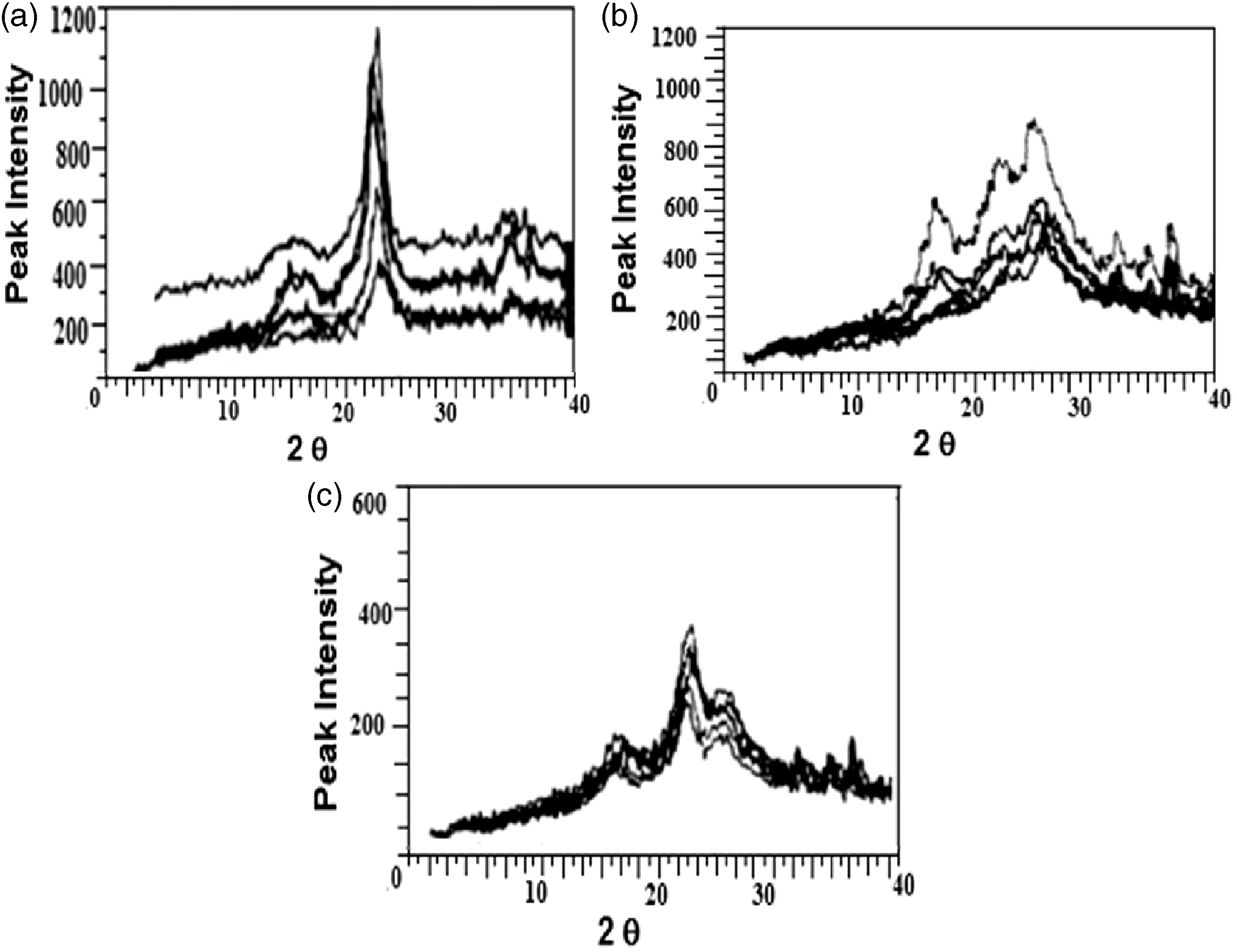
The change in crystalline parameters of the treated samples.

Note I: d-spacing, II: peak intensity, III: crystalline index, IV: half band width.
In considering polyester samples, (Figure 1(b) and Table 1(b)) two peaks appeared and centered at 2θ ∼ (23°,26°) for the blank and on treatment with ZnO, additional four peaks appeared at 2θ ∼ (18°, 32°, 34°, 36°). The appearance of these new peaks with treatment means that the presence of ZnO with different amounts can cause structural variation in the polymeric network of polyester [21]. Besides, all the peaks have broad bands, i.e. higher crystallinity. There was a continuous increase in peak intensity for all the bands with increasing [ZnO], i.e. continuous increase in crystallinity/amorphousity ratio; this trend agrees well with the d-spacing behavior, the increment in d-spacing is taken as an indication to the enhancement in compatibility between fabric and nanoparticles. The crystalline index showed a continuous decrease with increasing [ZnO], indicating a decrease in crystalline regions. The half band width showed slight decrease at higher [ZnO], i.e. a small increase in crystal size.
When examining XRD curves of blend samples (Figure 1(c) and Table 1(c) different bands appeared and centered at 2 ∼ (18°, 23°, 26°, 32°, 34°, 36°). It was found that nearly all the peaks have broad bands. The two bands centered at 2 θ ∼ {26°, 32°} are related to polyester and consequently appeared in blend, while the four bands centered at 2 θ ∼ (18°, 23°, 34°, 36°) are common in the three fabrics. Generally, the peak intensity values and d-spacing of the different bands for blank blend samples were lower than that of its constituents; also the behavior of the change in peak intensity and consequently d-spacing with different [ZnO] was somewhat different from its corresponding in each fabric. The two bands centered at 2 θ ∼ (18°, 23°) for treated blend samples followed the same behavior as that obtained in treated pure cotton, i.e. the highest intensity and d-spacing are observed at [ZnO] 0.25% followed by a decrease in both intensity and d-spacing as illustrated in Figure 2(a) and (b). The peak intensity and d-spacing of the two bands, centered at 2 θ ∼ (26°, 32°) were common in both blend and polyester, and they follow the same behavior as that of pure polyester, i.e. increase with increasing [ZnO]. The two bands centered at 2 θ ∼ (34°, 36°) were common bands for the three examined fabrics. The change in behavior with treatment for the blend samples as compared with its constituents can be interoperated and discussed on the light of hybridization theory, which states that both cotton and polyester fabrics can be considered as a hybrid structure of cotton and polyester fabrics blended together to form a system. The differences in their contribution towards the overall behavior of the structure can be related to: first the divers mechanical properties of these constituents fabrics. Second, the interaction between the two constituent will alter the nature of the fabric behavior especially during fracture. However, it has become widely accepted in many cases that, the hybrid structure of blended fabrics follows the rule-of-mixture behavior that is the resultant properties of the structure are the mean values of the volume fraction weighted properties of its constituents [22]. Although in critical cases there were deviations from this rule, the strength of blended fabric could be lower or higher than the strength of its constituent.
Comparison of the change in peak intensity values of the common bands in the three examined samples at different concentrations of ZnO.
The highest crystalline index is obtained at [ZnO] 0.25% following by a decrease in crystalline index; this behavior agrees well with that obtained in cotton samples. The crystalline index for the three samples followed the order: cotton > blend > polyester. The half bandwidth for the blank samples lays in between cotton and polyester, i.e. polyester > blend > cotton, and on treatment, the half width was nearly constant and its value is inversely proportional to particles size, the order of half band width with treatment was polyester > cotton ≥ blend.
Mechanical properties
Effect of nano [ZnO] on mechanical properties of the treated samples.
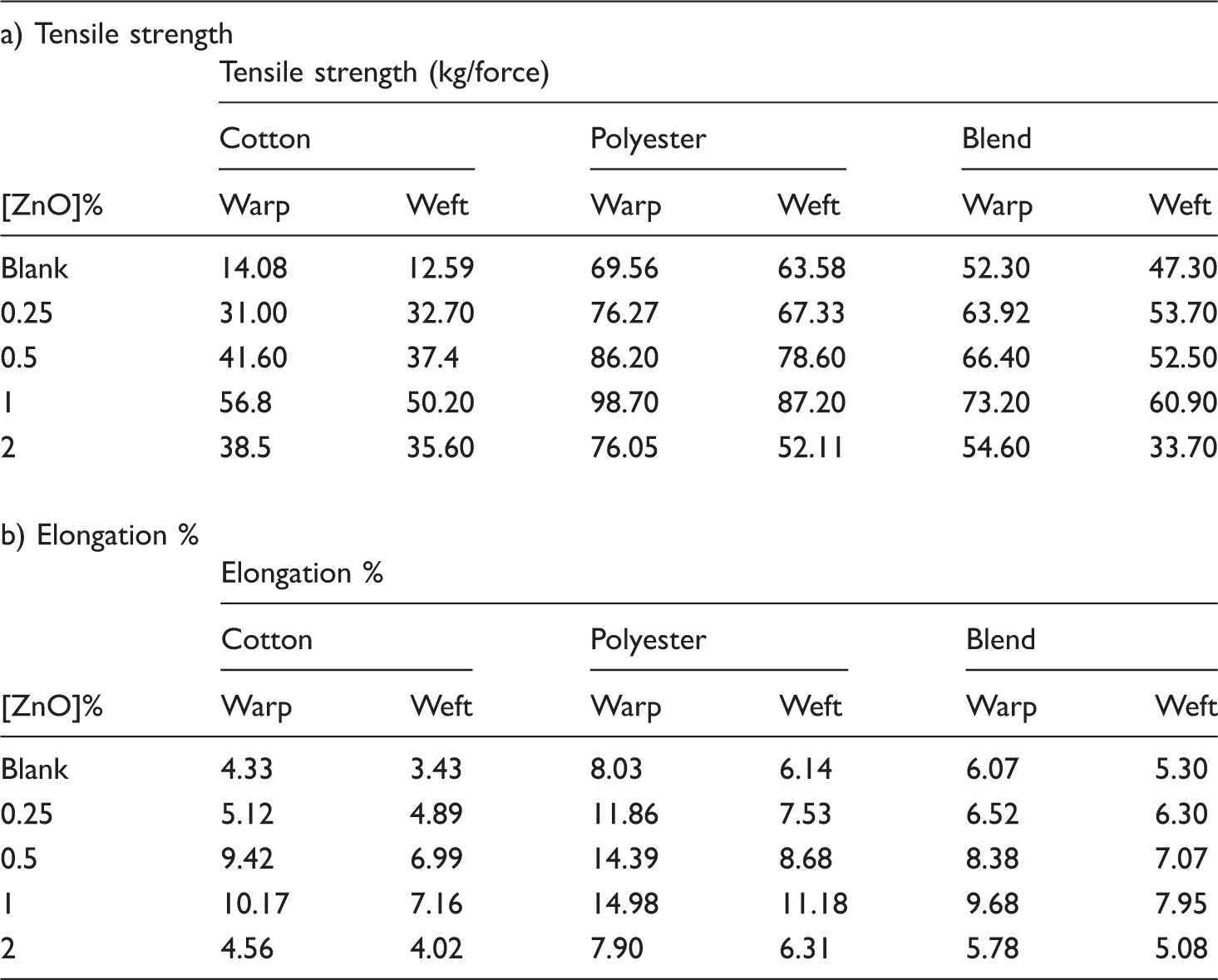
The weak mechanical properties of the blank cotton samples are due to their higher amorphousity [14], i.e. inelastic, highly formed hydrogen bonds which tend to hold the polymer system and prevent the fabric system from slippage. On the other hand, blank blend lays in-between, and this agrees with hybridization theory as previously mentioned.
The mechanical properties of the treated samples gradually increase by the increase of ZnO concentration up to 1%. By increasing the ZnO concentration to 2% the mechanical properties decrease as the ZnO accumulation on the sample surface leads to brittleness of the fabric [12]. The improvement in warp direction is higher than that in weft; this was in agreement with the weaving requirements. The order of improvement in mechanical properties for the examined samples was cotton > polyester > blend.
These results can be discussed by the fact that, the uniform distribution of the nanoparticles on the surface, increase surface area unit volume, so makes the coated samples more elastic. Also nano size can be embedded in the polymer matrix, especially with cotton that having higher amorphousity as explained in XRD, thus increasing the tensile properties.
Differential scanning calorimetry
DSC is the most commonly used method to determine transition temperatures such as glass transition, rate of decomposition and total heat flow [21]. Figure 3 (a) to (c) shows the DSC spectra of the examined samples while Table 3(a) to (c) clarifies the data of transition temperatures and heat of fusion of the samples. For cotton samples (Figure 3(a) and Table 3(a)), there were two endothermic peaks, the first one due to dehydration of cellulose, and the second endothermic peak shows depolymerization and oxidation of the of the cotton samples. The melting peaks shifted gradually to higher temperature with treatment, and the heat of fusion ( (a–c) DSC of the treated samples. DSC parameters of the treated samples.

In considering polyester fabrics, its thermal decomposition is initiated at above 260℃ by rupture of an alkyl-oxygen bond and the materials decompose via the formation of cyclic or open chain oligomers. Figure 3(b) and Table 3(b) show the data of DSC spectra of polyester samples. There were two endothermic peaks; these peaks were shifted to higher temperature with treatment, the first peak ∼ 260℃ due to the scission of alkyl-oxygen band this is due to oxidative reaction of the polymer and combustion of char residues. The heat release −ΔH decreased since ZnO nanoparticles interfere with crystallization; this enhances flame retardancy and slows down polymer degradation.
The thermal behavior of blend (cotton/polyester) was completely different due to the difference in physical and chemical properties of both cotton and polyester. During the thermal degradation, cotton begins to decompose at a temperature well below that required for thermal decomposition of polyester. Thus, the cotton acts as the initial source of ignition in the blend, the polyester which has melting point at ∼ 260℃, tends to wick on the cotton char resulting in the phenomenon called scaffolding [21]. The polyester component furnishes the additional fuel to the gas phase and as the polymer temperature is raised, the heat is produced from combustion of cotton decomposition products. Figure 3(c) shows the DSC spectra of blend samples while Table 3(c) clarifies the behavior of decomposition and heat of fusion during the different stages of combustion. It is clear that there were two stages of decomposition one pertaining to decomposition of cotton and the other pertaining to polyester. Also, there was shift in peak temperature with increasing treatment concentration. The heat of fusion decreased with increasing concentration of ZnO, and heat of fusion during the first stage was lower than that the second stage. The reduction in
Conclusion
Different concentrations of nano ZnO were used to coat some textile fabrics namely cotton, polyester and blend using the pad-dry cure method. The treated fabrics were evaluated through the following measurements: XRD, mechanical tests and DSC measurements. The crystal parameters of the cotton samples showed the highest improvement at [ZnO] 0.25% followed by a decrease; while the polyester showed continuous increase and the hybrid structure blend samples followed the mixture behavior rule. The improvement in mechanical properties followed the order: cotton > polyester > blend.
The DSC measurement showed a decrease in heat release with treatment and the heat of fusion for all samples followed the order: polyester > cotton > blend.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.

