Abstract
Silver-containing wound dressings are now commonly available with much publicised claims of antimicrobial activities against all kinds of pathogens including methicillin-resistant
Introduction
Human wounds preserve various types of microbial and viral infections from the surrounding environment that have negative impact on the healing process of wound and bodily functions [1–3]. Wounds are dressed to reduce the intrusions of these environmental infections. However, these dressing products play a very limited role in controlling the risk of these environmental infections without antimicrobials. A range of antimicrobial agents along with these wound dressing materials are now available to use and control these infections and improve the endurance of patients to infections [3, 4].
Silver, among the other antimicrobial agents, is the most common and well-known antimicrobial due to its broad antimicrobial effect. It has been found effective against a range of aerobic, anaerobic, Gram-positive, Gram-negative bacteria, fungi and viruses [5, 6]. Silver is active at very low concentrations; even 1 part per 100 million of elemental silver is effective in a solution as an antimicrobial and no bacteria resistance is developed to silver unlike other available antibiotics [7–12].
Silver-containing fibres, hydrogels and composites dressing are now abundantly available and used to reduce the risk of wound infections and complications. However, to maximise the effectiveness of silver for longer periods, slow and sustained release of silver is very important [13–16]. The natural polymeric materials, especially alginates have been found to be instrumental in achieving successful and long-lasting healing effects from silver antimicrobial drugs [17, 18]. Alginate is a major polysaccharide found in brown algae and is biocompatible, biodegradable material. It is non-toxic to human body and is used as moist healing material in wound management industry [19, 20]. Although its effectiveness in killing bacteria is limited, it can help to reduce the spreading of bacteria due to its unique gel-forming properties [21, 22]. Inclusion of natural water absorbing and gelling polysaccharides with ultra-high absorption and gelling properties like Psyllium or Aloe Vera into established natural polymers could possibly lead to a new breed of dressing materials with much enhanced properties. Such systems could perhaps contribute more efficiently to the wound exudate absorption and the healing process with possible advantage of carrying less antimicrobial agents through better dispersion and subsequent release mechanisms.
Psyllium is a natural polysaccharide obtained from the NMR spectra of Seed Husk Psyllium [23].
In this work, Psyllium is coextruded with sodium/calcium alginate as a carrier, together with silver compound to develop a novel, low-dose/slow-release silver-containing biomaterial for wound care applications. In vitro, antimicrobial assessment of the new fibre is made by exposure to known selection of pathogens. The average silver contents and their release characteristic are also tested and compared to the commercially available silver dressings. Comprehensive details of physical and mechanical properties as well as other characterisations of the developed fibres are described elsewhere [24, 25].
Materials and method
Food grade Psyllium husk (QARSHI Laboratories of Pakistan Limited, Karachi) was purchased from local UK supermarket. Sodium alginate was provided by Speciality Fibres and Materials (Coventry, UK), silver carbonate and calcium chloride were purchased from Sigma Aldrich Company Limited, (USA) and acetone from VWR International Limited (UK).
Acticoat active absorbent which is comfortable absorbent type 1 alginate dressing containing nanocrystalline silver was supplied by Smith & Nephew (UK) while Aquacel Ag which is made from sodium carboxymethyl cellulose (CMC) containing ionic silver was supplied by CONVATEC (UK).
Fibre extrusion method used
Silver carbonate, 0.05% w/v, was added to the Psyllium gel solution and stirred continuously to achieve adequate dispersion of silver carbonate into the Psyllium gel solution. Sodium alginate (4% w/v) was then added and continuously stirred for further 4 h. The solution was left overnight to remove air bubbles and extruded under pressure through a spinneret (20 holes, 76 µm diameter) into calcium chloride coagulation bath. The resultant fibres were drawn, collected, washed with water, rinsed and dried using acetone (50–100% v/v). The multifunctional pilot-size wet extruder used in this study is depicted in Figure 2.
Flow diagram of Multi-functional pilot-size wet extruder.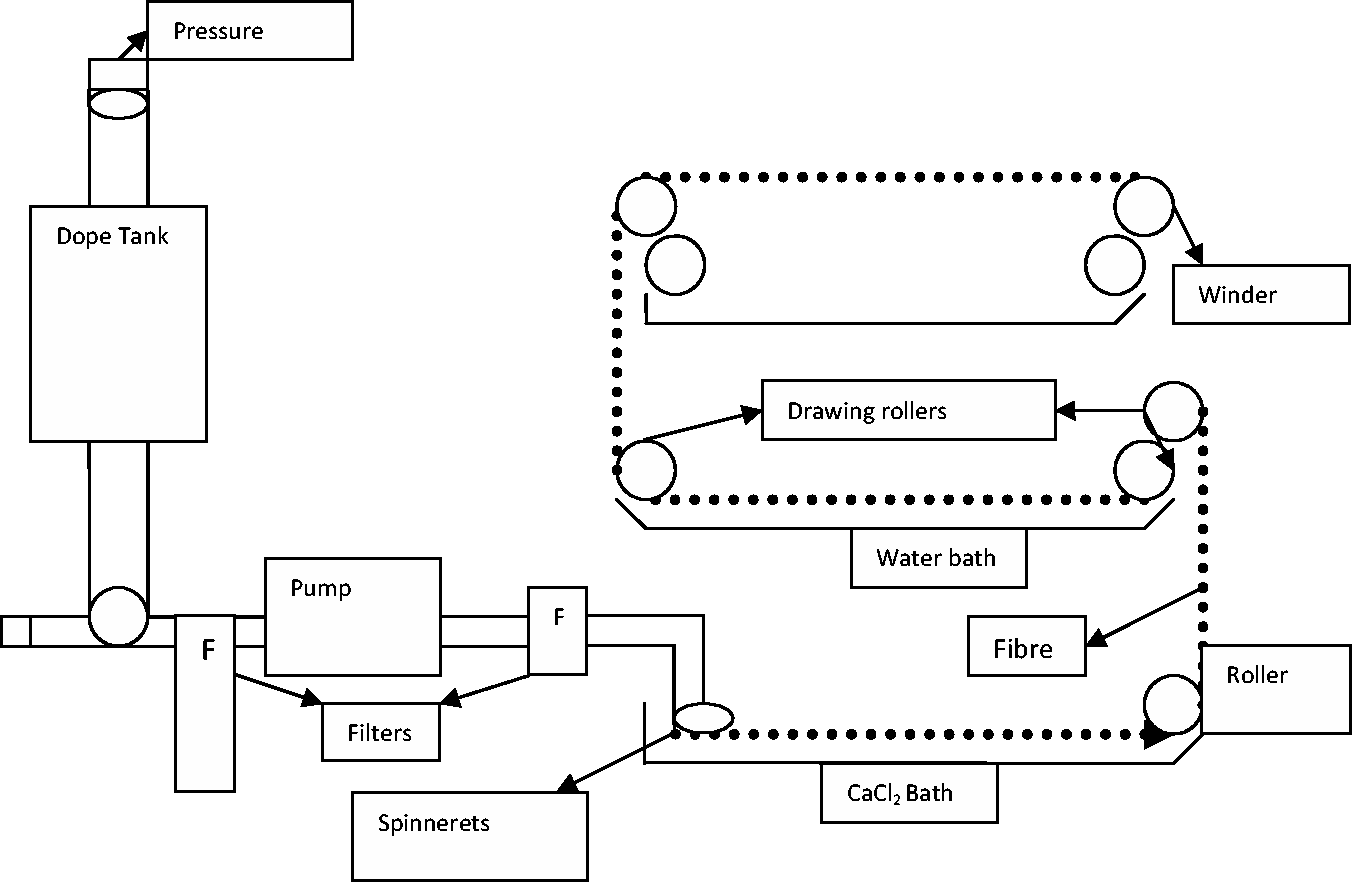
Assessment technique of produced fibres
Silver release
One gram from each dressing fibre was suspended in nutrient-brothed saline solution (a mixture of standard nutrient broth and saline solution, 1:1) at a ratio of 1:100 (w/v). The samples were placed at room temperature over three days. During this period, aliquots were removed at timed intervals of 1, 4, 24, 48 h and the liquid was topped up to maintain a constant volume. Samples were filtered, diluted as appropriate and analysed using atomic absorption spectrometry.
Silver content measurement
One gram of each sample dressing was dissolved in 100 ml of concentrated nitric acid and sulphuric acid solution. The solution mixture was then heated for 30 min at 60℃ for complete breakdown of the fibres/dressings. The solution was filtered using very fine nylon mesh to remove any possible residue. The filtered solution was further diluted (1/10) using deionized water to enable quantification of total silver in the fibres/dressings. The quantity of silver was also measured using the atomic absorption spectrometry.
Antimicrobial efficacy testing
The AATCC (American Association of Textile Chemists and Colorists) 2004-147 agar diffusion or Zone of Inhibition test was used to determine the antimicrobial efficacy of the test fibres using three selected species of bacterial cells. These test methods were purely qualitative.
Results and discussion
Liquid absorption properties of test dressings
Liquid absorption of the developed fibre (psyllginate) containing 0.05% silver and test dressings.

Saline solution and solution A are normally used for absorption testing to mimic human body fluids or blood composition. Hence, higher absorption capability of Psyllginate 0.75%Ag2CO3 0.05% w/v fibre outperforms both calcium alginate (SFM) and CMC (Aquacel) fibres when exposed to body fluids or extrudates.
Silver content
Average silver contents of Psyllginate fibre containing 0.05% silver and test dressings.
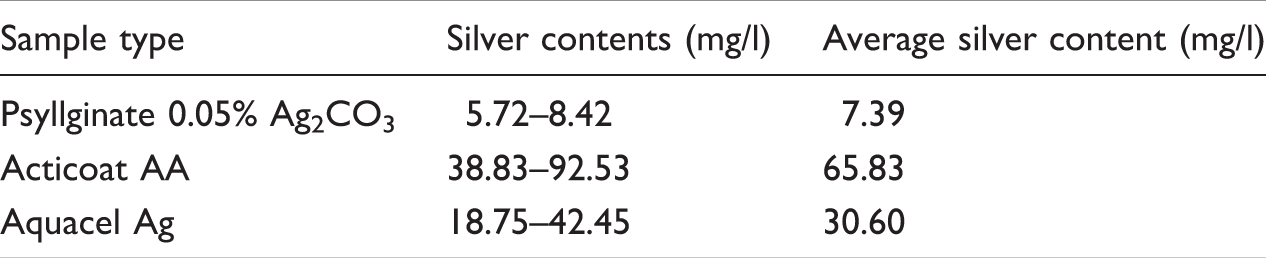
Silver release analysis
Silver release from Aquacel Ag and Acticoat AA is very fast and high in the first 24 h of immersion compared to that of Psyllginate fibre containing 0.05% silver carbonate. However, silver release from Aquacel Ag tends to level off after 24 h, whereas silver releases from Acticoat AA continues to increase after 24 h of immersion. Silver releases from Psyllginate fibre containing 0.05% silver carbonate follows a steady and slow rate of release up to and over 72 h. The rate of liquid diffusion into dressing matrix is normally expected to control the rate of silver release. The results in Figure 3 show the amount of silver released with time from the test fibres into aqueous medium through the diffusion process.
Average silver release as a function of time for the three test fibres.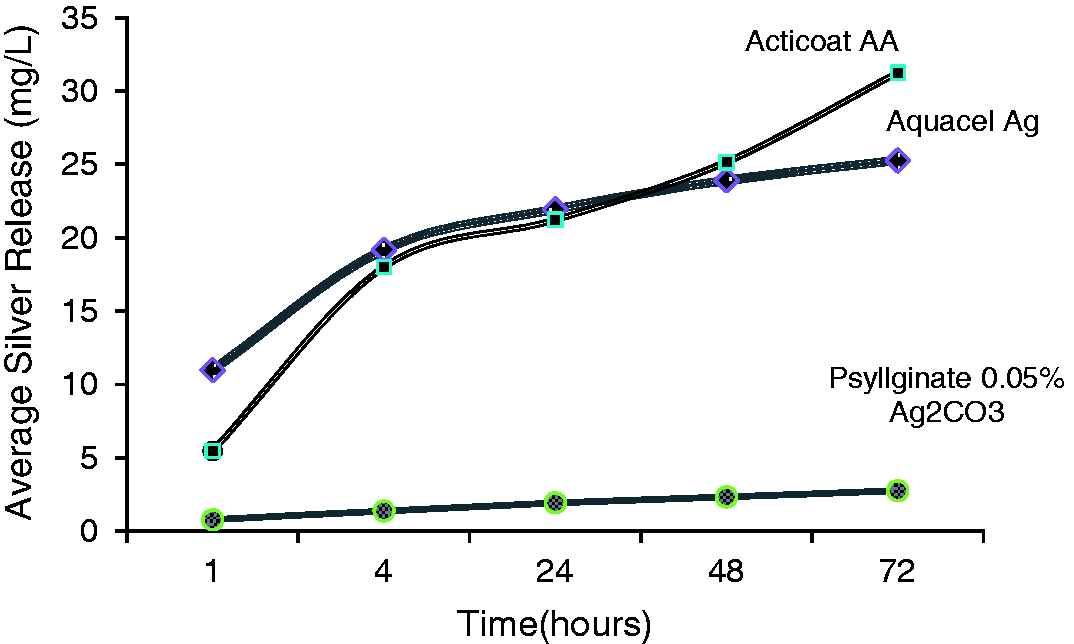
The results also suggest a very high rate of liquid diffusion into Aquacel Ag and Acticoat AA during the first 24 h; therefore, high quantities of silver are released from these two dressing materials in the first 4 h. After the initial 4 h, Aquacel Ag carries on releasing silver at a much lower rate and eventually stabilises at around 20–25 mg/l. Acticoat AA continues to release silver with time at slightly faster rate than Aquacel Ag and the rate of release tends to become faster after 48 h. Rate of liquid diffusion into Psyllginate containing 0.05% silver carbonate appears to be a lot slower over 72 h and hence quantities of silver release are much smaller, increasing only slightly with time. Slow release of silver from newly developed fibre is because of the excellent gelling properties of Psyllium. Silver is physically entrapped within gelatinous mass of Psyllium that prevents the silver from quick release.
Antimicrobial efficacy
The results of antibacterial sensitivity, as shown in Figures 4 to 6, show that Aquacel Ag, Acticoat AA and the Psyllginate 0.05% have all clear zones of inhibition against Gram-negative Appearances of inhibitory zone with Appearances of inhibitory zone with Appearances of inhibitory zone with MRSA 16.


The result shows that Psyllginate containing very low concentration of silver is highly effective against known Gram-negative, Gram-positive bacterium and MRSA 16.
Conclusion
In this study, a new biomaterial fibre has been developed from natural polymers with inbuilt ability to gel and absorb large quantities of pseudo exudates. The developed fibre contains up to six times less silver than the conventional silver dressings. The novel fibre also displays very slow rate of release whilst maintaining full potency over time against known microorganisms including MRSA. Silver is a premium material and therefore has a high cost value. Excessive use of silver not only increases manufacturing costs but can also initiate unnecessary complications in the wound healing process. The Psyllginate fibre containing silver developed in this work is much cheaper to produce and is highly effective against Gram-positive and Gram-negative bacteria with unlikely danger of causing discoloration, damage or staining based on its very low silver content. The study concludes that the developed fibre has long-lasting antimicrobial and gelling properties that are comparable, if not better, than Acticoat AA and Aquacel Ag, two commercially available silver dressings.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
