Abstract
A layer of TiO2 nanoparticles was deposited on the surface of cenosphere by using tetrabutyl titanate or titanium sulfate as the precursor under hydrothermal conditions. The morphology, chemical composition, crystal structure and optical properties of cenosphere before and after treatments were determined by scanning electron microscope, energy dispersive spectroscopy, X-ray diffraction and diffuse reflectance spectrum. The photocatalytic activity of methylene blue degradation was evaluated by using ultraviolet irradiation. The results showed that compared with tetrabutyl titanate, a thicker layer of TiO2 nanoparticles in anatase structure was deposited on the surface of cenosphere treated with titanium sulfate. The crystal size of TiO2 nanoparticle by using titanium sulfate as precursor was larger than that by using tetrabutyl titanate as precursor. The photocatalytic activity of TiO2-coated cenosphere by using titanium sulfate as precursor was slightly enhanced mainly because of the higher crystallinity of anatase TiO2. The formation mechanisms of TiO2 coating on cenosphere surface and photocatalytic degradation of methylene blue with ultraviolet irradiation were also discussed.
Introduction
Cenosphere, known as hollow ceramic aluminosilicate or borosilicate microsphere, has found a variety of potential applications in different fields due to its unique characteristics such as lightweight, good insulation, sound damping, good mechanical properties, thermal and chemical stabilities. The thickness of shell is generally in the range of 1–3 µm and the diameter is from 5 to 120 µm. The core–shell cenosphere has been prepared by coating different materials on its surfaces via electroless plating technique, sol–gel process, magnetron sputtering deposition, precipitation method, etc. For example, fly ash cenospheres were used as electromagnetic wave shielding and microwave absorption support to prepare the magnetic metal-coated functional composites by electroless plating technique [1,2]. Cenospheres were also used as the substrates to produce the industrial grade multi-walled carbon nanotubes by fluidized bed chemical vapor deposition [3]. Likewise, magnetic cobalt, γ-FeNi and binary alloy coated microspheres were prepared using heterogeneous precipitation thermal reduction method [4–6].
Because of its high surface area and low density, cenosphere can be employed as efficient catalyst support, which results in intensive investigation on the preparation of catalyst [7]. The TiO2-coated cenosphere can float on water surface, which simplifies its separation from the water through filtration process. The functional coatings, including ZnO, Fe2O3 and TiO2, are applied to cenosphere to break down organic compounds, pesticides, oils as well as dyes. It has been confirmed that TiO2 is a well-known semiconductor effective for photocatalytic reactions. Once TiO2 is exposed to UV irradiation with energy larger than its band gap, the electron will jump from the valence band to the conduction band. A pair of electron and hole is formed on TiO2 surface. The negative electron and positive hole will combine with dissolved oxygen and water to produce hydroxyls, peroxides and superoxides. These radicals are destructive to many organic pollutants in water. As known, the photocatalytic activity of TiO2 depends upon its crystal structure, surface area, size distribution, porosity, presence of dopants, and surface hydroxyl group density, etc. [8]. These factors directly influence the production of electron–hole pairs, the surface adsorption and desorption process and the redox process [9]. More recently, some research has been carried out to explore the fabrication of TiO2-coated cenospheres for degradation of various pollutants. For instance, Surolia et al. [10] reported the synthesis of TiO2-coated cenosphere using the sol–gel process, which could degrade methylene blue, p-nitroaniline, n-decane and n-tridecane under sunlight irradiation.
The photocatalytic properties of TiO2-coated cenosphere depend on the synthetic methods. There are many ways to enhance the photocatalytic activity of TiO2, such as doping ions, compound, dye sensitization, acid treatment and so on. To extend the photoresponse of TiO2 into the visible region as well as light absorption enhancement, the surface of TiO2-coated cenosphere was further modified by hydrogen peroxide [11]. TiO2 films were deposited on the surface of cenosphere using the modified magnetron sputtering equipment under different working conditions [12]. To improve the visible light absorption, some heteroatoms have been introduced to modify the electronic structure of TiO2. The transition metal ion Fe3+ was doped in the matrix of TiO2 deposited on fly ash cenosphere by sol–gel method. The photocatalytic activity of Fe-doped TiO2-coated cenosphere varied with the iron content and the calcination temperature [13]. The visible light photocatalytic activity of TiO2-coated cenospheres was obtained by the heteropolyacid modification [14]. It has been demonstrated that the higher visible light activity is attributed to the increased adsorption capacity, enhanced light absorption and better charge separation [15].
It is well known that synthetic dyes are very difficult to degrade by using biodegradation, adsorption and oxidation techniques. Owning to the toxicity and esthetic considerations, synthetic dyes are undesirable waste byproducts of textile industrial effluent. So far, there have been a few reports about the cenosphere photocatalyst for degradation of dyes. Koopman et al. [16] examined the effects of magnetic stirrer and aeration bubbler on the ambient solar photocatalysis of Procion Red dye. However, the TiO2-coated cenosphere has not been extensively considered as candidate materials for treatment of textile industrial wastewater.
The hydrothermal method has been found to be one of the best techniques to prepare TiO2 particles of desired size and shape with homogeneity in composition as well as a high degree of crystallinity. Its most important feature is that it favors a decrease in agglomeration among particles, narrow particle size distributions, phase homogeneity and controlled particle morphology [17]. In this study, we employed a hydrothermal processing to deposit pure anatase phase TiO2 nanoparticles on the surface of cenosphere using tetrabutyl titanate or titanium sulfate as precursor. A large specific surface area and good crystallinity of TiO2 nanoparticles are beneficial to the TiO2-coated cenosphere in waste water treatment. The coating coverage, element identification, phase structure and optical properties of cenosphere were characterized by scanning electron microscope (SEM), energy dispersive spectroscopy (EDS), X-ray diffraction (XRD) and diffuse reflectance spectrum (DRS). In addition, the photocatalytic activity of TiO2-coated cenosphere was examined on methylene blue dye using ultraviolet light. The resulting TiO2-coated cenospheres with low density and high surface area have a potential application for the photocatalytic degradation of other organic compounds.
Experimental
Materials
The white cenospheres were obtained from Shanghai FEP International Trade Co. Ltd. The particle sizes are from 15 to 85 µm with the mean diameter of 40 µm. The compressive strength is about 38 MPa and the actual density is around 0.38 g/cm3. The chemicals used for TiO2 nanoparticle preparation were tetrabutyl titanate (Ti(OC4H9)4), titanium sulfate (Ti(SO4)2), urea ((NH2)2CO), hydrochloric acid (HCl), acetone, anhydrous ethanol and distilled water. All reagents were used as received.
Catalyst immobilization on cenosphere
Two precursors, tetrabutyl titanate (scheme one) and titanium sulfate (scheme two), were used to immobilize the titanium dioxide on the surface of cenosphere under hydrothermal conditions, respectively. About 1.0 g of cenospheres was pretreated with 200 mL of a 10% HCl solution at room temperature for 30 min, followed by washing with the anhydrous ethanol solution for 15 min, and then were completely washed with distilled water prior to usage. For scheme one, approximately 0.8 g (0.5 mL) of tetrabutyl titanate was first dipped into 20 mL of anhydrous ethanol solution under vigorous stirring at room temperature. 0.5 g of pretreated cenosphere was then added to the above solution under stirring for 3 min. 20 mL of anhydrous ethanol was mixed with 20 mL of distilled water, and then was dropwise added to the tetrabutyl titanate and cenosphere solution within 10 min under stirring, followed by adding 20 mL of distilled water. A white precipitate of amorphous oxide mixed with cenospheres was produced and the mixture was transferred to a 100 mL PTFE sealed can, which was put into the stainless steel autoclave. The autoclave was placed in a furnace and was heated to 120℃ at a rate of 2℃/min. After 3 h, the PTFE sealed can was cooled down to room temperature. The buoyant cenospheres were successively washed with acetone, anhydrous ethanol and distilled water at room temperature for 10 min, respectively, and were subsequently dried in an oven at 80℃ for 48 h. The weight of cenosphere before and after treatment was measured with a balance. The pick-up of TiO2 nanoparticles deposited on cenosphere in weight relative to the untreated one was calculated.
For scheme two, approximately 0.96 g of titanium sulfate was first dissolved in 40 mL of distilled water at 60℃, followed by adding 0.48 g of urea under stirring. 0.5 g of pretreated cenosphere was then added to the above solution under stirring for 3 min, followed by adding 40 mL of distilled water. The hydrothermal reaction temperature was set at 140℃ and the reaction time was 2 h. The following procedures were the same as the scheme one.
Characterization and measurement
The surface morphology of the sample was observed with a JEOL JSM-6700 field emission scanning electron microscope. The chemical composition of the sample was also analyzed by using an energy dispersive spectroscopy of an emissive-type electron microscope. XRD spectrum was obtained by using Cu Kα1 radiation (
The photocatalytic activity of the sample was assessed after exposure to UV irradiation based on the decomposition of methylene blue dye. The irradiation was performed using 20 W (main wavelength 254 nm) quartz ultraviolet lamp. 0.04 g of the sample was added to 50 mL of methylene blue solution at a concentration of 10 mg/L at neutral pH. The lamp was placed above the solution at a distance of 10 cm. The absorbance of the characteristic peak of methylene blue at the maximum absorption wavelength (662 nm) was recorded using a UV-Vis spectrophotometer (Beijing Rayleigh Analytical Instrument Corp. UV-1600) every 20 min. The degradation rate 
Results and discussion
SEM observation
The SEM images of cenosphere before and after treatments are illustrated in Figure 1. The surface of the untreated cenosphere is relatively clean and smooth (Figure 1(a)). Many small caves are formed on the surface of cenosphere because of the chemical etching in HCl solution (Figure 1(b)). The surface of the tetrabutyl titanate modified cenosphere (scheme one) is very rough. A large number of particles in micrometer size are attached onto the cenosphere surface (Figure 1(c)). From the high resolution SEM image, the small particle is composed of aggregated nanoparticles with spherical shape and an average size of 30 nm (Figure 1(d)). The cross-section image indicates that the thickness of the coating is about 100 nm (Figure 1(e)). The primary nanoparticles are closely stacked together without interparticle voids and formed on the shell of cenosphere, which provide high surface area for photocatalytic degradation of dye [19]. For the titanium sulfate modified cenosphere (scheme two), a layer of substances is coated on its surface, which is dispersed with some micrometer-sized particles with respect to the tetrabutyl titanate modified one (Figure 1(f)). The coating is also constituted of aggregated nanoparticles with a mean size of 50 nm (Figure 1(g)). The corresponding thickness of the deposition is around 200 nm (Figure 1(h)). The pick-ups of the coating are 4.1% (w/w) for scheme one and 6.3% for scheme two, respectively. The difference in the TiO2 loading is probably caused by using different Ti-precursors and dosages, reacting at different temperatures and times.
Scanning electron microscopic images of cenosphere before and after treatments: (a) the untreated cenosphere 2000×; (b) the etched cenosphere 2000×; (c) 2000×; (d) 30,000× and (e) cross-section of cenosphere modified with tetrabutyl titanate; (f) 2000×, (g) 30,000×; and (h) cross-section of cenosphere modified with titanium sulfate and urea.
The reaction for Ti(OC4H9)4 (Ti(OR)4) as the precursor can be considered the following:



For Ti(SO4)2 and urea, the reaction is expected as following:



As for Ti(OR)4, it is first hydrolyzed (1) and then polymerized with each other through dehydration (2) or dealcoholization (3) reaction to produce ≡Ti–O–Ti≡ species. The hydrolyzed titania species are encapsulated over cenosphere via the catalytic self-condensation between the hydroxyl groups among titania species [20]. But for Ti(SO4)2 and urea, the Ti(OH)4 gel is first peptized and further recystallized into the TiO2 sol of nanoparticle in aqueous solution by a series of chemical reactions (4) to (6) [21]. So, more and more TiO2 nanoparticles are deposited on the surface of cenosphere via the self-assembly process with the hydrothermal reaction proceeding. In addition, these small caves play a fundamentally positive role in anchoring the TiO2 nanoparticles on the cenosphere surface.
EDS analysis
The EDS survey spectra of cenosphere before and after treatments are shown in Figure 2. The corresponding results are also given in Table 1. The compositions for the untreated cenosphere by mass percent are O 49.42%, Si 41.60% and Ca 8.98% (2.074, 9.434 keV ignorable). The corresponding atomic percents are O 64.43%, Si 30.89% and Ca 4.68%. It indicates that the cenosphere mainly comprises SiO2 and CaO, while other oxides of trace elements such as Al, K and Na are absent [22]. After treatments, the compositions by mass percent are O 52.95%, Si 34.23%, Ca 8.11% and Ti 4.71% for scheme one (2.074, 8.038, 9.421 keV ignorable) and O 66.75%, Si 15.34%, Ca 3.76% and Ti 14.15% for scheme two (2.067, 8.039, 9.431 keV ignorable), respectively. The corresponding atomic percents are O 68.54%, Si 25.24%, Ca 4.19% and Ti 2.03% for scheme one and O 83.54%, Si 9.61%, Ca 1.65% and Ti 5.20% for scheme two.
Energy dispersive X-ray spectroscopy survey spectra of: (a) the untreated and modified cenospheres with (b) tetrabutyl titanate and (c) titanium sulfate and urea. The compositions of the untreated and modified cenospheres.

XRD analysis
The XRD patterns of cenosphere before and after treatments are represented in Figure 3. It can be seen that there is no typical diffraction peak in the XRD pattern of the untreated cenosphere. After treatments, a series of characteristic peaks for both schemes are found at 2 X-ray patterns of: (a) the untreated and modified cenospheres with (b) tetrabutyl titanate and (c) titanium sulfate and urea.
DRS analysis
The diffuse reflectance spectra of cenosphere before and after treatments in the range of 200–800 nm are displayed in Figure 4. The results indicate a higher light-scattering and light-harvesting capability of TiO2-coated cenospheres in ultraviolet waveband. As compared with the untreated cenosphere, the average reflectances decrease about 23.1% in UVB (280–315 nm) region and 14.1% in UVA (315–400 nm) region for scheme one and 24.4% in UVB region and 17.3% in UVA region for scheme two, respectively. This is attributed to the band gap transition of TiO2 nanoparticles [25]. The absorption ability of TiO2 to visible light is slightly improved with respect to the untreated one. The mean reflectances in visible wavebands increase around 1.4% for scheme one and 2.2% for scheme two, respectively. By comparison scheme one with scheme two, the increase in the TiO2 coating seems to enhance the light-scattering capability to some extent. In addition, the relationship between ( Diffuse reflectance spectra of: (a) the untreated and modified cenospheres with (b) tetrabutyl titanate and (c) titanium sulfate and urea.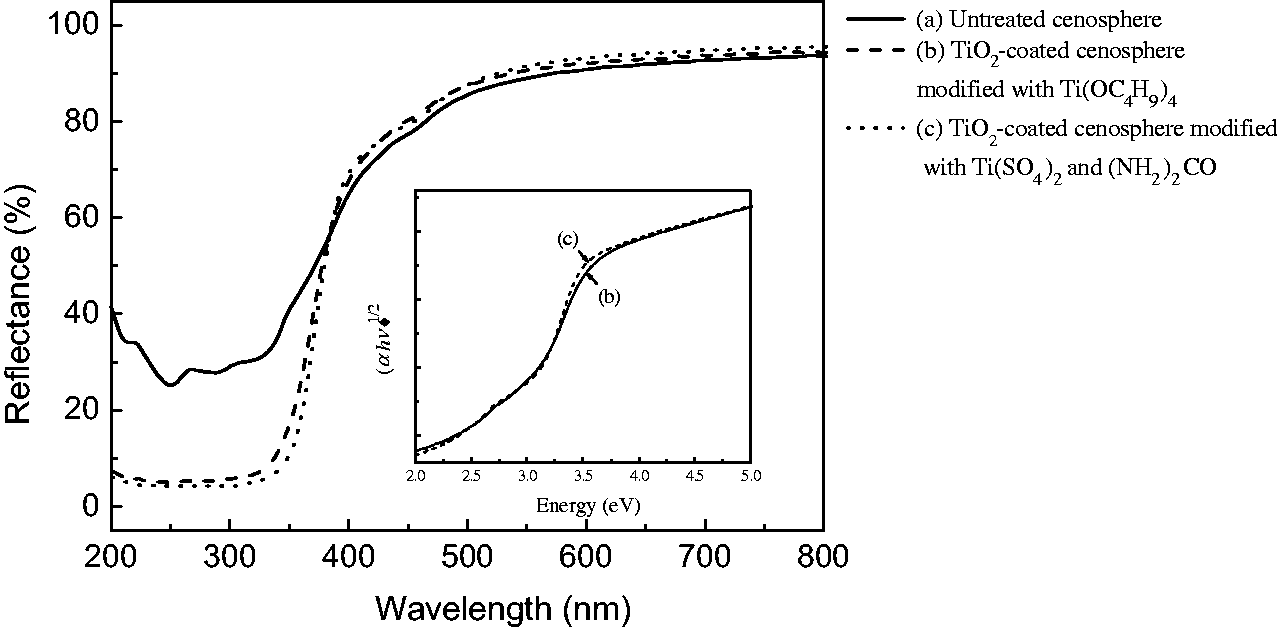
Photocatalytic activity
The transmission spectra of methylene blue solution exposed to UV irradiation at different times are exhibited in Figure 5. The transmission spectra of methylene blue solution vary with the irradiation time for three samples. The characteristic absorption peaks at 250 nm, 292 nm and 662 nm are gradually weakened with the increase of irradiation time. The changes of absorption peaks for both schemes are more significantly compared with the untreated one. The variation of degradation rate with irradiation time is revealed in Figure 6. It is clear that the degradation rate increases with the increase of irradiation time. After UV irradiation for 60 min, the degradation rates are 73.3% for the untreated cenosphere, 97.4% for scheme one and 97.6% for scheme two. It is clear that TiO2 surfaces become hydrophilic under UV irradiation [26]. When the TiO2 nanoparticle is irradiated by a ray equal or greater than the band gap energy (3.2 eV), an electron is transferred from capacity band to conduction band, which leads to a pair of holes and electrons on the surface of the TiO2 nanoparticle. The photo-generated electrons tend to reduce Ti4+ cations to the Ti3+ state and the holes oxidize O−2 anions. In this process, the oxygen atoms are thrown out and a group of oxygen vacancies is produced on the surface. The water molecules in the solution can occupy the empty sites and adsorbed OH groups are created on the surface, which increases the hydrophilicity of the TiO2 surface. At the same time, the photo-generated electrons react with the oxygen molecules in the solution and produce superoxide radical anions (O−2). The photo-generated holes react with water and (OH) radicals are produced. The molecules of methylene blue in water are absorbed on the surfaces of TiO2 nanoparticles. The strong oxidant reagents result in the deconstruction of the surface adsorbed dye molecules [27–29]. Therefore, the photocatalytic activity of TiO2-coated cenospheres can be attributed to the synergetic effects of hydrophilicity (hydroxyl groups), crystallinity and absorption in UV region [30].
Transmission spectra of methylene blue solution exposed to UV irradiation at different times. Variation of degradation rate with irradiation time.

The photodegradation of methylene blue follows approximately a first-order kinetic model by the linear transforms ln(
The results of the reduplicative photocatalytic degradation of methylene blue solution are presented in Figure 7. It can be seen that the degradation rate of methylene blue dye after being irradiated for 60 min decreases slightly with the increase of the experimental cycles. After 8 cycles, the degradation rate is about 87%, which implies the high photocatalytic activity of TiO2-coated cenosphere. By comparison with the tetrabutyl titanate modified cenosphere, the cenosphere modified with titanium sulfate has a higher degradation rate of above 90%. It also proves that the deposition of TiO2 nanoparticles has firmly attached to the cenosphere surface, not to be easily exfoliated from the cenosphere.
Effect of experimental cycle on the degradation rate.
Conclusions
TiO2 nanoparticles were immobilized on the surface of cenosphere using tetrabutyl titanate or titanium sulfate as the precursor by hydrothermal method. The SEM, EDS and XRD results revealed that anatase-type TiO2 nanoparticles were coated on the surface of cenosphere. The crystal size of TiO2 nanoparticle by using l titanate as the precursor was smaller than that by using titanium sulfate. The DRS and photocatalytic degradation of methylene blue dye results demonstrated that the enhancement to ultraviolet light of TiO2 nanoparticle prepared by titanium sulfate and urea had an advantage over that prepared by tetrabutyl titanate. The photocatalytic activity of TiO2 nanoparticle for methylene blue degradation by using titanium sulfate as the precursor was higher than that by using tetrabutyl titanate. The usage of cenosphere as catalyst support makes it potentially useful in a vast range of applications.
Footnotes
Funding
This work was supported by the Scientific and Technological Project (No. 2012R10012-09) from Department of Science and Technology of Zhejiang Province, China.
