Abstract
Cellulose acetate (CA) membranes and films were fabricated, respectively, by electrospinning and solvent casting. A systematic analysis of structure and properties was made to compare the differences between nanotexture and casting texture. Scanning electron microscopy, X-ray diffraction and thermal analysis were used to characterize the morphology, inner structure and thermal behavior of electrospun CA membrane, casted CA film as well as original CA powder. In addition, the pore size distribution and surface hydrophilicity of electrospun membrane and casted film were tested. It was found that the nanotexture produced by electrospinning process had a significant effect on the structure and properties of CA materials. The pore size distribution of electrospun membrane was more uniform, although the mean pore size was larger. In addition, the electrospun CA membrane possessed higher crystallinity and better thermal stability, compared with casted CA film. The results also showed that the electrospun membrane gave a contact angle of 127.2° and was observably more hydrophobic than the casted film, reporting a contact angle of approximately 74.4°.
Keywords
Introduction
As one of the most promising materials of the 21st century, nanofiber materials have attracted a great deal of attention in the past few decades. The electrospinning process is a straightforward method to produce fibers with nanometer scale diameters from polymer solutions or polymer melts, which endows the material with several amazing characteristics such as very large surface area to volume ratio and high aspect ratio compared with any other traditional materials. These perfect properties make polymer nanofibers optimal candidates in tissue engineering [1,2], wound dressing [3], drug release [4,5], sensor [6] and filter material applications [7,8].
Cellulose acetate (CA), an environment friendly and biodegradable regenerated cellulose material, can easily be fabricated as semipermeable membranes for separation processes and as fibers and films for textile and biomedical applications [9,10]. However, for CA films that are fabricated by solvent casting, the surface stability is poor and film structure is dense. Moreover, the casted films exhibit low chemical and thermal stabilities [10]. Therefore, the nanotexture produced by electrospinning may be adopted to improve the structure and properties of traditional casting texture.
As one of the most common biopolymers on earth, CA has tremendous potential use in electrospinning. Liu and Hsieh [11] produced ultrafine fibrous cellulose membranes by electrospinning and studied the deacetylation of ultrafine CA fibers. Smooth nanofibers with diameters ranging from 100 to 1000 nm were obtained. Han et al. [12] obtained electrospun CA nanofibers from a mixed solvent of acetic acid and water. The average diameters of the CA nanofibers could be controlled from 160 to 1280 nm by changing the composition of the mixed solvent. Tungprapa et al. [13] prepared ultrafine fibrous membranes by electrospinning from different solvent systems. The effects of solvent systems on the morphological appearance and size of electrospun CA products were thoroughly investigated. It was reported by Celebioglu and Uyar [14] that ribbonlike porous CA fibers were electrospun from a mixed solvent of dichloromethane and acetone. The morphology and the porous structure of the CA fibers were very much depending on the ratio of dichloromethane in the dichloromethane–acetone binary solvent system. More porous fibers were obtained due to the high volatility.
In this study, a comparative study of structure and properties on two different 2D platforms was made. CA platforms, both as electrospun membranes and as casted films, were systematically investigated to identify the differences, mainly pertaining to morphology, pore size distribution, degree of crystallization, thermal behavior and surface hydrophilicity. As a contrast, the crystallinity and thermal behavior of the original CA material were also tested.
Materials and methods
Materials
CA (white powder; degree of substitution, 2.43; degree of polymerization, 800–1000; molecular weight of single unit approximately, 303) was obtained from Nantong Acetate Fiber Company in Jiangsu Province, China.
The solvents used in this study were acetone (analytical grade; CH3COCH3; molecular weight, 58.08; boiling point approximately 56℃; volatile) and N,N-dimethylacetamide (DMAc; analytical grade; CH3CON(CH3)2; molecular weight, 87.12; boiling point approximately 160℃), all from Shanghai Lingfeng Chemical Reagent Co Ltd China. These chemicals were used as received.
Preparation of polymer solutions and CA films
The 8–13% (w/w) CA solutions were prepared using 2:1 (v/v) acetone/DMAc mixture solvent and gently stirred at room temperature until complete dissolution of polymer occurred.
The CA films were obtained from a 11% solution (w/w) of CA in 2:1 (v/v) acetone/DMAc mixture solvent. The films were cast by drawing on glass plates with a glass rod, followed by air drying for 48 h at room temperature.
Electrospinning setup
The experimental setup used for the electrospinning process is shown in Figure 1. The CA solution was poured into a syringe attached with a capillary tip of 1 mm diameter. A variable high power supply was used to produce a high DC potential across the capillary tip and an aluminum sheet used as the stationary screen collector. The electrode of positive polarity that was emitted was attached to the needle. The voltage used in the experiment was 15 kV and the current was adjusted to be constant. The flow rate was 0.8–1.0 mL/h and the distance between the tip and the collector was fixed at 20 cm. The electrospun nanofibers membranes were air-dried at room temperature for 48 h to remove residual solvent.
The schematic diagram of the electrospinning setup.
Measurements of CA materials
Scanning electron microscope test
The morphologies of the resulting membranes and films were examined by scanning electron microscopy (SEM; JSM-5600LV, Jeol Co, Japan) after gold sputtering. The average diameter of the electrospun nanofibers was obtained using an image analyzer (Photoshop CS). From each image, at least 100 different fiber segments were randomly selected and their diameters measured to generate the diameter of an average nanofiber.
Pore distribution test
The pore distribution of the resulting membranes and films was measured by a through-pore size analyzer (Porometer 3 G-zh). Porometry, a liquid expulsion technique, was used to measure the size and size distribution of through-pores. Pores are filled with a special wetting fluid (Quantachrome Porofil Wetting Fluid). Gas pressure emptied the pores according to size and the resulting flow through the pores was measured.
X-ray diffraction test
The microstructure changes in CA materials were studied by X-ray diffraction, using D/max-2550 PC X-ray diffractometer, which had the MDI jade software to evaluate the crystallinity of samples.
Thermogravimetric analysis and differential scanning calorimetric analysis
The thermal properties of CA materials were characterized by thermogravimetric analysis (TGA) (Netzsch TG 209 F1, Germany) and differential scanning calorimetry (DSC) (Netzsch 204 F1, Germany). For TGA, the heating rate was 10℃/min and the thermal degradation temperature ranged from 30℃ to 700℃. For DSC, the samples were tested at heating rate of 5℃/min in a nitrogen atmosphere at a gas flow rate of 20 mL/min.
Water contact angle measurement
The surface hydrophilicity of electrospun fibrous membranes and casted films was measured by a video contact angle instrument (OCA15EC, Germany). Deionized water (3 µL) was dropped onto the surface of samples and a photo was taken after the water droplet stabilized on the surface of the sample. Five readings were recorded for each sample and the average value was calculated.
Results and discussion
The morphology and pore distribution of electrospun membranes and casted films
When only acetone was selected as the solvent for electrospinning, the tip of the needle was easily blocked, which is attributed to the low boiling point of acetone. However, when pure DMAc was selected as the solvent, droplets of large size collected on the aluminum sheet. These phenomena were consistent with the results of Liu and Hsieh [11], who showed that both acetone and DMAc were not a desirable solvent for electrospinning CA. Therefore a mixture of acetone/DMAc solvent system at the ratio of 2:1(v/v) was used to dissolve CA and fabricate electrospun membranes.
Figure 2(a) to (f) shows the SEM images of CA-electrospun membranes fabricated from CA solutions of differing concentrations. When low CA concentrations (8–10%) were used for electrospinning, nanofibers with beads were generated. The numbers of beads decreased, the beads were larger in size and the distances between beads were increased, as the CA concentration increased. Meanwhile, the beads gradually changed in shape from an almost spherical to a spindle-like shape. It is known that the viscosity of the solution is one of the most important factors leading to the formation of beaded nanofibers [15,16]. Higher viscosity favored formation of fibers without beads. Improving CA concentration could lead to higher viscosity, so the beads of CA nanofibers would be reduced. In addition, Figure 2(c) shows that the nanofibers produced from 10% CA solution in the had an increased crosslinking phenomenon.
SEM images of electrospun CA membranes and casted films prepared from different concentrations of CA solutions: (a) 8% CA solution; (b) 9% CA solution; (c) 10% CA solution; (d) 11% CA solution; (e) 12% CA solution; and (f) 13% CA solution; and (g) casted film produced from 11% CA solution.
As shown in Figure 2(d) to (f), nanofibers without beads were generated. With the increasing of CA concentration, the diameter of the nanofibers showed a growth trend; however, the uniformity of the nanofibers reduced. Figure 3 shows that the average diameters of nanofibers increased from 333 to 373 nm with CA concentrations ranging from 11–13% (w/w). When a higher CA concentration was used, the error bar of the average diameter was larger, indicating that the CA nanofibers obtained from higher concentration were more uneven. So the electrospun membrane fabricated from 11% CA solution was selected to conduct a comparative analysis with casted film.
The average diameter of CA nanofibers produced from different concentrations.
The casted film was also produced from 11% CA solution. The morphology of casted film is shown in Figure 2(g). A certain number of pores were found to exist on the surface of casted film; however, the size of pores was uneven.
Figure 4 shows the pore size distribution of electrospun membrane and casted film, both produced from 11% CA solution. It was found that the casted film possessed a larger pore size distribution interval that ranged from 0.127 to 13.35 µm, which revealed that the pore size was extraordinarily uneven. Moreover, the main pore size of casted film ranged from 0.127 to 0.30 µm (as the inset shows in Figure 4(a)) and pore size more than 1µm accounted for only 0.1%. In contrast, the pore size distribution of electrospun membrane was more uniform, showing pore size distribution interval ranging from 1.884 to 5.185 µm (as shown in Figure 4(b)). As shown in Table 1, the mean pore size of electrospun membrane was larger than that of casted film. This shows that the pore size distribution of electrospun membrane was more uniform, although the mean pore size was larger.
The pore distribution of CA materials: (a) casted film; (b) electrospun membrane, both produced from 11% CA solution. The pore distribution parameters of electrospun membrane and casted film, both produced from 11% CA solution.

XRD characterization of CA materials
Figure 5 shows the X-ray diffraction patterns of three different CA materials. In the whole ranges of 2θ angle, it was found that the amorphous peak area of original CA material was significantly larger than that of casted film and electrospun membrane. To determine the degree of crystallization of three different CA materials, the conventional X-ray analysis was adopted and the XRD pattern was divided into two different contributions, amorphous and crystalline phases [17]. Van der Waals halo or amorphous halo, corresponding to the amorphous region of the material, was used to deconvolute the XRD pattern. It is known that the broad halo was usually located around 20° and presented in all organic polymers [18].
X-ray diffractograms of CA materials: (a) the original CA material without processing; (b) the casted film and (c) the electrospun fibrous membrane.
For original CA material (Figure 5(a)), when the 2θ angle ranged from 5° to 14°, there were three small intensive peaks, respectively, at 9°, 10° and 13°. In addition, one main diffuse peak exhibited at 20°. Therefore, the fibril of original CA material was constituted by crystalline and amorphous blocks and the maximum diffuse peak suggested that this material possessed basically amorphous characteristics. According to the calculation of software MDI jade, the crystallinity of original CA material was 28.24% and the ratio of corresponding amorphous region was 71.76%. Hydrogen bonding between the chains and water uptake seem to be responsible for this noncrystalline structure [19].
As shown in Figure 5(b), when the 2θ angle ranged from 5° to 60°, the casted CA film had a main intensive peak at 9° and an amorphous peak at 18°. According to the calculation of software MDI jade, the crystallinity of casted CA film was 34.41%. Compared with original CA material, the crystallinity increased by 6.71%, mainly due to the effect of the solvent. When CA was dissolved in the acetone/DMAc solvent mixture, hydrogen bonding between the chains was destroyed and recrystallization phenomenon occurred. Thus the crystallinity of casted CA film increased.
Figure 5(c) shows that the electrospun membrane had an obvious intensive peak at 9°, which was similar to that in the casted film. In addition, a diffuse peak exhibited at 24°. Through the calculation of software MDI jade, the crystallinity of electrospun fibrous membrane was shown to be 51.02%, which showed a dramatic improvement compared with the other CA materials. Kim et al. [17] and Zhao et al. [20] also obtained electrospun cellulose and cellulose derivative nanofibers with high crystallinity, i.e. 40–60%. There were two reasons for the increasing crystallinity of electrospun CA membranes. On the one hand, recrystallization occurred when CA was dissolved in the acetone/DMAc solvent mixture. On the other hand, the CA solution jet and the polymer macromolecule suffered more stretching in the electrospinning process; hence, the crystallinity increased. Qin and Wang [21] and Qin [22] also found that the stretching of electrostatic force is propitious to the formation of crystal of nanofibers.
DSC characterization of CA materials
The thermal data varied greatly for different materials depending on the methods of preparation and analysis [23], so investigations were undertaken under the same conditions. When the DSC test was adopted for the three samples, their initial weights, respectively, were 5.16 mg (Figure 6(a)), 5.39 mg (Figure 6(b)) and 5.44 mg (Figure 6(c)). The DSC thermograms of the three CA materials are shown in Figure 6.
DSC thermograms of CA materials: (a) the original CA material without processing; (b) the casted film and (c) the electrospun fibrous membrane.
Figure 6 shows that for the three samples a similar endothermic hump was observed between 30 and 110℃. This hump appeared at the onset temperature (
Unfortunately, pronounced glass transition was not observed in all three samples, mainly due to the disturbance of crystalline regions. The secondary heating curves (not shown in this study) showed a similar glass transition temperature of 185℃ for the three samples.
The melting peak and fusion enthalpy were observed in all three curves. It revealed that the three samples appeared approximately at the same
TGA of CA materials
When TGA was conducted for the three samples, their initial weights, respectively, were 8.4 mg (Figure 7(a)), 8.5 mg (Figure 7(b)) and 7.6 mg (Figure 7(c)). As shown in Figure 7, the three samples showed similar changing trend in the whole ranges of temperature. The decomposition appeared between 350℃ and 410℃, mainly due to a cellulose degradation process such as depolymerization, dehydration and the decomposition of glucosyl. Eventually, charred residues were formed [24,25].
TGA curves of CA materials: (a) the original CA material without processing; (b) the casted film and (c) the electrospun fibrous membrane.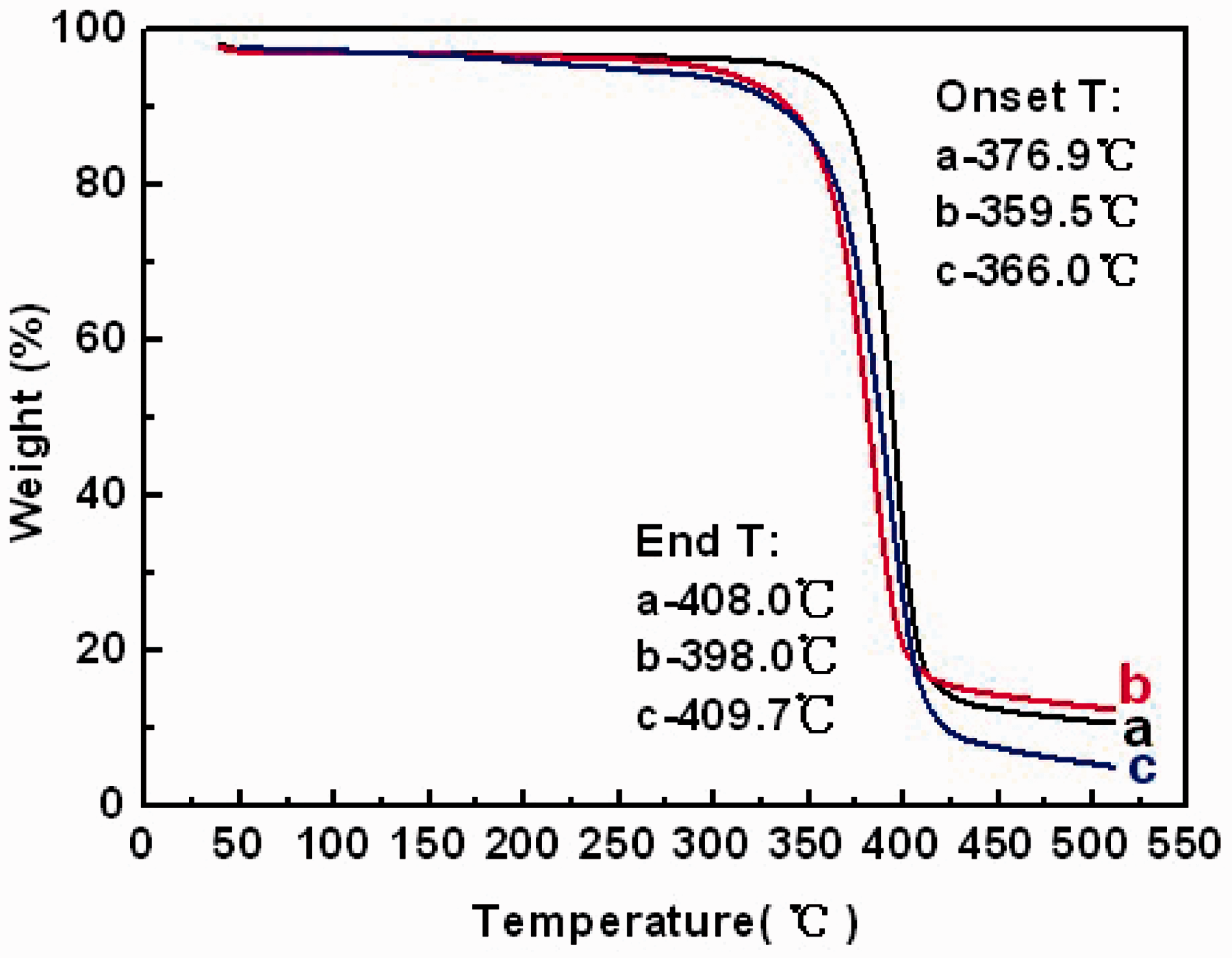
The characteristic parameters of thermoanalytical curves on TGA.

CA: cellulose acetate; TGA: thermogravimetric analysis;
As for the whole temperature range of decomposition (
The weight loss ratio could, only to some extent, reflect the change in internal structure and property within the material, but it was not entirely absolute. In the actual testing process, the weight loss ratio was affected by many factors, such as the initial weight and the testing temperature rate. Therefore, the weight loss rate was not used to measure the thermal stability in this study.
Surface hydrophilicity of electrospun membranes and casted films
The hydrophilicity of the CA electrospun membrane and casted film, which may affect their overall performance, was examined by contact angle measurement to compare the effect of nanotexture and casted texture on the surface hydrophilicity. Figure 8 shows that the surface of the casted film was hydrophilic, reporting contact angle of approximately 74.4°. However, the electrospun membrane gave a contact angle of 127.2° and was observably more hydrophobic than the casted film.
Contact angle of CA materials: (a) the casted film and (b) the electrospun fibrous membrane.
In theory, the wettability of liquid droplets on solid surfaces was governed mainly by both the chemical composition and the geometrical microstructure [26]. Conventionally, it was difficult to fabricate hydrophobic surface from hydrophilic materials. Herminghaus [27] reported that it was theoretically possible to construct a waterrepellent surface from a material using a material with a contact angle less than 90°. It was proved by Feng [28] and Feng’s group fabricated a superhydrophobic surface (Contact angle = 171.2°) from amphiphilic poly(vinyl alcohol).
In this study, the hydrophobic surface was obtained from hydrophilic CA material by electrospinning. This phenomenon may be attributed to the rearrangement of CA molecules during electrospinng process, which may be similar with the PVA molecular rearrangement found by Lin [28]. Hydrophobic groups were reoriented at the air/solid interface and the hydrogen bonds were formed in the interiors. The other reason was that the electrospun fibrous membrane possessed higher crystallinity (verified in Figure 5) compared with casted film, which was difficult for the diffusion and translation of water molecules. Therefore, it exhibited higher surface hydrophobicity.
Conclusions
In this study, CA membranes and films were fabricated successfully by electrospinning and solvent casting. A systematic analysis between nanotexture and casting texture showed that the nanotexture produced by electrospinning process had a significant effect on the structure and properties of CA materials.
The electrospun CA membrane with an average diameter of 333 nm exhibited more uniform pore size distribution, although the mean pore size was larger compared with the casted CA film. The electrospun CA membrane showed a contact angle of 127.2° and was observably more hydrophobic than the casted film, reporting a contact angle of approximately 74.4°. X ray diffraction test and DSC characterization showed that the crystallinity of electrospun CA membrane dramatically increased, compared with casted CA film as well as original CA powder. The thermal analysis indicated that the electrospun CA membrane possessed better thermal stability than did the casted CA film.
Footnotes
Funding
This study was partly supported by grants (50973014 and 11172064) from the National Natural Science Foundation of China and from the Foundation for the Author of National Excellent Doctoral Dissertation of P.R. China (200961), as well as sponsored by Shanghai Rising-Star Program in China (10QA1400100) and Fok Ying Tong Education Foundation (121071) to Prof. Xiaohong Qin. This study was also supported by Program for New Century Excellent Talents in University (NCET-10-0322) and the Fundamental Research Funds for the Central Universities as well as ‘Shu Guang’ (11SG33) project supported by Shanghai Municipal Education Commission and Shanghai Education Development Foundation. This work was also supported by Chinese Universities Scientific Fund (CUSF-DH-D-2013021) to Mr Shaohua Wu.
