Abstract
The cattail fiber assembly used as a sorbent for the sorption of engine and vegetable oils was investigated. The internal factors governing the oil sorption of cattail fibers were discussed. The analysis for the adsorption mechanism of cattail fiber assembly indicated that the tuft of cattail fibers has a down-like structure and, thus, its surface area and retaining space are large. The high surface waxiness and small surface free energy enabled the cattail fiber to have excellent oleophilic–hydrophobic property. The experimental results indicated that the oil-sorption rates of cattail fiber assemblies to engine oil and vegetable oil were 13.4 g/g and 14.6 g/g, respectively, and the oil retention capability to both engine oil and vegetable oil after 24 hours was over 95%. The cattail fibers have similar oil sorption capability with those of nonwoven wool materials, however, nature sorbents such as kapok fibers are superior to the cattail fibers.
Introduction
Oil is one of the most important energy sources in the world. However, when oil is explored, transported, stored and used, there will be spillage risk that may cause significant environmental damage [1]. The environmental pollution of water by oil has been a specific and serious problem [2,3]. Some advanced technologies have been developed and applied in oily water treatment such as microfiltration and ultra-filtration membranes [4–9], but the expensive cost limited the wide application of these filtration technologies. In recent years, considering the advantages of natural fibers as the low cost, biodegradability and reusability, the fibers such as cotton [10], kapok [11–15], cotton grass fiber [16], animal fibers [17,18] and other various vegetable fibers [19] were investigated.
Cattail fibers were from the fruits of cattail plants, and when cattail plant grew mature, the sheaths of cattail plant would automatically crack, and the cattail fibers would be blown away as flying wadding. Owing to the low cost and biodegradability, the cattail fibers were ever investigated as the use of biomass sorbents for oil removal from gas station runoff and polycyclic aromatic hydrocarbon removal from water [20,21]. The literatures indicated that cattail fibers had good capability of oil sorption, but the reasons were not given in detail. In addition, the literature regarding oil adsorption mechanism of cattail fibers is rather limited. In this paper, the oil sorption mechanism and capability of cattail fiber assembly as a biomass sorbent was investigated which would enlighten the application of the fiber in the field of removing oil from water.
Materials and methods
Cattail fibers
The cattail fibers were collected from the cattail plants growing near a lake of Songjiang, Shanghai, PR China. The mature spike of cattail and the cattail fibers are shown in Figure 1(a) and (b), respectively.
Cattail fibers: (a) the mature spike of cattail and (b) cattail fibers.
3-D rotational microscopy and the scanning electron microscopy (SEM) were used to observe the structures of cattail fibers, shown in Figure 2. Figure 2(a) showed the configuration of the tuft of cattail fibers something like the structures of down. The tuft of cattail fibers consisted of three parts including the stem, individual fibers and the seed. The individual fibers were divaricated from the stem at some points and the seed grows on the top of the stem. Figure 3(b) was the morphological structure of individual cattail fiber. The cross-section of cattail fiber was like belt which was sunken in intermediate and protruded on both sides something like the shape of “ The configuration of cattail fiber: (a) the tuft of cattail fibers and (b) individual cattail fiber. Oil on the surface of cattail fiber. ”.
”.


For the surface property of cattail fibers, the previous research indicated that cattail fiber was mainly composed of cellulose, hemicelluloses, pectin and a certain number of the surface wax. Soxhlet extractor method was ever used for the quantitative analysis of the wax on the cattail fibers. The result showed that the surface wax of the mature cattail fiber was up to 10.64% [22].
Adsorption mechanism of cattail fiber assembly
Oil sorption by cattail fibers could be explained by one or a combination of the following mechanisms: adsorption by interactions between waxes on fiber surface and oils and adsorption by physical trapping on the fiber surface through its irregular surface morphology.
The physical trapping were apparent for the cattail fibers. From Figure 2, the down-like structures and the open space of the cattail fibers supplied the large surface area and storage space for the liquid adsorption retention of oil. A great number of oil was trapped on the surface of cattail fiber, shown in Figure 3.
The parameters of oil liquids.

Waxes was generally deposited in the cuticle of cattail fibers, attractions between oil and the fiber surface were present through their hydrophobic interaction and van der Waals forces because both were hydrocarbons. Chemical compatibility between oil and the surface wax of cattail fibers led to minimum surface tension and contact angle for minimum energy barrier for oil to penetrate the fiber structure. After minimum energy barrier was overcome, void fraction availability or the presence of effective space inside the cattail fiber assembly would predominantly affect the sorption capacity of the fiber at specified packing density.
Oil liquids
The sample oils including engine oil and vegetable oil are shown in Figure 4. Dynamic contact angle tester (DCAT11) and SNB-2 digital spinning viscometer were used to test the properties of liquid. The tested data are shown in Table 1.
Oil liquids: (a) engine oil and (b) vegetable oil.
Oil-sorption of cattail fiber assembly
Oil sorption tests
The apparatus used in this test were beaker (500 ml), filter gauze, timer, electronic balance. The water sorption was tested at first as following. A total of 100 ml deionized water was firstly poured into the beaker, and then cattail fiber assembly weighing 1 g was placed in the liquid for 12 hours. The cattail fiber assembly was removed from the beaker with the filter gauze and then weighed with water sorption rate about 3% indicating the water repellency of cattail fiber assembly.
The existed literature ever investigated oil sorption for wool-based nonwoven material in pure oil medium and oil on aqueous medium, indicating wool-based nonwoven material had almost the same oil sorption capability [17]. Considering the water repellency of cattail fibers and the research in the existed literature [17], the oil sorption in pure oil medium was focused on in this paper.
The oil sorption test was done as following, 100 ml oil liquid was firstly poured into the beaker, and then the cattail fiber assembly weighing 0.5 g was placed in the oil liquid for 1 minute. Then, the assembly of cattail fibers was removed from the beaker with the filter gauze. The assembly with the oil sorption was stood on the filter gauze for 15 minutes and weighed as the maximum oil sorption of fiber assembly. And also, the mass of oil sorption of cattail fiber assembly was weighed after hanging for 24 hours. All tests were performed at 20 ± 2℃ and 65% ± 3% relative humidity.
Oil sorption
The oil sorption capability and oil retention capability were used to express the oil sorption of the assembly of cattail fibers.
The oil sorption capability was the rate of the maximum mass of oil sorption of cattail fiber assembly to the mass of cattail fiber assembly, shown as equation (1).

The oil retention capability was the rate of the mass of oil sorption of cattail fiber assembly hanging for the 24 hours to the mass of the maximum mass of oil sorption of cattail fiber assembly, shown as equations (2).
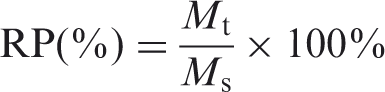
Results and discussion
Oil sorption of cattail fiber assembly.

From Table 2, it was found that, the oil sorption capability of cattail fiber assembly to engine oil and vegetable oil were 13.4 g/g and 14.6 g/g, respectively, and the oil retention capability after 24 hours both to engine oil and vegetable oil was over 95%. The results indicated that the assembly of cattail fibers had excellent characteristic of oil adsorption. As for the different oil liquids, the results showed a little difference due to the difference of oil liquids. Taking the engine and vegetable oil as an example, the capability of oil sorption was inversely proportional to oil viscosity, the higher the viscosity, the more difficult was the oil penetrating into cattail fibers.
Oil sorption capacity of some natural sorbents.
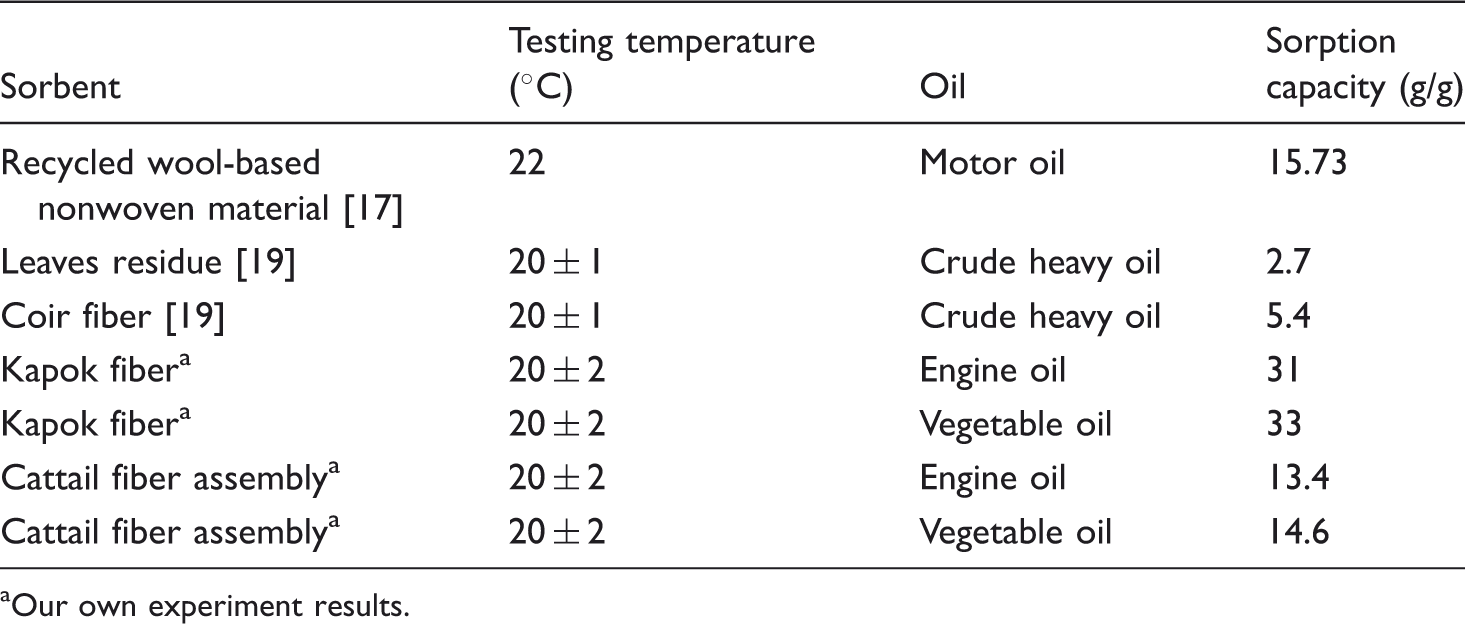
Our own experiment results.
From Table 3, it could be seen that the cattail fiber assembly as the sorbent was not as good as the kapok fiber but better than some vegetable fibers such as coir fiber and leaves residue. The oil adsorption of cattail fiber assembly was similar as that of the wool nonwoven material. But considering the oleophilic–hydrophobic characteristic, low cost and biodegradability, the cattail fiber as a natural sorbent could be well used in the field of removing oil from water.
Conclusions
The assembly of cattail fibers had the characteristic of good oil adsorption with the oil sorption capability to engine oil and vegetable oil 13.4 g/g and 14.6 g/g, respectively, and the oil retention capability to engine oil and vegetable oil after 24 hours was over 95%. The cattail fiber assembly as the natural sorbent had the capability of oil sorption approaching that of wool nonwoven material and fell between the levels of natural sorbents of leaves residue and kapok fiber.
The internal factors determining the oil sorption of fiber assembly were investigated. The results indicated that, the down-like structures of the tuft of cattail fibers could supply the porous structures of the assembly. And the special configuration of ‘ ’ determined the fiber has open space with large surface area and retaining space. The surface waxiness and the small surface free energy enabled the cattail fiber to have the excellent oleophilic–hydrophobic characteristic.
’ determined the fiber has open space with large surface area and retaining space. The surface waxiness and the small surface free energy enabled the cattail fiber to have the excellent oleophilic–hydrophobic characteristic.
Footnotes
Acknowledgements
The authors would like to thank Ms Wen-ying Cao and Ms Yun-hong Deng for their contributions in some of the experiments in this study.
Funding
This work was supported by ‘the Fundamental Research Funds for the Central Universities’.
