Abstract
A temperature-sensitive hydrogel was successfully grafted on the surface of non-woven poly(propylene) materials. This was carried out by the application of unmodified β-cyclodextrin and
Keywords
Introduction
Although there is a huge interest in the use of stimuli-responsive polymers for textile applications [1–3], this technology has not yet found many applications in textile science and technology. Temperature-sensitive polymers which become insoluble below or above a particular temperature, lower or upper critical solution temperature (LCST or UCST), respectively, are categorized as intelligent hydrogels. Among intelligent hydrogels, poly(
Cyclodextrins (CDs) are cyclic oligosaccharides which derive from starch. Among this cyclic oligosaccharides, α-, β-, and γ-CDs and a range of their chemical derivatives are commercially available [17]. CDs possess a relatively hydrophobic cavity and a hydrophilic outer shell. This structure causes the remarkable general ability to form inclusion complexes with guest molecules and provides vast applications, for example formulation of drugs in drug delivery systems [18–24] and controlled release experiments of fragrances and antibacterial compounds for textile applications [25–27].
Novel hydrogels were constructed by copolymerization of NIPAAm with modified CDs [28–33]. One study has indicated that CDs indeed can improve some properties of the formed hydrogels such as their low response rate [31]. Studies have indicated that a novel function can be achieved when a molecular recognition host (CD) and a stimuli-responsive polymer (PNIPAAm) are integrated into the same material [32–34]. Also, it has been reported that the binding constants of CD with guest molecules are influenced dramatically by temperature variation across the LCST [35]. By the use of a plasma-graft pore-filling polymerization technique, preparation of a temperature-dependent molecular-recognizable membrane has been carried out by grafting of PNIPAAm and modified β-CD onto the pores of a gating membrane [36,37]. In these studies, the modification of β-CD has been done before the grafting step, resulting in a prolonged and expensive process. These modified membranes have been used as a separating system for organic liquids. A systemic study on single-step simultaneous employment of hydrogels and unmodified CDs on textiles has not been reported previously. Thus, in this study, unmodified β-CD was used instead of modified β-CD and the possibility of the formation of a hydrogel which includes PNIPAAm and unmodified β-CD was investigated onto the surface of non-woven poly(propylene) (PP) materials. With respect to this, PNIPAAm and unmodified β-CD were grafted onto the surface of plasma-treated non-woven PP materials by the use of graft polymerization technique. Thereafter, the existence of β-CD molecules in grafted PNIPAAm on the non-woven PP surface was investigated. The main aim of this investigation was to achieve a temperature-sensitive textile with an ability to include the guest molecules such as fragrances and antibacterial molecules.
Experimental
Materials
Plasma treatment
This study employed a direct current pulsed plasma treatment for the modification of materials. The apparatus (Glow discharge Plasma from Fanavar Amin Company, Iran) consisted of a DC power supply, matching box, rotary pump, horizontal chamber, gas supply with appropriate pressure gauges, current and voltage probes, and digital oscilloscope. The chamber was cylindrical (60 cm in diameter, 65 cm in length) with a cathode (27 cm in diameter) that was powered through the matching box. Plasma formed between the cathode and the wall of the chamber. We used non-woven PP with a thickness of 0.14 mm and weight 40 g/m2. Plasma treatment was applied to produce free radicals on the non-woven PP surface [38]. Samples were cut into circles (7 cm diameter) and washed in ethanol at room temperature for 24 h. Washed non-woven PP was dried in a vacuum oven at room temperature for 24 h and weighed immediately. Afterward, non-woven PP was placed inside of plasma instrument, on the platform at the center of the chamber. Pressure (6.6 × 10−5 bar) was adjusted by a continuous flow of argon gas (>99.9%), and the plasma treatment was initiated at 480 W for 30 min at room temperature.
Graft polymerization
Graft polymerization on the plasma-treated non-woven PP materials was carried out according to the previously published method [39]. First, 10 mL of an aqueous solution of NIPAAm (900 mg), β-CD (300 mg), and BIS (35 mg) were prepared and stirred for 8 h to allow the chemicals to dissolve and mix completely. APS (11 mg) and TEMED (14 mg) were added to the solution to initiate free radical polymerization. To prepare the non-woven PP materials for grafting with PNIPAAm and unmodified β-CD (PNIPAAm/β-CD-g-PP), the plasma-treated non-woven PP materials were swiftly transferred to the polymerization solution. The process was repeated for the preparation of non-woven PP materials grafted with PNIPAAm (PNIPAAm-g-PP), which was made without β-CD. The mixtures were allowed to react for 24 h at room temperature, and subsequently, the samples were rinsed by several portions of distilled water for 6 days in order to remove unreacted homopolymers and non-attached materials. Thereafter, washed non-woven PP was dried in a vacuum oven at room temperature for 24 h and weighed immediately. The grafting yield (GY) of non-wovens was determined using equation (1):

Water uptake
For investigating the temperature response of the samples, their water uptake (WU) behavior was studied by soaking them in a temperature-controlled water bath at different temperatures (10℃, 25℃, 30℃, 32℃, 40℃ and 50℃). After 4 h of soaking, the samples were removed from the water bath and gently blotted with filter paper to remove surface water, followed by immediate weighing. The WU% was calculated by equation (2):
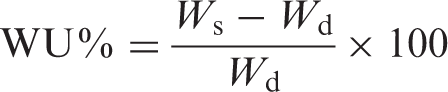
Wicking ability
To examine the effects of the grafting, the wicking ability of the non-woven PP materials were evaluated by a vertical wicking test using water baths set at 20℃ and 50℃, respectively. Samples were cut into the strips (5 cm long × 2 cm wide) and taped vertically with a string with about 2 cm of the strip immersed in water and weighted with a paper clip. The test was carried out with water at room temperature (20℃) and water warmed with a hot plate (50℃). The height to which the water was taken up along the strip was measured at 5, 10, 15, 20 and 25 min intervals.
Instrumental analyses
The grafted non-woven PP surfaces were characterized by Fourier transform infrared attenuated total reflection (FTIR-ATR) spectra (BOMEM Hartmann and Braun, MB-series). The LCST was determined by differential scanning calorimetry (DSC; DSC 2010 calorimeter, TA Instruments) in samples swelled in deionized water, and at a heating rate of 1℃/min. The onset point of the endothermic peak was used for the LCST determination. The surface morphologies of the samples were observed by scanning electron microscopy (SEM, Philips-XLC) after coating with gold using SEM coating equipment. We used a Cary Eclipse fluorescence spectrophotometer (Varian) to confirm the presence of β-CD in hydrogel grafted on the surface of non-woven PP by changes of the fluorescence intensity of ANS. First, the plasma-treated non-woven PP, PNIPAAm-g-PP, and PNIPAAm/β-CD-g-PP were immersed in 30 mL of ANS at a concentration of 5 × 10−5 M at room temperature for 24 h. Then, the fluorescence intensity of ANS absorbed by the samples was measured on the fluorescence spectrophotometer with excitation wavelength 350 nm, and the emission rate measured in the range 400–600 nm.
The content of β-CD in PNIPAAm/B-CD-g-pp was determined by elemental analysis (Perkin Elmer 2400 series II CHNS Analyzer) using equations (3) and (4).

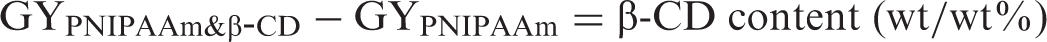
Dynamic contact angle measurements were carried out using a dynamic Wilhelm plate technique (Dataphysics Instruments GmbH. SCAT) at 20℃ and 40℃. The speed and immersion depth were 0.05 mm/s and 5 mm, respectively, and six readings were averaged.
Results and discussion
FTIR-ATR results
Figure 1(a) indicates the characteristic peaks of unmodified non-woven PP materials. The absorption band of C–C stretching at 1164 cm−1, CH3 bending at 1375 cm−1, and CH2 bending at 1454 cm−1 appeared. Furthermore, absorption bonds of CH2 and CH3 stretching can be seen at 2949, 2868 and 2837, 2916 cm−1, respectively [40]. For PNIPAAm-g-PP, several peaks at 1648, 1521, and 3400 cm−1 were found, which can be attributed to the characteristic peaks of C = O stretching, N–H deformation (amide I, amide II), and –NH stretching, respectively (Figures 1(b) and 2(a)). These peaks confirm the presence of PNIPAAm groups on the surface of the non-woven PP materials [33]. Figure 2(b) illustrates characteristic peaks of the observed bond around 1040 cm−1, which can be assigned to characteristic stretching vibrations of C–OH of β-CD [28], indicating the presence of β-CD in the synthesized hydrogel on the surface of non-woven PP.
FTIR-ATR spectra of unmodified non-woven PP materials (a), PNIPAAm-g-PP (b), and PNIPAAm/β-CD-g-PP (c). FTIR-ATR spectra of PNIPAAm-g-PP (a) and PNIPAAm/β-CD-g-PP (b).

Water uptake
Figure 3 shows WU% for unmodified non-woven PP materials, plasma-treated non-woven PP materials, PNIPAAm-g-PP, and PNIPAAm/β-CD-g-PP at different temperatures. As expected, no change in WU% was observed for unmodified and plasma-treated non-woven PP materials. However, for PNIPAAm-g-PP and PNIPAAm/β-CD-g-PP, the WU decreased with increasing temperature. This indicates that PNIPAAm/β-CD-g-PP as well as PNIPAAm-g-PP still possess the intrinsic properties of PNIPAAm. PNIPAAm/β-CD-g-PP exhibited thermo-sensitive response similar to PNIPAAm-g-PP with almost identical LCST. Both of these hydrogels are swollen below their LCST and shrunken above their LCST. However, the WU% of PNIPAAm/β-CD-g-PP is higher than that of PNIPAAm-g-PP at the temperature above the LCST. It may be attributed to the presence of the β-CD component which can effectively prevent the PNIPAAm component from forming a more compact structure [28].
The WU% of unmodified non-woven PP materials, plasma-treated non-woven PP materials, PNIPAAm-g-PP, and PNIPAAm/β-CD-g-PP as a function of temperature.
Morphological analysis by SEM
Figure 4(a) to (c) displays the morphology of non-woven PP materials before and after grafting with PNIPAAm and PNIPAAm/β-CD, respectively. The porous structure of non-woven PP is clearly seen in Figure 4(a). The grafting layers of PNIPAAm (GY% = 15.71) and PNIPAAm/β-CD (GY% = 6.92) can be observed vividly on the surface of non-woven PP in Figure 4(b) and (c), respectively. However, the grafted PNIPAAm/β-CD layer is not smooth and exhibits a different structure from the grafted PNIPAAm layer structure. One possibility is that this structure is the result of the removal of unbound β-CD from the hydrogel after the washing of the samples. A hydrogel with a porous structure can be formed as a result. Another possibility is that the way of polymerization of NIPAAm on non-woven PP materials has been influenced with the introduction of β-CD in NIPAAm monomer. Previous studies have revealed a net-shaped structure for PNIPAAm grafted on non-woven PP [32]; therefore, this contradiction in morphologies could be due to the presence of β-CD in the newly synthesized hydrogel system.
SEM micrographs of unmodified non-woven PP materials (a), PNIPAAm-g-PP (GY% = 15.7%) (b), and PNIPAAm/β-CD-g-PP (GY% = 6.92) (c).
Wicking
As the wicking ability of PNIPAAm changes with temperature, it is expected that the wicking ability of non-woven PP materials grafted with PNIPAAm alters with temperature similarly. Figure 5(a) and (b) presents the wicking time of unmodified non-woven PP, plasma-treated non-woven PP, PNIPAAm-g-PP, and PNIPAAm/β-CD-g-PP at 20℃ and 50℃, respectively. The results indicate that the wicking of unmodified non-woven PP materials and plasma-treated non-woven PP was almost zero at 20℃ and 50℃ after 25 min. However, the plasma-treated non-woven PP materials were expected to have higher wicking in comparison with unmodified PP due to the formation of −OH groups on the surface. In contrast, the wicking time of PNIPAAm-g-PP and PNIPAAm/β-CD-g-PP increases with increasing temperature from 20℃ to 50℃. This is due to the thermo-sensitive behavior of both of these hydrogels, as they are hydrophilic at temperatures below their LCST (20℃) and hydrophobic at temperatures above their LCST (50℃) [13,41,42]. At temperatures below the LCST, the hydrogel grafted on the surface of the non-woven PP is swollen and this narrows the diameter of the capillaries between the fibers in the non-woven PP materials, and consequently, narrower capillaries decrease the wicking time. On the other hand, at temperatures above the LCST, the shrinking of the hydrogels results in larger capillary diameters which enhances the wicking time [43]. Therefore, the swelling and deswelling of the hydrogel are the main reasons for changes in the wicking of the grafted samples.
The wicking rate of unmodified non-woven PP materials (black line), plasma-treated non-woven PP materials (red line), PNIPAAm-g-PP (yellow line) and PNIPAAm/β-CD-g-PP (blue line) at 20℃ (a) and 50℃ (b) as a function of time.
Furthermore, for the plasma-treated non-woven PP materials, the presence of capillaries with a large diameter is expected due to the absence of hydrogel. Most likely, this is the reason for their wicking inability. In addition, Figure 5(a) and (b) illustrates the faster wicking time of NIPAAm/β-CD-g-PP than that of PNIPAAm-g-PP at 20℃ and 50℃. Since water molecules can diffuse faster into PNIPAAm/β-CD-g-PP than PNIPAAm-g-PP, the total wicking ability of PNIPAAm/β-CD-g-PP is higher than that of PNIPAAm-g-PP. This can be attributed to the differences in the hydrophilic nature of the two components (CD and PNIPAAm).
Contact angle
Advancing contact angle of unmodified non-woven PP, PNIPAAm-g-PP, and PNIPAAm/β-CD-g-PP as a function of temperature.

Three individual samples were used.
PP: poly(propylene); PNIPAAm-g-PP: PP materials grafted with poly(
DSC measurements
The endothermic peaks observed in Figure 6(a) and (b) have been attributed to the unique and rapid alteration of the hydrophilic and hydrophobic states of the thermo-responsive non-woven PP materials [4,5]. In other words, at temperatures below the LCST, hydrogen bonds between the hydrophilic groups of the hydrogel and the water molecules are formed and at temperatures above the LCST, these hydrogen bonds are broken [4,5]. The LCST was determined by DSC to be about 31.9℃ and 33.2℃ for PNIPAAm-g-PP and PNIPAAm/β-CD-g-PP, respectively (Figure 6(a) and (b)). These temperatures are close to the LCST of pure PNIPAAm, suggesting that the incorporation of CD did not affect the thermal transition of PNIPAAm. However, the results of wicking and contact angle tests indicated higher hydrophilicity of PNIPAAm/β-CD-g-PP in comparison to PNIPAAm-g-PP. These results confirm that β-CD is probably entrapped between the hydrogel chains and there is no chemical bond between β-CD molecules and hydrogel. Therefore, the almost identical LCST of PNIPAAm-g-PP and PNIPAAm/β-CD-g-PP can be explained by the fact that each component can keep its own property independently.
Thermal analysis by DSC for PNIPAAm-g-PP (a) and PNIPAAm/β-CD-g-PP (b).
Fluorescence spectrophotometer measurements
The existence of CD molecules can be further demonstrated indirectly by fluorescence measurements of ANS. High fluorescence intensity implies a more hydrophobic environment such as the cavities of β-CD. As shown in Figure 7, the increase in fluorescence intensity of ANS in the PNIPAAm/β-CD-g-PP can be attributed to the presence of β-CD. As fluorescence is observed from the surface of a solid sample (non-woven PP materials) placed at a 45° angle on the incident light path, light scattering is also recorded. This is shown from the intense and broad shoulder from 400 nm, which is due to elastic scattering (Raleigh scatter) of the incident beam at 350 nm. The weak peak at 554 nm could be due to inelastic scattering (Raman scatter), which is typically orders of magnitude less than Raleigh scatter. However, the putative corresponding Stokes shift of 554 − 350 = 204 nm (wavenumber = 10,600 cm−1) would be too large to fit any known vibrational frequencies, in the mid-IR range. Therefore, it was concluded that the weak peak at 554 nm comes from fluorescent molecules in the non-woven PP materials itself. Traces of TiO2 particles could be the reason for this fluorescence. In the context of our experiment, the peak at 554 nm is not important, as it does not interfere much with the ANS signal. As Figure 7 shows, there is an Fluorescence spectral changes of ANS in plasma-treated non-woven PP materials (a), PNIPAAm-g-PP (b), and PNIPAAm/β-CD-g-PP (c).
Elemental analysis
Elemental analysis result for plasma-treated non-woven PP, PNIPAAm-g-PP, and PNIPAAm/β-CD-g-PP.

PP: poly(propylene); PNIPAAm-g-PP: PP materials grafted with poly(
Conclusion
In this study, β-CD-incorporated hydrogel was synthesized onto the surface of plasma-treated non-woven PP materials using unmodified β-CD and NIPAAm monomer through graft polymerization technique. There are two possibilities for associating the unmodified CD molecules in hydrogel (PNIPAAm). They could be either attached on NIPAAm polymeric chains and textile with a chemical bonding or physically entrapped. The results showed that PNIPAAm/β-CD-g-PP possesses improved the wicking ability and the hydrophilic properties as well as the capability of inclusion complex formation with ANS as a guest molecule, confirming the existence of unmodified β-CD molecules between NIPAAm polymer chains. Since there is no remarkable change in the LCST of the grafted hydrogel, it seems that unmodified β-CD has been entrapped between the PNIPAAm chains without creation of a chemical bonding. On the other hand, the possibility for the formation of a chemical bonding with unmodified β-CD is weak due to its inactivity. It can be concluded from the results of the performed studies that the second possibility is most probable. The content of β-CD in this study by elemental analysis was determined to be 97 µg/cm2. The PNIPAAm/β-CD-g-PP due to its temperature-sensitive properties provides new applications of hydrogels in textile technology.
Footnotes
Acknowledgment
The authors thank Fibertex Co. for the preparation of non-woven PP materials.
Funding
We wish to express our gratitude to the Research Affairs Division Isfahan University of Technology (IUT), Isfahan, for Support. Further partial Financial support from the research Affairs Division Aalborg university (aau), Demmark is gratefully acknowledged.
