Experimental section
Reagents and instruments
Allylamine (99%), diallylamine (98%), acryloyl chloride (98%), mono-propargylamine (98%), D-penicillamine (99%), allyl glycidyl ether (99%), 10-undecylenoyl chloride (99%), and methacryloyl chloride (99%) were purchased from Xiya Reagent Co. Ltd. (Shan Dong, China). 1,2-Epoxy-5-hexene (>96.0) was purchased from Tokyo Chemical Industry Co., Ltd. and 1, 2-hydroxy-7-octene (97%) was purchased from Thermo Fisher Scientific Co., Ltd. (China). β-Cyclodextrin (β-CD) was purchased from Shandong Binzhou Zhiyuan Bio-Technology Co., Ltd. (Binzhou, China). A FA2004 type electronic balance (Shanghai Hengping Scientific Instrument Ltd.) and XMTD-204 digital thermostat water bath equipment (Jiangsu Jintan Huacheng Kaiyuan Experimental Instrument Factory) were used in the experiments. An EQUINOX 55 Fourier transform infrared spectrometer (German Bruker Optics Company) and an Agilent Technologies 6540 UHD Accurate-Mass Q-TOF LC/MS (Agilent Technologies Inc) were used for product characterization. NMR spectra were obtained on a Bruker AV-600 MHz nuclear magnetic resonance spectroscopy, using DMSO-d6 or D2O as solvents. The chemical shifts (δ) are given in ppm relative to the TMS signal at 0.00 ppm as an internal reference, coupling constants (J) are given in Hertz (Hz).
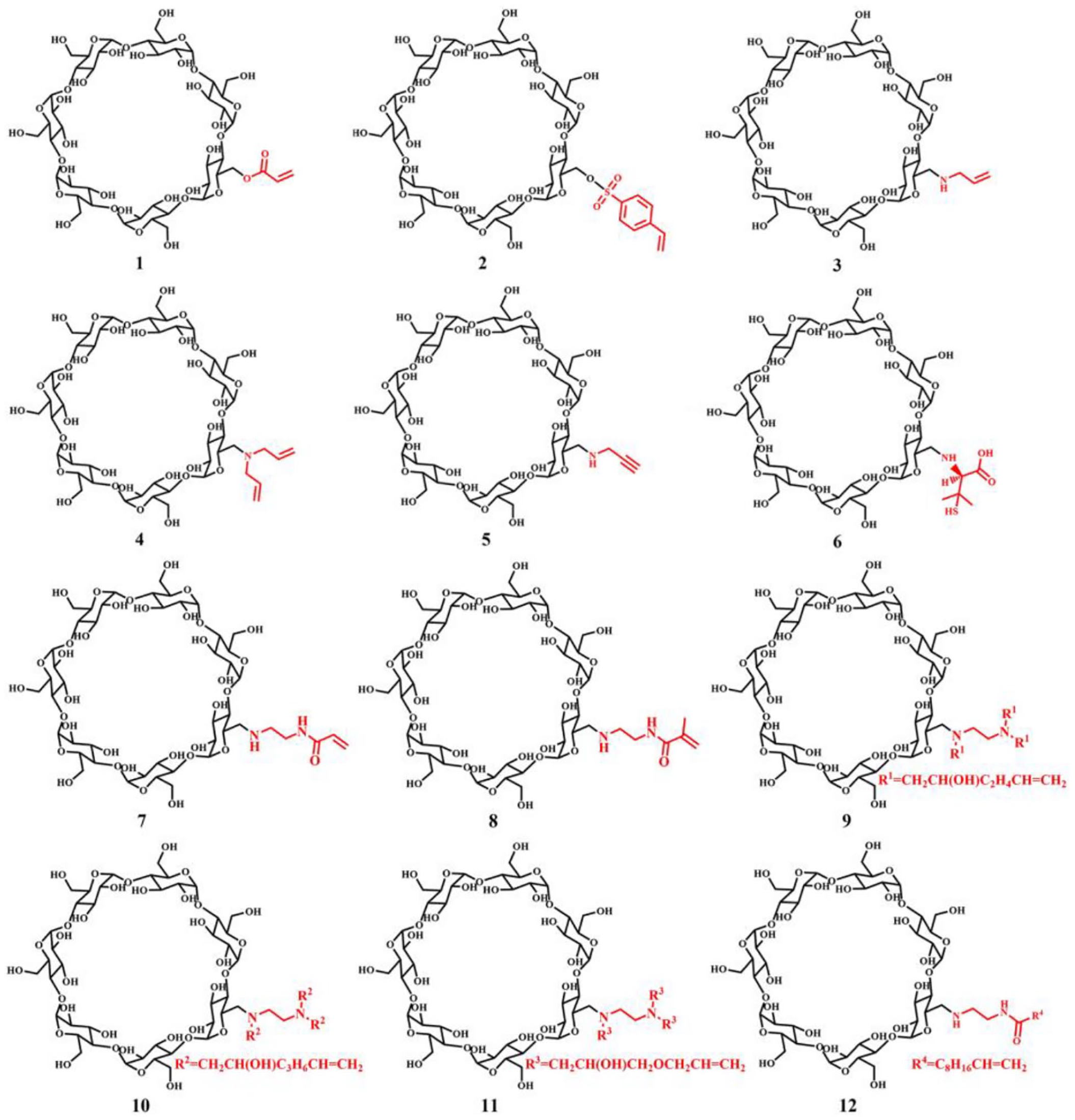
Synthesis of mono-(6-O-acryloyl-6-deoxy)-β-CD (1)
β-CD (7.9 g, 7.0 mmol) was added to a 500 mL round-bottomed flask. Next, NaOH (4.3 g, 0.11 mol) and distilled water 250 mL were added. After the solids had dissolved, the flask was placed in an ice-water bath (1.5 °C). Acryloyl chloride 10 mL was added to the flask by using a constant-voltage funnel. After stirring for 30 min, the solution was filtered and dried under reduced pressure at 40 °C. Then crude product was dissolved in N,N-dimethylformamide 20 mL, and the solution filtered to remove sodium chloride. Acetone 200 mL was added and the product precipitated and was collected. It was dissolved in methanol 20 mL and acetone 200 mL was added and the precipitate collected. The resulting product was dried under reduced pressure at 40 °C for 5 h.
Off white powder (yield 63%); m.p. 280–283 °C. The molecular weight of compound 1 is 1189.3803, with the found mass-charge ratio of 1157.3579 corresponding to [M – acryloyl + Na]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3418, 2935, 1719, 1638, 1514, 1441, 1300, 1156, 1030, and 867; 1H NMR (600 MHz, D2O): δ 5.64 (s, 1H), 5.33 (s, 1H), 5.06 (s, 1H), 3.88 (m, 3H), 3.68 – 3.49 (m, 2H), 1.85 (s, 3H), 1.67 (s, 1H), 1.45 (s, 1H), 1.31 (s, 1H), 1.04 (d, J = 6.8 Hz, 1H); 13C NMR (151 MHz, D2O): δ 177.53, 142.21, 122.03, 120.57, 101.93, 81.06, 76.20, 75.07, 72.92, 72.03, 60.28, 45.99, 28.55, 25.21, 24.05, 19.02, 17.59.
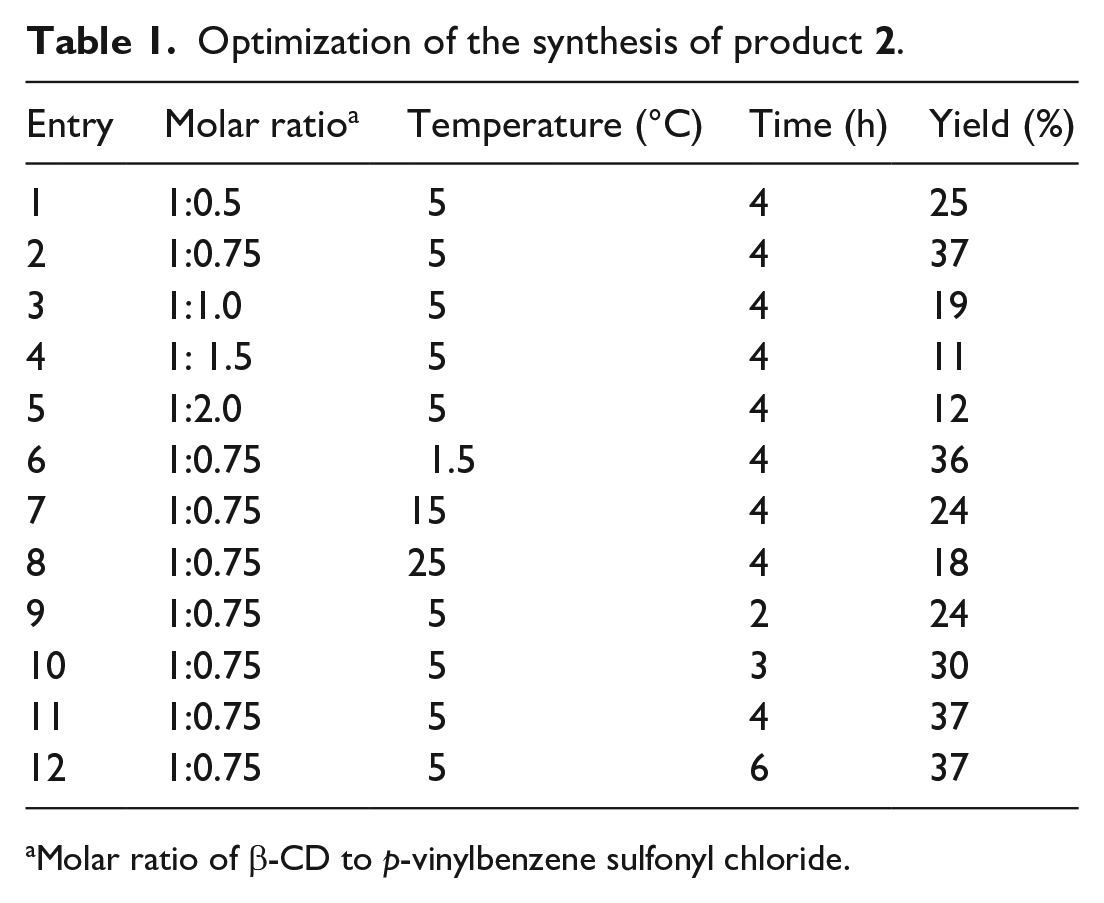
Synthesis of mono-(6-O-p-vinylbenzene sulfonyl-6-deoxy)-β-CD (2)
β-CD (22.7 g, 20 mmol) was added to a 250 mL three-necked flask. Next, NaOH (4 g, 0.1 mol) and distilled water 200 mL were added. After the solids had dissolved, the flask was placed in an ice-water bath (1.5 °C). Then p-vinylbenzene sulfonyl chloride (2.9 g, 15 mmol) was dissolved in acetonitrile 8 mL and added to the flask by a constant-voltage funnel. After stirring for 4 h, the pH of solution was adjusted to 1.5 with 1 mol/L HCl and the flask was placed in a refrigerator at 4 °C for overnight. Then the solution was filtered and recrystallized. The obtained product was dried under reduced pressure for 5 h at 55 °C.
White powder (yield 37%); m.p. 230–233 °C. The molecular weight of compound 2 is 1323.3677, with the found mass-charge ratio of 1324.3762 corresponding to [M + Na]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3417, 2923, 1637, 1417, 1367, 1193, 1177, 1157, 1079, 1029, 937, 836, 757, 663, and 578; 1H NMR (600 MHz, DMSO-d6): δ 7.84 (d, J = 8.1 Hz, 2H, Ph-H), 7.70 (d, J = 8.1 Hz, 2H, Ph-H), 6.83 (dd, J = 17.5, 11.0 Hz, 1H, -CH=CH2), 6.05 (d, J = 18.0 Hz, 1H, -CH=CH2), 5.51 (d, J = 10.9 Hz, 1H, -CH=CH2), 4.82 (d, J = 8.9 Hz, 5H), 4.75 (d, J = 2.8 Hz, 2H), 4.35 (d, J = 10.1 Hz, 1H, -CH2), 4.23 – 4.18 (m, 1H, -CH2); 13C NMR (151 MHz, DMSO-d6): δ 143.04, 135.52, 134.69, 128.55, 127.39, 119.27, 102.69, 102.41, 82.12, 81.97, 81.75, 81.29, 73.50, 73.33, 73.21, 72.85, 72.50, 72.32, 69.37, 60.33, 60.01, 59.76.
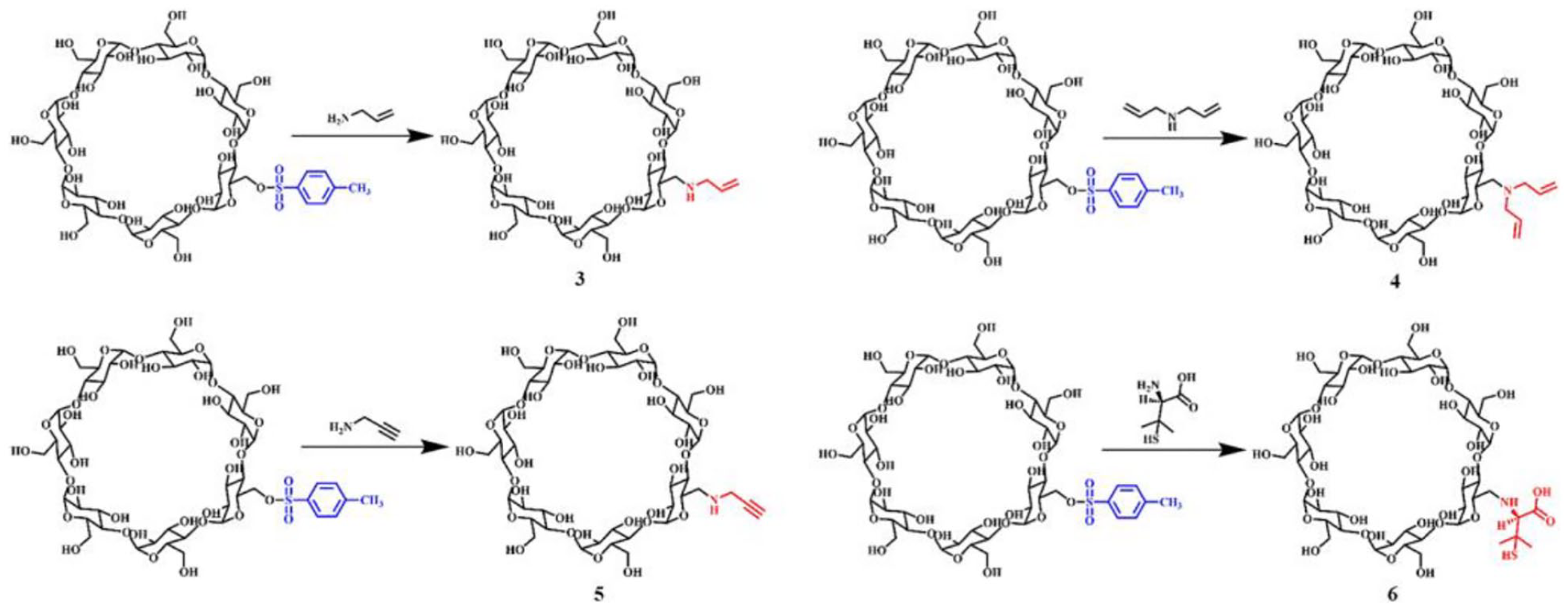
Synthesis of mono-(6-N-allylamino-6-deoxy)-β-CD (3)
The mono-(6-N-allylamino-6-deoxy)-β-CD (3) was obtained after optimization of the synthetic conditions based on the literature.
30
The reaction solvent was allylamine, and the ratio of reactants was 1:225. The reaction time was 4.5 h, while the reaction temperature was 60 °C. After the reaction, the solution was collected and cooled to room temperature. Then methanol 20 mL was added to dilute this solution, and acetonitrile 80 mL was also added. Then the solution was filtered and the white solid was collected. The above purification process was repeated 3 times. The resulting product was dried under reduced pressure at 40 °C for 2 h.
White powder (yield 84%); m.p. 222–224 °C. The molecular weight is 1174.4204, with the found mass-charge ratio of 1174.4245 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3385, 2926, 1645, 1557, 1417, 1298, 1157, 1155, 1079, and 1031; 1H NMR (600 MHz, DMSO-d6): δ 5.91 – 5.56 (m, 2H), 5.89 – 5.57 (m, 2H), 4.75 (s, 1H, -CH=CH2), 4.75 (s, 1H, -CH=CH2), 4.44 (dd, J = 13.9, 5.6 Hz, 1H), 4.44 (dd, J = 13.9, 5.6 Hz, 1H), 3.59 – 3.52 (m, 2H), 3.55 (d, J = 9.8 Hz, 2H), 3.48 (d, J = 9.6 Hz, 1H), 3.48 (d, J = 9.6 Hz, 2H), 3.23 (s, 1H), 3.07 (d, J = 4.8 Hz, 1H), 2.43 (s, 1H, -CH2NH), 2.43 (s, 1H, -CH2NH); 13C NMR (151 MHz, DMSO-d6): δ 137.63, 128.63, 125.94, 115.97, 102.65, 102.39, 102.24, 84.24, 81.94, 81.63, 73.47, 72.80, 72.44, 70.58, 60.32, 51.93, 49.33.
Synthesis of mono-(6-N-diallylamine-6-deoxy)-β-CD (4)
Compound 4 was synthesized with a method similar to that used for compound 3. The reaction solvent was diallylamine, and the ratio of reactants of synthesis compound 4 was 1:225. The reaction time was 4 h while the reaction temperature was 80 °C. After the reaction, the solution was collected and cooled to room temperature. Then methanol 20 mL was added to dilute this solution, and acetonitrile 120 mL was also added. Then the solution was filtered and the white solid was collected. The above purification process was repeated 3 times. The resulting product was obtained after optimization and dried under reduced pressure at 40 °C for 2 h.
White powder (yield 79%); m.p. 212–215 °C. The molecular weight of compound 4 is 1214.4517, with the found mass-charge ratio of 1214.4560 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3385, 2926, 1645, 1419, 1370, 1298, 1215, 1157, 1079, and 1029; 1H NMR (600 MHz, DMSO-d6): δ 5.74 (s, 1H), 5.69 (d, J = 6.5 Hz, 3H), 5.66 (s, 1H), 5.04 (d, J = 17.1 Hz, 1H, -CH=CH2), 4.99 (d, J = 10.3 Hz, 1H, -CH=CH2), 4.75 (s, 4H), 4.44 (d, J = 2.5 Hz, 2H), 3.54 (d, J = 8.8 Hz, 8H), 3.47 (d, J = 9.3 Hz, 5H), 3.22 (s, 5H), 2.42 (s, 2H, -CH2N); 13C NMR (151 MHz, DMSO-d6): δ 145.35, 138.53, 133.02, 130.36, 128.66, 128.04, 127.05, 125.93, 102.64, 102.36, 101.67, 82.61, 81.92, 81.52, 81.11, 73.49, 73.12, 72.80, 72.45, 70.20, 69.34, 60.33, 59.92, 59.64, 21.67, 21.28.
Synthesis of mono-(6-N-propargyl-6-deoxy)-β-CD (5)
Compound 5 was synthesized with a method similar to that used for compound 3. The reaction solvent was propargylamine. The ratio of reactants of synthesis compound 5 was 5:1. The reaction time was 5 h, and the reaction temperature was 85 °C. After the reaction, the solution was collected and cooled to room temperature. Then methanol 10 mL was added to dilute this solution, and acetonitrile 100 mL was also added. Then the solution was filtered and the white solid was collected. The purification process was repeated until the filtrate was colorless. The resulting product was obtained after optimization and dried under reduced pressure at 50 °C.
White powder (yield 84%); m.p. 290–291 °C. The molecular weight of compound 5 is 1172.4048. The mass-charge ratio of 1172.4087 corresponded to [M + H]+. The FTIR spectra of the compound revealed vibration bands at νmax cm–1 3383, 2927, 1643, 1417, 1371, 1245, 1156, 1080, and 1031; 1H NMR (600 MHz, DMSO-d6): δ 5.81 – 5.61 (m, 2H), 4.79 (d, J = 3.2 Hz, 1H), 4.55 – 4.41 (m, 1H), 3.63 – 3.57 (m, 2H), 3.52 (d, J = 9.1 Hz, 1H), 3.27 (s, 1H, -C≡CH), 2.47 (s, 1H); 13C NMR (151 MHz, DMSO-d6): δ 102.42, 84.15, 83.71, 82.02, 74.08, 73.50, 72.86, 72.48, 71.11, 60.34, 48.61, 36.28.
Synthesis of mono-(6-N-D-penicillamine-6-deoxy)-β-CD (6)
Mono-(6-toluenesulfonyl)-β-CD (1.3 g, 1.0 mmol) and penicillamine (0.45 g, 3.0 mmol) were added to a 250 mL three-necked flask. Then 20 mL triethanolamine and distilled water 30 mL were added. After the solids had dissolved, the flask was fitted with a reflux condenser and was placed in a water bath at 94 °C. After 10 h, the pH of the solution was adjusted to 2 with 1 mol/L HCl. The solution was dried under reduced pressure at 45 °C. Then it was dissolved in water 20 mL and filtered. The solution was collected and placed in a refrigerator at 4 °C for overnight; the solution was filtered and collected. The purification process was repeated 3 times. Then the pH was adjusted to 9 with 1 mol/L sodium bicarbonate and dried under reduced pressure at 45 °C. Then the crude product was dissolved in N,N-dimethylformamide 5 mL. The solution was filtered to remove sodium chloride. After that, the product was dissolved in methanol 10 mL. Acetone 100 mL was added and the product precipitated and was collected. The purification process was repeated twice. The product was dissolved in water 3 mL and methanol 10 mL was added. Finally, acetone 100 mL was added and the precipitate was collected. The obtained product was dried at 45 °C for 5 h.
White powder (yield 84%); m.p. 242–245 °C. The molecular weight of compound 6 is 1265.4102, with the found mass-charge ratio of 1265.4188 corresponding to [M]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3419, 2928, 1642, 1393, 1032, and 579; 1H NMR (600 MHz, DMSO-d6): δ 7.90 (s, 1H), 4.94 (s, 1H, -NH-), 4.79 (s, 7H), 3.60 (s, 18H), 3.53 (s, 9H), 2.85 (s, 1H), 2.69 (s, 1H), 2.46 (s, 3H), 2.04 (s, 3H), 1.37 (s, 3H, -S-C-CH3), 1.27 (s, 1H), 1.14 (s, 3H); 13C NMR (151 MHz, DMSO-d6): δ 145.32, 133.08, 130.37, 128.07, 102.68, 102.39, 101.72, 82.09, 81.94, 81.58, 81.17, 73.51, 73.16, 72.86, 72.49, 72.30, 70.19, 69.35, 60.34, 59.94, 59.68, 21.68.
Synthesis of mono-(6-ethylenediamine-(N-acryloyl)-6-deoxy)-β-CD (7) and mono-(6-ethylenediamine-(N-methylacryloyl)-6-deoxy)-β-CD (8)
Compounds 7 and 8 were synthesized as follows. Mono-(6-ethylenediamine-6-deoxy)-β-CD (4 g, 3 mmol) was added to a 250 mL three-necked flask. Next, NaOH (4 g, 0.1 mol) and distilled water 200 mL were added. After the solids had dissolved, the flask was placed in an ice-water bath (–1.5 °C). Acryloyl chloride (2.8 g, 0.0306 mol; or 3.2 g methacryloyl chloride) was added to the flask by a constant-voltage funnel. After stirring for 2 h, the flask was placed in a refrigerator at 4 °C for overnight. The solution was filtered and pH adjusted to 7 with 1 mol/L HCl. Then it was evaporated under reduced pressure at 40 °C. The crude product was dissolved in N,N-dimethylformamide 20 mL. The solution was filtered and precipitated by acetone 200 mL. The solid was collected and then dissolved in methanol 20 mL. The methanol solution was precipitated by acetone 200 mL again. The purification process was repeated 3 times. The obtained product was dried under reduced pressure for 3 h at 40 °C.
Compound 7 is white powder (yield 69%); m.p. 272–273 °C. The molecular weight of compound 7 is 1231.4419, with the found mass-charge ratio of 1231.4438 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3416, 2928, 1645, 1416, 1153, and 1031; 1H NMR (600 MHz, D2O): δ 6.15 (s, 1H), 6.13 (s, 1H), 6.13 (s, 1H), 5.93 (s, 1H, -CH=CH2), 5.68 (d, J = 6.9 Hz, 1H, -CH=CH2), 5.55 (d, J = 10.2 Hz, 1H), 4.97 (s, 1H), 3.86 (d, J = 7.1 Hz, 1H), 3.77 (d, J = 11.5 Hz, 2H), 3.55 (d, J = 7.9 Hz, 1H), 3.50 (d, J = 8.6 Hz, 1H), 3.25 (s, 1H), 2.91 (s, 1H), 2.76 (s, 1H), 2.67 (s, 1H), 1.81 (s, 1H), 1.09 (s, 1H); 13C NMR (151 MHz, D2O): δ 180.23, 178.87, 164.47, 133.59, 126.52, 101.76, 83.28, 83.03, 81.01, 72.95, 71.90, 71.65, 70.31, 70.01, 60.09, 49.71, 48.82, 45.08, 44.56, 36.77, 33.65, 33.01, 31.27.
Compound 8 is white powder (yield 72%); m.p. 268–270 °C. The molecular weight of compound 8 is 1245.4575, with the found mass-charge ratio of 1245.4608 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3389, 2928, 1655, 1555, 1451, 1407, 1234, 1154, and 1037; 1H NMR (600 MHz, DMSO-d6): δ 7.88 (s, 1H), 7.77 (t, J = 5.4 Hz, 1H), 6.16 (s, 1H), 5.55 (m, 1H, -CH=CH2), 5.23 (s, 1H, -CH=CH2), 4.97 (s, 1H), 4.76 (s, 1H), 4.50 (s, 1H), 3.57 (s, 2H), 3.50 (d, J = 7.0 Hz, 1H), 3.23 (s, 1H), 2.82 (s, 1H), 2.66 (s, 1H), 2.44 (s, 1H), 2.02 (s, 1H), 1.77 (s, 1H), 1.70 (s, 1H), 1.03 (d, J = 3.5 Hz, 1H), 0.84 (s, 1H); 13C NMR (151 MHz, DMSO-d6): δ 144.54, 140.35, 119.70, 118.53, 102.41, 81.98, 73.56, 72.84, 72.45, 60.30, 20.49, 19.01.
Synthesis of mono-(6-N-ethylenediamine (N-(2-bis(2-hydroxy-5-hexene))) (N-(2-hydroxy-5-hexene))-6-deoxy)-β-CD (9) and mono-(6-N-ethylenediamine (N-(2-bis(2-hydroxy-7-octene))) (N-(2-hydroxy-7-octene))-6-deoxy)-β-CD (10)
Compounds 9 and 10 were synthesized as follows. Mono-(6-ethylenediamine-6-deoxy)-β-CD (3.5 g, 3 mmol) and 0.1 mol/L sodium bicarbonate 40 mL were added to a 250 mL single neck round-bottom flask. And 1,2-epoxy-5-hexene (1.8 g, 17.88 mmol; or 2.3 g 1,2-hydroxy-7-octene) was added. Then the flask with a reflux condenser was placed in a water bath at 70 °C. After 10 h, the pH of the solution was adjusted to 7 with 1 mol/L HCl. The solution was dried under reduced pressure at 45 °C. Then the crude product was dissolved in N,N-dimethylformamide 20 mL. The solution was filtered to remove sodium chloride. After that, ether 100 mL was added. The DMF collected and acetone 100 mL was added. The product was precipitated and was collected. Then it was dissolved in 70 °C water 10 mL. Ether 100 mL was added and the water phase was collected. Next, acetone 100 mL was added and the precipitate collected. The purification process was repeated twice. The product was dissolved in methanol 10 mL and placed in a water bath at 70 °C. Finally, acetone 100 mL was added at room temperature and the precipitate collected. This purification process was repeated twice again. The obtained product was dried at 45 °C for 7 h.
Compound 9 is off white powder (yield 51%); m.p. 228–232 °C. The molecular weight of compound 9 is 1471.6508, with the found mass-charge ratio of 1471.6543 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3400, 2925, 1645, 1413, 1363, 1244, 1157, and 1035; 1H NMR (600 MHz, DMSO-d6): δ 5.85–5.74 (m, 1H, -CH=CH2), 5.04–4.95 (m, 1H, -OH), 4.83 (s, 2H, -CH=CH2), 3.73–3.51 (m, 8H), 3.45 (d, J = 7.8 Hz, 1H), 3.41–3.34 (m, 2H), 3.30 (s, 3H), 3.17 (s, 1H), 2.08 (s, 2H), 1.41 (s, 1H), 1.33 (s, 1H), 1.09 (t, J = 7.0 Hz, 1H); 13C NMR (151 MHz, DMSO-d6): δ 139.28, 139.15, 115.30, 115.04, 102.43, 85.03, 81.93, 81.68, 73.49, 72.84, 72.31, 67.64, 67.47, 67.02, 62.60, 60.15, 34.12, 29.88.
Compound 10 is yellow powder (yield 37%); m.p. 236–239 °C. The resulting product was dried under reduced pressure at 45 °C for 7 h. The yield was 37%. The molecular weight of compound 10 is 1555.7447, with the found mass-charge ratio of 1555.7481 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3384 (υ O-H), 2827, 2854, 1645, 1421, 1365, 1340, 1249, 1155, 1082, and 1029; 1H NMR (600 MHz, DMSO-d6): δ 6.02 (s, 1H), 5.71 (m, 2H), 5.00–4.85 (m, 1H, -OH), 4.76 (s, 1H, -CH=CH2), 4.71 (s, 1H, -CH=CH2), 4.54 (s, 1H), 4.50–4.41 (m, 1H), 4.30 (s, 1H), 3.98 (s, 1H), 3.98 (s, 1H), 3.71 (s, 1H), 3.55 (d, J = 9.1 Hz, 2H), 3.49 (d, J =6.8 Hz, 2H), 3.31 (s, 1H), 3.23 (s, 1H), 3.02 (s, 1H), 2.78 (s, 1H), 2.45 (dd, J = 15.1, 13.4 Hz, 2H), 2.33 (d, J = 11.1 Hz, 1H), 2.18 (m, 1H), 1.99–1.89 (m, 1H), 1.38–1.21 (m, 2H), 1.18 (d, J = 14.2 Hz, 1H), 1.02 (t, J = 7.0 Hz, 1H); 13C NMR (151 MHz, DMSO-d6): δ 139.11, 115.36, 102.45, 81.91, 73.51, 72.85, 72.46, 72.33, 60.18, 34.79, 33.63, 28.72, 24.90.
Synthesis of mono-(6-ethylenediamine-(N-(2-bis(3-allyloxy-2-hydroxy-propyl)-(N-(3-allyloxy-2-hydroxy-propyl)-6-deoxy-β-cyclodextrin) (11)
Compound 11 was synthesized as follows. Mono-(6-ethylenediamine-6-deoxy)-β-CD (3.5 g, 3 mmol) and 0.1 mol/L sodium bicarbonate 40 mL were added to a 250 mL single neck round-bottom flask. Allyl glycidyl ether (1.9 g, 16.5 mmol) was added. Then the flask with a reflux condenser was placed in a water bath at 70 °C. After 10 h, the pH of the solution was adjusted to 7 with 1 mol/L HCl. The solution was dried under reduced pressure at 45 °C. Then the crude product was dissolved in N,N-dimethylformamide 20 mL. The solution was filtered to remove sodium chloride. After that, acetone 100 mL was added. The product was precipitated and was collected. Then the product was dissolved in methanol 10 mL. Then acetone 100 mL was added and the precipitate collected. This purification process was repeated twice. And it was dissolved in methanol 10 mL. Ether 100 mL was added and the methanol phase was collected. Finally, acetone 100 mL was added and the precipitate was collected. The obtained product was dried at 45 °C for 7 h.
White powder (yield 50%); m.p. 209–212 °C. The molecular weight of compound 11 is 1518.6355, with the found mass-charge ratio of 1519.6399 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3404, 2924, 1645, 1634, 1418, 1369, 1337, 1300, 1247, 1207, 1155, 1080, 1032, 945, 864, 756, 708, 611, and 584; 1H NMR (600 MHz, DMSO-d6): δ 5.89 (dt, J = 11.5, 8.2 Hz, 1H, -CH=CH2), 5.31 (s, 2H, -CH=CH2), 5.28 (s, 1H, -CH=CH2), 3.93 – 3.76 (m, 19H), 3.69 (s, 5H), 3.60 (d, J = 8.9 Hz, 7H), 3.57 – 3.44 (m, 10H), 3.29 (s, 1H), 2.17 (s, 1H), 1.14 (t, J = 7.1 Hz, 1H); 13C NMR (151 MHz, DMSO-d6): δ 135.67, 116.93, 102.75, 102.38, 81.80, 73.50, 73.24, 72.87, 72.38, 71.70, 68.23, 67.88, 60.20, 58.74.
Synthesis of mono-(6-ethylenediamine-(N-undecylenyl)-6-deoxy)-β-CD (12)
Compound 12 was synthesized with a method similar to that used for compounds 7 and 8. Mono-(6-ethylenediamine-6-deoxy)-β-CD (2 g, 3 mmol) was added in a 250 mL three-necked flask. Then NaOH (4 g, 0.1 mol) and distilled water 200 mL were added. After the solids had dissolved, the flask was placed in an ice-water bath (0 °C). Then 10-Undecenoyl chloride 0.02039 mol was added to the flask by a constant-voltage funnel. After stirring for 2 h, the flask was placed in a refrigerator at 4 °C for overnight. The solution was filtered and pH adjusted to 7 with 1 mol/L HCl. Then it was evaporated under reduced pressure at 50 °C. The crude product was dissolved in N,N-dimethylformamide 20 mL. The solution was filtered and precipitated by acetone 200 mL. The solid was collected and then dissolved in water 20 mL. The solution was precipitated by acetone 200 mL again. The purification process was repeated 4 times. The solid was collected and then dissolved in methanol 20 mL. The methanol solution was precipitated by acetone 200 mL again. The obtained product was dried under reduced pressure for 3 h at 40 °C.
Off white powder (yield 70%); m.p. 243–247 °C. The molecular weight of compound 12 is 1343.5671, with the found mass-charge ratio of 1343.5695 corresponding to [M + H]+. The FTIR spectrum of the compound revealed vibration bands at νmax cm–1 3406, 2923, 2854, 1638, 1561, 1418, 1157, 1080, 1031, 947, and 560; 1H NMR (600 MHz, D2O): δ 5.77 (s, 2H), 4.77 (s, 1H, -CH=CH2), 4.47 (s, 1H), 3.58 (s, 3H), 3.25 (s, 2H), 2.45 (s, 1H), 1.96 (s, 1H), 1.43 – 1.07 (m, 2H); 13C NMR (151 MHz, D2O): δ 183.05, 166.12, 139.00, 138.26, 114.94, 103.70, 102.82, 101.90, 81.74, 81.44, 81.02, 73.88, 73.13, 72.86, 72.43, 71.87, 60.76, 60.03, 59.29, 39.13, 38.29, 37.83, 32.78, 29.23, 28.88, 28.46, 28.04, 26.90, 26.18, 25.69.