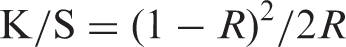Abstract
Antibacterial activities and UV-blocking properties of polyethylene terephthalate (polyester) containing fabrics are easily achieved by surface modification via alkaline hydrolysis, to generate surface carboxyl groups on polyester component, followed by treatment with certain basic dyes, metal salts, or antibiotic. The results showed that the improvement in antibacterial activities and anti-UV-B protection properties are governed by the type of substrate (polyester > polyester/viscose > polyester/cotton), the pretreatment history (alkali-treated > untreated), and type of basic dye (C.I. Basic Blue 9 > C.I. Basic Red 24). On the other hand, the extent of improvement in both the antibacterial efficacy against Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli) bacteria and the UV-protection properties are governed by the nature of loaded metal ion and followed the decreasing orders (Zn > Cu) and (Cu > Zn), respectively. Additionally, the results proved that post-treatment of modified substrates with Doxymycin® antibiotic brings about a significant enhancement in antibacterial activity along with an improvement in the UV-blocking properties regardless of the used substrate. After 10 washing cycles, the imparted functional properties show some reduction. Possible reaction mechanisms have been given.
Keywords
Introduction
Polyethylene terephthalate (PET) or polyester fibers have been widely used in textile industries because of their low production costs, high strength, easy-care properties, dimensional stability, high thermostability and chemical resistance, as well as, suitability for blending with other hydrophilic natural fibers, e.g. cellulosic fibers. However, PET-fiber has certain undesirable properties such as lack of hydrophilicity, oleophilicity, and tendency for static electricity build up [1,2]. Extensive research efforts have been dedicated to minimize the aforementioned deficiencies and to upgrade the performance properties of PET-fibers and fabrics through controlled alkaline treatment [3–5], aminolysis [3,6–8], using chitosan [9,10], plasma or laser treatment [11,12], grafting of hydrophilic monomers [2], introduction of sugar onto PET fabrics [13], and/or blending with hydrophilic natural fibers [10,13,14].
In the recent years, functionalization of textiles has become extremely important for maintaining current properties, creating new material properties, as well as producing high value-added textiles with better performance properties to meet consumer demands in terms of comfort, easy-care, health and hygiene, along with protection against chemical, biological, and thermal attacks [15–18]. Today, multifunctional textiles are used in a wide range of potential applications, e.g. apparel, household, furnishing, medical, industrial, and technical uses [15,16]. Functionalization of textiles can be obtained either by (a) incorporation of functional additives into the polymer melt before spinning, (b) chemical grafting of reactive additives onto the fiber surface directly or by means of crosslinkers, and (b) functional coating [15–22].
Accordingly, the main task of this study was to generate surface carboxyl groups on PET-containing fabrics, via alkaline hydrolysis, which can serve as an anchor sites for binding and fixing of certain reactive additives to impart multifunctional properties.
Experimental
Materials
In this study, 100% scoured and bleached plain weave PET (168 g/m2), polyester/cotton (PET/C) blend (65/35, 120 g/m2), and polyester/viscose (PET/V) (70/30, 96 g/m2) fabrics were used.
Zinc acetate, copper acetate, sodium hydroxide, and acetic acid were of laboratory reagent grade.
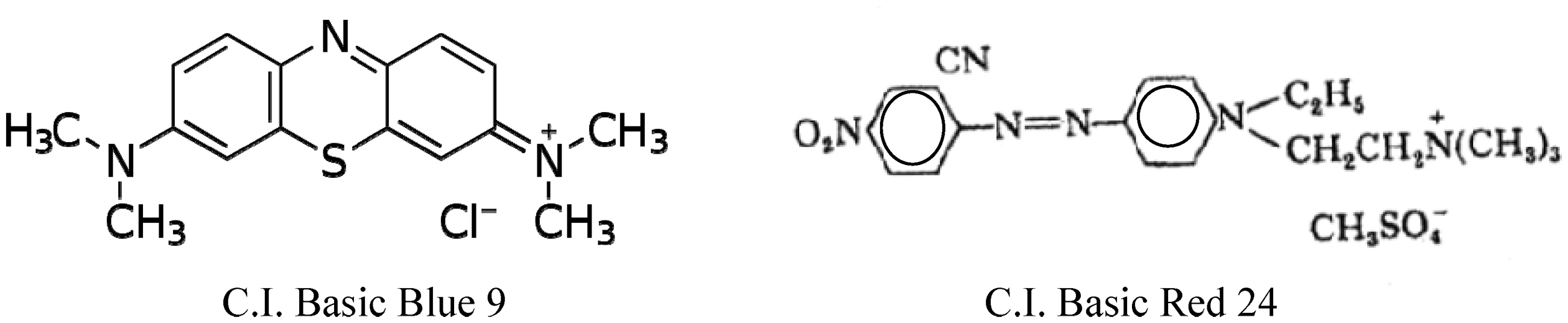
Basic dyes namely C.I. Basic Blue 9 (Merk) and C.I. Basic Red 24 (DyStar) were used as supplied.
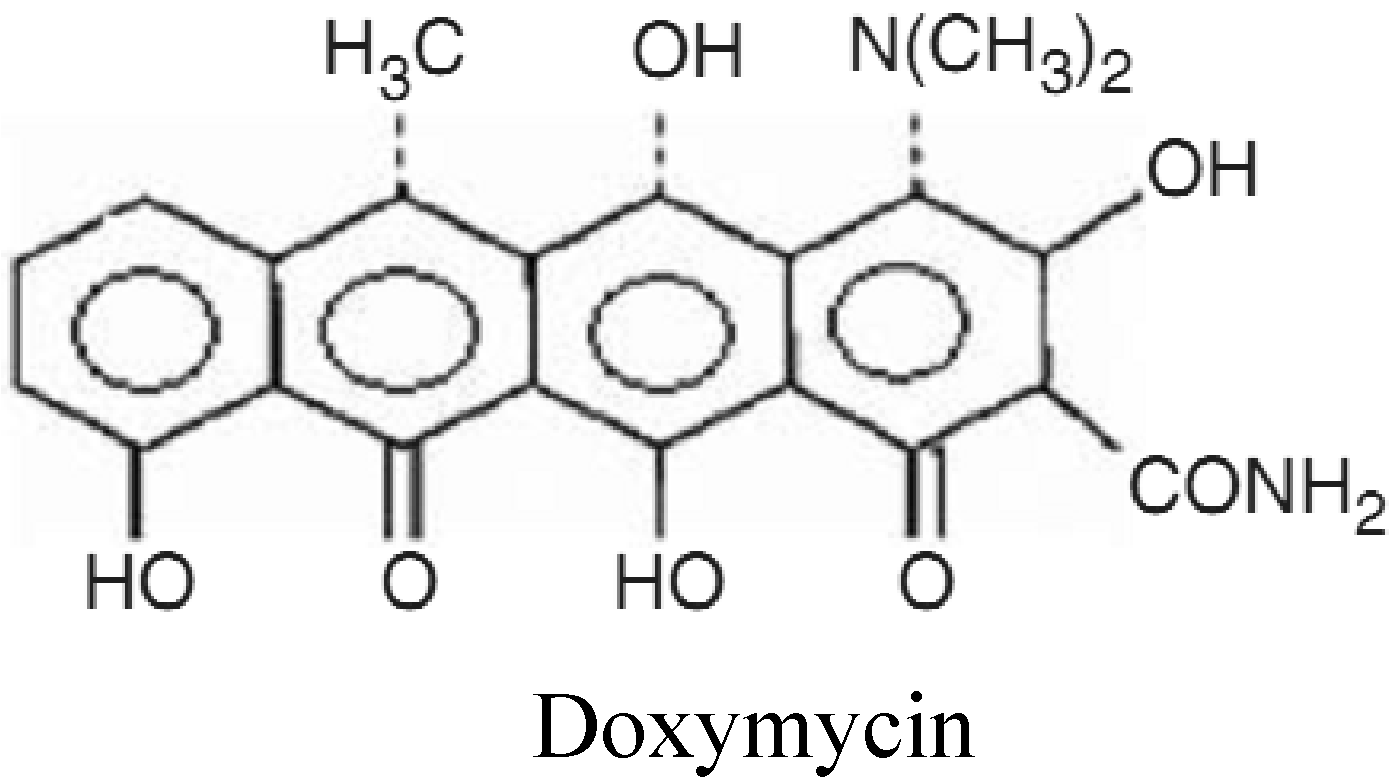
Doxymycin® antibiotic was purchased from Nile Co. for Pharmaceutical and Chemical Industries, Cairo, Egypt and used as supplied. Hostapal® CV-ET (a non-ionic wetting agent based on alkyl aryl polyglycol ether, Clariant) was of commercial grade.
Methods
Alkali treatment
The alkali treatment of the used PET-containing fabrics was carried out using an aqueous alkali solution (0–2% owf), liquor ratio (LR, 1/10–1/40) for 15–75 min at 95°C in launder Ometer cups with stirring. After the alkaline treatment, the fabric samples were rinsed thoroughly with distilled water, neutralized with acetic acid, rinsed, and finally air dried at room temperature.
Basic dyeing
Portions of alkali-treated fabric samples were post-dyed using the nominated basic dyes according to the conventional exhaustion method. Basic dyeing was carried out using the positively charged basic dyeing (2% owf), non-ionic wetting agent (2 g/L), LR (1/20) at pH 9 for 45 min at 95°C. The dyed fabric samples were thoroughly washed, neutralized using acetic acid, then rinsed with distilled water, and finally air dried at room temperature.
Metallic salts treatments
Two kinds of metallic salts, i.e. Cu acetate and Zn acetate, were used. The alkali-treated fabric samples were then treated with metallic salt solution (4% owf) at a LR (1/20), pH 9, and temperature 50°C for 60 min. The treated fabric samples were then rinsed with distilled water five times and finally air dried.
Antibiotic treatment
Portions of the alkali-treated fabric samples were post-treated with Doxymycin® at a concentration of (2% owf), a LR (1/20), and temperature 65°C for 60 min. The treated samples were then rinsed with distilled water five times and finally air dried.
Measurements
The percentage of weight loss (WL) was calculated with the following equation:
Nitrogen content (N%) was determined according to the Kjeldahl method [23].
The color strength (K/S) is commonly used to represent the extent of dye interaction and fixation into/onto the substrate. The K/S values were determined from the reflectance measurements using the Kubelka–Munk equation [24]:
The amount of metal content in the post-treated fabric samples was determined by a flame atomic absorption spectrophotometer (GBC-Avanta, Australia).
UV-protection factor (UPF) was determined according to the Australian/New Zealand Standard (AS/NZS 4399-1996). Fabric can be rated as providing good, very good, and excellent protection if their UPF values range 15–24, 25–39, and above 40, respectively.
Antibacterial activity assessment against Gram-negative bacteria (Escherichia coli) and Gram-positive (Staphylococcus aureus) was assessed according to AATCC Test Method (147-1988) and expressed as zone of growth inhibition (nm).
Durability to wash was determined according to AATCC test method 61-1994.
All determinations were done in triplicate and the average was taken as final results.
FT-IR spectra of the fabric samples were recorded using Nexus 670 FTIR spectrometer from Nicolet, USA. The FT-IR spectra were recorded between the ranges 4000 cm−1 and 400 cm−1 with spectra resolution of 4 cm−1.
A scanning electron microscopic (SEM) examination was carried out by mounting the samples on stub with double-stick adhesive tape and coated with gold in a S150A sputter coater unit (Edwards, UK). The gold film thickness was 150 Å. The samples were then viewed in a JEAOL JXA-840A electron probe microanalyzer.
Tensile strength (TS) was determined, in the warp direction, using Instron Corporation 4435 Series IX Automated Materials Testing System, USA. According to D 5034-1995. The tensile strength loss (TSL) was determined according to the following equation:
Results and discussion
In this study, an attempt has been made to impart antibacterial and UV-blocking functional properties on PET-based textiles via surface modification along with generation of new active sites, −COOH groups, by alkaline hydrolysis to enhance their subsequent interactions with certain active materials, i.e. basic dyes, antibiotic and metal salts, and consequently upgrade their functionalities. Results obtained along with their appropriate discussion follows.
Alkaline hydrolysis
In this part, factors affecting surface modification of the nominated substrates, expressed as WL and dyeability with basic dyes, such as alkali concentration, material-to-liquor ratio as well as treatment time were investigated.
Alkali concentration
Figures 1 to 3 show the variation in the WL and dyeability with C.I. Basic Red 24 and C.I. Basic Blue 9, respectively, as a function of alkali concentration. For a given set of treatment conditions, it is clear that (a) increasing NaOH concentration from zero up to 2% owf results in a significant increase in the fabric WL, regardless of the used substrate (Figure 1); (b) the extent of WL is determined by the type of substrate and follows the decreasing order: PET > PET/V > PET/C; (c) the decrease in the mass of the treated fabrics reflects the impact of alkali treatment on surface modification of the PET component via partial hydrolysis, decreasing the residual impurities in the cellulosic component and/or partial degradation of some cellulosic fibers due to the formation of oxycellulose under the used conditions [1,3–5]; (d) the effectiveness of alkaline hydrolysis depends on the fabric weight and construction, PET/cellulose ratio, as well as ease of penetration; and (e) the partial hydrolysis of PET under alkaline conditions initially causes the shortening of surface chains along with the generation of hydrophilic/functional carboxylic acid and hydroxyl groups as follows [4,25]
Effect of NaOH concentration on WL. WL: weight loss; LR: liquor ratio. Alkali treatment: NaOH (0–2% owf); LR (1/20) at 95°C for 45 min. Effect of NaOH concentration on post-dyeing with C.I. Basic Red 24. LR: liquor ratio. Post-basic dyeing: dye (2% owf); non-ionic wetting agent (2 g/L); pH 9; LR (1/20) at 95°C for 45 min. Effect of NaOH concentration on post-dyeing with C.I. Basic Blue 9. LR: liquor ratio. Post-basic dyeing: dye (2% owf); non-ionic wetting agent (2 g/L); pH 9; LR (1/20) at 95°C for 45 min.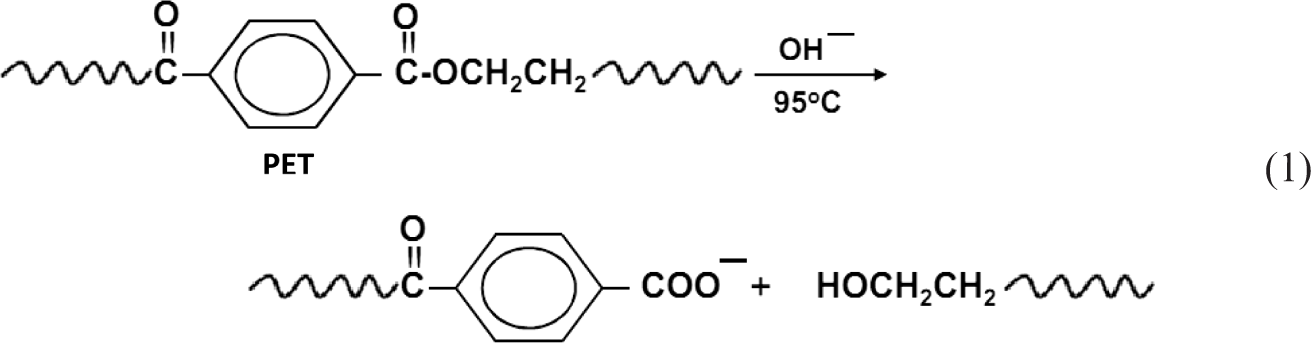



On the other hand, to assess the extent of creation of carboxyl groups on the treated fabric surfaces, which then served as anchor sites, post-dyeing with C.I. Basic Red 24 (Figure 2) and C.I. Basic Blue 9 (Figure 3) was studied. It is clear that within the range examined: (a) increasing alkali concentration up to 2% owf brings about an improvement in the extent of basic dye uptake, expressed as K/S value; (b) the enhancement in the dye uptake confirms the generation of –COOH groups at the PET surface along with an adequate number of carboxyl groups of cellulosic component which are formed during pretreatments [1,4]; (c) the extent of interaction and fixation of the used basic dyes follows the decreasing order: PET > PET/V > PET/C irrespective of the used dye, reflecting the differences among the treated substrates in extent of surface modification, availability and accessibility of dye sites, i.e. −COOH groups, as well as ease of adsorption and penetration; and (d) the K/S values of the obtained dyeings follow the decreasing order C.I. Basic Blue 9 > C.I. Basic Red 24 most probably due to differences in molecular size and structure, basicity and functionality, ease of penetration, affinity and extent of interaction, as well as in chromogen responsible for the color [14].
Figure 4 illustrates the surface morphology of the untreated and alkali-treated PET, PET/V, and PET/C fabrics. The SEM micrographs of the untreated substrates (Figure 4(a), (c) and (e), respectively) demonstrate that the untreated fabrics have a smooth surface. Following the saponification with NaOH, one can notice the appearance of pits, holes, and pores along the PET and PET/V substrates (Figure 5(b) and (d), respectively) while elongated cracks parallel to the fiber axis in case of PET/C (Figure 5(f)).
SEM of the untreated and alkali-treated PET, PET/V, and PET/C fabrics. PET: polyester, PET/V: polyester/viscose; and PET/C: polyester/cotton. FT-IR of the untreated and alkali-treated PET, PET/V, and PET/C fabrics. PET: polyester, PET/V: polyester/viscose; and PET/C: polyester/cotton.

From the FT-IR spectra of untreated and alkali-treated samples (Figure 5), the following was assured.
A sharp and strong absorption in range from 1712 to 1714 cm−1 due to the C=O stretch. The intensity of C=O bond for all the alkaline-treated fabric samples has been increased from 61.78 to 67.71 in case of PET, from 86.3 to 89.81 in case PET/C, and from 77.6 to 81.59 in case of PET/V. In case of PET, (Figure 5(b)) there is a weak peak at 3429.78 cm−1 corresponds to the OH stretch. In the case of PET/C and PET/V, there is a broad absorption peak range 3330–3340 cm−1 due to OH stretch. The intensity of OH peak in alkaline-treated PET/V and PET/C fabric samples was also slightly increased, Figure 5(d) and (f).
As a result, there is an increase in the extent of picking up and fixation of the used basic dyes, expressed as K/S values (Figures 2 and 3), of the alkali-treated samples as a direct consequence of the increase in the active polar groups especially −COOH groups on the fabric surface.
Material-to-liquor ratio
It is clear from Figures 6 to 8 that within the range examined, LR 1/20 to 1/40, increasing the LR up to 1/40 is accompanied by gradual decrease in WL (Figure 6) regardless of the used substrate, i.e. the lower the LR, the higher the WL. This implies that increase of the LR not only decrease the alkali concentration and its availability in the vicinity of the so-called amorphous regions of PET but also minimizes the extent of subsequent attack on the PET-fiber surfaces and subsequent chain scission, i.e. lower extent of surface modification and generation of terminal active sites (−COOH groups). On the other hand, the decreasing trend of basic dye uptake (Figures 7 and 8) at higher LR is most probably due to a shortage in number of generated-accessible active sites (−COOH groups) which serve as centers for picking up and fixing the cationic dye molecules onto the modified fabric surfaces [26].
Effect of material-to-liquor ratio on WL. WL: weight loss; LR: liquor ratio. Alkali treatment: NaOH (0–20 g/L); LR (1/20) at 95°C for 45 min. Effect of material-to-liquor ratio on post-dyeing with C.I. Basic Red 24. LR: liquor ratio. Post-basic dyeing: dye (2% owf); non-ionic wetting agent (2 g/L); pH 9; LR (1/20) at 95°C for 45 min. Effect of material-to-liquor ratio on post-dyeing with C.I. Basic Blue 9. LR: liquor ratio. Post-basic dyeing: dye (2% owf); non-ionic wetting agent (2 g/L); pH 9; LR (1/20) at 95°C for 45 min.


Treatment time
As for the change in the WL values (Figure 9) and subsequent basic dye uptake, K/S, (Figures 10 and 11) as a function of treatment time, it is clear that prolonging the treatment time from 15 to 60 min results in a significant increase in both the WL and K/S values regardless of the used substrate. It is understandable that proper alkaline hydrolysis time is needed for opening up and swelling of the used substrates, adsorption of alkali at the fiber surface, and attacking the electron-deficient carbonyl carbons of the PET to form an intermediate anion followed by chain scission and generation of carboxyl (reactive groups) and hydroxyl groups on the PET surface, thereby enhancing the extent of surface modification, WL, and dyeability with basic dyes [1,4,26].
Effect of treatment time on WL. WL: weight loss; LR: liquor ratio. Alkali treatment: NaOH (0–20 g/L); LR (1/20) at 95°C for 45 min. Effect of treatment time on post-dyeing with C.I. Basic Red 24. LR: liquor ratio. Post-basic dyeing: dye (2% owf); non-ionic wetting agent (2 g/L); pH 9; LR (1/20) at 95°C for 45 min. Effect of treatment time on post-dyeing with C.I. Basic Blue 9. LR: liquor ratio. Post-basic dyeing: dye (2% owf); non-ionic wetting agent (2 g/L); pH 9; LR (1/20) at 95°C for 45 min.


Functional finishes
This part is focused on upgrading the antibacterial activity and anti-UV properties of alkali-treated PET-containing fabrics via (a) post-dyeing with C.I. Basic Red 24 and C.I. Basic Blue 9; (b) subsequent treatment with metallic salts namely Cu acetate and Zn acetate; or (c) post-treatment with Doxymycin® antibiotic. Results obtained along with appropriate discussion follows.
Basic dyes
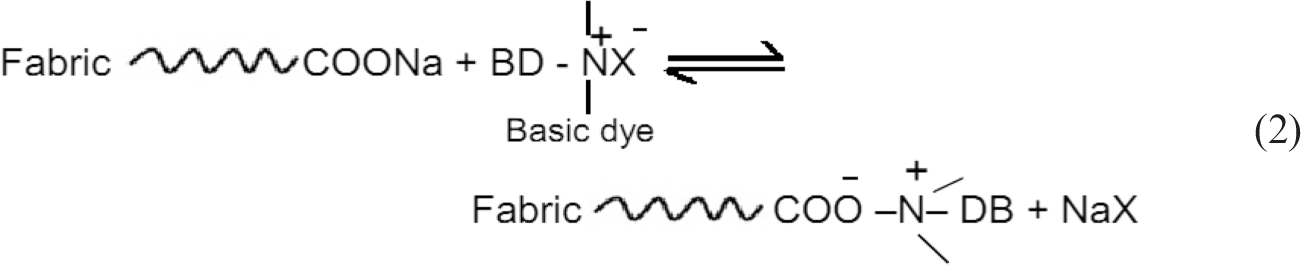
Effect of alkaline hydrolysis followed by subsequent basic dyeing on the functional properties of the modified substrates.

WL: weight loss (%); N: nitrogen content (%); K/S: color strength; UPF: UV-protection factor; ZI: zone of inhibition; TSL: tensile strength loss (warp direction); PET: polyester, PET/V: polyester/viscose; and PET/C: polyester/cotton.
Alkali treatment: NaOH (2% owf); LR (1/20) at 95°C for 45 min.
Basic dyeing: dye (2% owf); non-ionic wetting agent (2 g/L); pH (9); LR (1/20) at 95°C for 45 min.
Values between parentheses correspond to the untreated substrates that have no antibacterial efficacy.
TS: tensile strength of the untreated substrates PET, PET/V, and PET/C are 89, 72, and 75 kgf, respectively.
On the other hand, the decrease in the imparted anti-UV and antibacterial properties after 10 washing cycles is mostly due to the partial release of the fixed basic dyes.
Metallic salts

Effect of alkaline hydrolysis followed by subsequent metal salt treatment on the functional properties of the modified substrates.
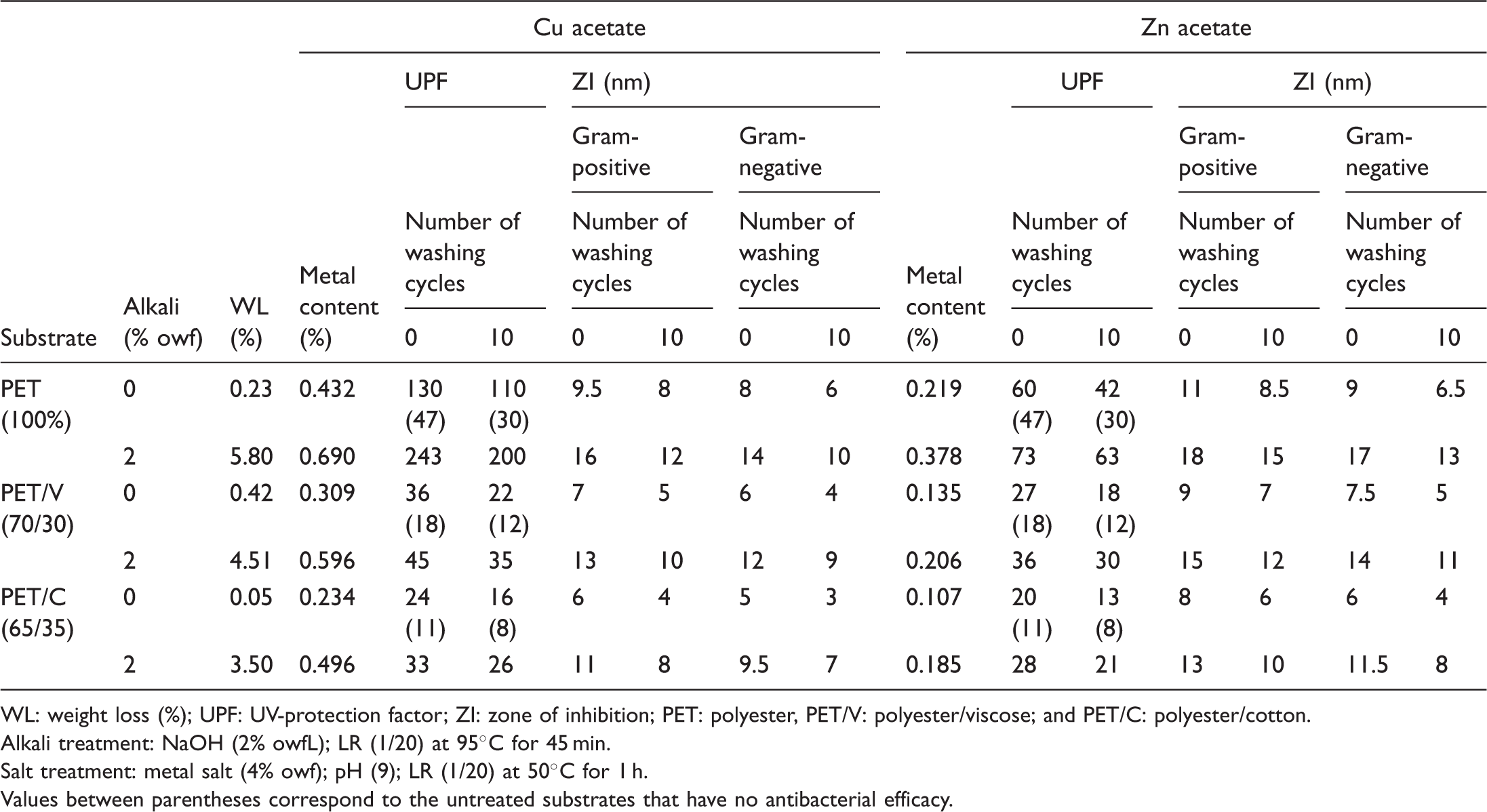
WL: weight loss (%); UPF: UV-protection factor; ZI: zone of inhibition; PET: polyester, PET/V: polyester/viscose; and PET/C: polyester/cotton.
Alkali treatment: NaOH (2% owfL); LR (1/20) at 95°C for 45 min.
Salt treatment: metal salt (4% owf); pH (9); LR (1/20) at 50°C for 1 h.
Values between parentheses correspond to the untreated substrates that have no antibacterial efficacy.
The variation in the imparted properties using the nominated metal salts could be discussed in terms of variation in molecular weight, molecular size, extent of metal ion uptake by the modified substrates, metal ion selectivity, location and extent of distribution, UV-blocking properties and UV-B absorbing capacity [21,23], as well as in their ability to react with cellular, coagulate cytoplasmic proteins and/or catalyze the production of oxygen radicals as follows [21,33–37]

Antibiotic treatment

Effect of alkaline hydrolysis followed by subsequent antibiotic treatment on the functional properties of the modified substrates.

WL: weight loss (%); N: nitrogen content (%); UPF: UV-protection factor; PET: polyester, PET/V: polyester/viscose; and PET/C: polyester/cotton.
Alkali treatment: NaOH (2% owf); LR (1/20) at 95°C for 45 min.
Antibiotic treatment: antibiotic (2% owf); pH (9); LR (1/20) at 65°C for 1 h.
Values between parentheses correspond to the untreated substrates that have no antibacterial efficacy.
On the other hand, increasing the number of washing cycles up to 10 results in a reasonable decrease in the imparted functional properties, i.e. antibacterial and anti-UV.
Conclusion
Antibacterial and anti-UV-B functionalities of PET-containing fabrics are easily achieved using a mild alkaline treatment conditions (sodium hydroxide (2% owf), LR (1/20) at 95°C for 45 min) for creating more hydrophilic surfaces with accessible active sites, i.e. −COOH groups, followed by subsequent treatment with proper basic dyes such as C.I. Basic Blue 9 and C.I. Basic Red 24, metallic salts, i.e. Zn acetate and Cu acetate, or Doxymycin® antibiotic. The extent of surface modification and subsequent functionalization are determined by type of substrate as well as kind and quantity of loaded-active ingredient in the post-treatment steps. Moreover, the given options for surface modification and functionalization of PET, PET/V, and PET/C fabrics are very simple, applicable, and quite cost-effective. Repeated washing cycles (10 cycles) of the post-treated fabric samples result in some decrease in their functional properties.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.