Abstract
Pretreatment of polyester-containing fabrics with chitosan, polyethylene glycol along with N-methylol crosslinking agent (Arkofix® NDW, DMDHEU) in the presence of MgCl2. 6H2O/citric acid as a mixed catalyst was carried out for enhancing their printability with disperse dyes as well as upgrading their ultraviolet-protecting properties. The results showed that the changes in the depth of the obtained prints are governed by the type of substrate, chitosan concentration and steaming conditions. On the other hand, incorporation of polyethylene glycol in the pretreatment bath results in a remarkable improvement in the depth of the post-printed fabric samples. Scanning electron microscope images showed the presence of chitosan-based deposits firmly attached to the surface of the pretreated fabric samples. The mechanism of surface modification via loading of chitosan with its amino groups along with polyethylene glycol moieties onto and/or into the finish/fabric matrix was proposed.
Keywords
Introduction
Among manmade fibers, polyester exhibits excellent properties such as dimensional stability, wrinkle resistance, chemical resistance, quick-drying properties and blendability with other natural fibers such as cotton and wool. However, polyester has high crystallinity, low moisture regain and due to its hydrophobic nature, it will exhibit static problems along with low level of comfort. It also has no functional groups, in comparison with natural fibers, and cannot interact with anionic or cationic dyes [1,2].
Disperse dyes are the only proper class for coloration of polyester, and due to their nonionic nature and the absence of ionizable groups, they have the tendency to sublime without decomposition and are strongly adsorbed by the hydrophobic polyester structure [1,3,4].
On the other hand, the potential applications of modified biopolymers in functional finishing of textiles as well as in improving dye uptake have attracted a great deal of scientific and industrial interests. Among various available biopolymers, the polysaccharide-based cationic biopolymer chitosan (Figure 1) is highly recommendable, due to its polyamine character, biocompatibility, biodegradability, non-toxicity and availability [5–7].
Structure of chitosan.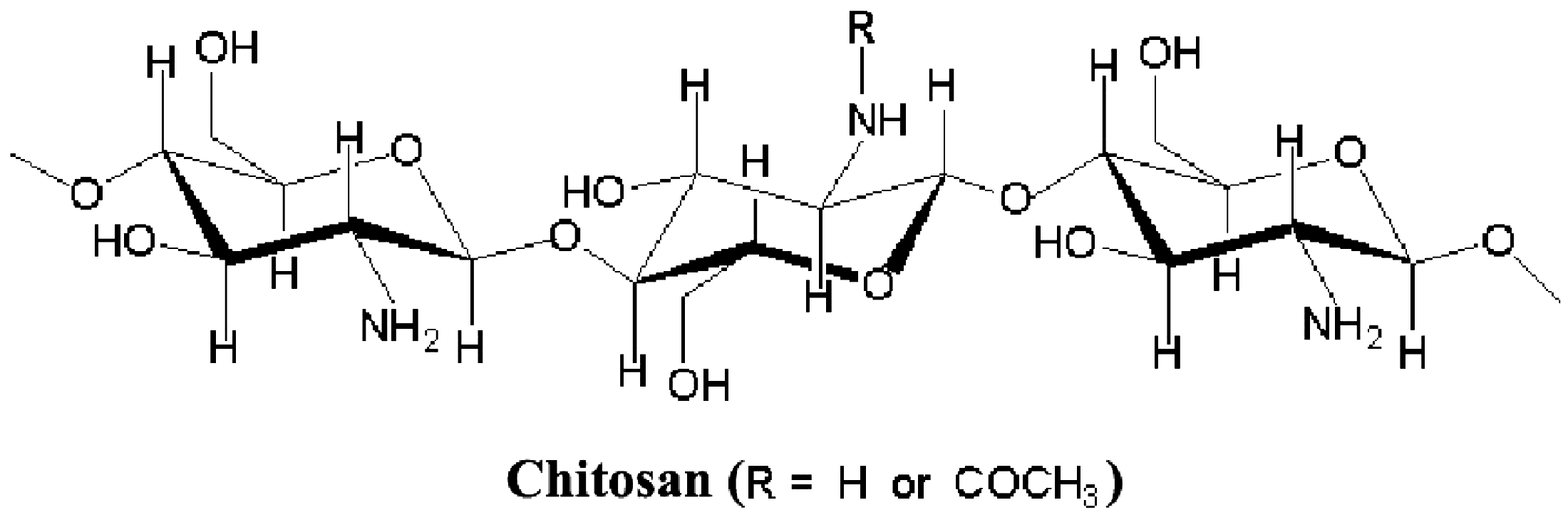
A number of studies have focused on improving the printability of polyester with disperse dye using different solvents [8] as well as on modifying the ultraviolet (UV)-blocking properties of conventionally and alkaline-disperse dyed polyester fabrics [9].
Very recently, we have investigated the technical feasibility of (a) union disperse printing and UV protecting of wool/polyester blend using a reactive beta cyclodextrin [10], (b) enhancing transfer printing and UV-blocking properties of polyester-based textiles using monochloro triazine-beta cyclodextrin, chitosan and ethylenediamine [11] and (c) upgrading printing and UV-blocking properties of polyester and polyester/wool fabrics by aminoloysis [12]. In this paper, we report the multieffect of chitosan, as an amino polysaccharide along with certain additives, on enhancing the extent of subsequent disperse printing as well as UV-blocking properties of pretreated polyester-containing fabrics.
Experimental
Materials
Mill-scoured and bleached 100% polyester (PET, 220 g/m2), polyester/wool (PET/W, 70/30–230 g/m2), polyester/cotton (PET/C, 70/30–370 g/m2) and polyester/ viscose (PET/V, 70/30–380 g/m2) woven fabrics were used.
Chitosan with degree of deacetylation of 82.9% and molecular weight of 140,500 Dalton, (Vanson, Inc. USA), DIAL GIN® LV 100 (Na-alginate of low-viscosity BFGoodrich Diamalt GmbH, Germany), Arkofix® NDW, modified dimethylol dihydroxyethylene urea (DMDHEU, 45%, BASF), Disperse Red 74, Disperse Blue 77 and Disperse Orange 29 (Sinochem Ningbo, China) were of commercial grade (Figure 2).
Disperse dyes structure.
Polyethylene glycols (PEG-200, 400, 600 and 800), citric acid, acetic acid and magnesium chloride hexahydrate (MgCl2·6H2O) were of laboratory grade reagents.
Methods
Pretreatment with chitosan (HO-CHT-NH2)
Fabric samples were impregnated by the aqueous solution containing chitosan (0–1.5 g/L), DMDHEU (0–30 g/L), MgCl2·6H2O (4 g/L)/citric acid (1 g/L) as a mixed catalyst and PEG-600 (10 g/L), roll squeezed to a wet-pickup of 70%, thermofixed at 120°C for 3 min, thoroughly rinsed and dried before being printed with disperse dyes.
Disperse printing
Printing paste components.

Printed fabric samples were then dried at 85°C for 5 min and steam fixed at 120°C for 15 min using Arilot® CSL-steamer-Italy, rinsed thoroughly, soaped to 15 min at 60°C in the presence of 2 g/L Leomin® W (nonionic wetting agent and detergent, 60% BASF), then rinsed well and finally dried at 85°C for 5 min.
Testing
Nitrogen content (N%) was determined according to the Kjeldahl method. Depth of the disperse prints, expressed as K/S value, was measured at the wavelength of the maximum absorbance using an automatic filter spectrophotometer, and calculated by the kubelka Murnk equation: K/S = (1 − R)2/2R, where K is the absorption coefficient; S is the scattering coefficient and Rλmax is the reflectance value of the fabric at maximum wavelength. Fastness properties to washing, perspiration, crocking and light of printed samples were evaluated according to American Association of Textile Chemists and Colorists (AATCC) tests methods (61-1972), (15-1973), (8-1972) and (16-1998), respectively. UV protection factor (UPF) was calculated according to the Australian/New Zealand standard (AS/NZS 4399-1996). A scanning electron microscope (SEM) examination was carried out by mounting the untreated and chitosan-treated fabric samples on sub with double stick adhesive tape and coated with gold in a S150A sputter coater unit (Edwards, UK), the gold film thickness was 150 Å, then viewed in a JEAOL JZA-840A electron probe microanalyses. All the determinations in this study were performed in triplicate.
Results and discussion
Chitosan concentration
Figure 3 shows that (a) increasing concentration up to 1.25 g/L is accompanied by a gradual increase in the K/S values of post-printed fabric samples, (b) the extent of improvement in the depth of the obtained prints is governed by the type of substrate, the extent of fixation of chitosan and the ability of modified fabric surface to pick-up, interact and/or entrap the disperse dye molecules in the subsequent steam fixation step, (c) the improvement in K/S values follows the descending order: polyester (PET) > polyester/wool (PET/W) > polyester/cotton (PET/C) > polyester/viscose (PET/V), keeping other parameters constant and (d) further increase in chitosan concentration, that is beyond 1.25 g/L, has practically slight or no effect on the depth of the obtained prints.
Effect of pretreatment with different chitosan concentration on the K/S values of the disperse prints. (i) Pretreatment conditions: DMDHEU (15 g/L), mixed catalyst (5 g/L), chitosan (0–1.5 g/L), padding to a wet pick-up (70%) followed by direct fixation at 120°C for 3 min. (ii) Post-printing conditions: Disperse Red 74 (30 g/kg); stock-thickening agent 10% (500 g/kg); acetic acid 30% (15 g/kg), drying at 85°C for 5 min, steaming at 120°C for 15 min. DMDHEU = dimethylol dihydroxyethylene urea.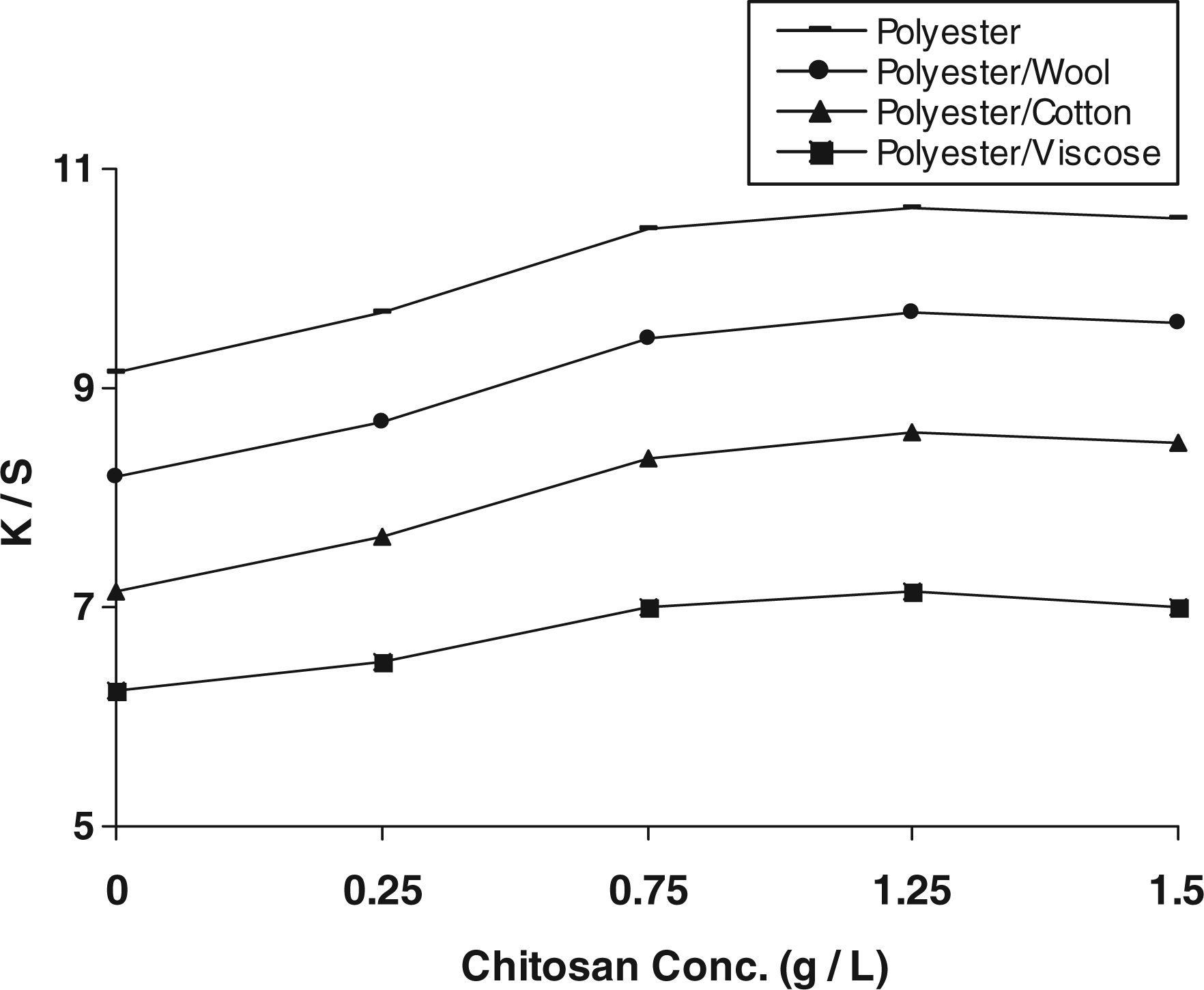
The improvement in the K/S values of the pretreated substrates is a direct consequence of surface modification of the used substrates (Figure 4) via (a) reaction of negatively charged carboxyl groups of wool, cellulose and/or terminal carboxyl groups of polyester with the positively charged amine groups of chitosan at acidic pH, thereby modifying the fabric surface [6].
Tentative mechanism of surface modification of the used substrate.
This in turn modifies the fabric surface thereby enhancing the extent of disperse dye fixation and accommodation, that is higher K/S values.
Increasing chitosan concentration up to 1.5 g/L results in an increase in the viscosity of the pretreatment bath along with a side interaction with the used DMDHEU thereby decreasing the extent of further fixation of disperse dye.
On the other hand, the SEM images in Figure 5 showed that pretreatment of polyester and wool/polyester fabrics with chitosan and DMDHEU resulted in the formation of surface deposits based on chitosan and/or crosslinked chitosan compared with the untreated ones and the extent of surface modification was governed by the type of substrate, its functional groups and the extent of loading and fixation of chitosan onto the fabric surface.
SEM of untreated and treated polyester and wool/polyester fabrics. SEM: Scanning electron microscope.
Finishing agent concentration
As far as the change in K/S values of post-printed fabric samples as a function of DMDHEU concentration in the pretreatment formulation along with chitosan (1.25 g/L) and the mixed catalysts (5 g/L), Figure 6 shows that increasing DMDHEU concentration up to 15 g/L has a positive effect on increasing the extent of post-printing, most probably due to the modification of the fabric surface and the introduction of free amino groups of chitosan onto the finish/fabric matrix thereby increasing the cationicity of the surface, enhancing the electrostatic attraction between the negatively charged groups, that is phenolic-OH groups of dye molecules and protonated amino groups of the pretreated substrates and/or changing the hydrophobicity/hydrophilicity ratio due to crosslinking, that is high affinity for disperse printing [14].
Effect of DMDHEU concentration in the pretreatment step on the K/S of post-printed fabric samples. (i) Pretreatment conditions: DMDHEU (0–30 g/L), mixed catalyst (5 g/L), Chitosan (1.25 g/L), padding to a wet pick-up (70%) followed by direct fixation at 120°C for 3 min. (ii) Post-printing conditions. DMDHEU: dimethylol dihydroxyethylene urea.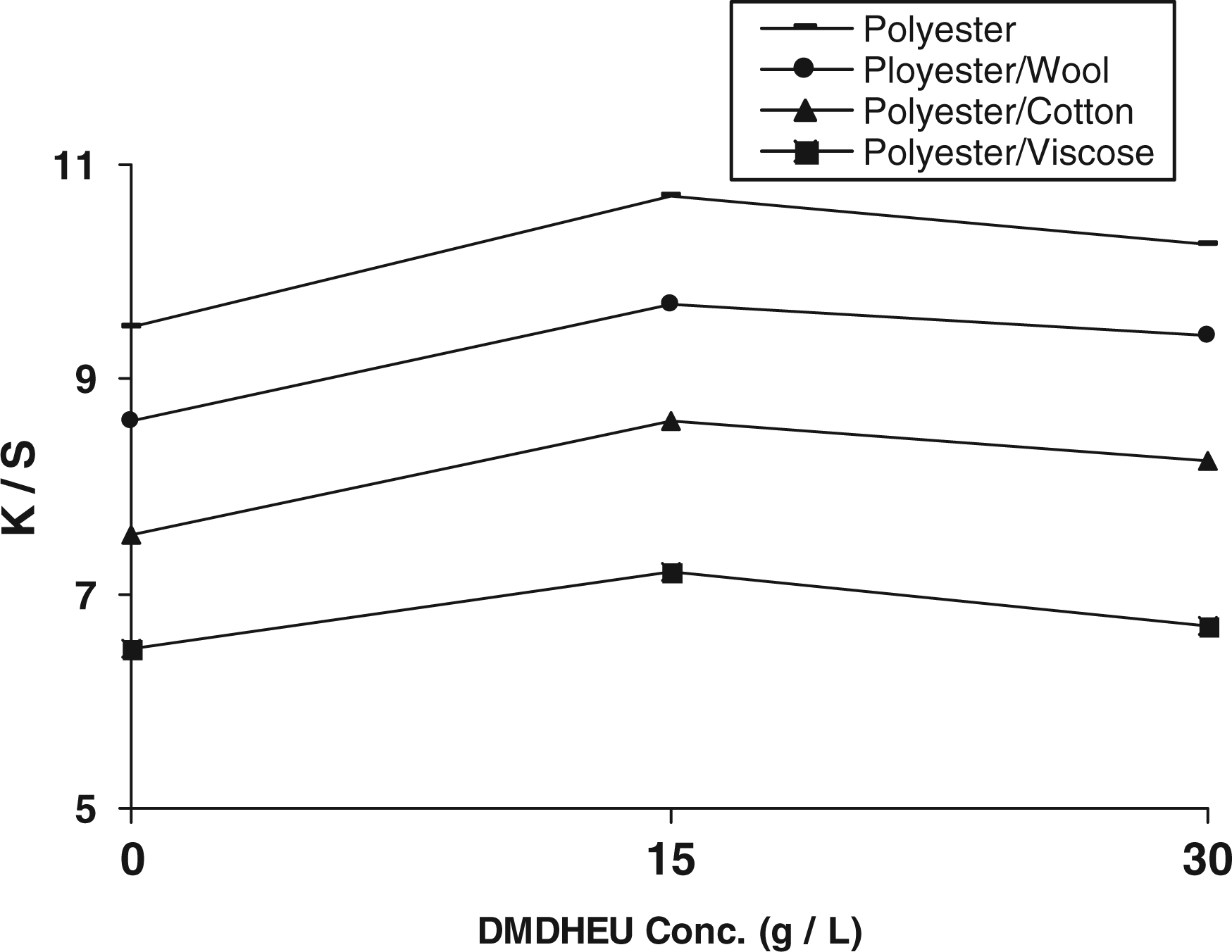
On the other hand, further increase in DMDHEU concentration up to 30 g/L results in a slight decrease in the K/S of the post-printed fabric samples, regardless of the treated substrate, which could be discussed in terms of higher extent of crosslinking, side interactions with the chitosan functional groups and reduction in the number of accessible cationic sites thereby giving rise to lower depth of shades.
Steaming temperature
Figure 7 reflects the effect of steaming temperature on the depth of the post-printed fabric samples. For a given set of pretreatment and disperse printing formulations, the results showed that increasing the steaming temperature up to 120°C for 15 min results in a gradual increase in the depth of the obtained prints, then slows down with further increase in steaming temperature, that is beyond 120°C, irrespective of the printed substrate.
Effect of steaming temperature on the K/S values of the disperse prints. (i) Pretreatment conditions: DMDHEU (15 g/L) , mixed catalyst (5 g/L), Chitosan (1.25 g/L), padding to a wet pick-up (70%) followed by direct fixation at 120°C for 3 min. (ii) Post-printing conditions : Disperse Red 74 (30 g/kg); stock-thickening agent 10% (500 g/kg); acetic acid 30% (15 g/kg), drying at 85°C for 5 min, steaming at (100–130°C) for 15 min. DMDHEU: dimethylol dihydroxyethylene urea.
This enhancement in K/S values could be discussed in terms of facilitating the dye release from the thickener film, improving the swellability of the pretreated substrates and availability of their active sites thereby providing more possibilities for adsorption, accommodation and fixation of disperse dye molecules, that is better depth of shade [15].
On the other hand, the decrease in K/S values beyond 120°C might be due to partial hydrolysis of adhered and loaded chitosan and/or crosslinked chitosan onto/within the fabrics surface, and the increase in the swellability of the thickener film thereby hindering the extent of dye release to the modified substrates, and/or undue diffusion and penetration of the released dye into the highly swelled substrates, that is lighter depth of shades.
Needless to say, the K/S of the obtained prints is determined by the type of substrate as discussed before.
Steaming time
Figure 8 reflects the effect of steaming time on the depth of the obtained disperse prints. It is clear that prolonging the steam fixation time up to 15 min at 120°C is accompanied by an improvement in the K/S values of the obtained prints. This could be discussed in terms of enhancement in the extent of dye release and sublimation as well as the adsorption, diffusion and retention of dye molecules onto and/or within the modified substrates thereby enabling more due fixation [15]. Further increase in fixation time, that is up to 20 min at 120°C has practically a slight negative impact on the depth of the obtained prints. This reduction in color strength, at prolonged steaming time, could be discussed in terms of adversely affecting the thickener film properties and/or the modified surface of the treated substrates thereby reducing the extent of coloration [16].
Effect of steaming time on the K/S values of the disperse prints. (i) Pretreatment conditions: DMDHEU (15 g/L), mixed catalyst (5 g/L), Chitosan (1.25 g/L), padding to a wet pick-up (70%) followed by direct fixation at 120°C for 3 min. (ii) Post-printing conditions: Disperse Red 74 (30 g/kg); stock-thickening agent 10% (500 g/kg); acetic acid 30% (15 g/kg), drying at 85°C for 5 min, steaming at 120°C for (5–20 min). DMDHEU: dimethylol dihydroxyethylene urea.
Molecular weight of PEG
Figure 9 shows the variation in the K/S values of the disperse printed fabric samples as a function of molecular weight of PEG in the pretreatment step. For a given set of pretreatment formulation as well as subsequent disperse printing, it is clear that (a) incorporation of any of nominated PEGs, except PEG-800, along with chitosan, DMDHEU and the mixed catalysts is accompanied by an improvement in the extent of post-printing with the nominated disperse dye, (b) increasing PEG molecular weight up to 600 results in a dramatic increase in K/S values of both the polyester and the polyester/wool blend fabrics along with a reasonable improve in K/S values of both the PET/C and the PET/V substrates, (c) further increase in PEG molecular weight, that is beyond molecular weight 600, brings about a decrease in the extent of post-printing, (d) the enhancement in K/S values by incorporating PEG in the pretreatment bath reflects its positive impacts on decreasing the extent of crosslinking via direct competition with the −OH and −NH2 groups of chitosan and/or the blended substrates for the accessible methylolated groups of the DMDHEU, (e) its ability to keep the structure of pretreated substrates swell and open during the thermofixation step thereby enhancing the extent of: adsorption of the released dye, interaction, retention and fixation, that is better printability and (f) the decrease in K/S values of the disperse printed samples pre-loaded with PEG-800 could be discussed in terms of blocking and/or reducing accessibility of the amino groups of grafted chitosan, enhancing the hydrophilicity and minimizing the access of the released dye molecules to the treated fabric surface, decreasing the extent of dye adsorption, dye–fiber interaction and fixation, as follows (Figure10) [10,17].
Effect of including PEG with different molecular weights on the K/S values of the disperse prints. PEG: Polyethylene glycol. (i) Pretreatment conditions: DMDHEU (15 g/L), mixed catalyst (5 g/L), Chitosan (1.25 g/L), PEG (200–1000) (10 g/L), padding to a wet pick-up (70%) followed by direct fixation at 120°C for 3 min. (ii) Post-printing conditions: Disperse Red 74 (30 g/kg); stock-thickening agent 10% (500 g/kg); acetic acid 30% (15 g/kg), drying at 85°C for 5 min, steaming at 120°C for 15 min. DMDHEU: dimethylol dihydroxyethylene urea; PEG: polyethylene glycol. Possible interactions of PEG with other ingredients. PEG: Polyethylene glycol.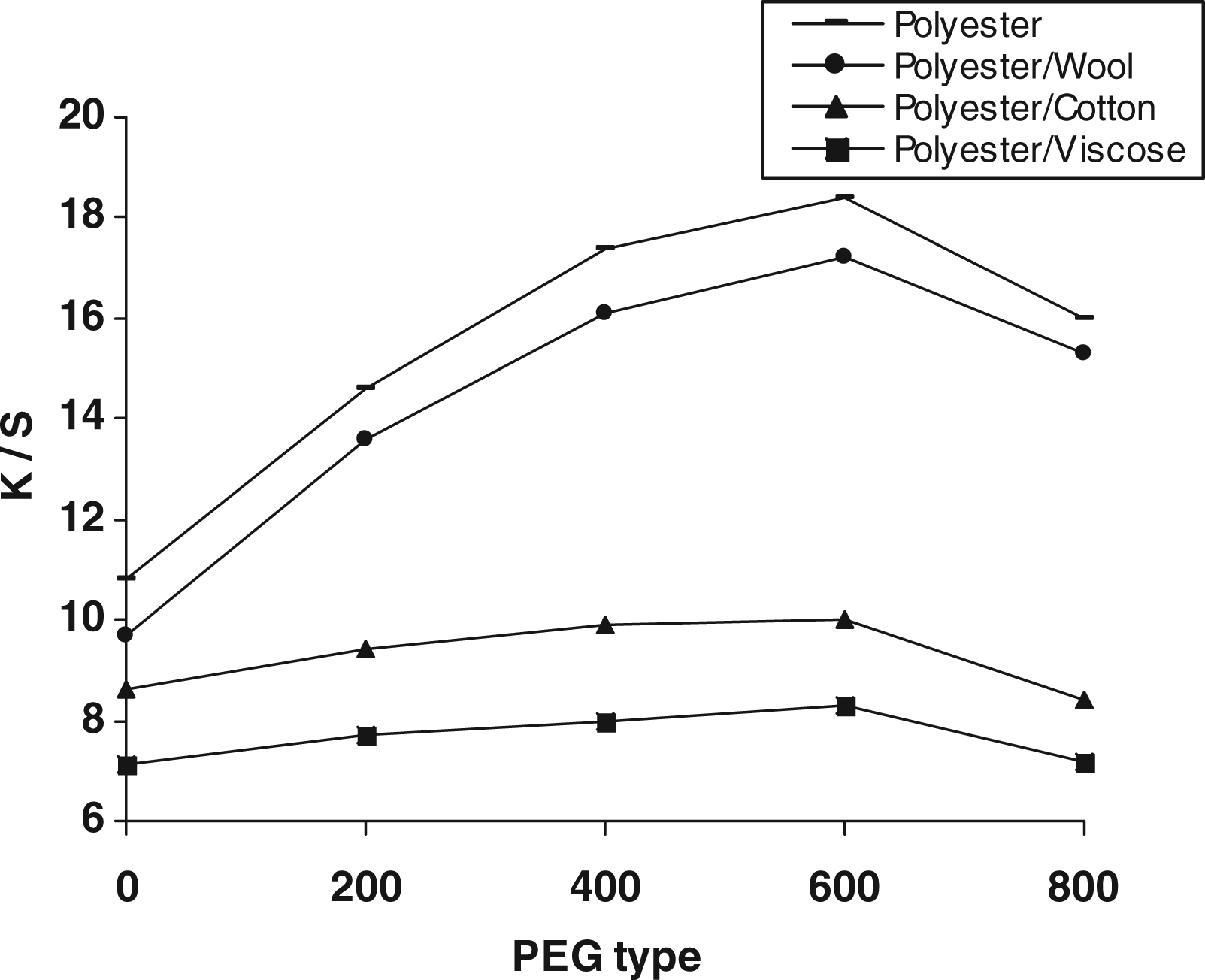

Disperse printing and UV-protecting properties

The impact of pretreatment and type of disperse dye on disperse printing and UV-protecting properties.
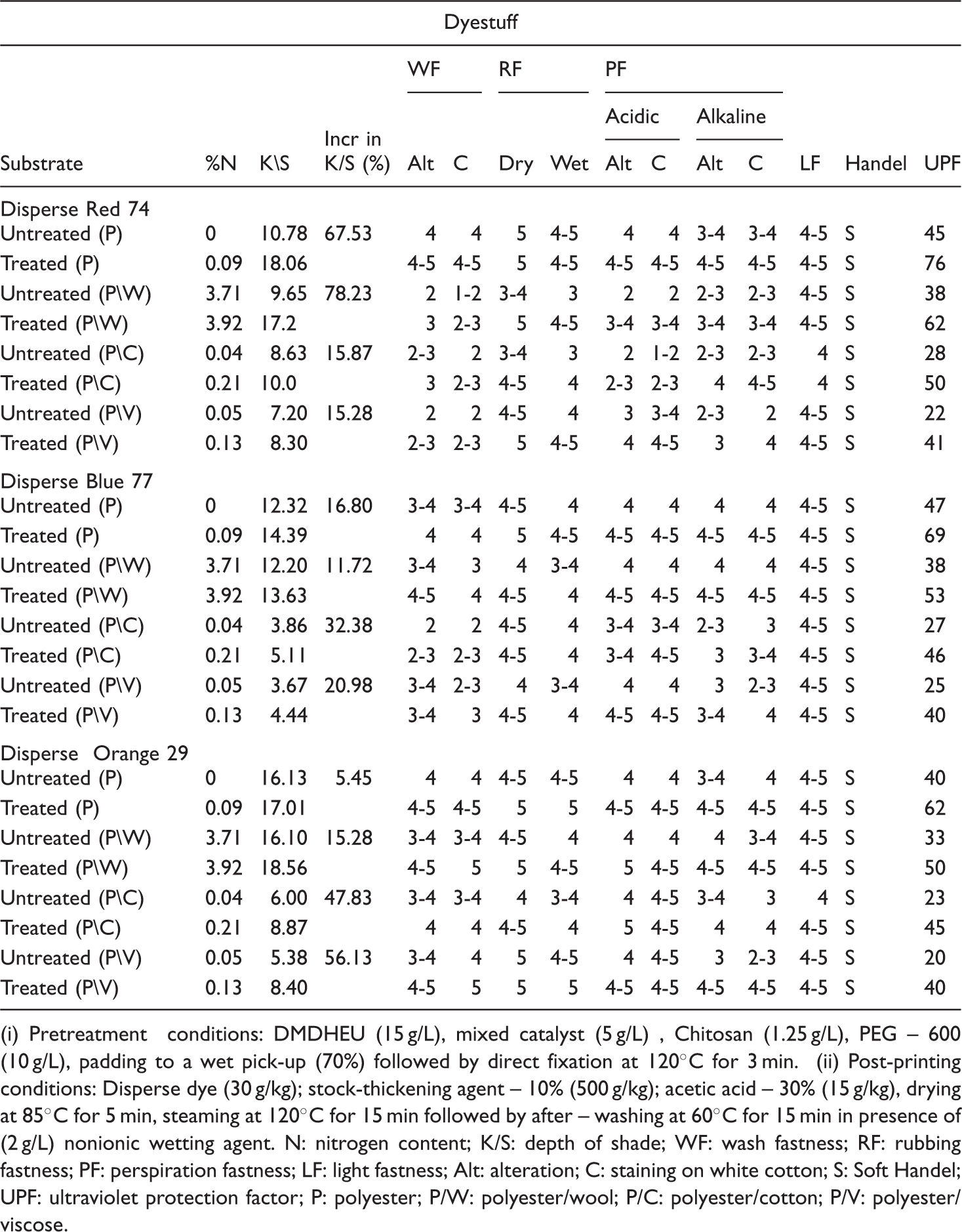
(i) Pretreatment conditions: DMDHEU (15 g/L), mixed catalyst (5 g/L) , Chitosan (1.25 g/L), PEG – 600 (10 g/L), padding to a wet pick-up (70%) followed by direct fixation at 120°C for 3 min. (ii) Post-printing conditions: Disperse dye (30 g/kg); stock-thickening agent – 10% (500 g/kg); acetic acid – 30% (15 g/kg), drying at 85°C for 5 min, steaming at 120°C for 15 min followed by after – washing at 60°C for 15 min in presence of (2 g/L) nonionic wetting agent. N: nitrogen content; K/S: depth of shade; WF: wash fastness; RF: rubbing fastness; PF: perspiration fastness; LF: light fastness; Alt: alteration; C: staining on white cotton; S: Soft Handel; UPF: ultraviolet protection factor; P: polyester; P/W: polyester/wool; P/C: polyester/cotton; P/V: polyester/viscose.
Conclusions
Chitosan, which is an amino-polysaccharide, can be used to enhance the affinity of polyester-containing fabrics, that is PET, PET/W, PET/C and PET/V, for disperse printing. It is possible to get union disperse printing of the above mentioned substrates after chitosan/PEG/DMDHEU pretreatment. The suggested pretreatment results in a significant modification of the treated substrates via introducing of chitosan primary amino groups as well as PEG moieties onto and/or into the fabric structure thereby improving, the extent of disperse printing as well as upgrading washing, rubbing and perspiration fastness properties of the obtained prints. With respect to the surface modification by pretreatment, the SEM photographs revealed the deposition of chitosan on the treated fabrics. Type of substrate, PEG and disperse dye significantly affected the depth of the obtained prints. The influence of pretreatment on both the depth of the obtained prints and on their UV-protection capacities is very significant. The highest UPF values appear with pretreated and disperse printed PET and PET/W fabrics, regardless of the used disperse dye.
Upgrading printing properties and antibacterial functions of polyester-containing fabrics via using chitosan along with other additives will be discussed in a future publication.
