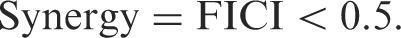Abstract
Surgical site infection due to biofilm formation is a common clinical complication in post-operative patients. Surgical site infection is the main complication in the case of gastrointestinal (appendectomy, genitourinary), colorectal, vaginal or abdominal hysterectomy, Caesarean section, abortion, traumatic wound and ophthalmic surgeries. Hence, the main aim of this study was to prepare antibacterial suture materials to prevent the biofilm formation. Surface characterization of drug-coated sutures was chemically analysed by Fourier transform infrared spectroscopy. Antibacterial activity of drug-coated sutures was analysed qualitatively (Agar diffusion test) and quantitatively (bacterial adherence test). Persistence of drugs in the sutures after the elution period was analysed by serial plate transfer test. Fourier transform infrared spectroscopy analysis showed two peak values for drugs (1450.52 and 2870.17). Largest inhibition zone for Staphylococcus aureus (39 mm) was reported; quantitative analysis confirmed significant reduction (p < 0.05) of adhered bacteria in drug-carrier-coated sutures than that of carrier-coated sutures (p > 0.05). Serial plate transfer test indicated the persistence of drug after 5 weeks. In conclusion, the antimicrobial activity and persistence property of these drug-eluting silk sutures could be of great interest for the prevention of surgical site infections.
Keywords
Introduction
Surgical site infections (SSIs) are a major cause of nosocomial infections [1], which accounts for 14% to 16% of hospital-acquired infections [2]. Depending on the type of surgery and patient characteristics, SSI rates range of 0.5% to 13% was reported [3]. SSIs, if prevented, help in lowering the rate of mortality of patients and length of hospital stay [4]. Commonly used measures to prevent SSIs are appropriate use of antibiotics against the common pathogens [5] and drug-loaded suture materials which include triclosan-coated silk sutures [6], ibuprofen-incorporated surgical sutures and polyglactin-coated silk suture [7]. Generation of resistance is one of the major problems associated with straightforward antimicrobial loading. This may enhance virulence, delay the administration of appropriate therapy and subsequent recovery [8]. Due to the generation of resistance, various types of antimicrobial agents used for coating were not considered as biocompatible although they have proven antibacterial activity [9]. Biofilms are the major cause of antimicrobial resistance. These are complex, hydrated matrix of polysaccharide, protein and organized communities of bacteria that grow in association with a wide array of biotic and abiotic surfaces [10]. These are embedded within a self-produced matrix of extra cellular polymeric substances (EPS) and within the high dense bacterial population, efficient horizontal transfer of resistance and virulence genes takes place [11]. The accepted clinical practice to prevent suture-associated infections was the use of combination therapy in which two or more antimicrobials were blended at different combinations, so that broader spectrum of activity was achieved at a lower concentration resulting in more effective therapy and decreased resistance [12]. According to Boeckh et al. [13], biofilm formation can be prevented by combining quinolones with other antibacterial agents. Fluoroquinolone and nitroimidazole drugs exhibit synergism since they inhibit DNA synthesis and replication by acting on the DNA of bacteria [14]. These problems led us to study the effects of introducing a fluoroquinolone drug and a nitroimidazole drug with a biodegradable carrier (tocopherol acetate) at the surface level of a suture material. Tocopherol acetate is biocompatible and has antioxidant property since they scavenge free-radicals and inhibit platelet adhesion [15]. Hence, in the present study it was used as a coating material to bind the antibacterial agents and also to enhance the antibacterial activity.
The objective of our work was to develop an antibacterial coating of biomedical sutures for preventing SSI. The specific objectives are to assess the synergistic effect of the fluoroquinolone and nitroimidazole drug, to explore tissue reaction of drug-coated suture material, to characterize the drug-coated surfaces of suture material using chemical method and to evaluate the release kinetics, antibacterial activity and persistence of synergistic drug-coated suture material.
Materials and methods
Suture material
Commercially available silk suture was procured (Ethicon. Inc, India) and used as test sample for further processing in the present study. Sample size of 7.5 cm was cut and pre-weighed before coating with antibacterial drugs.
Antimicrobial compounds
Medical grade fluoroquinolone (levofloxacin) and nitroimidazole compounds (tinidazole) were purchased from Sigma chemical Co. The drugs were checked for their purity based on their specific wavelength using UV-VIS spectrophotometer.
Drug carrier – tocopherol acetate
Drug carrier used for the present study was tocopherol acetate (TA; Merck, India).
Structure of tocopherol acetate (C31H52O3)

Test organisms
Clinical isolate of biofilm-forming strains of Staphylococcus aureus and Staphylococcus epidermidis, Escherichia coli, Klebsiella pneumonia and Pseudomonas aeruginosa obtained from a clinical laboratory was used for the in vitro studies. All the strains were cultured to late logarithmic growth phase on blood agar plates at 37°C for 18 h before testing. Bacterial cells were then re-suspended in normal saline and adjusted to 2 × 105 colony forming unit (CFU)/mL by visual comparison with a 0.5 McFarland standard.
Pre-treatment of silk suture (scouring)
Scouring is a process of removal of natural and added impurities present in suture materials, in order to improve the absorbency. The suture material was scoured in 1% by weight of sodium hydroxide (for 1 min) at room temperature (27°C) to eliminate impurities and washed with de-ionized water [16].
Assessing the synergistic activity of selected drugs
Minimal inhibitory concentration
The minimal inhibitory concentration (MIC) of each drug was determined by a standard agar dilution method as described by Bharadwaj et al. [17]. Subsequently by doing a checkerboard titration [18], the combined action of these set of drugs on each test organism was also studied to assess the synergistic effect of the drugs using fractional inhibitory concentration index (FICI).

where A and B are the two antimicrobials under investigation,
Interpretation
Synergy: Synergistic action of a combination of antibiotics is present if the effect of the combination exceeds the additive effects of the individual components.
Carrier grafting with antibacterial drugs
Preparations of levofloxacin and tinidazole suspension with drug carrier were made according to the method described by Matl et al. [19]. In brief, the antibacterial agents were suspended in 0.1 N acetone (Sigma chemicals). To decrease the particle size, suspensions were homogenized for 5 min under sterile conditions. The resulting suspension was added to the drug carrier (TA), for building up a drug concentration of 10%.
Suture coating with antibacterial drugs
The silk suture was cut into pieces (length-7.5 cm) and sterilized in the autoclave at 121°C. Tocopherol acetate used as a drug carrier for incorporating levofloxacin and tinidazole (drugs) at a concentration of 10%. Sterile suture pieces were coated with the drug carrier combination by dip-coating method [19], ensuring a regular polymer coating, and referred to below as drug-carrier-coated suture. To determine the antibacterial efficiency of drug-carrier-coated sutures, another set of suture pieces (length-7.5 cm) was coated in the same way with the carriers alone without drugs; these control sutures are referred to below as carrier-coated sutures (ccs).
FTIR analysis of drug-coated silk sutures
The Fourier transform infrared spectroscopy (FTIR) absorption spectra of the uncoated and drug-carrier-coated silk sutures were recorded in the range of 400–4000 cm−1 by KBr disc method using FTIR spectrophotometer. Briefly, the coated materials (uc and dcc) were mixed with KBr (200 mg) to avoid moisture and made into discs (1 cm dia). Each disc was dried under radiation to remove excess moisture content. The peaks for .C=O stretching and O–H stretching of .COOH and the drugs were analysed [20].
Release kinetics of synergistic drugs
The release profile of synergistic drugs from the drug-carrier-coated suture surface was studied for 120 h. The release profiles of the synergistic drugs were evaluated using UV-VIS spectrophotometer at their respective wavelengths. The concentration of each drug was obtained from the calibration curves of the respective drugs [21].
Tissue response of chick chorio-allantoic membranes to suture material
To understand the hypersensitive reactions of drug-carrier-coated suture material on the live tissues, the materials were placed on the surface of chorio-allantoic membrane (CAM) of embryonated chick eggs. Uncoated suture was used as a control for differentiating the tissue reaction. The experiment was carried out based on the method described by Valdes et al. [22].
Antibacterial activity
Tests for qualitative antibacterial activity were carried out on silk of 5 cm in length (cut from 7.5 cm drug-carrier-coated suture, ccs and uncoated sutures). The sutures were all rinsed twice in phosphate buffer saline (PBS) before testing to remove any surface accumulation of drug. Antibacterial activity was expressed in terms of zone of inhibition around the suture [23].
Quantitative analysis was carried out by the method described by Elayarajah et al. [24]. Briefly, a 50 µL aliquot of bacterial suspension (2.5 × 105 CFU/mL) was added to test tube containing 1 mL PBS and two suture pieces. To calculate the amount of bacteria added (inoculum dose), 100 µL samples of bacterial suspension were plated on nutrient agar plates after 1:10 serial dilutions. After 3 h of incubation, the adherent bacteria were detached with a water-bath sonicator. The amount of detached bacteria (DB) was calculated as CFU/mL after the 1:10 dilution series were plated onto nutrient agar plates.
To see whether the drug-carrier-coated sutures could inhibit bacterial growth during the 3-h incubation period, remaining bacteria were calculated as CFU/mL after a 1:10 dilution series. The ratio of CFU of the remaining bacteria to CFU of the inoculum dose was calculated. Similar set of experiment was done parallel for the ccs to check the efficiency of antimicrobial agent.
Statistical analysis
SPSS for Windows 7.0 was used as the statistical package and chi-square-nonparametric test was used to calculate the differences in bacterial adherence studies in which p < 0.05 was considered significant.
Persistence test-serial plate transfer test
The persistence test-serial plate transfer test (SPTT) is a test used to assess both the presence of antibacterial activity and its duration. It has also been useful in detection of resistance in adherent bacteria after prolonged exposure. The test was continued for 5 weeks [25].
Results
Synergistic activity of levofloxacin and tinidazole
Assessment of synergy of levofloxacin and tinidazole on test organisms.
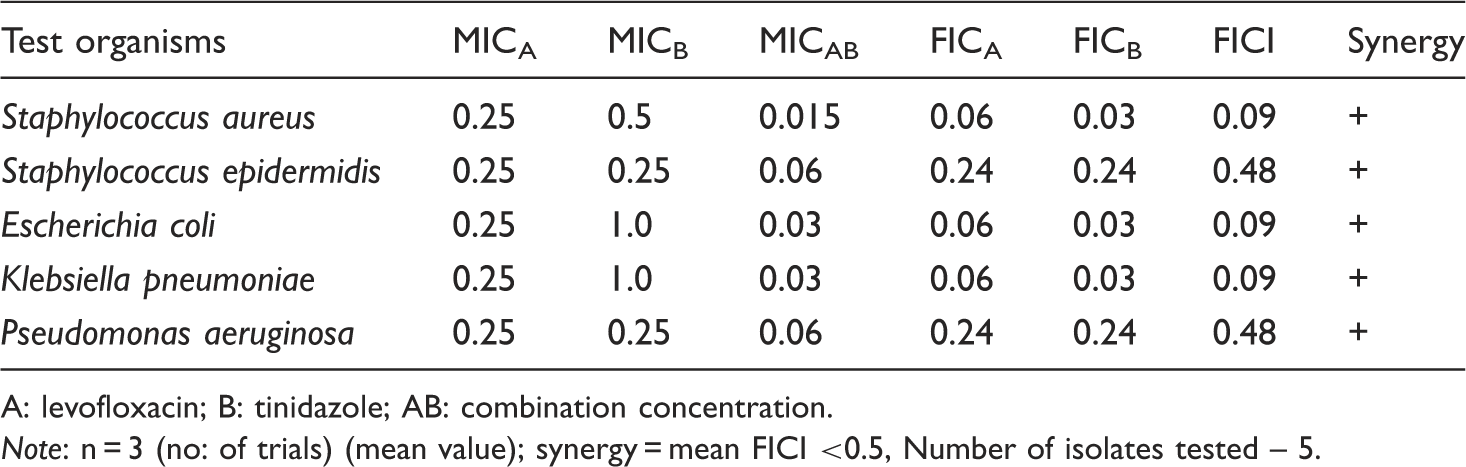
A: levofloxacin; B: tinidazole; AB: combination concentration.
Note: n = 3 (no: of trials) (mean value); synergy = mean FICI <0.5, Number of isolates tested – 5.
FTIR analysis of drug carrier coated silk sutures
The FTIR spectra of uncoated suture and drug-carrier-coated sutures were recorded. The results were shown in Figures 1 and 2. In the FTIR spectrum of the uncoated suture sample, the presence of the trace amount of COOH (carboxyl) functional group was evident from the absorption in the region 3200 cm−1 to 2800 cm−1 (O-H stretch) and in the region 1500 cm1 to1200 cm−1 (C=O stretch). FTIR analysis showed two common peaks for uncoated and drug-carrier-coated silk sutures at C=O stretching (1311.64) and O–H stretching (2955.04) of COOH. The other peaks representing the functional group of silk suture were replaced by the peak values of drugs (1450.52 and 2870.17). This indicated the presence of synergistic drugs on the coated suture surface.
FTIR analysis of uncoated suture. FTIR analysis showing two common peaks for uncoated and drug-carrier coated sutures at O-H stretching (2955.04) of COOH and C=O stretching (1311.64). FTIR analysis of drug-carrier coated suture. Peaks representing the functional group of silk suture were replaced by the peak values of drugs (1450.52 and 2870.17).

Release kinetics of synergistic drugs
After the dissolution studies, the concentration of released drug from each suture material (for each drug) was determined from the calibration graph (Figure 3). Thus, it was observed that the levofloxacin showed maximum release between 65 and 130 µg and Tinidazole between 70 and 135 µg. The drug-carrier combination showed best results with initial burst period during the first few hours of release. The amount of drug released from the material found static after 96 h of time with slow and sustained release of drugs for a longer duration. Only few amount of drug was produced after 120 h, it may continue its release many more hours. The amount released after dissolution study were proportional to the drug coated on the suture surface. The weight of material after coating revealed that an average of 4 mg was coated on each material. The release kinetics was carried out thrice to ensure the efficacy of drug-carrier-coated suture.
Drug release analysis of drug-carrier coated sutures. Levofloxacin showed maximum release between 65–130 µg and Tinidazole between 70–135 µg.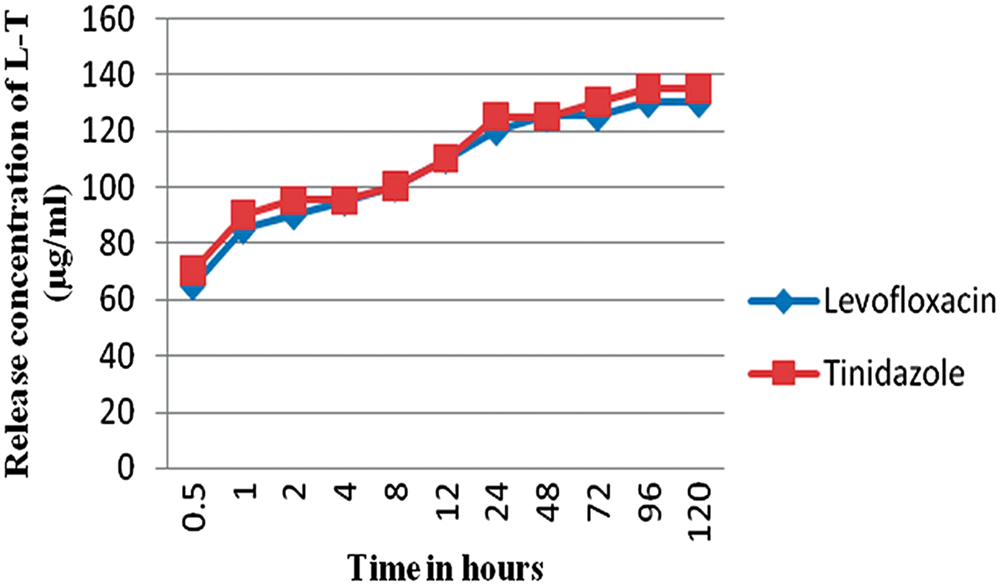
Tissue response of CAM to drug-coated silk sutures
Bright-field microscopic analysis was done after the incubation period of 7 days. The observation showed no degenerative cells for drug-carrier-coated when compared to uncoated silk suture on the CAM (Figure 4).
Tissue reactions of drug coated suture on CAM. No degenerated cells were seen on CAM exposed to drug-carrier coated sutures.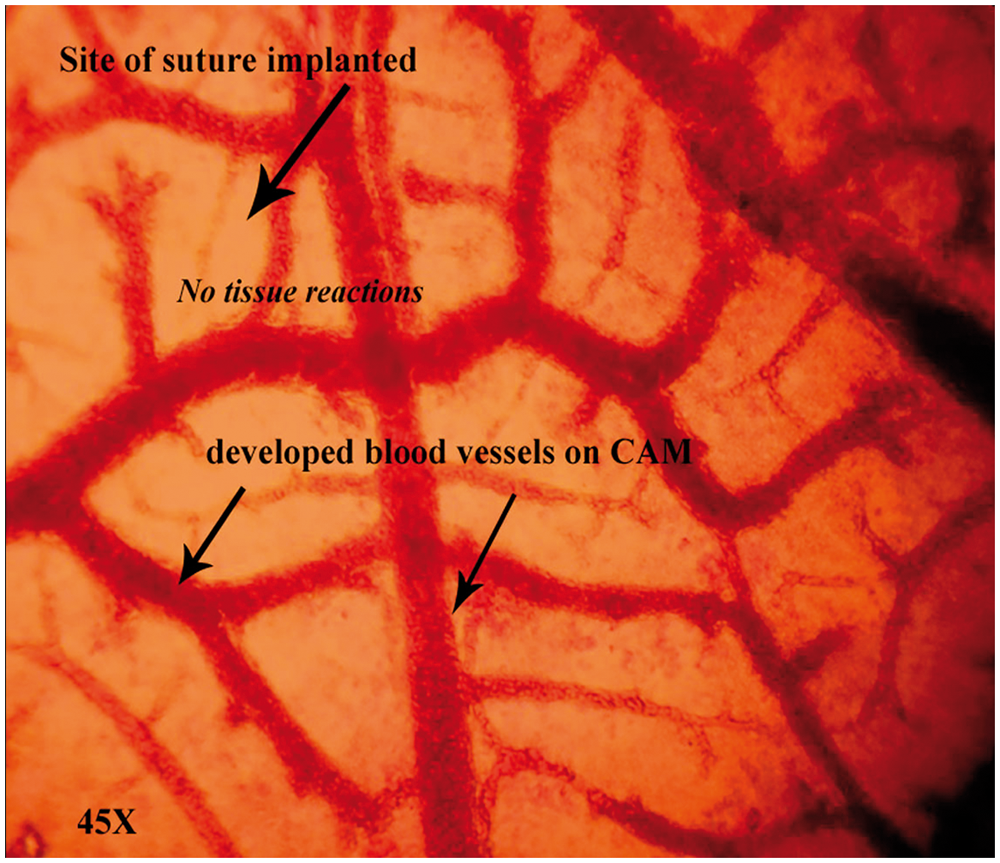
Antibacterial activity
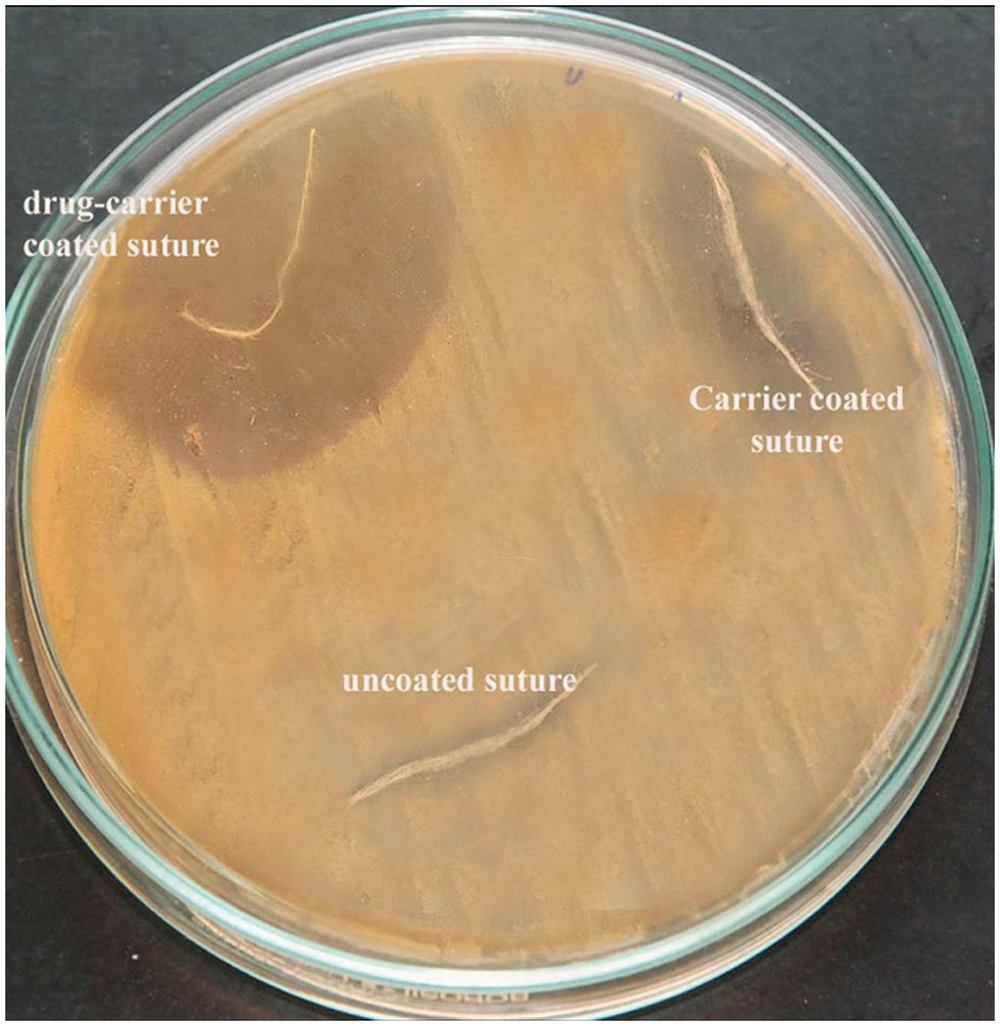
The diffusing ability of the antimicrobial drugs from the coated suture pieces to retard the growth of test cultures seeded on Muller-Hinton Agar (MHA) plate was calculated based on the zone of inhibition. The largest zone of inhibition was observed for Staphylococcus aureus (39 mm). The zone of inhibition produced by S. epidermidis, E. coli, K. pneumoniae and P. aeruginosa were 33, 36, 38 and 37 mm, respectively. The uncoated sutures showed no zones of inhibition (Table 2). Figure 5 represents the qualitative antibacterial activity of fabricated sutures against S. aureus.
Antibacterial activity of sutures. Drug-carrier coated sutures showing more antibacterial activity against S. aureus when compared with carrier-coated and uncoated sutures. Agar diffusion test against test organisms. dccs: drug-carrier-coated sutures; ccs: carrier-coated sutures; uc: uncoated. Note: n = 3 (no: of trials) (mean value).

Drug-coated-carrier sutures (dccs) and ccs were inoculated with S. aureus, S. epidermidis, E. coli, K. pneumoniae and P. aeruginosa separately and adherence was studied in vitro based on the effect of inoculum dose and also to see whether the dccs could inhibit bacterial growth during 3-h incubation period. The remaining bacteria was calculated based on the ratio of CFU of remaining bacteria to CFU of inoculum dose and compared simultaneously for ccs.
Bacterial adhesion studies of drug-coated-carrier sutures and carrier-coated sutures.

Note: n = 3 (no: of trials) (mean value).
Serial plate transfer test
Serial plate transfer test (SPTT).

Note: n = 3 (no: of trials) (mean value).
Discussion
SSIs account for about one third of all the nosocomial infections. The pathogens come from various sources such as members of the surgical team, contaminated instruments or infected site in the patient’s body [26]. The common bacteria causing SSIs are S. aureus, Enterococcus species, S. epidermidis and E. coli [27]. Bharadwaj et al. [17] found out the antibacterial efficacy of quinolone-nitroimidazole drug combination against both aerobic and anaerobic bacterial infections. In comparison with individual effect of ofloxacin and ornidazole, ofloxacin-ornidazole combination exhibit increased efficacy against aerobic bacteria [28]. Their work suggests that ofloxacin-ornidazole combination can be used to prevent mixed microbial infection of aerobic bacteria, anaerobic bacteria and pathogenic protozoan. Hence, these observations agree with the present study which involves the use of synergistic drug-eluting sutures capable of inhibiting the bacterial growth effectively with sustained release, thereby preventing biofilm formation for a longer duration.
Patel et al. [20] obtained major peaks such as 1663.19, 2843.01 nm, 2975.69 and 821.53 nm for Gatifloxacin sesquehydrate present in drug-loaded ocular inserts. This confirmed the presence of drug in the polymer without any interaction. The peak for the .C=O stretching of .COOH was obtained at 1281.59 nm. In our study, similar assumption was made. Peak values for drugs were 1450.52 nm and 2870.17 nm whereas the peak for .C=O stretching of .COOH was obtained at 1311.64 nm. The release kinetics of synergistic drugs showed sustained release of drugs after initial bursting. This is similar to the observation made by Mashru and Saikumar [21] in which the dissolution of ofloxacin and ornidazole was studied. The effect of tissue response of the drug-carrier-coated suture was determined by examining the chorio-allantoic membranes of embryonated eggs against the uncoated suture. There were no visible signs of tissue reactions on the CAM even after incubating for a period of 7 days. Similar work was carried out by Valdes et al. [22] to determine the tissue response of CAM against different dental implant materials. In their study, epoxy resins showed a mild response with minimal alteration of tissue elements; and metal implant showed a mild response of some loss of ectoderm and increased fibrous depositions under the membrane.
In the qualitative tests, the antibiotic combination exhibits synergism that effectively inhibits the kinetic development of test organisms over the MHA plates. Similar work was done by Shanmugasundaram et al. [29] in which drug-loaded poly-lacticacid suture inhibits the growth of S. aureus and Proteus bacteria. The quantitative test involving bacterial adhesion revealed a significant reduction in viable counts which is similar to the observation of Gollwitzer et al. [14] in which the combination of poly D-L-lactic acid with either gentamicin or teicoplanin or both antibiotics on the implant together indicated reducing viable counts to almost undetectable levels.
The persistence of antibacterial agents on the sutures has been proved by means of SPTT. In the present work, the persistence of drug with a reduction in antibacterial activity was observed after 5 weeks. This observation agree well with the work of Bayston et al. [25] in which the loss of activity in terms of zone size was between 33% and 40% for the Staphylococci and >75% for E. coli. The findings in our study revealed that two synergistic drug combinations can strongly bind to suture materials and inhibit the growth of test organisms.
Conclusion
The present research with levofloxacin-tinidazole-TA combination showed good antibacterial and persistence activity against both Gram positive and negative test organisms. So this combination may help to reduce the SSIs caused particularly by S. aureus, K. pneumoniae and P. aeruginosa. SSIs could be prevented by using this type of drug-carrier-coated sutures that provide a sustained release of drugs at the operated site, so that the chance of portal entry at the operated site by any microrganisms can be prevented. Other synergistic drugs may be used in a similar manner so that more research could be expanded to coat different types of suture materials like silicone, nylon and polyester. Thus, the fabricated drug-eluting silk sutures could be of great interest for the post-operative patients for the prevention of SSIs if these results are confirmed in vivo. It can be concluded that the drug-carrier-coated sutures are quite suitable for wound healing in addition to wound closing.