Abstract
In this research, the non-formaldehyde wrinkle-resistant treatment of cotton fabrics has been investigated using the citric acid (CA) as a cross-linking agent and sodium hypophosphite (SHP) as a catalyst together with nano-titanium dioxide (nano-TiO2) as a co-catalyst compound. The effect of the changes in the concentrations of CA, SHP, and nano-TiO2 on the wrinkle recovery angle (WRA), tensile strength, tearing strength, whiteness index, and flexural rigidity of cotton fabrics was studied using Box–Behnken design. It was found that the addition of nano-TiO2 could enhance the wrinkle resistance and decrease the flexural rigidity of the cotton fabric with little effect on the whiteness index, and tearing and tensile strengths of the treated fabric. The developed empirical models are found to be in good correlation with the selected variables (r2 ≥ 0.8). From this study, it was concluded that 10% CA, 10% sodium dihydrogen phosphate, and 0.1% nano-TiO2 were the optimum concentrations required to enhance the WRA without adversely affecting the other physical properties of the treated cotton fabrics.
Introduction
The purpose of textile finishing is to furnish the textile materials suitable for their end uses as well as to meet certain customer’s expectation by the application of innovative chemical finishes based on the aspects of comfort, safety, esthetics, and functional performance. The use of novel finishes and innovative technologies to modify natural materials can be a cheaper route to achieve a higher quality and technical performance rather than the use of high-cost fiber with inherent built-in performance properties.
Cotton is a well-known cellulosic fiber, which is a kind of polysaccharide. The cellulose molecules are linearly arranged and pass through the crystalline and amorphous regions of the cotton fibers. In the crystalline region, the cellulose chains are closely packed. On the other hand, in the amorphous regions, they are temporarily held together with weak hydrogen bonds and easily broken when distortion force is applied. When a force of sufficient magnitude is applied onto the fibers, slippage occurs between the cellulose chains or between larger structural units of the fiber. The hydrogen bonds present between the cellulose molecules tend to resist or prevent the slippage. However, when slippage occurs, the hydrogen bonds reform at new locations and tend to maintain the fiber in the bent or wrinkled state [1]. As a result, fabrics become wrinkled in appearance. In addition, cotton fiber is hydrophilic and can easily absorb water. The absorption of water breaks the hydrogen bonds and allows the fiber or fabric to shrink. As a result, 100% cotton easily wrinkles and has the potential to shrink after laundering.
One of the most important factors that influence the quality of fabric is the ability of fabrics to recover from induced wrinkles. Many cotton fabrics are treated with chemicals to reduce wrinkling. This crease-resistant finish to cotton fabrics improves the comfort, ease of maintenance, dimensional stability, and pilling performance [2]. The disadvantages of conventional urea-formaldehyde resin finishing include release of excess free formaldehyde during drying, pungent odor, toxicity, reproducibility, and loss of tensile strength of the treated fabric. Non-formaldehyde finishes such as dimethylol dihydroxyethylene urea and polycarboxylic acid (PCAs) have been used as the major conventional wrinkle finishing agent because of their easy availability, high stability to acid and chlorine, ease of reaction, cost effectiveness, and the absence of bad odor. However, excessive fabric strength loss remains a serious problem [2–4].
Nanotechnology controls the material in this size range having unique properties that enable novel applications. Nano-particles are significant compared to the larger particles due to their higher surface area and the attractive forces between nano-particles and the substrates [5]. The advancement of nanotechnology in textile finishing area brought so many innovative functional finishes, thus overcoming the major problems associated with the conventional finishes. The application of titanium dioxide (TiO2) or nano-TiO2 as a catalyst or co-catalyst to improve the crease recovery property has been found to be feasible [6–8]. Hitherto, numerous research studies have been done on the application of carboxylic acids especially the use of 1,2,3,4-butanetetracarboxylic acid as the cross-linking agent for wrinkle resistance [9–16]. Very little research has been done on the effectiveness of the nano-particles with the citric acid (CA) with TiO2 as the co-catalyst [17,18]. The wrinkle-resistant property of cotton specimens treated by CA and catalyzed by sodium hypophosphite (SHP) in the presence of nano-TiO2 has been evaluated in this study. Different concentrations of CA, SHP, and nano-TiO2 used in the wrinkle-resistant finishing have been evaluated in order to determine their optimum concentrations using Box–Behnken method. Surface morphology of cotton specimens has also been investigated to evidence the presence of metal oxide on the fiber surface.
Materials and methods
Materials
A 100% semi-bleached plain-weave cotton fabric (EPI × PPI 96 × 96; yarn count 42s Ne), with fabric weight 112 g/m2 and size of 30 × 30 cm2 was used. Synthesis of nano-particles was carried out by a precursor titanium iso-propoxide. Cross-linking agent CA, catalyst SHP (NaH2PO2), co-catalyst TiO2 nano-particles, and Lissapol-N (Imperial Chemical Industries, UK), used as surfactant for the dispersion of nanoparticles in acrylic binder (Texacryl Binder SLN) were used to impart crease resistant finish.
Methods
Synthesis of TiO2 nano-particles
Titanium iso-propoxide (AR Grade), mol. Wt: 284.26, purchased from M/s Sigma–Aldrich chemical company, USA, was used as precursors.
Ti(OC3H7)4, ethanol, and acetic acid were maintained in a molar ration of 1:100:0.05, respectively. Ti(OC3H7)4 was hydrolyzed using glacial acetic acid at room temperature. The required amount of ethanol was added under vigorous stirring for 2 hour until a clear solution of TiO2 nano-crystals was formed. The pH value of the sol was adjusted to 8–9 using sodium hydroxide as gelling reagent and the sol droplets were formed during the process. The sol was further subjected to stirring for 2 hour, and then the gel particles were separated, which was followed by intermittent washing with acetone thrice. The washed gel particles were dried in the oven at 80°C. During the sol–gel synthesis of nano-TiO2, high ethanol ratio was kept to enhance the nucleophilic attack of water on Ti(OC3H7)4 and to suppress the fast condensation of Ti(OC3H7)4 species to yield TiO2 nano-crystals [19]. The chemical reactions that are expected to take place during this synthesis are shown below:
Characterization of nano-particles using X-ray diffraction
The crystallinity of the nanoparticles was determined by X-ray diffraction (XRD) using a Shimadz U model XRD6000 X-ray Diffractometer equipped with a Cu Kα (λ = 1.7889 Å) source (applied voltage 40 kV, current 40 mA). About 0.5 g of the dried particles were deposited, in the form of a randomly oriented powder, onto a Plexiglass sample container, and the XRD patterns were recorded at angles between 10° and 80°, with scan rate 5°/minute, sampling pitch 0.02°, and preset time 0.24 second. The peak width at half maximum in the XRD has been used to determine the crystal diameter as per the following Deby–Scherrer formula:
CA two-bath pad-dry-cure treatment
Two-bath method was adopted for treating the cotton fabric specimens [7]. Different concentrations of CA, SHP, and nano-TiO2 solutions of various concentrations from 0.01% to 0.1% were prepared.
In the first bath, the specimens were dipped and padded with a 100-mL mixture of CA and SHP solution until wet a pick-up of 100% was achieved at room temperature. The specimens were then dried at 85°C for 5 minute. In the second bath, the dipping and padding processes were performed again using 100-mL nano-TiO2 solution with various concentrations along with 0.5% Lisaapol-N (surfactant) and 1% acrylic binder. The two-bath padded specimens were subsequently dried at 85°C for 5 minute and then cured at 170°C for 3 minute. Finally, the fabrics were then exposed to an intensity of UV light source of 0.37 Mw/cm2 for 1 hour.
Scanning electron microscopy
The surface morphology of cotton specimens was examined by a JEOL JSM-6360 scanning electron microscope (SEM) with accelerating voltage 20 kV and current 10 µA at a high magnification power up to ×2000.
Evaluation of physical properties
The physical properties of the woven fabric such as wrinkle recovery angle (WRA; AATCC Test Method 66-2003), tensile strength and elongation (ASTM Test Method D 5035), tearing strength (ASTM D 1424-96), bending length (ASTM D1388-2002), and whiteness index (ASTM E313) values of both untreated and treated samples, were evaluated using the standard testing procedures and equipments after conditioning the specimens at 65% RH and 21 ± 1°C for 24 hour by bringing them to approximate moisture equilibrium in the standard atmosphere for preconditioning textiles as directed in Practice D 1776 in an environmental chamber (ASTM 2008).
Development of empirical model
Response surface methodology is an empirical modelization technique devoted to the evaluation of the relationship of a set of controlled experimental factors and observed results. It requires a prior knowledge of the process to achieve statistical model. Basically, this optimization process involves three major steps, performing the statistically designed experiments, estimating the coefficients in a mathematical model, and predicting the response and checking the adequacy of the model [20].
Box–Behnken design with three variables such as concentrations of CA, catalyst, and TiO2 at three different levels was studied to identify a significant correlation between the effects of these variables to the amount of desired wrinkle recovery and other physical parameters.
In this experiment, significant variables such as concentrations of CA and SHP (catalyst) have been selected based on the literature [21]. The concentrations of nano-TiO2 (co-catalyst) have been selected based on the preliminary work done by us. The nano-TiO2 concentrations have been kept at different levels 0%, 0.01%, 0.03%, 0.05%, 0.1%, 0.15%, 0.2%, 0.3%, 0.4%, and 0.5%. WRA of the finished fabrics was analyzed. From the preliminary work, it was observed that the nano-TiO2 beyond 0.1% does not show significant improvement in WRA and there was a decrease in the improvement of WRA as the concentration of nano-TiO2 was kept increasing. This was mainly due to the increase in the amount of catalyst that promoted the reversible cross-linking of cellulose molecules, resulting in the diminishing of the wrinkle recovery effect [8].
The levels of variables chosen for the trials

Computation was carried out using multiple regression analysis using the least squares method. In a system involving three significant independent variables X1, X2, and X3, the mathematical relationship of the response on these variables can be approximated by the quadratic (second degree) polynomial equation:

A multiple regression analysis is done to obtain the coefficients and the equation can be used to predict the response. The design is preferred because relatively few experimental combinations of the variables are adequate to potentially estimate complex response functions. A total of 15 experiments were necessary to estimate the 10 coefficients of the model using multiple linear regression analysis.
Results and discussion
Characterization of nano-TiO2 particles
The synthesis of uniform and stable nano-particles of TiO2 is of significant importance in determining the photo-catalytic activity. The XRD patterns of synthesized nano-TiO2 particles using Ti(O − iPr)4 precursor sample are similar to those reported in the literature [22], suggesting the formation of TiO2 nano-particles in anatase form (JCPDS card no. 21-1272)[23]. The identified XRD patterns of these synthesized samples give distinctive peaks at 25.39, 38.11, 48.47, 54.5, and 62.53, which resemble the Jung et al. study. From Figure 1, it is observed that the crystalline peaks are relatively broad compared to those normally obtained for bulk material, indicating that the crystal sizes are smaller.
XRD patterns of nano-TiO2 synthesized by sol–gel method.
The Scherrer equation is used to determine the average crystal size of the TiO2 nano-particles. According to Debye–Scherrer formula, the XRD peaks corresponding to the diameters of TiO2 particles are in range 6–10 nm (β = 1.3292, 2θ = 26.80). This indicates that the prepared nano-particles are closer to single crystal morphology.
Characterization of nano-coated fabric with SEM
The surface morphology of the nano-sol coated cotton fabric was investigated using SEM. In Figure 2, SEM micrograph shows the nano-scaled TiO2 particles on cotton samples. The nano-particles are well dispersed on the fiber surfaces and finely dispersed and embedded. The particle size plays an important role and it is showed that the particles, after washing, have penetrated and adhered strongly into the fabric matrix. The SEM results confirm that the nano-particles are uniformly distributed over the fiber surface.
SEM analysis results for CA- and nano-TiO2-treated 100% cotton fabrics.
Optimization of process parameters
In this investigation, nano-TiO2 treated fabric was optimized by Box–Behnken method for the improvement in WRA. The influence of three factors such as concentrations of CA, SHP, and TiO2 were investigated with three different levels.
Physical properties of treated and untreated samples using different process parameters

Regression equations for various physical properties

Effect of process parameters on WRA
Contour plot in Figure 3 represents the relation maximum WRA versus critical process parameters.
Optimization of: (a) CA with SHP, (b) CA with TiO2, and (c) SHP with TiO2 on WRA.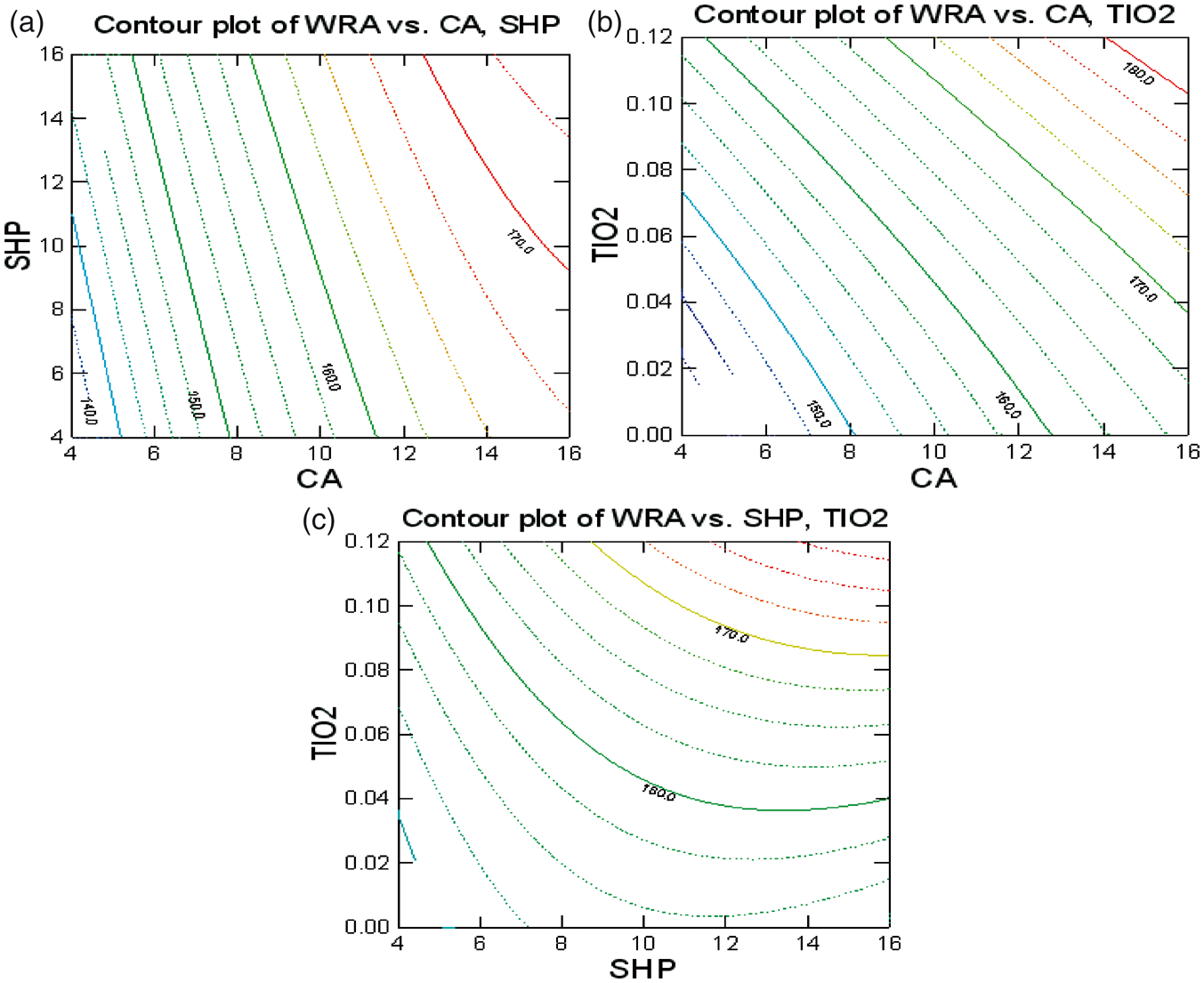
Figure (3a) shows the influence of CA and SHP on WRA for TiO2 concentration 0.05%. WRA increases with the increase in concentration of the CA and also with the increase in the concentration of SHP. The reaction between the PCA and cellulose hydroxyl group is composed of two stages. The first stage is the formation of a five-member cyclic anhydride intermediate, while the second stage is the formation of the ester bond between the cyclic anhydride ring and the hydroxyl group on the cellulose. Since the anhydrides are reactive and able to esterify cotton cellulose without a catalyst, the chief role of the catalyst is to accelerate the formation of anhydrides from PCAs [26]. The increased concentrations of CA and SHP increase the amount of cross-linking and hence the WRA.
Figure (3b) shows the influence of CA and TiO2 on the WRA for the SHP concentration of 10%. WRA increases with the increase in concentration of the CA, as mentioned above and also with the increase in the concentration of nano-TiO2. In the case of nano-TiO2, optimum wrinkle recovery could be achieved when concentrations of range 0.05–0.1% were added. Further increase in the amount of nano-TiO2 could not enhance the wrinkle recovery. As the particle size of the TiO2 is in nano-scale, the nano-TiO2 could fill the amorphous region of the cellulose and hence the presence of nano-TiO2 inside the fiber would probably restrict the molecular movement of cellulose. Hence, the reduction of the wrinkle recovery ability of the cotton fabric would possibly be the result. Furthermore, the increase of nano-TiO2 would promote the reversible cross-linking of cellulose molecules and so it might diminish the wrinkle recovery effect [8].
From the predominant parameters of the experimental results shown in the article, the cross-linking reactions of cotton fabric by CA in the presence of nano-TiO2 under UV irradiation are indicated in Schemes 1 and 2. The cross-linking mechanism of cellulose with PCAs can be followed in the presence of proton and/or electron. The cross-linking mechanism of cotton through proton attraction is more likely. This is because of the inclination of oxygen in the carbonyl group to attract protons (H+). PCA molecules absorbed on the surface of the nano-TiO2 particles are subjected to the reaction of photo reduction under UV irradiation. Therefore, as shown in Scheme 1, first, the nano-TiO2 under irradiation forms the pair of electron (e−) and hole (H+) (Equation (1)). The hole has a positive charge but PCAs are electron donors; so, the h+ activates the oxygen of the carbonyl group. This stage is represented in Equation (2). In the next stage, the nucleophilic reaction occurs. Oxygen, as a nucleophilic portion of cellulose, is attached to carbon with positive charge (Equation (3)). The next stage is elimination. Here, the produced intermediate loses one water molecule. The resulting compound has two resonance forms (Equation (4)). In the last stage, hole (h+) will separate from the product and lead to the ester linkage among carboxyl group and cellulose (Equation (5)) and the cross-linking of cellulose polymer chains is carried out. The mechanism occurring during the electron attack can be suggested (Scheme 2) the oxygen radical attack on hydroxyl groups of cellulose. This provides the nucleophilic needed for reaction with carbonyl groups of PCAs. The esterification of cellulose occurs and can be followed with other hydroxyl groups of cellulose and carbonyl groups of PCAs [27].
Mechanism of cellulose cross-linking with PCAs using nano-TiO2 photo catalyst through proton attraction. Mechanism of cellulose cross-linking with PCAs using nano-TiO2 photo catalyst through electron attacks.


Figure (3c) shows the influence of SHP and TiO2 on WRA for CA concentration of 10%. As explained above, the WRA increases with the increase in the concentration of SHP up to 10% and also with the increase in the concentration of TiO2 up to 0.1%. The concentration of SHP above 10% does not show significant improvement in WRA.
The results show that the WRA of the woven fabric is increased by increasing the concentration of CA, and SHP acts as the best catalyst at all concentrations of CA with TiO2 as the co-catalyst, whose effect maximizes till 0.1% concentration, after which it has no positive effect on the WRA.
Effect of process parameters on tensile strength
Contour plot in Figure 4 represents the relation maximum WRA versus critical process parameters.
Optimization of: (a) CA with SHP, (b) CA with TiO2, and (c) SHP with TiO2 on tensile strength.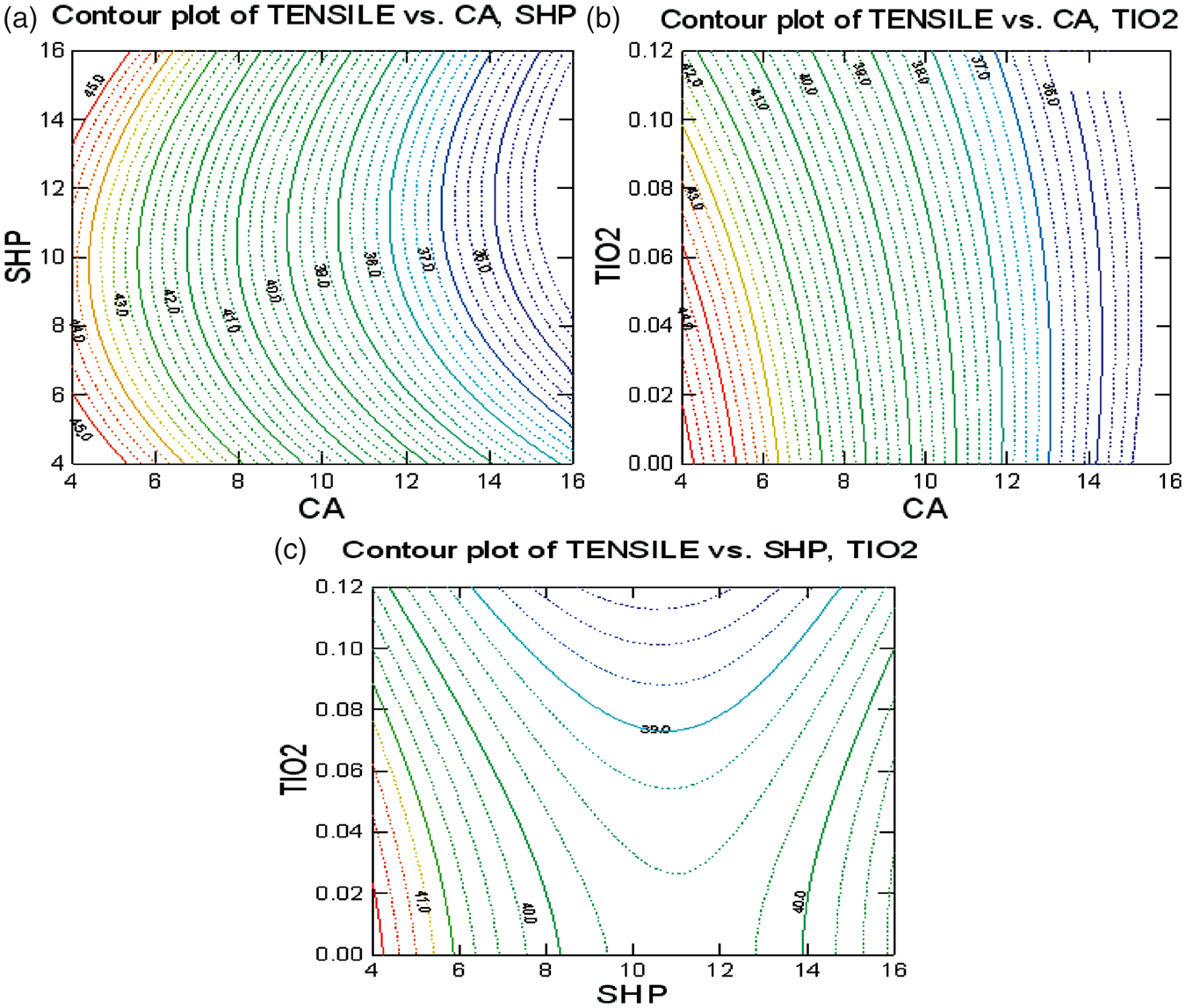
Figure (4a) shows the influence of CA and SHP on the tensile strength for TiO2 concentration 0.05%. The tensile strength of the treated fabric gradually reduces as the concentrations of CA and SHP increase up to 10%. The loss in tensile strength of cotton fabric treated with PCA is a result of the acid-catalyzed depolymerization of cellulose molecules [28].
Figure (4b) shows the influence of CA and TiO2 on the tensile strength for SHP concentration 10%. The tensile strength of the treated fabric gradually reduces as the concentrations of CA and TiO2 increase. At elevated temperatures, PCAs depolymerize cellulose. Cross-linking of cellulose molecules increases the brittleness of cotton fibers and in turn reduces the strength of the cross-linked fabrics. The magnitude of the tensile strength loss is enhanced with the increase in the degree of cross-linking. As a result, for most durable press treatments of cotton, there will be a decrease in the tensile strength of the fabric as the WRA is increased [29].
Figure (4c) shows the influence of SHP and TiO2 on tensile strength for CA concentration 10%. The increase in the concentrations of SHP and nano-TiO2 leads to a decrease in the tensile strength of the treated fabric as the catalyst and co-catalyst increased in the amount of the cross-linking agent reacting with the fabric, hence rendering more brittleness. However, the concentration of nano-TiO2 above 0.1% leads to reversible cross-linking of cellulose molecules, which diminishes the wrinkle recovery effect and hence increases the tensile strength of the fabric.
Effect of process parameters on tearing strength
Contour plot in Figure 5 represents the relation maximum tearing strength versus critical process parameters.
Optimization of: (a) CA with SHP, (b) CA with TiO2, and (c) SHP with TiO2 on tearing strength.
Figure (5a) shows the influence of CA and SHP on the tearing strength for TiO2 concentration 0.05%. The tearing strength of the treated fabric is proportionately decreased by the increase in concentration of CA. In the case of SHP, the tearing strength decreases up to 10%, which might due to the result of the increase in cross-linking of cellulose molecules, which, in turn, reduced the tearing strength of the cross-linked fabric as mentioned before.
Figure (5b) shows the influence of CA and TiO2 on the tearing strength for SHP concentration 10%. The tearing strength of the treated fabric was proportionately decreased by an increase in the concentration of CA. In case of nano-TiO2, the tearing strength decreases up to 0.06% and a slight increment in tearing strength was noticed, which may be due to presence of excess amount of TiO2 within the fibers and yarns. This might induce inter-fiber and inter-yarn frictions or make the yarns adhere more and more to one another, which would resist the yarn slippage resulting in the improvement of tearing strength compared to 0.05% concentration of nano-TiO2.
Figure (5c) shows the influence of SHP and TiO2 on the tearing strength for CA concentration 10%. The tearing strength of the treated fabric was proportionately decreased by an increase in the concentration of SHP up to 10%, which might due to the result of the increase in cross-linking of cellulose molecules. Increase of nano-TiO2 concentration beyond 0.1% enhanced the tearing strength as explained before.
Effect of process parameters on whiteness index
Contour plot in Figure 6 represents the relation maximum whiteness index versus critical process parameters. Figure (6a) shows the influence of CA and SHP on the whiteness index for TiO2 concentration 0.05%. The lower concentration of the CA gives higher whiteness index values but the increment in concentration of SHP has a very meager effect on it. However, a major drawback of CA is the yellowing problem observed on the finished fabrics. The reason is that unsaturated PCAs are produced during the curing, generating a conjugated double bonding system as a chromophore. The contribution of SHP on whiteness index is insignificant.
Optimization of: (a) CA with SHP, (b) CA with TiO2, and (c) SHP with TiO2 on whiteness index.
Figure (6b) shows the influence of CA and TiO2 on the whiteness index for SHP concentration 10%. It is apparent that lower concentration of CA gives good whiteness index as explained above. The influence of concentration of nano-TiO2 on the whiteness index is insignificant.
Figure (6c) shows the influence of SHP and TiO2 on the whiteness index for the CA concentration of 10%. The whiteness index decreases with the increase in the concentrations of SHP and nano-TiO2. The reason for the yellowness of CA-treated fabric is that unsaturated PCAs are produced during the curing, generating a conjugated double bonding system as a chromophore. The effect of SHP and nano-TiO2 on yellowing of fabric is insignificant and mainly due to the CA.
Effect of process parameters on flexural rigidity
Contour plot in Figure 7 represents the relation maximum flexural rigidity versus critical process parameters. Figure (7a) shows the influence of CA and SHP on flexural rigidity for TiO2 concentration 0.05%. The flexural rigidity (which is proportional to the bending length) of the fabric reduces with the increase in the concentrations of CA and SHP. However, there is a constant trend observed after the concentrations 10% and 8% of CA and SHP, respectively.
Optimization of: (a) CA with SHP, (b) CA with TiO2, and (c) SHP with TiO2 on flexural rigidity.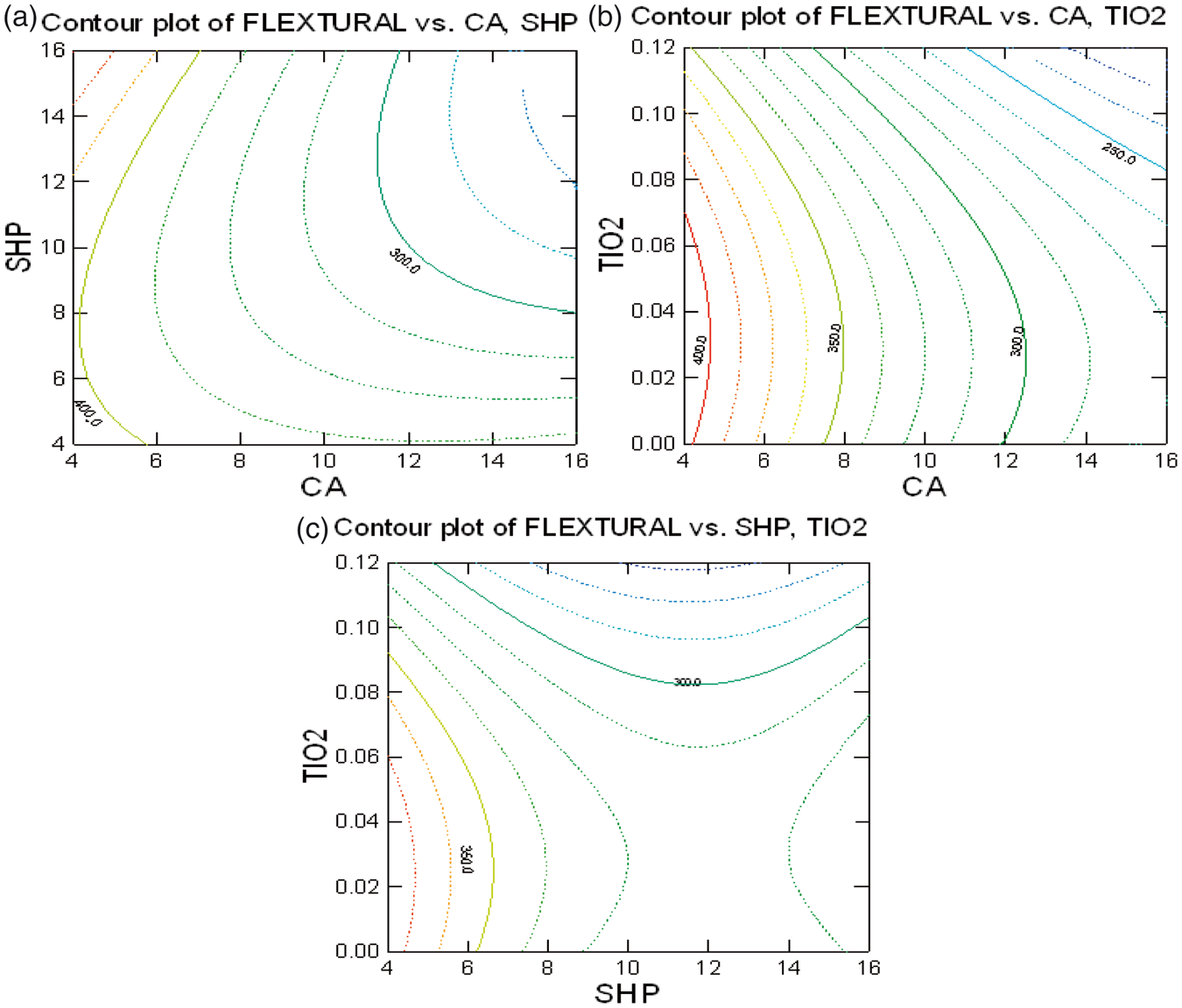
The decrease in flexural rigidity of the fabric might be due to the reduction of cross-sectional area of the cotton fibers after the CA treatment. When the treated fibers were dried during the curing step, the CA molecules present inside the fibers reacted with the cellulose surfaces. As the cross-links introduced between the hydroxyl groups were covalent bonds that held the cellulose molecules together, they would function as a swelling restraint. When the fiber cross-sectional increases and other factors remain constant, the stiffness increases [30]. The swelling restraint induced by the cross-links caused the reduction of the cross-sectional areas of the cotton fibers, which consequently led to the decrease in flexural rigidity of the fabrics after the CA treatment.
Figure (7b) shows the influence of CA and TiO2 on the flexural rigidity for SHP concentration 10%. The flexural rigidity decreases with the increase in concentration of citric as explained above. Moreover, the addition of nano-TiO2 generally decreases the flexural rigidity of the CA-treated fabrics. This might be due to the presence of nano-TiO2 between the fibers that restricted the swelling of the fibers, thereby reducing the flexural rigidity of the fabrics as mentioned before.
Figure (7c) shows the influence of SHP and TiO2 on the flexural rigidity for CA concentration 10%. The flexural rigidity decreases with the increase in the concentrations of nano-TiO2 up to 0.1%. The influence of concentration of SHP is less significant though the flexural rigidity reduces as the concentration of SHP increases mainly because it accelerates the formation of anhydrides along with the cross-linking agent.
Consolidating, we can observe that the selected parameters like concentrations of CA, SHP, and nano-TiO2 have a major influence on the wrinkle recovery property of cotton fabric. The major role of SHP as a catalyst is to accelerate the formation of anhydrides from CA, which in turn esterifies the cotton cellulose and hence provides better WRA. The influence of SHP concentration on other physical properties was found to be insignificant with respect to the concentrations of CA and nano-TiO2. The optimum regions of concentrations of CA, SHP, and nano-TiO2, considering the maximum WRA without adversely affecting the other physical properties of the treated fabric were found to be 10%, 10%, and 0.1%, respectively. However, it is possible to have an optimum level of selected variables to achieve the best possible particular fabric characteristic.
Conclusions
The study proposed a way to employ nano-TiO2 as a co-catalyst in the CA treatment aiming to enhance the wrinkle recovery performance of the cotton fabrics. It was found that the addition of nano-TiO2 particles in the treatment could enhance the wrinkle resistance of the cotton fabrics through its photocatalytic property. The addition of nano-TiO2 in the CA treatment also imparted a reduction in stiffness and softer feel to the treated fabric. The optimum concentrations of CA, SHP, and nano-TiO2 added in the system were 10%, 10%, and 0.1%. However, there was a slight decrease in the tensile and tearing strengths of cotton fabrics as a result of addition of nano-TiO2. Furthermore, a slight reduction in whiteness of the treated fabrics was observed, which is mainly due to the CA treatment.


