Abstract
Recycled poly(ethylene terephthalate) (RPET) multifilament yarns are used in carpet manufacturing as a way to reduce plastic waste. The conventional RPET carpet is however susceptible to bacterial accumulation. As a result, this research experimentally doped RPET with nano-structure titanium dioxide (nano-TiO2) to produce RPET/nano-TiO2 bicomponent multifilament yarns with antibacterial property. The experimental multifilament yarn structure consisted of two parts: neat RPET core and RPET/nano-TiO2 shell. The nano-TiO2 content in the shell was varied between 1 and 3 wt% and the core/shell (C/S) ratios between 90/10, 70/30, and 50/50 w/w. The effects of C/S ratio and nano-TiO2 content on the mechanical and antibacterial properties of bicomponent multifilament yarns were determined. The experimental results indicated that the C/S ratio had no effect on the tenacity and elongation at break. Meanwhile, the tenacity and elongation at break of bicomponent fibers increased with nano-TiO2 content in the shell. The TiO2-doped RPET bicomponent yarns effectively inhibited the growth of Escherichia coli and Staphylococcus aureus. The 90/10 bicomponent multifilament fiber with 3 wt% TiO2 achieved the highest antibacterial activity. The very high antibacterial activity was attributable to greater deposition of nano-TiO2 particles near and on the shell surface.
Keywords
Introduction
Plastic waste recycling helps reduce the burden on manufacturing resources, conserve energy, and save the environment. Poly(ethylene terephthalate) (PET) is a polyester used in a variety of applications ranging from water bottles, packaging, fibers, films to textiles. Extensive use non-biodegradable PET contributes to post-consumer plastic waste problems. As a result, many techniques have been proposed to recycle PET waste, including mechanical recycling, chemical recycling, and energy recovery.
In mechanical recycling, RPET is blended with other polymers. The advantages of mechanical recycling include ease of operation, economy, and superior mechanical properties of the blends [1]. Chemical recycling is also used to solve PET waste problems [2–4]. Chemically-treated RPET filaments could substitute virgin plastic fibers in many engineering applications [5–7].
Titanium dioxide (TiO2) is a colorless, non-toxic, thermally-stable, chemically-inert, and insoluble inorganic material. TiO2 exists in three crystalline phases: rutile which is the most stable and most abundant form, anatase (octahedrite), and brookite (orthorhombic) [8,9]. TiO2 is an inorganic filler commonly used in polymers. Meanwhile, nano-structure TiO2 is commonly used in high-performance applications, including in solar cells [8], as catalyst [10], for antibacterial effect, and photocatalytic activity [11,12]. In [13], titanate nanofibers were synthesized with natural ilmenite mineral (FeTiO3) as the starting material. The as-synthesized nanofibers possessed fiber-like morphology with 20–90 nm in diameter and high photocatalytic property. Anatase TiO2 nanotubes from natural leucoxene mineral exhibited high photocatalytic activity under UVA, UVB, and visible light [14,15].
A multifilament yarn is made up of a bundle of long ultrathin threads or filaments. There are many multifilament configurations, and the core/shell (C/S) cross-section bicomponent is the most common. In the C/S bicomponent filament, the core offers the desirable physical characteristics while the shell provides specific functionality to the filament. Furthermore, fillers could be added to the bicomponent multifilament fibers during spinning to achieve specific functionalities, e.g., conductivity, flame retardant [16].
To recycle plastic waste, this research experimentally doped RPET with nano-TiO2 to produce RPET/nano-TiO2 bicomponent multifilament yarns with antibacterial property for carpet manufacturing. Nano-TiO2 is effective in inhibiting bacterial growth under visual and ultraviolet light. The experimental multifilament yarn structure consisted of two parts: neat RPET core and RPET/nano-TiO2 shell. The nano-TiO2 content in the shell was varied between 1 and 3 wt% and the core/shell (C/S) ratios between 90/10, 70/30, and 50/50 w/w. The effects of C/S ratio and nano-TiO2 content on the mechanical and antibacterial properties of bicomponent multifilament yarns were determined.
Experimental materials and methods
Experimental materials
RPET pellets from PET plastic bottles were from Negoro Sangyo (Japan), a Thailand-based carpet manufacturer. Nano-powder titanium (IV) dioxide of grade P25 of anatase form with particle size less than 25 nm (nano-TiO2) was from Sigma-Aldrich Corporation and used as received.
RPET/nano-TiO2 master batch preparation
The master batch was prepared by compounding RPET with 10 wt% nano-TiO2 using a twin screw extruder at 260–270°C. The density of neat RPET at 25°C was 1.40 g/cm3, and that of RPET/10 wt% nano-TiO2 (master batch) was 0.85 g/cm3.
Multifilament yarn preparation
Prior to fiber processing, the master batch (RPET/10 wt% nano-TiO2) was blended with RPET pellets for RPET/nano-TiO2 with 1 and 3 wt% TiO2 (total weight 300 g); and oven-dried at 150°C for 5 h. The rationale behind first preparing the master batch and then the experimental compounds (RPET with 1 and 3 wt% nano-TiO2) was to improve the distribution of TiO2 nanoparticles in the RPET matrix. The blend was then processed by melt-spinning through 24-hole die (0.5 mm hole diameter) using a scale bicomponent extruder fed spinning machine (LBS-100, Hills, Inc., USA) (Figure 1). The core/shell (C/S) ratio of bicomponent filament was varied between 90/10, 70/30, and 50/50 (w/w). The neat RPET fiber (0 wt% nano-TiO2) was used as control. The bicomponent multifilament yarns were spooled at a winding speed of 1,000 m/min. Figure 2 illustrates the structure of core/shell multifilament fiber.

A bicomponent extruder fed spinning machine.

Core/shell multifilament fiber structure.
Tensile properties
The tensile strength of multifilament fibers was determined in accordance with ASTM D 3822-01 standard. The diameter of fibers (Denier) was measured using Fiber Fineness tester by winding nine rounds and weighing (g) with four digits. The measurement was carried out in triplicate and results averaged. The fiber diameters were determined by comparing the measured weights against the weight of 9000-meter fiber strand. The diameters were then used to calculate the tensile strength of fibers.
In the tensile strength test, the distance between C-shaped clamps was 250 mm. The tensile strength was measured using 10 samples each of the experimental bicomponent multifilament fibers and results were averaged. The samples were subjected to 50 kN load cell.
Morphology
Scanning electron microscopy (SEM) measurement was carried out using JEOL, JSM-S410LV (Japan) with 20 kV accelerating voltage. All samples were coated with an ultrathin gold layer using sputter coating system.
X-ray diffraction (XRD) analysis
The XRD analysis of bicomponent multifilament fibers was carried out using X-ray diffractometer (PW3040/60 Panalytical X’Pert Pro) with Cu Kα radiation (λ = 1.54°A) generated at 40 kV and 30 mA. The scanning was performed at 2θ of 5–80° with the scan step and time per step of 0.01° and 0.5 s.
Energy dispersive X-ray spectrometer (EDX) analysis
Energy dispersive X-ray spectrometer (EDX) analysis was performed using a scanning electron microscope operating at an accelerated voltage of 15 kV and equipped with an EDX probe (Energy dispersive X-ray spectrometer, Oxford, X-Max (IE-350)). The elemental mapping analysis (MAP) was reported to confirm the presence of nano-TiO2 in the shell surface of the bicomponent fibers.
Antibacterial tests
The antibacterial activity was determined by using JIS (Japanese Industrial Standard) Z 2801:2010 method [8]. The weight of bicomponent multifilament fiber samples was 0.3 g. Prior to the test, E. coli and S. aureus were transferred into 100 ml liquid culture and incubated at 37°C for 24 h with 150 rpm constant shaking. The fiber samples were then placed in E. coli and S. aureus liquid culture and incubated under UVA/UVB light (365 and 312 nm wavelength for UVA and UVB and 1.35 and 1.80 mW/cm2 intensity at 15 cm, respectively) at 37°C for 72 h. The antibacterial activity was determined at the start and every 24 h for 3 days (i.e., 24, 48, and 72 h) by transferring 0.1 ml liquid culture with E. coli and S. aureus onto solid feed and counting the bacterial colony.
Results and discussion
Diameter of bicomponent multifilament fibers
Figure 3 presents the cross-sectional SEM images of 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers, given 3 wt% nano-TiO2 in the shell part. Figure 4 compares the diameters of 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers, given 1 and 3 wt% nano-TiO2 in the shell. The doping of RPET with nano-TiO2 had no significant effect on the fiber diameter. Specifically, the diameters of 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers were statistically insignificant, independent of nano-TiO2 content in the shell (1 and 3 wt%), as shown in Figure 4.

Cross-sectional SEM images of bicomponent multifilament fibers, given 3 wt% nano-TiO2 in the shell: (a) 90/10, (b) 70/30, (c) 50/50 C/S ratio.

Diameters of 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers given 1 and 3 wt% nano-TiO2 in the shell part.
Morphology of bicomponent multifilament fibers
Figure 5 illustrates the surface morphology of 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers, given 1 and 3 wt% nano-TiO2 in the shell. The circles indicate the presence of TiO2 nanoparticles on or near the shell surface. In the figure, bulging TiO2 nanoparticles could be observed in the surface of the bicomponent multifilament fibers, and the TiO2 nanoparticles were more evident in the fibers containing 3 wt% nano-TiO2. This was consistent with [14,17] who reported that TiO2 particles were mostly deposited on the surface of polypropylene (PP) media. Meanwhile, nanosols containing modified TiO2 were deposited on polyester fabric by dip-coating; and chemically or plasmatically activated for antibacterial applications [18]. In practice, TiO2 nanoparticles embedded on the core/shell multifilament would stay for longer periods, vis-à-vis the dip-coating method [14]. The photocatalytic test of polypropylene (PP) meltblown fibers coated with TiO2 nanoparticles using layer-by-layer deposition technique showed decline in methylene blue reduction rate with loss of TiO2 particles from the substrate outer surface [17].

SEM images of 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers given 1 and 3 wt% nano-TiO2 in the shell.
XRD analysis of bicomponent multifilament fibers
Figure 6 shows the XRD patterns of neat RPET, 90/10 with 1 wt% nano-TiO2, 90/10, 70/30, and 50/50 (C/S) with 3 wt% nano-TiO2 bicomponent multifilament fibers. Since the XRD patterns of 90/10, 70/30, and 50/50 (C/S) fibers (with 1 wt% nano-TiO2) resembled those of bicomponent fibers containing 3 wt% nano-TiO2, this research thus showed that of 90/10 with 1 wt% nano-TiO2.
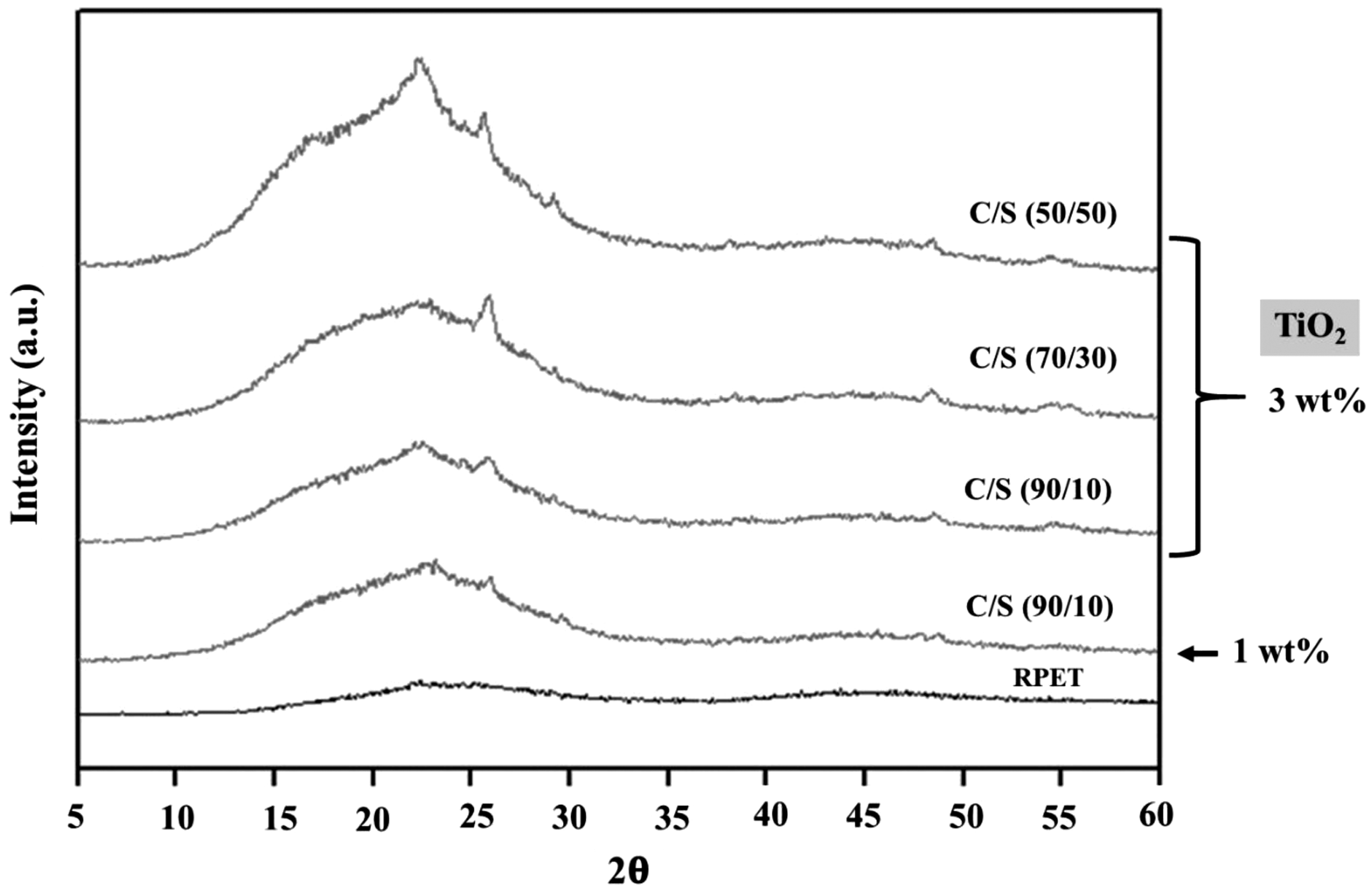
XRD analysis of neat RPET, 90/10 with 1 wt% nano-TiO2, 90/10, 70/30, and 50/50 (C/S) with 3 wt% nano-TiO2 bicomponent multifilament fibers.
In Figure 6, a broad amorphous peak was present in all samples. The XRD peaks at 2θ = 22.64° and 25.43° indicated the diffraction of crystal plane (116) and (101) of anatase crystal, while the peak of crystal plane of rutile TiO2 (110) at 2θ = 27.49° was not observed indicated that TiO2 was appeared in the anatase crystal structure [19,20]. The XRD peaks of PET crystallization (at 2θ = 16.8 and 25.2°) from nuclei position of phenylene and coplanar ester groups was not observed [21]. Therefore, neat RPET, 90/10 with 1 wt% nano-TiO2, 90/10, 70/30, and 50/50 (C/S) with 3 wt% nano-TiO2 bicomponent multifilament fibers were presented in amorphous structure [22].
EDX analysis of bicomponent multifilament fibers
Figure 7 illustrates the energy dispersive X-ray spectroscopy (EDX) analysis of neat RPET, TiO2 and 90/10 (C/S) with 3 wt% nano-TiO2 bicomponent multifilament fibers. Given the similarity between the EDX patterns of 90/10, 70/30, and 50/50 (C/S) with 1 and 3 wt% nano-TiO2 fibers, this section discussed those of and 90/10 with 3 wt% nano-TiO2 bicomponent multifilament fibers.

EDS analysis of (a) neat RPET, (b) TiO2 and (c) 90/10 with 3 wt% nano-TiO2 bicomponent multifilament fibers.
In Figure 7, the EDX pattern of RPET indicated the C and O compositions [23]. The EDX patterns of 90/10 bicomponent multifilament fibers with 3 wt% nano-TiO2 appeared in low amount. The TiO2 intensity was clearly observed in the elemental mapping analysis (MAP), confirmed the presence of nano-TiO2 in the shell surface of the bicomponent fibers (Figure 8). The MAP results implied that TiO2 nanoparticles showed a uniform and homogeneous structure [24–27].

The elemental mapping analysis (MAP) of (a) neat RPET, (b) TiO2 and (c) 90/10 with 3 wt% nano-TiO2 bicomponent multifilament fibers.
Mechanical property measurement
Figure 9 shows the tenacity of neat RPET and 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers, given 1 and 3 wt% nano-TiO2 in the shell. The tenacity of bicomponent multifilament fibers increased with increase in TiO2 in the shell as a result of the nucleating effect of nano-TiO2, resulting in increased crystallinity and enhanced tenacity of the fibers. Meanwhile, variation in the C/S ratio had minimal effect on the tenacity of bicomponent fibers. The tenacity of fibers is in agreement with the corresponding percent of crystallinity (Figures 6 and 9).

Tenacity of neat RPET and 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers, given 1 and 3 wt% nano-TiO2 in the shell.
Figure 10 compares the elongation at break of neat RPET and 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers, given 1 and 3 wt% nano-TiO2 in the shell. The elongation at break of the biocomponent fibers with 3 wt% nano-TiO2 in the shell part was higher than that containing 1 wt% nano-TiO2. Increased elongation at break could be attributed to energy-dissipating mechanism (mobility of the particles) induced by TiO2 nanoparticles in the RPET. However, given identical TiO2 content, the elongation at break decreased with increase in the shell thickness. Specifically, the elongation at break of 50/50 (thick shell) bicomponent multifilament fiber was lower than that of 90/10 (thin shell) bicomponent fiber. This could be attributed to non-uniform distribution of TiO2 as the shell thickness increased.

Elongation at break of neat RPET and 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers, given 1 and 3 wt% nano-TiO2 in the shell.
Antibacterial tests
Figure 11 shows the cumulative loss of cell viability of E. coli and S. Aureus in neat RPET (0 wt% TiO2; control) and 90/10 (C/S) bicomponent multifilament fibers after 24, 48, and 72 h, given 1 and 3 wt% TiO2 in the shell part. To initially optimize the TiO2 content in the shell, the C/S ratio remained constant at 90/10 given the highest elongation at break (Figure 10), while varying the TiO2 content. The results showed that TiO2 effectively inhibited the growth of E. coli and S. Aureus, as evidenced by increase in the loss of cell viability as time increased from 24 to 72 h. By comparison, the antibacterial activity of bicomponent fibers containing 3 wt% TiO2 were higher than that with 1 wt% TiO2. As a result, 3 wt% TiO2 was used in the shell part.

Cumulative loss of cell viability of E. coli and S. aureus (%) in neat RPET and 90/10 (C/S) bicomponent multifilament fibers, given 1 and 3 wt% nano-TiO2 in the shell.
Figure 12 illustrates the cumulative loss of cell viability of E. coli and S. aureus (%) in neat RPET (0 wt% TiO2; control), 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers after 24, 48, and 72 h, given 3 wt% TiO2 in the shell (i.e., optimal TiO2 content). The loss of cell viability of E. coli and S. aureus increased with time from 24 to 72 h. The bactericidal effect of nano-TiO2 particles resembled silver nano-particles (AgNP) whose mechanisms included: interference with cell wall synthesis; inhibition of protein synthesis; interference with nucleic acid synthesis; and inhibition of a metabolic pathway [28,29].

Cumulative loss of cell viability of E. coli and S. aureus (%) in neat RPET, 90/10, 70/30, and 50/50 (C/S) bicomponent multifilament fibers after 24, 48, and 72 h given 3 wt% nano-TiO2 in the shell
In Figure 12, the 90/10 with 3 wt% TiO2 bicomponent multifilament fiber achieved the highest cumulative antibacterial activity for both E. coli and S. aureus. The very high antibacterial activity of 90/10 bicomponent multifilament fiber (thin shell) was attributable to greater deposition of nano-TiO2 particles near and on the shell surface, compared with the 70/30 and 50/50 bicomponent fibers (thicker shell). As a result, the optimal C/S ratio and TiO2 content of the bicomponent multifilament fiber for antibacterial activity were 90/10 with 3 wt%.
Conclusion
This research doped RPET with nano-TiO2 to produce bicomponent multifilament yarns with antibacterial property. The structure of the experimental bicomponent multifilament yarns consisted of neat RPET core and RPET/nano-TiO2 shell. The nano-TiO2 content in the shell was varied between 1 and 3 wt%, and the core/shell (C/S) ratios between 90/10, 70/30, and 50/50 w/w. The results showed that the C/S ratio had no effect on the tenacity and elongation at break. Meanwhile, the tenacity and elongation at break were positively correlated with TiO2 content in the shell. The TiO2-doped RPET bicomponent fibers effectively inhibited the growth of E. coli and S. aureus. Specifically, the 90/10 bicomponent multifilament fiber with 3 wt% TiO2 (thin shell) was most effective in bacterial inhibition due to greater deposition of TiO2 nanoparticles on and near the shell surface, vis-à-vis 70/30 and 50/50 bicomponent fibers (thicker shell). Apart from recycling plastic waste, the TiO2-doped RPET bicomponent multifilament fibers, given 90/10 C/S ratio and 3 wt% nano-TiO2, possess commercial potential for antibacterial carpet manufacturing.
Footnotes
Acknowledgement
The authors would like to extend deep gratitude to the Thai Negoro Co., Ltd., for material support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to extend deep gratitude to the Research and Researcher for Industry (RRi), the Thailand Research Fund (TRF), code: MSD60I0120.
