Abstract
Purpose:
To evaluate the feasibility, technical success, and safety of ultrasound (US)-guided direct thrombolytic injection in patients with acute and early-subacute extremity arterial occlusions.
Materials and Methods:
This retrospective, single-center study included 24 patients treated between July 2023 and January 2025 for acute (≤14 days) or early-subacute (14-21 days) arterial occlusions (Rutherford I-IIb). Under real-time US guidance, alteplase (2-10 mg total) was injected into the thrombus at 2 to 3 cm intervals using a 27-gauge (40-mm) or, for deeper targets, a 22-gauge (90-mm) needle. Doppler US was performed at 2, 4, and 6 hours to assess recanalization. The primary endpoint was Doppler US-based recanalization category at 6 hours (complete/partial/none) prior to any adjunct endovascular revascularization. Secondary endpoints included adjunct angiography/endovascular procedures, complications, and follow-up primary-assisted and secondary patency at 1, 3, and 6 months.
Results:
At 6 hours, complete recanalization was observed in 12/24 (50.0%), partial in 4/24 (16.7%), and no recanalization in 8/24 (33.3%). The median alteplase dose was 6 mg (range, 2-10 mg). Diagnostic angiography was performed in 15/24 (62.5%), and adjunct endovascular procedures were performed as indicated. Minor complications occurred in 10/24 (41.7%); no major bleeding or procedure-related mortality occurred. Two patients with Rutherford IIb ischemia and symptom duration >14 days ultimately underwent major amputation after refusing further revascularization. Primary-assisted patency rates were 66.7%, 58.3%, and 41.7%, and secondary patency rates were 87.5%, 79.2%, and 75.0% at 1, 3, and 6 months, respectively. In multivariable ordinal regression, shorter time to alteplase administration was independently associated with a more favorable 6-hour Doppler recanalization category (OR 15.67; 95% CI, 1.88-130.59; p=0.011), with wide confidence intervals indicating limited precision.
Conclusions:
US-guided direct intrathrombus alteplase injection appears feasible with an acceptable safety profile and provides an early Doppler-defined response in selected patients. Prospective comparative studies are needed to define its role and refine patient selection.
Clinical Impact
Ultrasound-guided direct intrathrombus alteplase injection is a feasible, minimally invasive option for selected patients with acute and early-subacute arterial occlusions. The innovation is localized, real-time US-targeted delivery of low-dose tPA without catheter placement or prolonged infusion, combined with serial Doppler assessment to guide repeat dosing and timely escalation to angiography and adjunct endovascular therapy when indicated. For clinicians, this provides a practical workflow with close non-ICU monitoring, enabling early identification of residual thrombus, distal embolization, or underlying stenosis. The study also reinforces that shorter symptom duration strongly influences early recanalization.
Keywords
Introduction
Acute limb ischemia (ALI) is a vascular emergency that can cause irreversible tissue damage, loss of function, and amputation due to sudden arterial occlusion.1,2 It is most often caused by embolic events, thrombosis of atherosclerotic lesions, or graft/stent occlusions. Its incidence ranges from 1 to 1.5 per 10 000 person-years, with higher rates observed in elderly populations and patients with peripheral artery disease.1,3,4 Prompt diagnosis and early intervention are critical to limb salvage and survival.
Traditional treatment strategies for ALI include systemic anticoagulation, catheter-directed thrombolysis (CDT), open surgery, and endovascular revascularization. 5 While CDT remains a widely accepted approach for selected patients, its long infusion times, risk of bleeding, and intensive care unit (ICU) requirements pose significant clinical challenges. 6
Recent studies on percutaneous thrombolysis for thrombosed arteriovenous fistulas (AVFs) have demonstrated high technical success rates and favorable safety profiles.7–9 Despite its promising outcomes in the dialysis access context, the application of this technique in arterial occlusions of the limbs has not been systematically investigated.
Ultrasound (US)-guided direct intrathrombus injection of recombinant tissue plasminogen activator (tPA) is a minimally invasive approach that enables localized thrombolytic delivery without the need for prolonged infusion. This localized delivery strategy may facilitate clot lysis while limiting systemic exposure and complications. Building on these potential advantages, this study aimed to evaluate the feasibility, technical success, and safety profile of US-guided percutaneous thrombolytic injection in acute and early-subacute arterial occlusions. To our knowledge, this is the first study to evaluate this technique specifically in the management of ALI.
Materials and Methods
Patients
The local ethics committee approved this retrospective study (Decision no: 2024/334, Date: December 03, 2024), which was conducted between July 2023 and January 2025. Twenty-four patients (20 males, 4 females; mean age, 69.2 ± 10.7 years) were included. The location and length of occlusion were determined by B-mode and Doppler US or computed tomography angiography (CTA). Patients were included in the study if they had acute (≤14 days since symptom onset) or early-subacute (14-21 days) arterial occlusions and were classified as Rutherford categories I, IIa-b, indicating a salvageable limb. 10 Exclusion criteria included chronic total occlusions, Rutherford category III, which required immediate surgical revascularization, or patients with contraindications to thrombolytic therapy. Demographic data, clinical characteristics, and procedural outcomes of the patients were obtained from their medical records. Post-procedural outcomes were also documented, including primary-assisted and secondary patency rates, the number of adjunctive endovascular interventions, and any observed complications. Written informed consent was obtained from all patients before treatment.
Procedure
All procedures were performed in the interventional radiology suite under sterile conditions and local anesthesia by 3 interventional radiologists, each with ≥7 years of experience, using a standardized protocol. With the patient in the supine position, a 27-gauge, 40-mm needle attached to a 10-mL syringe was advanced percutaneously into the thrombus under real-time US guidance (Philips Affiniti 70; Samsung RS85). For distal femoropopliteal artery injections, if the subcutaneous tissue was thick or the thrombus was deeply located, a curvilinear transducer was used to improve visualization, and a 22-gauge, 90-mm needle was used to ensure adequate thrombus penetration.
Recombinant tissue plasminogen activator (alteplase, Actilyse, Boehringer-Ingelheim, Germany) was injected directly into the thrombus along its length at 2 to 3 cm intervals. Approximately 1 to 2 mg of alteplase was administered at each puncture site, with the per-puncture dose adjusted based on vessel caliber and thrombus burden, resulting in a total dose of 2 to 10 mg primarily determined by thrombus length. During injection, the needle was gently moved back and forth within the thrombus to ensure uniform distribution of tPA (Movie 1). In cases of in-stent thrombosis, punctures were made through the stent struts under real-time US guidance at points where thrombus was clearly visualized (Movie 2).
After thrombolytic injection, patients were observed in a non-ICU setting (ambulatory suite) under close clinical monitoring. All patients received therapeutic-dose subcutaneous enoxaparin sodium (1 mg/kg every 12 hours) during the post-procedure observation period to reduce the risk of re-thrombosis. Doppler US examinations were performed at 2, 4, and 6 hours and additionally if ischemic symptoms worsened or shifted distally. If Doppler US demonstrated partial recanalization with residual thrombus at 6 hours, an additional 1 to 5 mg of alteplase was injected into the remaining clot using the same technique, followed by an additional 2 to 6 hours of observation and repeat Doppler US evaluation (Figure 1). If distal embolization occurred, selective low-dose alteplase was administered directly into the distal thrombus under US guidance, with continued clinical monitoring and reassessment.
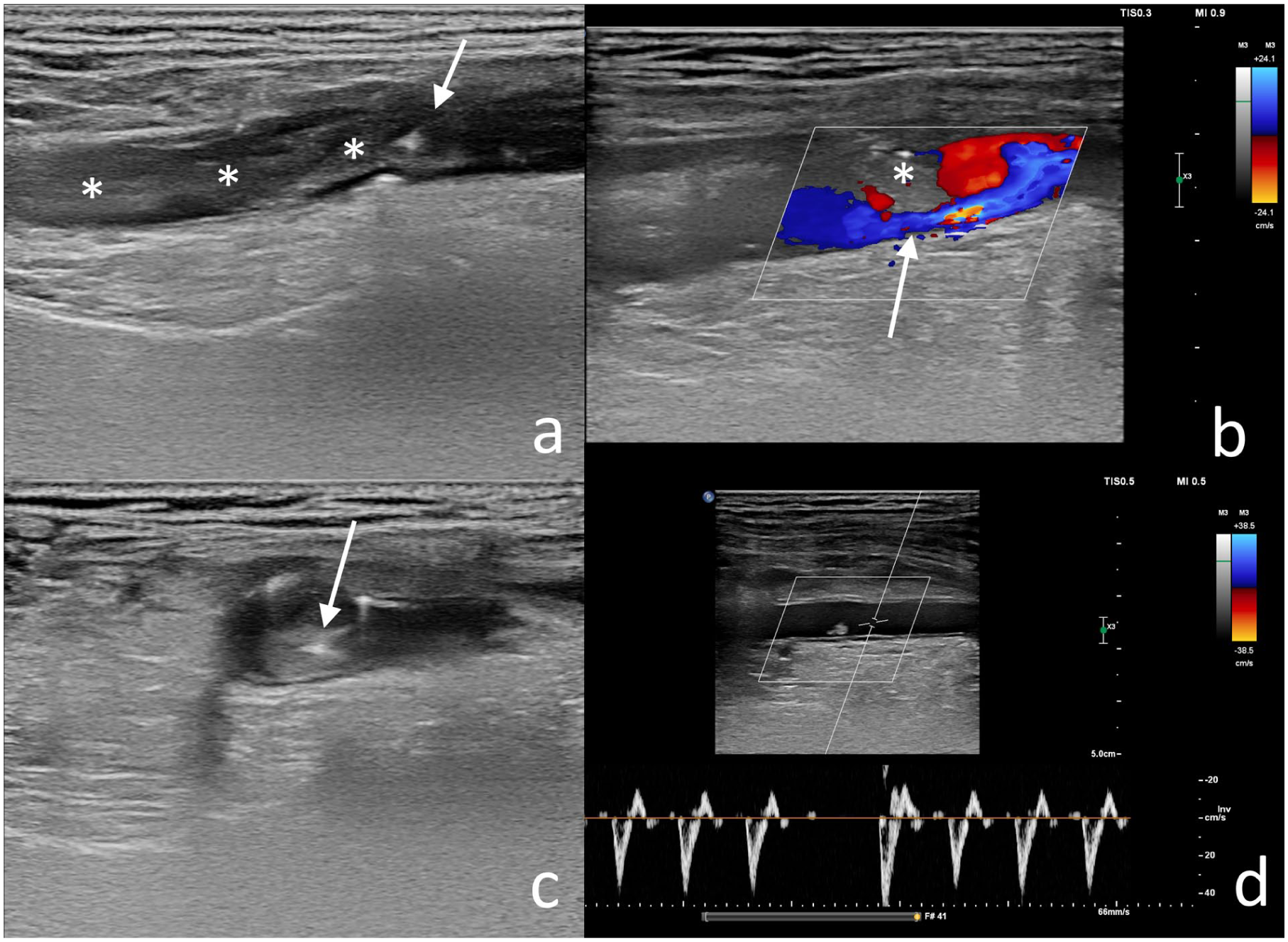
Ultrasound (US)-guided percutaneous thrombolytic injection and follow-up imaging in a patient with acute arterial occlusion. (a) Injection of tPA into an acute thrombus (asterisk) in the right popliteal artery using a 27-gauge needle (arrow). (b) Partial thrombus (asterisk) and revascularization (arrow) were observed at 2 hours of Doppler US monitoring. (c) Additional tPA was administered directly into the residual thrombus (arrow) under US guidance. (d) At the 4-hour follow-up Doppler US, complete arterial recanalization was achieved.
Patients with residual thrombus or abnormal flow on Doppler US after the 6-hour assessment underwent diagnostic angiography the following day to assess residual thrombus burden and to identify any underlying arterial pathology (e.g., atherosclerotic stenosis/occlusion) that could warrant adjunctive treatment. Based on angiographic findings, adjunct endovascular treatments (balloon angioplasty, mechanical thrombectomy, atherectomy, or stent placement) were performed when indicated.
Definitions
Technical success was defined as total recanalization or <30% residual stenosis in the treated arterial segment following percutaneous tPA injection and before any adjunct endovascular procedure. Clinical success was defined as restoring arterial flow sufficient to relieve ischemic symptoms without immediate additional intervention. Recanalization status on Doppler US was classified as complete (restoration of continuous flow with normal spectral pattern), partial (incomplete or reduced flow through residual thrombus), or no recanalization (absent flow signal within the treated segment).
The primary endpoint was Doppler US-based recanalization status at 6 hours after tPA administration, selected to capture the early hemodynamic effect prior to any adjunct endovascular revascularization. Secondary endpoints included angiographic findings, adjunct procedures, and follow-up primary-assisted and secondary patency rates. Primary-assisted patency was defined as uninterrupted vessel patency achieved after successful thrombolysis and maintained, with or without adjunct endovascular procedures performed to prevent re-occlusion. Secondary patency referred to the restoration of vessel patency after re-occlusion, requiring repeat intervention.
Complications were classified as minor or major according to the Society of Interventional Radiology reporting standard. 11 Major complications included those requiring hospitalization, additional intervention, or resulting in long-term sequelae or mortality. Minor complications were defined as those requiring no or minimal intervention and resolving without significant clinical consequences.
Follow-up
All patients were followed up with Doppler US at 1, 3, and 6 months after the procedure. During follow-up, the presence of symptoms and the need for additional intervention were assessed. Further imaging was performed in symptomatic cases with CTA or angiography.
Statistical Analysis
Continuous variables were presented as medians (interquartile range) and compared using the Mann-Whitney U test. Categorical variables were expressed as counts and percentages and analyzed using Fisher’s exact test. Ordinal logistic regression was employed to assess the association between clinical variables and occlusion status (Complete recanalization, Partial recanalization, and No recanalization) at 6 hours after tPA administration. Both univariable and multivariable analyses were conducted, with odds ratios (OR) and 95% confidence intervals (CI) reported. Variables with p<0.1 in univariable analyses were included in the multivariable model. Multivariable ordinal logistic regression was conducted to assess independent predictors of occlusion status, adjusting for clinically relevant covariates. Statistical significance was set at α<0.05. Analyses were performed using R software (version 4.4.3), and the polr function from the MASS package was used for ordinal regression analysis.
Results
A total of 24 patients (20 males, 4 females; mean age 69.2 ± 10.7 years) were included in the study. Among them, 20 patients had lower limb arterial occlusions, while 4 had upper limb arterial occlusions. The distribution of occlusion sites was as follows: superficial femoral artery (SFA) (n=10), popliteal artery (n=8), crural arteries (n=2), and upper-extremity arteries (n=4). A history of in-stent occlusion was present in 8 patients (33.3%) (Figure 2, Movie 2).
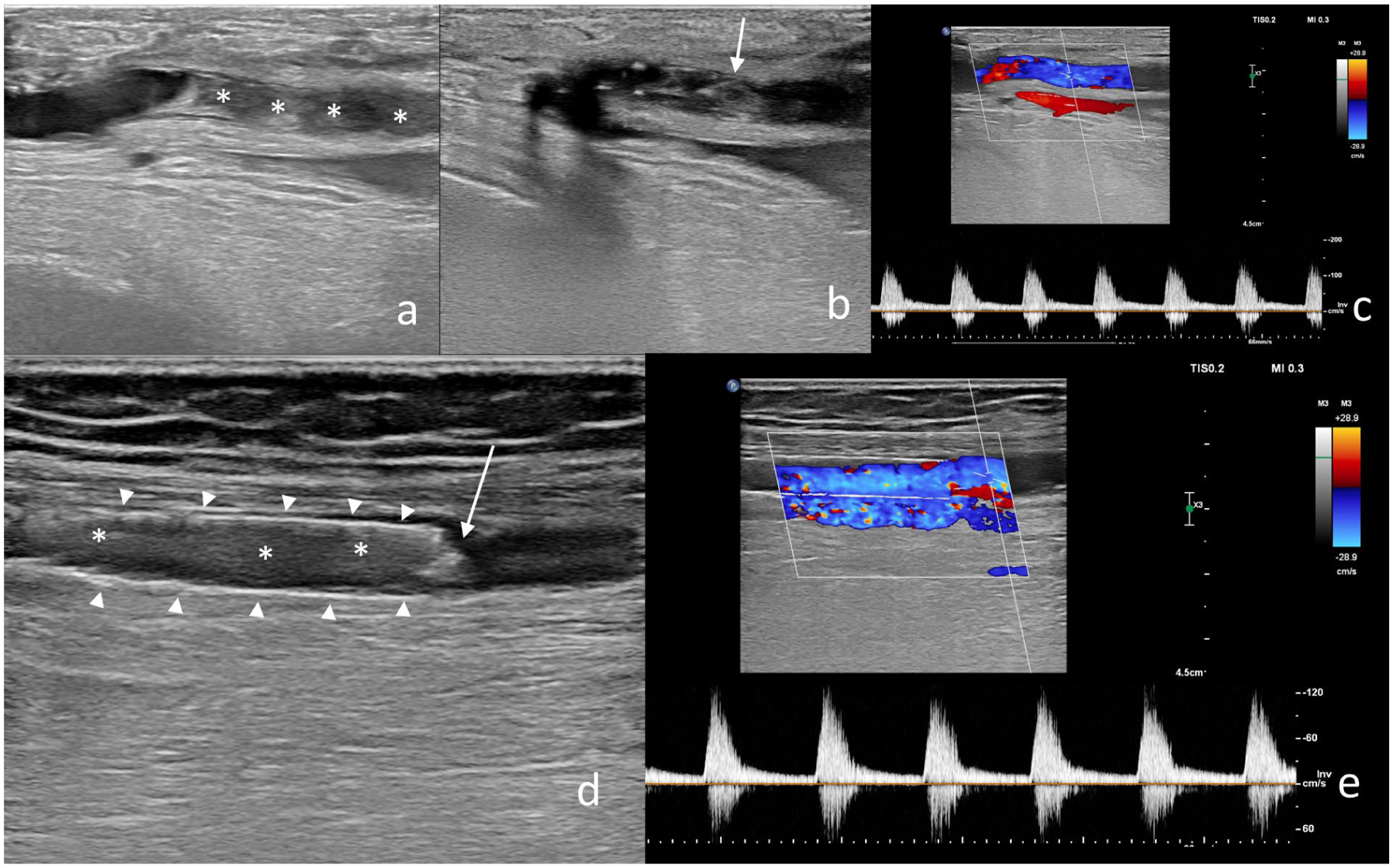
Ultrasound (US)-guided direct thrombolytic injection in an 83-year-old female patient with long-segment superficial femoral artery (SFA) occlusion involving a stented segment. (a) Gray-scale US image demonstrating an intraluminal thrombus (asterisk) at the origin of the SFA. (b, d) US-guided direct thrombolytic injection into the thrombus (arrow) and distal to the stented segment (arrowhead) of the SFA using a 27-gauge needle (arrow). (c, e) Color and spectral Doppler US images demonstrating complete arterial recanalization with restored flow at both the SFA origin and distal to the stented segment following thrombolysis.
The occlusion length ranged from 8 to 28 cm (median, 14 cm; IQR: 10-20 cm). All treated thrombi were clearly delineated under US before injection. Eleven patients were classified as Rutherford category I, 8 patients as IIa, and 5 patients as IIb. The median time from symptom onset to tPA administration was 7 days (range 1-21 days). Regarding tPA dosing, the median dose was 6 mg (range 2-10 mg).
Recanalization status was evaluated at 2, 4, and 6 hours following tPA administration (Figure 3). At 2 hours, 3 patients (12.5%) demonstrated complete recanalization, 9 (37.5%) had partial recanalization, and 12 (50%) showed no recanalization. By 4 hours, complete recanalization was achieved in 5 (20.8%) patients, partial in 10 (41.7%), and no recanalization in 9 (37.5%). At 6 hours, 12 patients (50%) had complete recanalization, 4 (16.7%) had partial, and 8 (33.3%) showed no recanalization.
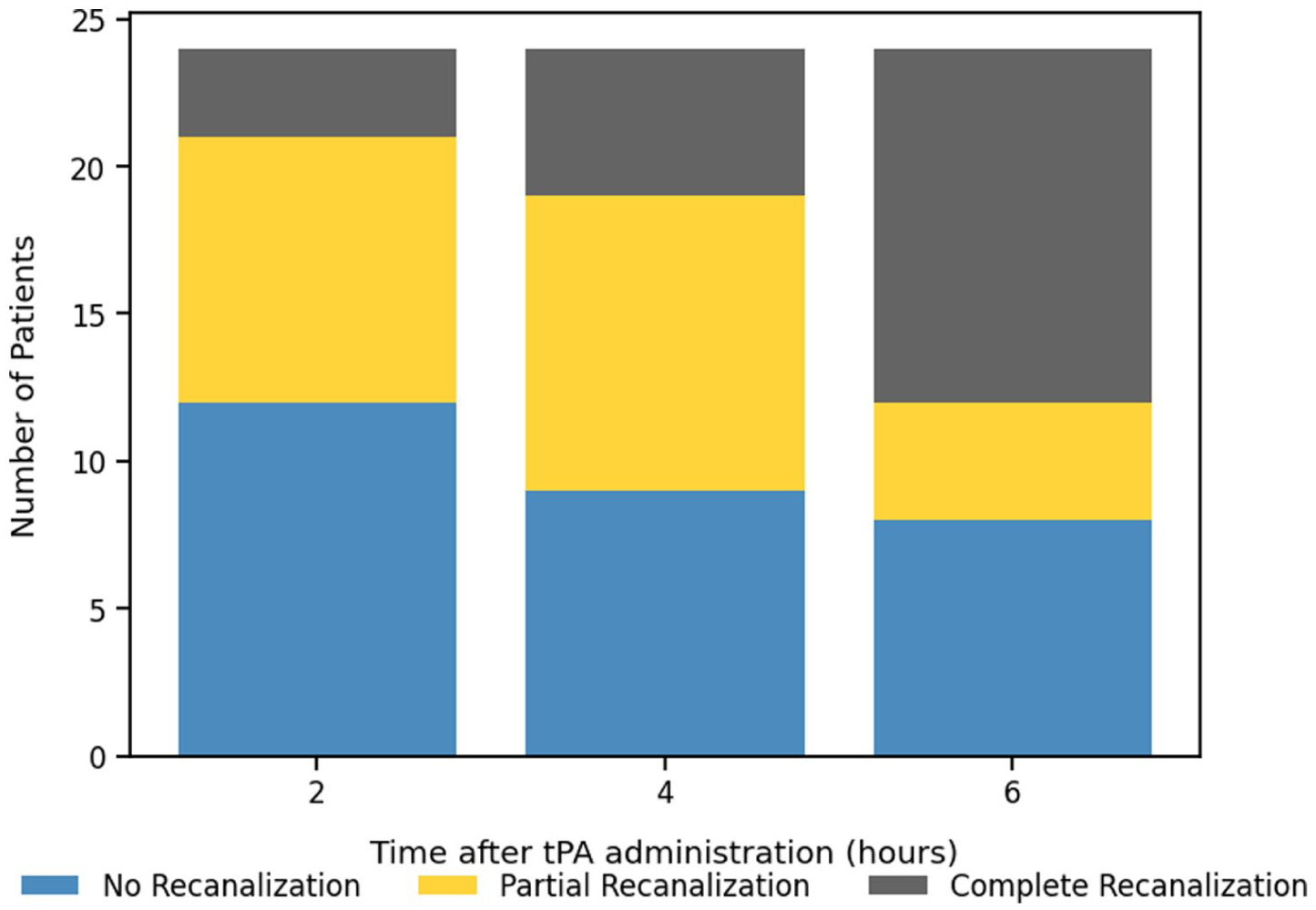
Recanalization status over time after tPA administration.
Diagnostic angiography was performed in 15 patients (62.5%) after thrombolysis. Residual thrombus was detected in 6 patients, underlying chronic atherosclerotic occlusive disease in 4, and focal stenosis in 5. Additional endovascular procedures were performed, including balloon angioplasty (n=9), mechanical thrombectomy (n=4), stent implantation (n=2), and atherectomy (n=2). After adjunct endovascular treatment, complete patency on angiography was achieved in 12 of the 15 patients (80%).
Minor complications were noted in 10 patients, including localized pain (n=5), ecchymosis (n=4) (Figure 4), and groin hematoma (n=1). No major bleeding or procedure-related mortality occurred. Two patients with Rutherford category IIb ischemia with symptom duration exceeding 14 days eventually underwent major amputation. Both patients had refused diagnostic angiography and surgical revascularization, did not respond to percutaneous tPA injection, and showed no recanalization on follow-up Doppler US. Additionally, 2 patients developed acute hand pain approximately 4 hours after injection, consistent with distal embolization on Doppler US; both were treated with targeted additional low-dose tPA injection, resulting in complete symptom resolution and restoration of flow.
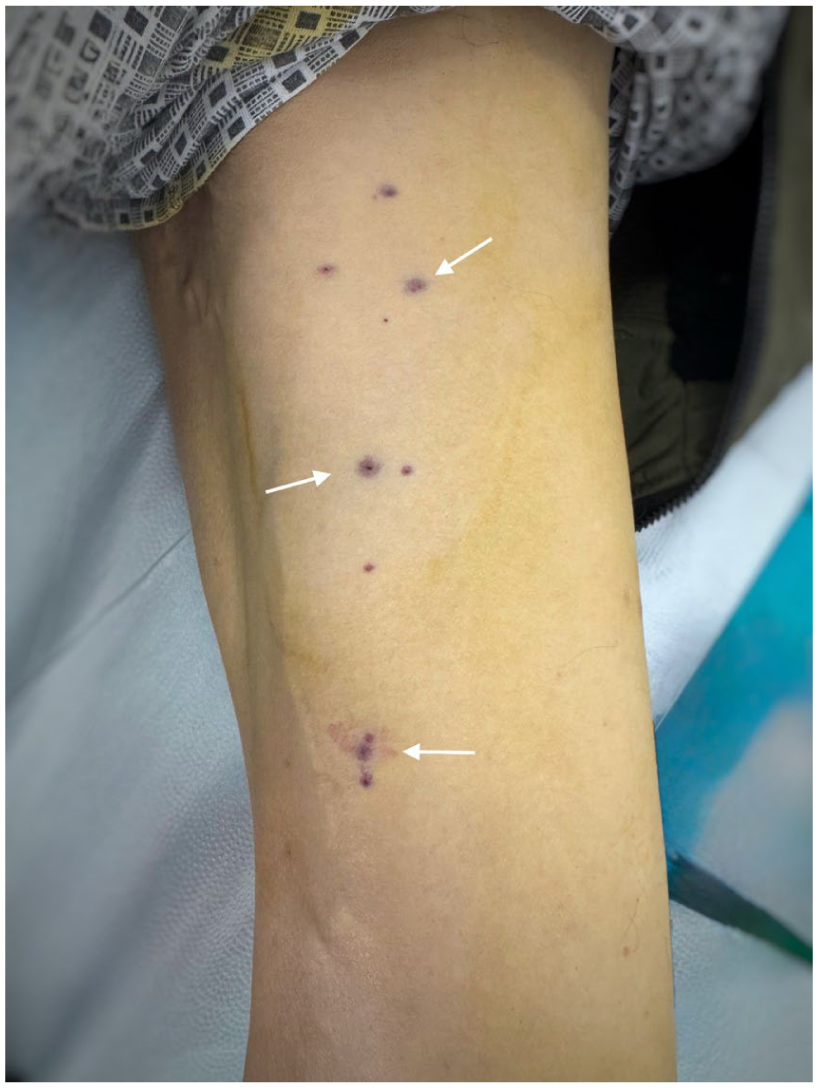
Minor access-related complication. Clinical image showing localized ecchymosis at puncture sites (arrows), after percutaneous intrathrombus injection, managed conservatively without additional intervention.
All patients were followed up with Doppler US at 1, 3, and 6 months after the procedure. Primary-assisted patency rates were 66.7%, 58.3%, and 41.7%, while secondary patency rates were 87.5%, 79.2%, and 75.0% at the same intervals, respectively (Figure 5).
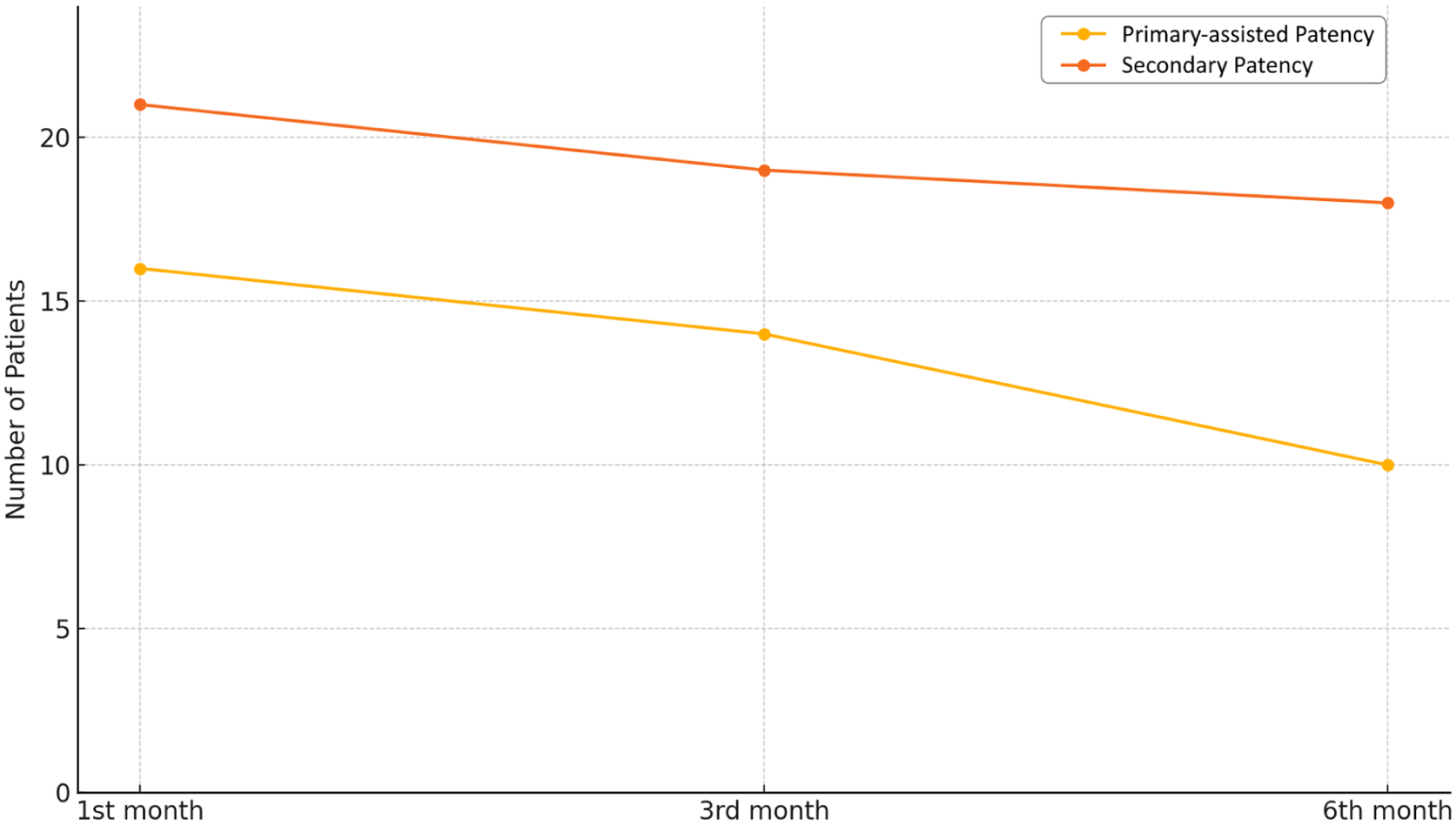
Primary-assisted and secondary patency rates over a 6-month follow-up.
Thrombus age demonstrated a strong association with Doppler recanalization category at 2, 4, and 6 hours (p<0.05) (Table 1). At 6 hours, all patients with no recanalization (8/8) had received tPA more than 14 days after symptom onset, whereas 75% (9/12) of patients with complete recanalization were treated within 1 to 6 days (p<0.001). Diabetes mellitus was also significantly associated with occlusion severity at 6 hours (p=0.025), as 50% (4/8) of the no-recanalization group and 75% (3/4) of the partial-recanalization group were diabetic, compared to only 8.3% (1/12) in the complete-recanalization group.
Baseline Demographic and Clinical Characteristics Across Recanalization Groups at 2, 4, and 6 Hours After tPA Administration.
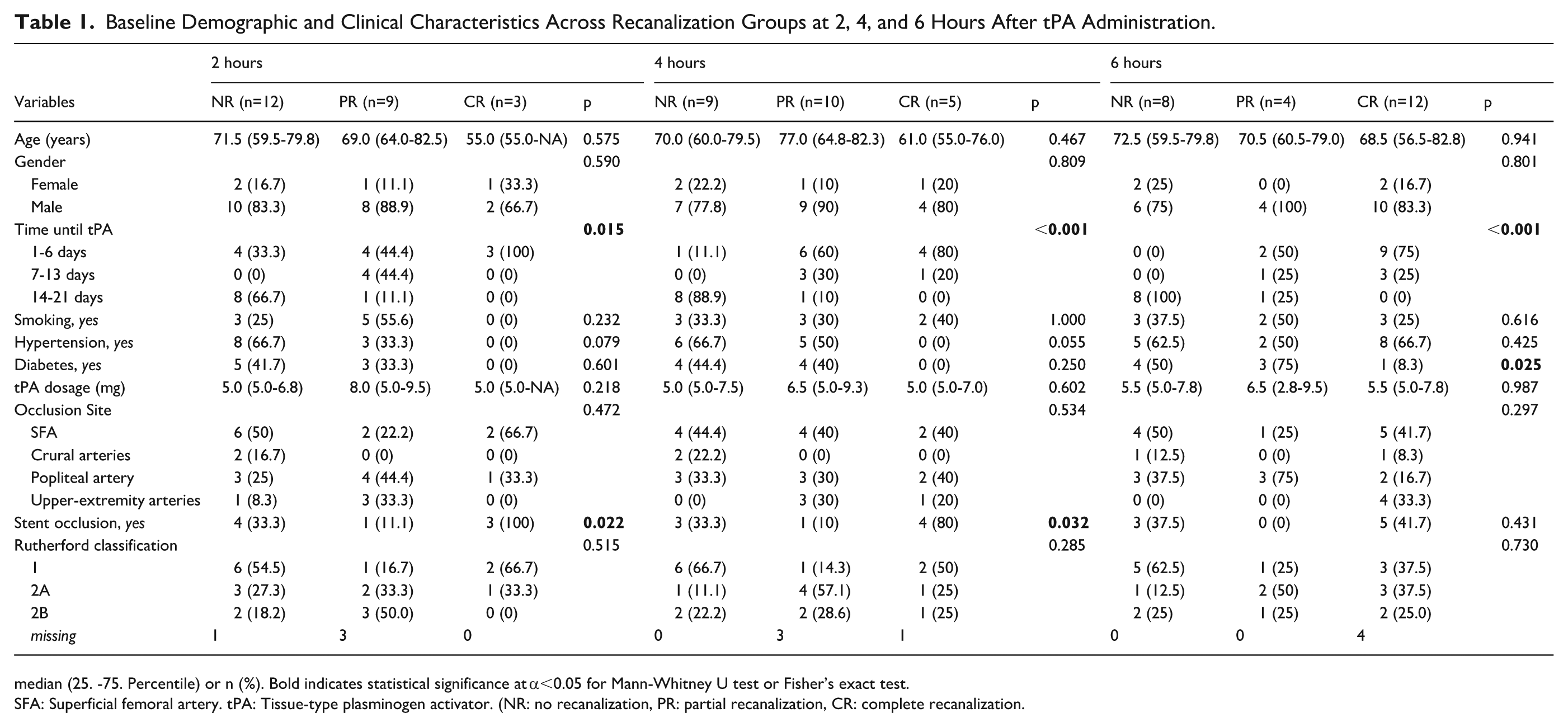
median (25. -75. Percentile) or n (%). Bold indicates statistical significance at α<0.05 for Mann-Whitney U test or Fisher’s exact test.
SFA: Superficial femoral artery. tPA: Tissue-type plasminogen activator. (NR: no recanalization, PR: partial recanalization, CR: complete recanalization.
On univariable analysis, delayed tPA administration (≥7 days) was associated with a significantly higher risk of failed recanalization (OR, 21.55; 95% CI, 2.90-160.18; p=0.003) (Table 2). Diabetes mellitus was also identified as a significant factor (OR, 5.90; 95% CI, 1.10-31.53; p=0.038). In the multivariable model, time to tPA administration remained independently associated with the 6-hour Doppler recanalization category (OR, 15.67; 95% CI, 1.88-130.59; p=0.011), whereas diabetes lost statistical significance. No other variables demonstrated significant associations. Wide CI indicate limited precision of the effect estimates, likely reflecting the small sample size and sparse data in some strata.
Univariable and multivariable ordinal logistic regression for predictors of recanalization status at 6 hours after tPA administration.
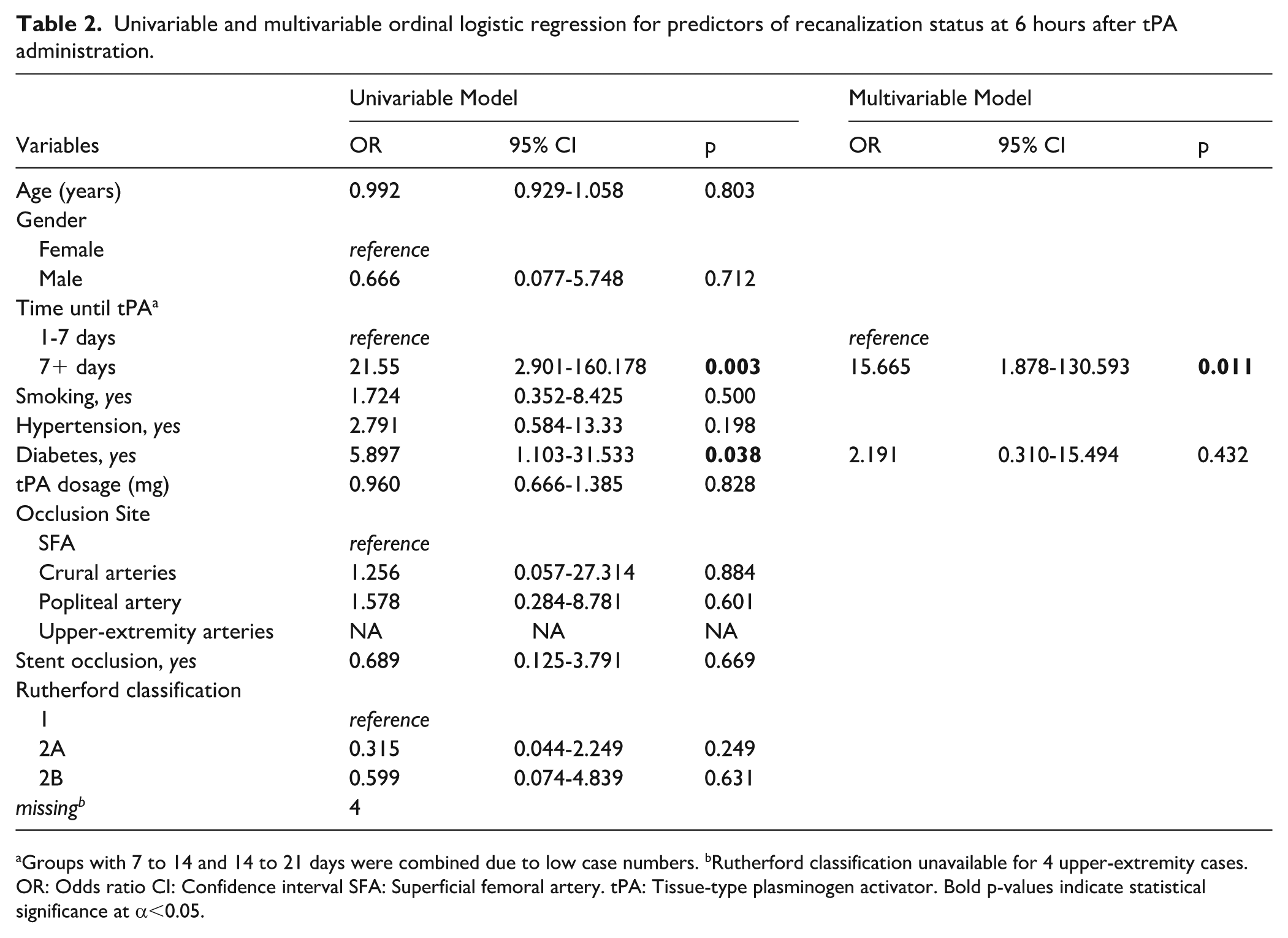
Groups with 7 to 14 and 14 to 21 days were combined due to low case numbers. bRutherford classification unavailable for 4 upper-extremity cases. OR: Odds ratio CI: Confidence interval SFA: Superficial femoral artery. tPA: Tissue-type plasminogen activator. Bold p-values indicate statistical significance at α<0.05.
Discussion
Percutaneous thrombolytic injection has previously been described in the treatment of thrombosed arteriovenous fistulas (AVFs), demonstrating high technical success and favorable safety profiles.12–15 For instance, Durmaz and Birgi 9 reported a 100% technical success and 94.1% clinical success rate using US-guided percutaneous thrombolytic injection with 5 to 10 mg of tPA in thrombosed native hemodialysis fistulas. The Direct Percutaneous Thrombolysis (DPT) technique, as described by Prasad et al., 8 involves direct puncture of the thrombosed segment under US guidance and localized delivery of the thrombolytic agent, achieving approximately 84% procedural success in thrombosed AVFs without underlying stenosis. These AVF studies support the broader concept that direct, US-guided intrathrombus delivery can enable localized thrombolysis with limited systemic exposure.
Traditionally, CDT, mechanical thrombectomy, and surgical revascularization have been used in the treatment of ALI. 10 Large randomized studies, such as the TOPAS, STILE, and Rochester trials, reported recanalization rates of 65% to 82%, but also major bleeding in 8.8% to 12.5% of patients and the need for intensive monitoring.16–18 The systematic review by Morrison 19 and Güneş et al. 20 highlighted that CDT often requires 12 to 36 hours of infusion, catheter manipulation under fluoroscopy, and is associated with access-site and systemic bleeding complications. Theodoridis et al. 21 further reported that up to 28.7% of CDT-treated patients experience complications, 79.9% of which are major and potentially life-threatening. In our cohort, we used a single-session, US-guided percutaneous tPA injection without catheter placement. All procedures were performed in a non-ICU setting (ambulatory suite) under close clinical monitoring, and no major bleeding or mortality occurred. Minor complications such as subcutaneous pain or ecchymosis were relatively common, likely related to multiple puncture sites, but all were self-limited and required no additional intervention.
Although no major bleeding occurred in our cohort, safety findings should be interpreted cautiously, given the small sample size and single-center design. Prior reports of direct percutaneous thrombolytic injection largely involve thrombosed AVFs, where puncture sites are typically superficial and bleeding—if present—can often be controlled with local measures. In contrast, application to distal femoropopliteal arteries may involve a different risk profile, as bleeding within deeper soft tissues may be more challenging to control with external compression alone and may warrant prompt imaging and angiographic evaluation if hemostasis is not readily achieved. In our protocol, bleeding risk was mitigated by using small-gauge needles (27-gauge for most targets; 22-gauge for deeper targets when required) and real-time US guidance to maintain intrathrombus needle-tip visualization. We also note that the surrounding soft tissue may provide a degree of tamponade for small-caliber punctures; however, clinically significant hemorrhage remains a theoretical concern in deeper arterial targets.
Distal embolization was observed in 2 patients with upper-extremity occlusions approximately 4 hours after tPA administration, manifested by worsening hand pain during post-procedure monitoring. This event likely reflects thrombus fragmentation during lysis, with migration of clot material into the distal arterial bed when runoff is initially patent—a recognized risk of thrombolytic therapy, including CDT. Therefore, we inform patients before the procedure that distal ischemic symptoms may worsen as lysis progresses, and we maintain close clinical monitoring with prompt Doppler US reassessment if symptoms change. In these 2 upper-extremity cases, Doppler US localized new thrombus at the radial artery near the wrist, and selective additional low-dose tPA was administered directly into the distal thrombus under US guidance, resulting in complete symptom resolution and restoration of patency on follow-up Doppler US. Although based on a small number of events, this experience suggests that when distal embolization occurs in a patent distal vascular bed, timely symptom-triggered imaging and targeted distal thrombolysis may be an effective management strategy in selected patients.
From a technical perspective, US-guided direct thrombolytic injection enables targeted drug delivery and real-time assessment of thrombus dissolution and flow restoration, facilitating additional dosing or escalation to adjunct endovascular intervention when indicated. Adequate intrathrombus access was achieved using a 27-gauge (40-mm) needle for most targets and a 22-gauge (90-mm) needle for deeper lesions, without the need for catheter placement for thrombolytic infusion. When incomplete patency or abnormal flow persisted, early angiographic evaluation allowed timely transition to adjunctive procedures (e.g., balloon angioplasty, stenting, mechanical thrombectomy, or atherectomy). Our protocol used a low total alteplase dose (mean 6.8 mg), consistent with prior DPT reports (5-10 mg) and lower than most CDT regimens,22–24 which may help limit systemic exposure.
Based on our experience, feasibility is highest in arterial segments that are readily accessible to high-frequency linear US, including upper-extremity arteries, the proximal thigh, the popliteal fossa, and distal crural segments. Conversely, deeper distal femoropopliteal segments—particularly in patients with increased subcutaneous adiposity—may require a curvilinear transducer and a longer needle, which can increase technical complexity compared with more superficial targets. Marked arterial calcification with acoustic shadowing may limit thrombus and needle-tip visualization and can restrict applicability; however, we did not encounter cases in which shadowing fully precluded treatment in our cohort. Occlusion length is also relevant because multiple punctures are required at 2 to 3 cm intervals; longer lesions may increase the number of punctures and procedure time.
This technique may be considered a complementary strategy for carefully selected patients with ALI, potentially serving as a bridging approach alongside established treatments. In practice, our findings suggest that the most suitable candidates are those with salvageable limb ischemia, particularly Rutherford category I and IIa, shorter symptom duration, and thrombus-dominant lesions with clear US visualization. Conversely, this approach should be avoided in cases requiring immediate revascularization (e.g., Rutherford category IIb with motor deficit), in the presence of organized thrombus, or when adequate US visualization and a safe percutaneous access route cannot be established.
In our descriptive analyses, anatomic territory/occlusion site did not show a consistent association with recanalization at 2, 4, or 6 hours, and the early association observed with in-stent thrombosis at 2 and 4 hours was not sustained at the 6-hour endpoint nor in multivariable modeling (Tables 1, 2). Consistently, administering tPA within 6 days of symptom onset was associated with a higher likelihood of 6-hour Doppler-defined recanalization, whereas treatment at ≥14 days was associated with persistent occlusion. Ordinal logistic regression identified time to tPA as the strongest independent predictor of the 6-hour Doppler recanalization category. However, CI was wide, indicating limited precision, which may reflect the small sample size and sparse data in some strata. Nevertheless, these findings align with prior studies reporting improved thrombolytic efficacy with earlier intervention. For instance, Moon et al. 25 reported that early thrombolysis (<7 days) was superior to late thrombolysis (7-14 days). Similarly, Palfreyman et al. 26 emphasized that delayed thrombolysis (≥14 days) significantly reduces the likelihood of complete revascularization.
This study has several limitations. First, its retrospective, single-center nature, small sample size, and absence of a control group restrict the ability to draw definitive comparisons with CDT or surgery. Second, the inclusion of both acute and early-subacute occlusions may have introduced heterogeneity, as thrombus age/organization and underlying pathophysiology can influence thrombolytic response; therefore, subgroup findings should be considered exploratory/descriptive and are not powered for formal hypothesis testing. Third, as US-guided injection procedures are operator-dependent, the observed outcomes may not be directly generalizable to other practice settings. Inter-operator variability and learning-curve effects were not assessed. Despite these limitations, the technique appeared feasible with an acceptable safety profile in our cohort.
Conclusions
The US-guided direct thrombolytic injection is a feasible, minimally invasive option for selected patients with ALI, with an acceptable safety profile and an early hemodynamic response. Early tPA administration was associated with more favorable recanalization, highlighting the importance of timely intervention. Prospective comparative studies are needed to define the role of this approach compared with CDT and to refine patient selection.
Supplemental Material
sj-docx-1-jet-10.1177_15266028261429915 – Supplemental material for Ultrasound-Guided Direct Thrombolytic Injection for Acute and Early-Subacute Arterial Occlusions: Feasibility and Early Outcomes
Supplemental material, sj-docx-1-jet-10.1177_15266028261429915 for Ultrasound-Guided Direct Thrombolytic Injection for Acute and Early-Subacute Arterial Occlusions: Feasibility and Early Outcomes by Samet Genez, Hamza Özer, Yunus Yılmazsoy, Oya Kalaycıoğlu, Onur Taydaş and Ömer Faruk Ateş in Journal of Endovascular Therapy
Footnotes
Acknowledgements
The authors have no acknowledgments to declare.
Ethical Considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required. This study was approved by the Institutional Review Board of Bolu Abant Izzet Baysal University Non-Interventional Clinical Research Ethics Committee Approval (decision no: 2024/334).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
