Abstract
Purpose:
Open aortic arch repair is the gold standard in the treatment of diseases involving the ascending aorta and aortic arch. However, due to the invasive nature of open repair, high-risk patients with multiple comorbidities are often not suitable candidates for open surgical repair. While endovascular aortic repair is far less invasive, endovascular arch repair remains a difficult challenge due to the aortic arch diameter and angulation, origin of the supra-aortic arteries, and the lack of commercially available thoracic branched devices in the United States.
Case Report:
Here we describe palliation of a mycotic aortic arch pseudoaneurysm with a physician-modified endograft and in situ laser fenestration. Our technique allowed for rapid repair of the pseudoaneurysm with minimal physiologic disturbances and no perioperative complications in a high-risk surgical patient.
Conclusion:
Physician-modified endografts are feasible and may be an effective treatment option for palliation of acute aortic arch lesions in high-risk surgical patients.
Keywords
Introduction
Open surgical repair is the gold standard in the treatment of diseases involving the ascending aorta and aortic arch. However, conventional surgical techniques for open surgery are highly invasive, involving sternotomy, cardiopulmonary bypass, and hypothermic circulatory arrest. Therefore, patients with multiple comorbidities are often deemed unfit for surgery and denied open repair. In contrast, hybrid and endovascular repair techniques allow surgeons to treat arch pathology in a less-invasive way. For pathologies of the descending aorta up to the distal arch, thoracic endovascular aneurysm repair (TEVAR) has prevailed as the treatment of choice. 1 However, endovascular repair of the aortic arch remains a challenge as the aortic diameter, angulation, and origin of the supra-aortic arteries pose anatomical difficulties for the deployment of endografts. 2
The most common minimally invasive approach in high-risk patients with aortic arch disease is a hybrid procedure, consisting of supra-aortic debranching and revascularization followed by stent-graft deployment. 3 Alternatively, endovascular repair using chimney or parallel endografts, in situ fenestrations, or custom-made fenestrated and branched stent grafts have all been proposed as additional options to achieve endovascular solutions in high-risk patients. Here, we describe the endovascular repair of a mycotic arch pseudoaneurysm with a physician-modified stent graft and in situ laser fenestration and propose physician-modified endografts as a feasible and effective treatment option for aortic arch lesions in high-risk surgical patients.
Case Report
A 73-year-old female with dementia, hypertension, diabetes mellitus, severe deconditioning, and protein calorie malnutrition presented as a transfer from her nursing home with blood cultures positive for gram-positive cocci. One year prior, she had suffered a vertebral fracture complicated by infection and long-term methicillin-resistant staphylococcus aureus (MRSA) lumbar discitis, treated with long-term intravenous (IV) daptomycin and surgical fixation. As part of her infectious workup, she underwent a chest radiograph that demonstrated a widened mediastinum with rightward mediastinal shift. Subsequent computed tomography of the chest revealed a large pseudoaneurysm extending from the lesser curve of the aortic arch on the left and measuring 3.4 × 2.3 × 2.5 cm (Figure 1), associated with a pericardial effusion, bilateral pleural effusions, and early echocardiographic evidence of tamponade. She was dyspneic at rest, afebrile, and hemodynamically stable. She was started on esmolol for impulse control. Given the patient’s significant comorbidity burden, limited rehab potential, and limited life expectancy due to dementia, deconditioning, and long-term discitis, she was considered at prohibitive risk for open surgical repair. The patient and her daughter both indicated that cardiopulmonary resuscitation and prolonged intubation were outside of her goals of care, but that she wanted relief of her dyspnea. Using an interdisciplinary approach with representatives from palliative care, vascular surgery, and cardiothoracic surgery, a detailed conversation was had with the patient and her family regarding the best therapeutic option. On one hand, the patient’s frailty and limited life expectancy predisposed her for postoperative complications and endograft infection. On the other hand, without intervention, the patient was at risk for rupture and fulminant sepsis. Furthermore, the patient primarily wanted relief of her dyspnea, which was thought to be related to both her pericardial effusion and arch aneurysm. It was felt her dyspnea would not resolve with non-operative management alone, and we feared early drainage of the pericardial fluid before addressing the arch aneurysm could have predisposed her to aneurysm rupture. We therefore offered physician-modified TEVAR with large-vessel fenestration followed by creation of a pericardial window as a palliative measure for her mycotic pseudoaneurysm and effusion.
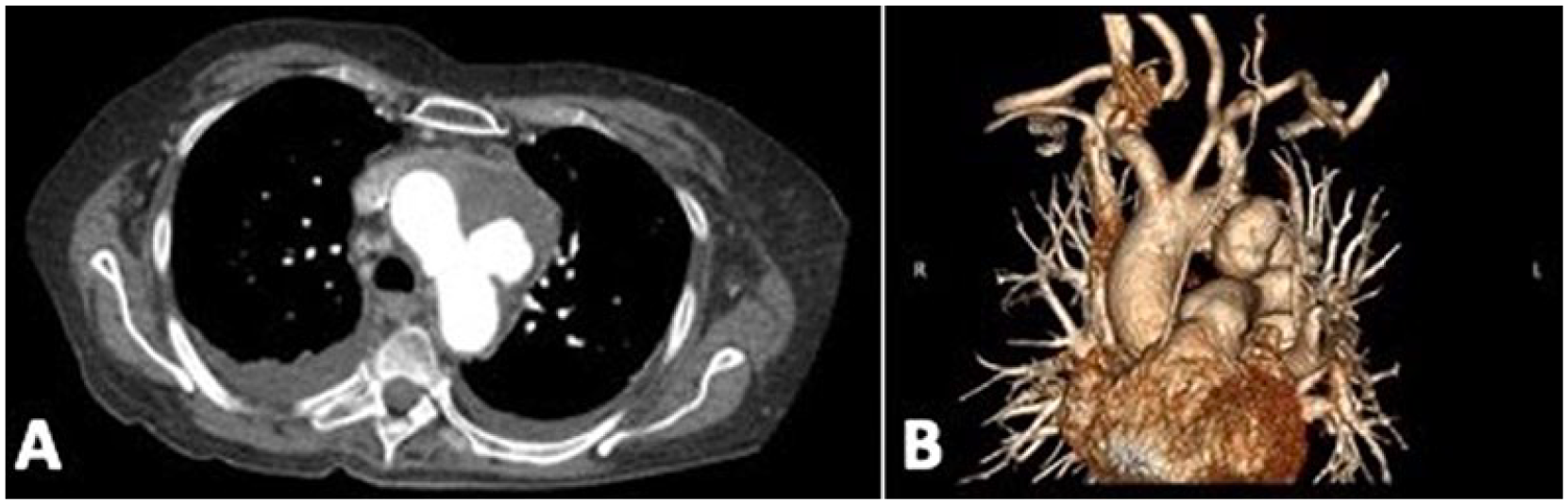
Preoperative axial (A) and volume rendered (B) computed tomography (CT) shows a 3.4 × 2.3 × 2.5 cm pseudoaneurysm extending from the lesser curve of the aortic arch.
While general anesthesia was being induced, a 34 × 28 mm Cook Alpha thoracic stent graft was deployed on the back table (Figure 2). A fine blade and cautery were used to create a large “island technique” fenestration approximately 2 × 2 cm on the greater curvature of the device for revascularization of the innominate artery and left common carotid artery. This was reinforced with a radiopaque soft 0.014 wire and secured with a running 4-0 braided polyethylene suture. A 0.014 guidewire was used to precannulate the graft. The device was then restrained back into its original sheath.

Intraoperative photograph of the physician-modified endograft.
The primary access was obtained through the right common femoral artery. Additional bilateral radial access was gained for hemodynamic monitoring, through and through access, and in situ laser fenestration of the left subclavian artery. The patient was then heparinized, and the activated clotting time was monitored for a target of >350 seconds. The femoral access was preclosed with 2 PerClose devices in the usual fashion, and an 8-Fr sheath was placed. Rapid pacing via transfemoral temporary pacing wires was performed.
A 0.035” intravascular Philips ultrasound probe was navigated from the femoral artery to the aortic root. A double-curved Lunderquist wire was placed. The arch vessels were marked and the aortic dimensions were measured at zone 0 and zone 4. The 8-Fr sheath was exchanged for an 18-Fr hemostatic sheath.
A pigtail catheter was placed via the left radial access into the ascending aorta. A support catheter was used to pass a stiff hydrophilic wire for through-and-through access from the right radial artery access to the femoral sheath. The catheter tip was left externalized and the wire was removed.
A flush aortogram was then performed to confirm the location of the arch vessels. The large-bore sheath was removed, and the physician-modified graft with a prewire fenestration was loaded onto the double-curved Lunderquist wire. The fenestration wire was passed through the support catheter and then externalized out the radial artery sheath. 4 The catheter was removed. The fenestrated device was then advanced under fluoroscopic guidance, making sure to orient the fenestration appropriately on the greater curvature. Once the device was in position, an additional flush aortogram was performed, and the device was deployed, spanning from zone 0 to zone 4. There was no change in cerebral oximetry or right radial artery pressure. The delivery system was removed and a large-bore sheath replaced.
The pigtail was removed back into the left subclavian artery, and this was exchanged for a laser catheter. The laser catheter was then passed up the left subclavian artery until it was abutting the origin of the left subclavian artery. The laser was then activated, and a hydrophilic wire was passed through the in situ fenestration and then externalized from the femoral sheath. Intravascular ultrasound (IVUS)-assisted the fenestration procedure. Over this wire, a 4-mm balloon was passed and inflated to dilate the fenestration. A 7 × 29 mm VBX stent was then deployed into the left subclavian artery. Completion angiography revealed excellent filling of all arch vessels with no filling of the aortic pseudoaneurysm (Figure 3A). All access sheaths were removed and hemostasis was obtained.
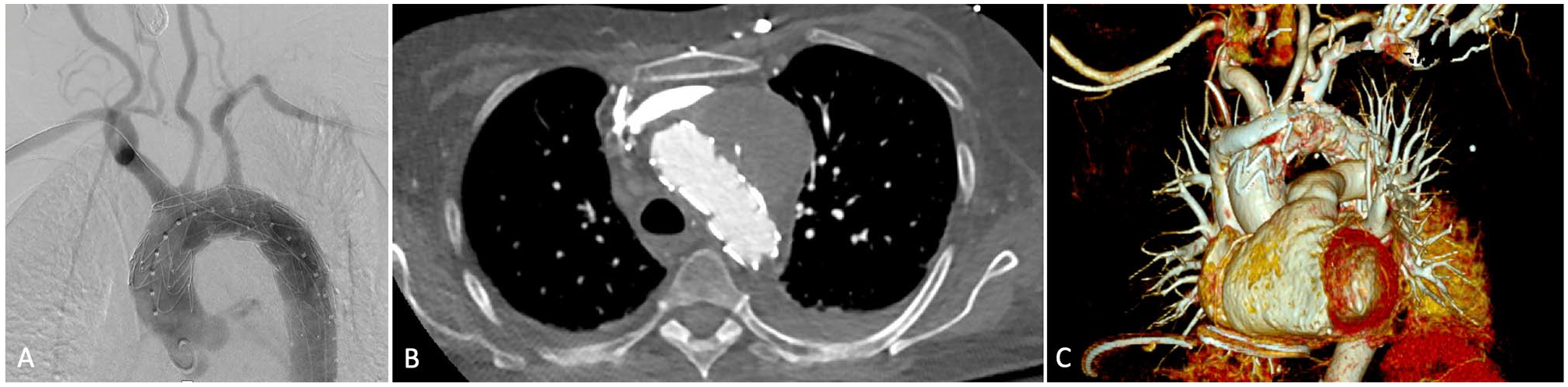
Completion angiography (A) shows resolution of the pseudoaneurysm and filling of all arch vessels. Confirmed by axial- (B) and volume-rendered (C) computed tomography (CT).
Following this, a subxiphoid pericardial window was created. Purulent fluid was suctioned from the pericardial space. This was cultured and loculations were lysed with blunt dissection. The space was irrigated with antibiotic solution and a drain was placed. A right pleural chest tube was also placed due to continued mild respiratory acidosis while under general anesthesia. Intraoperative cultures of the pericardial fluid grew methicillin-resistant Staph aureus.
Postoperatively, the patient made an unremarkable recovery and was discharged back to her nursing facility on postoperative day 10, following consultations with palliative care to avoid subsequent hospitalizations. Postoperative computed tomography confirmed an excellent technical result (Figure 3B). The patient had negative blood cultures at discharge, and she was started on 6 weeks of IV vancomycin and lifetime rifabutin to prevent endograft infection.
Discussion
Diseases of the aortic arch pose a formidable surgical challenge for high-risk surgical patients as the anatomic and hemodynamic characteristics of the arch make endovascular repair difficult. Since the first endovascular aortic arch repair performed by Chuter in 2003, several advances, specifically techniques involving fenestrated and branched stent grafts, have evolved from experimental procedures to clinically accepted approaches for aortic arch disease in high-risk surgical patients. 5 Some have suggested that branched grafts may have superior long-term durability and patency of the aortic branches due to the movement of the aortic arch during the cardiac cycle. 2 However, branched stents require a larger diameter sheath and involve higher complexity deployment. In contrast, fenestrated grafts are technically simpler and faster to deploy, require less manipulation of the arch, and have a lower risk of microembolic strokes. 6 In addition, fenestrated procedures can address pathologic processes of the arch without requiring a landing zone in the ascending aorta, which could prove advantageous in a dilated or aneurysmatic ascending aorta. 2
While commercially available fenestrated and branched stent grafts exist and can be custom-made according to a patient’s specific anatomy, they are available only to a small number of centers in the United States, and patients with rapidly expanding or symptomatic aneurysms often cannot await the time required to manufacture custom-made stent grafts. Physician-modified endografts are therefore a very useful technique in treating high-risk patients with a spectrum of aortic arch pathology. In fact, physician-modified endografts have been found to have similar outcomes to off-the-shelf fenestrated and branched grafts, including technical success of 91% and 95%, mortality of 3.5% and 1.1%, and target vessel patency of 97% and 98%, respectively. 7
For these reasons, we decided to use a physician-modified endograft with a single fenestration for the common origin of the innominate and left common carotid arteries followed by laser fenestration of the more remote left subclavian artery in our patient with a mycotic pseudoaneurysm of the aortic arch. While stenting the innominate and left common carotid following the large fenestration may have been preferred for long-term patency, it was felt this was not necessary given the palliative nature of the repair.
An alternative option in this patient may have been to use the double-fenestrated physician-modified stent graft as described by Canaud and collegues.8,9 It was decided not to use this technique as it was felt a single large fenestration was easier to align than a double-fenestrated graft, and we sought to avoid as much manipulation of the graft in the arch as possible. Furthermore, we have more experience with in situ laser fenestration at our institution and have found deployment of a VBX stent following in situ laser fenestration to be an effective and technically feasible approach for arch vessel revascularization. 10 Specifically, a 2.3-mm Spectranetics laser is used as opposed to a smaller 0.9 mm laser to create a larger initial fenestration and decrease the risk of fabric tear with dilation. Overall, the combination of laser fenestration with a single premade fenestration simplifies the deployment with lower risk of malalignment while still ensuring continuous cerebral perfusion.
Conclusion
Here we describe palliation of a mycotic aortic arch pseudoaneurysm with a physician-modified endograft and in situ laser fenestration. Our technique allowed for rapid repair of the pseudoaneurysm with excellent filling of all arch vessels and no perioperative complications in a high-risk surgical patient. While the long-term durability and stability of physician-modified endografts require further elucidation, the use of physician-modified endografts for aortic arch repair is promising and may be an excellent option for carefully selected high-risk surgical patients with aortic arch disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
