Abstract
Purpose:
The Manta Vascular Closure Device is a novel collagen-based vascular closure device that has been designed specifically for closure of large-bore percutaneous arterial accesses. The aim of this retrospective study is to evaluate the immediate and 30-day outcome of Manta at the completion of endovascular aneurysm repair (EVAR) or thoracic endovascular aortic repair (TEVAR). The hypothesis is that Manta is not inferior in obtaining hemostasis compared with the Perclose ProGlide Suture-Mediated Closure System device.
Materials and Methods:
We recruited all the percutaneous accesses for (T)EVAR performed from January 2021 to April 2023 by all the Italian Divisions of Vascular Surgery using Manta at the time of data collection (May 2023). The primary outcome is to evaluate the incidence of complications at the puncture site after Manta implantation and at 1 month, and compare this with ProGlide. We applied the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) criteria for observational studies.
Results:
Overall, 524 consecutive femoral accesses for (T)EVAR procedures were collected: 355 in the Manta cohort and 169 in the ProGlide cohort, respectively. The size of the sheath was 17.2±2.7 Fr for Manta, 15.7±2.3 Fr for ProGlide (p<0.001). No statistically significant differences between the groups regarding age, sex, body mass index, ultrasound-guided access, femoral calcifications, intraoperative, and 30-day complications. Successful arterial closure at groin puncture sites for (T)EVAR using Manta is 90.5% and 93.1% using ProGlide. Freedom for any reintervention for any complication is 95.5% for Manta and 96% for ProGlide.
Conclusion:
The 2 vascular closure devices have proved to be similar in terms of complications, without any statistically significant difference, although the median size of the sheaths for (T)EVAR was statistically significantly larger when Manta has been used, compared with ProGlide.
Clinical Impact
Manta® is effective in the hemostasis of the access sites following the completion of (T)EVAR in this multicenter, retrospective, case-control study on 524 percutaneous femoral accesses. Compared to the more popular Proglide®, the average size of the introducers in the Manta® group was significantly larger than in the Proglide® group.
Keywords
Introduction
In patients affected by abdominal aortic aneurysm (AAA) or thoracic aortic aneurysm/type B aortic dissection (TAA/TBAD) with suitable aorto-iliac anatomy, endovascular aneurysm repair (EVAR) or thoracic endovascular aortic repair (TEVAR) should be, respectively, considered as the preferred treatment modality. 1
These procedures can be performed to obtain vascular access with a dual modality: surgical approach to the vessels or percutaneous approach.
Surgical cut-down of both common femoral arteries (CFAs) during (T)EVAR procedure is associated with an increased risk of complications. Several studies have reported complications related to surgical incision in the groin, such as hematomas, lymphoceles, and infections. Furthermore, surgical access to the femoral artery can lead to significant morbidity reported as 10% to 22%. The surgical approach also requires longer operating time, increased hospitalization, greater probability of surgical site infections, and iatrogenic lesions of the femoral nerve. 2
Nowadays, most endovascular procedures are performed with a percutaneous approach. Various closure devices have been developed, with different techniques that ensure hemostasis and closure of the CFA access.
Abdominal aortic aneurysm, TAA, and TBAD suitable for (T)EVAR are performed with modular stent grafts with a range of delivery system profiles from 12 to 20 Fr. Even more complex aneurysms are increasingly being treated with endovascular technique with use of complex stent-graft systems that include fenestrated and branched (T)EVAR devices, which may require larger sheaths.
Considering the increase in percutaneous procedures, in recent years, biomedical engineering has heavily focused on the development and improvement of devices for closing percutaneous accesses. One of these percutaneous techniques is the pre-closure technique using 2 Perclose ProGlide Suture-Mediated Closure System devices (Abbott Vascular, Abbott Park, Illinois). The Manta Vascular Closure Device (Teleflex, Wayne, Pennsylvania) is a novel collagen-based vascular closure device that consists of a resorbable polylactic-co-glycolic acid (PLGA) surface within the artery, connected to a resorbable bovine collagen plug outside the artery. It has been designed specifically for closure of large-bore arterial accesses. 3
In the current literature, the use of Manta is mainly described for cardiological procedures (transcatheter aortic valve implantation [TAVI]). The aim of this study is to collect the Italian multicenter experience in the use of Manta for (T)EVAR procedures, describing its primary outcome in the short term.
Materials and Methods
Patient Population
We recruited all the percutaneous accesses for (T)EVAR performed from January 2021 to April 2023 by all the Italian Divisions of Vascular Surgery using Manta. We investigated demographic data, body mass index (BMI), the external sizes of the dedicated sheaths, possible ultrasound (US)-guided punctures, common femoral artery (CFA) calcifications (described as: moderate, <50% of the circumference, and severe, >50% of the circumference), intraoperative and 30-day complications related to the vascular closure device, and related reinterventions.
We compared the results of Manta with those of ProGlide: both devices have been used in all the centers at the surgeons’ discretion at same time of data collection.
Institutional review board approval and patient publication consent were waived. The current Italian legislation on observational studies (our study falls under this category) does not request the above-mentioned documents when clinical data are anonymized (Official Gazette of the Italian Republic #76, 31 March 2008). We applied the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) criteria for observational studies. 4
Study Outcomes
The primary outcome is the incidence of complications at the puncture site after Manta implantation (bleeding treated with manual compression [MP], bleeding requiring surgery [RS], arterial occlusion, or severe stenosis) and at 1 month (pseudoaneurysm, infections), compared with ProGlide. Secondary outcomes are the correlation of these complications with the presence of CFA calcifications, US-guided access for device placement, and sheath caliber.
Device Description
Manta is available in 2 calibers, 14 and 18 Fr, and finds indications for sheaths from 10 to 25 Fr in diameter. The Manta vascular closure device consists of the Manta closure, an insertion sheath with introducer and a depth locator. The Manta closure consists of a release handle containing an absorbable collagen pad, a stainless steel locking component, and an absorbable polymer anchor connected by a non-absorbable suture. Hemostasis is achieved primarily by the mechanical means of the anchor-arteriotomy-collagen sandwich, to which are added the coagulation-inducing properties of collagen. The extravascular radiopaque block fixes and marks the position of the resorbable unit. The external surface and the internal plug are joined by a non-absorbable polyester suture and fixed with a radiopaque stainless steel block.
The release Manta is divided into the following steps:
Before starting the procedure, it is necessary to determine the depth of the arterial access using the depth locator, inserting this device on a 0.035 wire until blood comes out.
Subsequently, the depth locator must be retracted until the bleeding stops and the measurement at skin level is noted by adding 1 cm to define the depth of the delivery system.
After endovascular procedure, the Manta sheath must be inserted on the wire and the sheath removed.
At this point, the Manta system can be inserted until the “double click” and must be retracted up to the distance measured at the beginning of the procedure, deployment occurs at a 45 degrees angle to the skin.
The deployment lever must be rotated to release the anchor in the vessel and the system must be kept under tension. (Figure 1). The correct pressure required to ensure the 2 surfaces adhere well is indicated with a green indicator on the handle of the device. 5
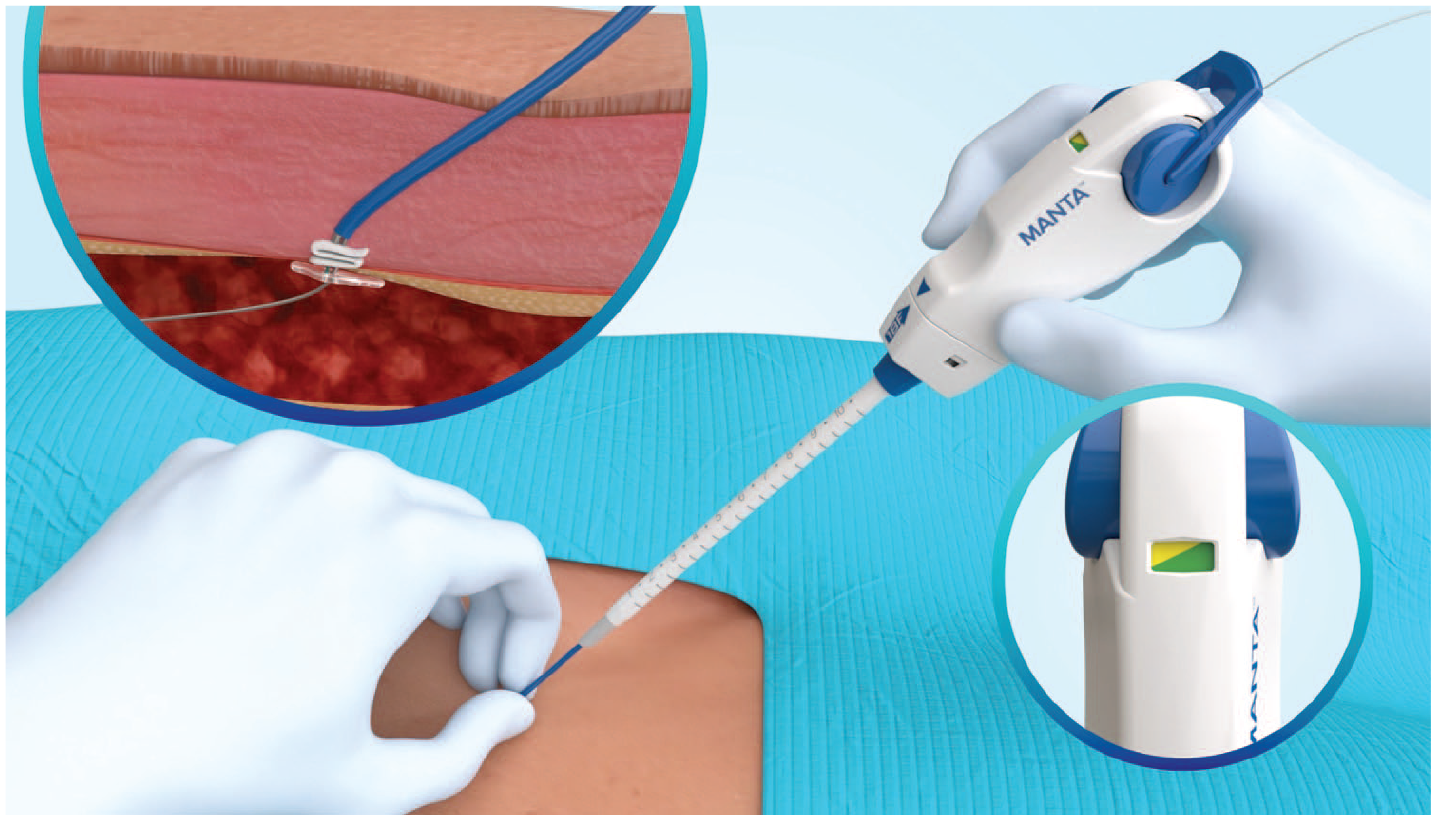
Manta vascular closure device during deployment 6 ; print color requested.
Unlike Manta, 2 ProGlide devices are required for effective closure of large-bore CFA access. ProGlide is a 6 Fr device designed to close CFAs puncture by delivering a single monofilament polypropylene suture.
The positioning of the ProGlide vascular closure system is divided into the following steps:
Blood flow through the sheath is restricted by a hemostasis valve with or without the guide wire being in place.
The guide houses the needles and the foot, and precisely controls the placement of these needles around the puncture site.
The plunger advances the needles and retrieves the suture. The guide also contains a marker lumen that allows a pathway for back-bleeding from the artery to ensure proper device positioning.
The device is slid on the wire until it enters the artery. Correct positioning of the device is indicated by the filling of a small tube on the side of the device with blood.
Maintaining an angle of 45 degree with respect to the vessel axis, rotate the device with respect to the vessel.
Once rotated, the front black lever is pulled, and the needles are anchored to the artery wall.
Subsequently, once raised to a 30-degree angle, the upper piston is pressed and the monofilament polypropylene suture is pulled to recover the free ends (Figure 2).
Once all this is done, a reference is placed on the ends of the monofilament polypropylene suture and the same procedure is continued on the opposite side, this time rotating the device on the opposite side.
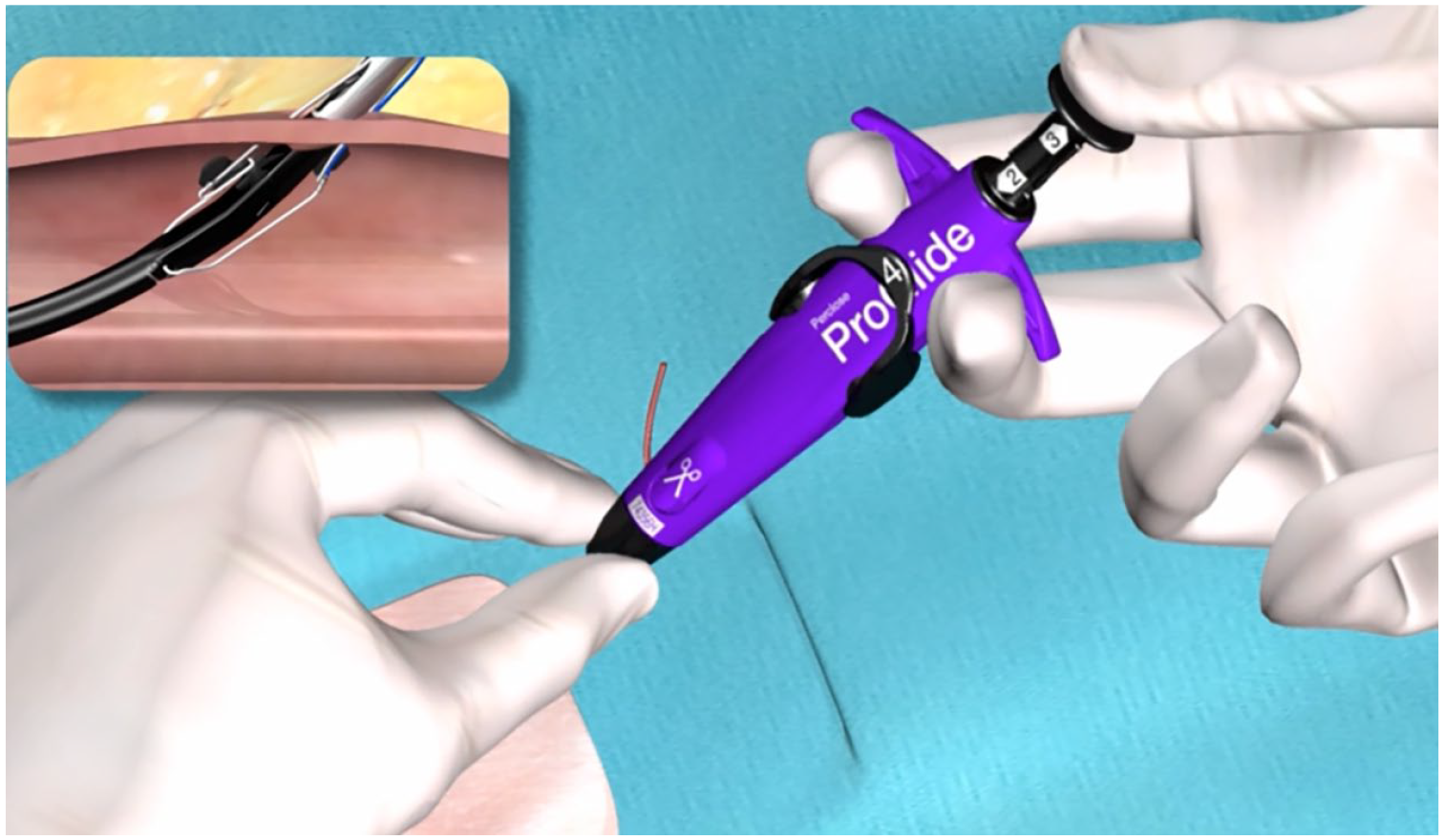
ProGlide vascular closure device 8 ; print color requested.
This procedure is carried out before the surgical procedure, once the procedure is completed, before removing the sheath and guide, the knots are progressively tightened. One end of each suture is released from the grip of the landmark and pulled using an instrument called a knot pusher to slide the knot up to the artery wall. The knot pusher accessory accomplishes the positioning of the tied suture knot to the top of the CFA access, and the trimming of the trailing limbs of the suture. 7
Statistical Analysis
Statistical analysis was carried out with version 1.4.1106© 2009 to 2021 RStudio (Integrated Development for R. Rstudio, PBC, Boston, Massachusetts). Continuous variables (age, sheath size) were analyzed with a Welch 2-sample t test, while categorical variables (males, US-guided access, femoral anterior calcification) with a 2-sample test for equality of proportions with continuity correction. Outcomes (intraoperative and 30-day complications) were analyzed with Fisher’s test to obtain odds ratio and a 95% confidence interval. A p value of less than 0.05 was considered statistically significant.
Results
Ten Italian Divisions of Vascular Surgery were using Manta at the time of data collection and joined the study. Overall, 524 consecutive femoral accesses for (T)EVAR procedures were collected: 355 in the Manta cohort and 169 in the ProGlide cohort, respectively, from January 2021 to April 2023. The 14 and 18 Fr Manta were always chosen properly, according to the external size of the introducers.
The baseline characteristics are shown in Table 1.
Clinical and Technical Characteristics.

Abbreviations: BMI, body mass index. CFA, common femoral artery. US, ultrasound. Continuous data are presented as the means±standard deviation; categorical data are given as the counts (percentage).
Mean age was 73.7±7.6 years, and 93.8% were males, without statistically significant differences between the groups. Use of US-guided puncture were similar (97.7% for Manta and 100% for ProGlide), as well as the presence of moderate calcifications of the CFA (53.5% for Manta and 52.1% for ProGlide). The BMI for the 2 groups was similar (26.8±2.8), without statistically significant differences (p=0.258). Severe calcifications of the CFA were more frequent in the ProGlide group (1.4% vs 4.7%) but, despite being close to being so, this is not statistically significant (p=0.046, which is approximately p=0.05). Instead, the introducer size during the procedures was significantly larger in the Manta group (17.2±2.7 Fr for Manta and 15.7±2.3 Fr for ProGlide, p<0.001).
Table 2 reports the main outcomes: none of these events was statistically significant.
Intraoperative and 30-Day Complications.
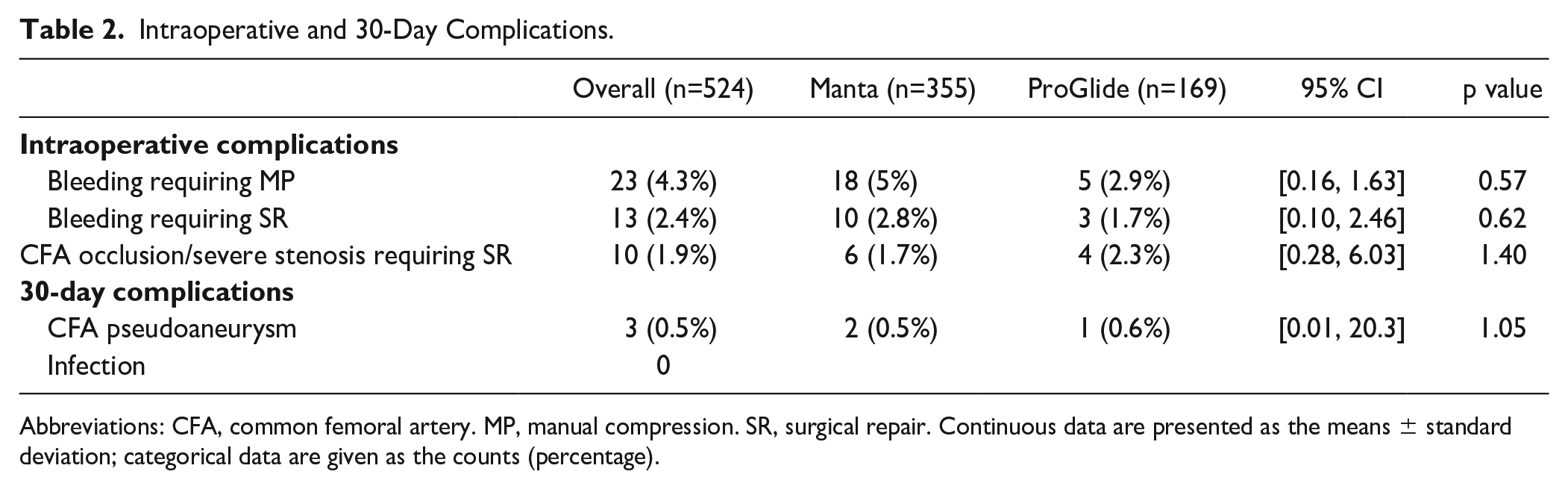
Abbreviations: CFA, common femoral artery. MP, manual compression. SR, surgical repair. Continuous data are presented as the means ± standard deviation; categorical data are given as the counts (percentage).
Intraoperatively, bleedings from the CFA access site were more frequent when using Manta (bleeding requiring MP 5% vs 2.9%; bleeding requiring SR 2.8% vs 1.7%), and CFA occlusion or severe stenosis were more frequent when using ProGlide (1.7% vs 2.3%). None of these differences were statistically significant, and the same applies to 30 days (CFA pseudoaneurysm 0.5% vs 0.6%).
We also performed a sub-analysis of the intraoperative complications in the 2 groups, according to the severity of the calcifications: however, the differences between Manta and ProGlide were not statistically significant (Table 3).
Intraoperative Complications According to the Severity of CFA Calcifications.
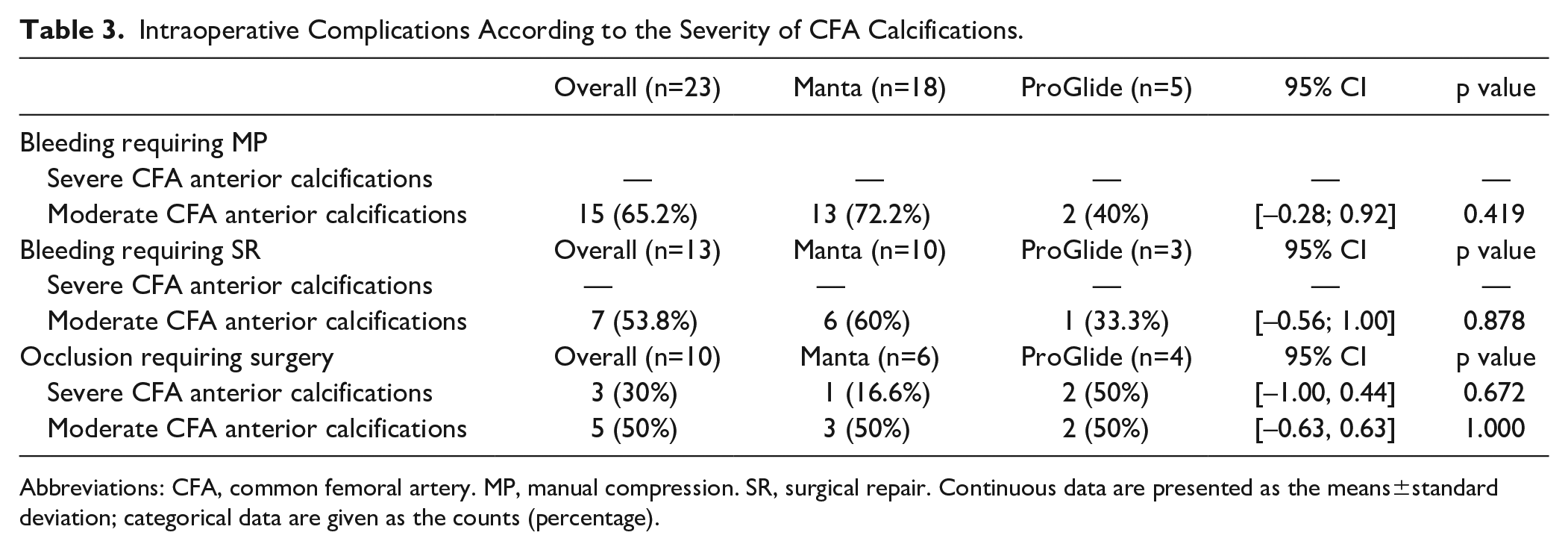
Abbreviations: CFA, common femoral artery. MP, manual compression. SR, surgical repair. Continuous data are presented as the means±standard deviation; categorical data are given as the counts (percentage).
Discussion
Several studies have reported complications related to the surgical incision in the groin, such as hematomas, lymphoceles and infections, and open surgical access of the CFAs can lead to significant morbidity reported between 10% and 22%.1,2,9 The EVAR 1, DREAM, OVER, and ACE international trials have led to these conclusions. 10
To date, most endovascular procedures are performed with totally percutaneous accesses. 11
In some studies, ProGlide was found to be more effective than open surgical access in guaranteeing hemostasis, in reducing intervention times, invasiveness, and the number of complications related to the procedure. The pre-operative and post-operative ankle-brachial index evaluation highlighted a non-inferiority of this percutaneous method compared with the surgical approach in the prevention of lower limbs ischemia, without reporting suffering to the peripheral circulation. 12
A systematic review has demonstrated the safety and effectiveness of ProGlide, making this device the most used and the one with the greatest safety margins. 8
In recent years, Manta has appeared on the market as a new dedicated closure device specifically for large-bore vascular accesses. In our study we investigated the use of Manta in (T)EVAR, while other papers in the literature have reported its use during cardiological procedures, such as TAVI. Among these works that of Eftychiou et al 13 showed no substantial difference in mortality, cardiovascular mortality, access site-related complication, periprocedural vascular surgical interventions, bleeding, or transfusion rate between Manta and ProGlide in TAVI procedure. Several recent studies, such as that of Kmiec et al 14 have investigated a large series of patients in whom Manta was implanted in TAVI procedures and observed 5.6% major and 1% minor vascular complications related to Manta. Also, they identified female gender, vessel angulation at the puncture site, and moderate calcification of the dorsal CFA as independent predictors for major complications.
The presence of several works in the literature on the use of Manta in TAVI starts from the evidence that this closure system device was created for closure of large-bore vascular accesses. The pre-closure technique that uses 2 ProGlides versus a single Manta device is used to close accesses of the same size. For these reasons, we decided to carry out our study comparing these devices on smaller vascular accesses, such as those of (T)EVAR, compared with TAVI.
In the current literature, there are fewer works on the use of the Manta in (T)EVAR or with a lower number of cases compared with our research, such as Chaudhuri studied 76 patients undergoing (T)EVAR: successful hemostasis was achieved in 120 (91.6%), and failures occurred in 11 (8.4%) groins, reintervention was necessary in 8 patients. 15 Also, Moccetti et al reported, in his series of 100 access, 11% vascular complications (7% major and 4% minor) associated with Manta, occurring more frequently in patients with peripheral arterial disease and <6 mm CFA. 3 Krajcer et al reported 53 patients who underwent (T)EVAR treated with Manta, and demonstrated that the latter needs a shorter time to obtain complete hemostasis and carries lower complication rates compared with other percutaneous closure devices, with technical success of 98%. The mean time to hemostasis for those patients in the primary analysis cohort was 35±91 seconds (0.58 minutes), with a median time to hemostasis of 19 seconds (0.32 minutes). 16 However, these studies, although they show interesting results on the use of Manta in (T)EVAR procedures, recognize the main limitation in the fact that they were conducted without a control arm when compared with other vascular closure devices.
In the present study, we report the cumulative immediate and short-term outcome of Manta after (T)EVAR of all the Italian Divisions of Vascular Surgery using this device at the time of data collection (April 2023). Our study population (355 Manta) has been compared with 169 ProGlide implanted in the same centers in the same time period (Figure 3).
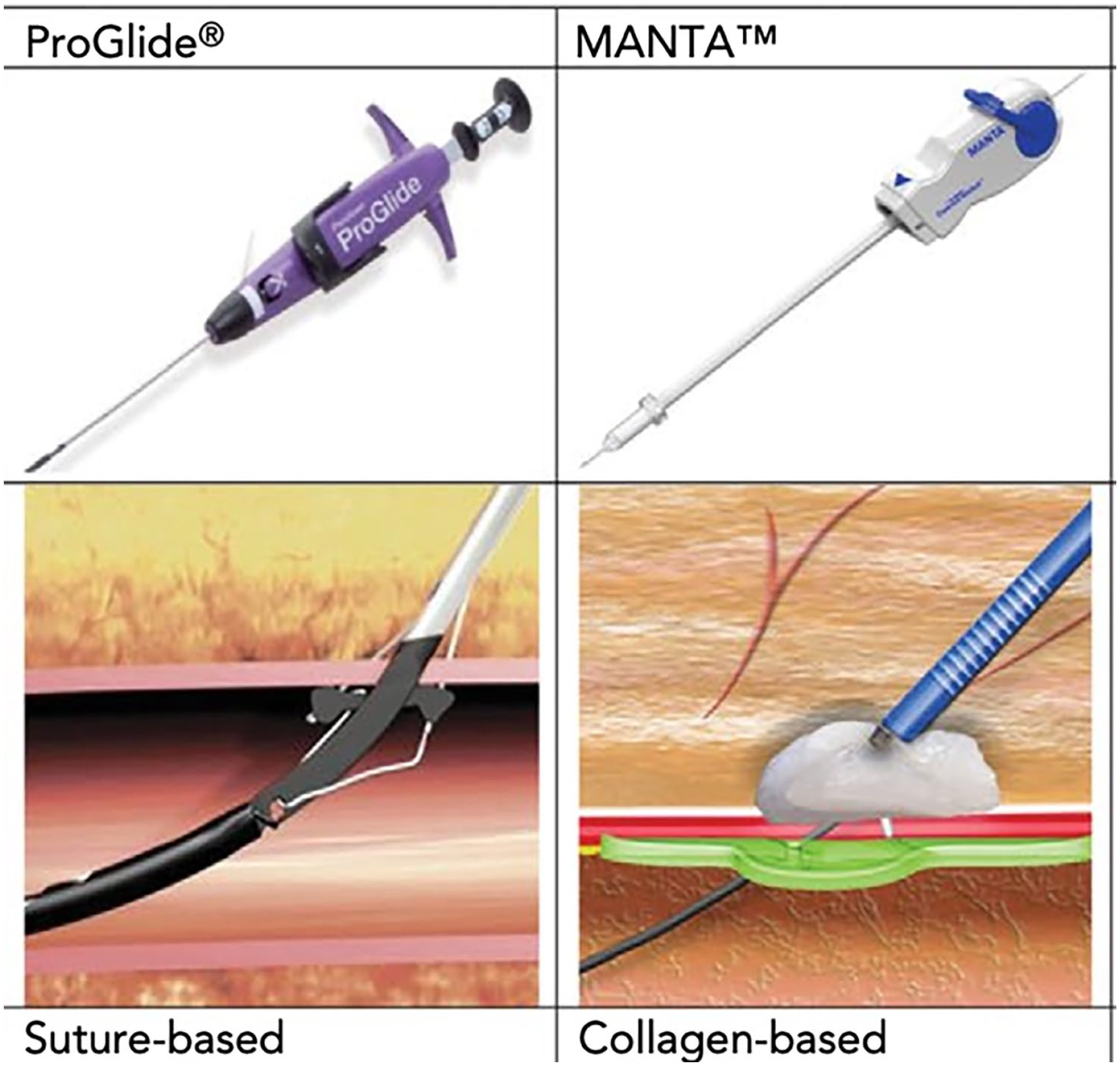
ProGlide versus Manta, suture-based versus collagen-based vascular closure devices 5 ; print color requested.
Each center reached the minimum requirements for the learning curve in the use of Manta. It should be noted that all the Italian centers, which introduced Manta in their procedures during the study period were included. Therefore, although ProGlide in these centers was historically used, in the study window examined these centers preferred to use Manta to gain experience and evaluate the effectiveness of the new device.
Our data show 90.5% successful arterial closure at groin puncture sites for (T)EVAR using Manta, and 93.1% using ProGlide. Freedom for any reintervention for any complication is 95.5% for Manta and 96% for ProGlide. The 2 vascular closure devices have proved to be similar in terms of complications, without any statistically significant difference, although the median size of the sheaths for (T)EVAR was statistically significantly larger when Manta has been used, compared with ProGlide. These results are comparable with those reported from other studies on ProGlide. 11
Other works have compared the use of Manta with ProGlide obtaining results similar to ours. Košak et al collected 189 patients undergoing TAVR and (T)EVAR. A double ProGlide was used on 91 patients (48%), while Manta was used on 98 patients (52%). The ProGlide cohort was associated with a significantly higher rate of milder complications, while the Manta cohort had a higher rate of major bleeding complications, requiring more complex treatment with potentially greater impact on quality of life. 17 Still, our data stem from the largest study sample.
However, access site hematomas, pseudoaneurysms, arteriovenous fistulas, and bleedings are common complications of the percutaneous access site for endovascular procedures. Their 30-day incidence is reported up to 13% (78.6% hematomas, 18.9% pseudoaneurysms, 1.4% arteriovenous fistulas, 1.1% overt bleedings), and about 10% of all vascular access site complications necessitate open surgical repair. 18
As reported in the MAUDE (Manufacturer and User Facility Device Experience) registry, the most common complication of Manta failure is the persistent bleeding (48.8%) and CFAs’ occlusion or stenosis (29.6%). Most complications require surgical intervention (40.4%). The most reported mechanism of failure is the failure in the deployment (22%), followed by subcutaneous deployment (7.6%), intraluminal deployment (4.8%), and detachment of collagen (2.8%). Access site infection is rare (1.2%). The 18 Fr Manta was associated with a lower risk of failure of deployment compared with the 14 Fr device but was associated with a higher risk of vessel occlusion or stenosis (32.4% vs 16.3%, p=0.04) and thrombosis (11.6% vs 0%, p=0.03). 19
In our study, the presence of CFA calcifications is not associated with an increased risk of access complications in the 2 groups. However, the association of calcifications and non-US-guided access is a negative prognostic factor for the occurrence of complications, no matter if Manta or ProGlide is used as closure device. Kalish et al 20 examined more than 7000 CFA accesses and demonstrated the superiority of US-guided access, with significant reduction in the rate of complications, compared with non US-guided access.
Manunga et al conducted a study on 752 patients undergoing EVAR using vascular closure devices and reported significant technical success in patients with <50% CFA anterior wall calcification compared with those with >50% calcifications. In their series, CFA calcification was a major determinant of failure and open surgical conversion was required in 14 cases. 21
The study by Ali et al reported the comparison of the effectiveness, safety, and costs of Manta and ProGlide during percutaneous transfemoral access during TAVI in the United Kingdom. They included in this study a total of 136 patients: 86 in the ProGlide group and 50 in the Manta group. No statistically significant difference was found between the 2 groups with respect to the average cost per patient. When considering the use of additional vascular closure device, protamine administration and the need for red blood cell transfusions and surgical repair, the average cost per patient in the ProGlide, group was £568.79, and in the Manta group was £599.55. 22 Although it is not possible to state this with the available data, we believe that the same results can be obtained when applied to the use of the same devices during (T)EVAR. Since ours is a multicenter study, and since Italy has a regional-based health care system, it has not been possible to carry out a global cost analysis with the data at our disposal. The purchase of the devices varies, depending on regional agreements. For instance, the cost of Manta is set at €580 in Campania, while at €410 in Lombardy. Regarding the incidence of intraoperative and 30-day complications requiring SR, results between Manta and ProGlide were similar in our study, and therefore would not affect the cost-benefit analysis between these 2 devices.
The main limitation of our work is the implantation of Manta by different operators, with variable learning curve and experience. In fact, while it is true that ProGlide implantation had taken place routinely for many years, it is also evident that percutaneous access procedure with vascular closure devices could not be standardized. Especially, the selection and assessment bias due to the way of selecting the access method (surgical or percutaneous access) based on the characteristics of the artery or patient, the use of US-guided access, the experience in terms of procedures carried out by the same operator, and consequently a certain learning curve with Manta (not all centers started at the same time the use of Manta and carried out the same number of procedures).
Another limitation of our study concerns the sample size. In the literature, the failure rates reported for Manta and ProGlide during EVAR/TEVAR procedures using large accesses (>22 Fr) are similarly low (6.9% and 8.9%, respectively). 23 Consequently, setting up a study to evaluate significant differences between the 2 closure devices would require a sample size greater than 5700 (p1=0.069, p2=0.089, sig. level=0.05, power=0.80->n=2854.2 in each group). Currently, it is complex to organize a study with such a large sample size, given the lower diffusion of Manta compared with ProGlide in clinical practice. The aim of our work is to demonstrate the non-inferiority of the Manta compared with the ProGlide. In addition, to the best of our knowledge, this study contains the highest reported number of Manta implants during EVAR in available literature. The post hoc power of our study using an effect size of 0.2, a value adequate to highlight small effect size according to Cohen, is almost 60% (h=0.2, n1=355, n2=169, sig. level=0.05>power=0.58).
In our work, we have not carried out a cost-effective analysis regarding the use of Manta and ProGlide devices during (T)EVAR procedures. There are different reasons behind this choice. First of all, we have not found any work in the literature that could support this research. However, the main reason is the inter-regional variety of costs relating to these 2 devices, which did not allow a unitary and statistical evaluation in terms of costs. In fact, we collected the data in a multicentric fashion from different Italian regions with different health care management in terms of device costs, without taking into account the difference in costs with which these devices are marketed abroad.
Our retrospective work did not allow us to collect BMI data from the patients involved. We believe that it should be taken into consideration for future work, as it can highlight new evidence regarding the compared devices. However, based on our experience, we believe that Manta can reduce complications compared with ProGlide in patients with high BMI, since the technical characteristics of Manta and in particular the use of the depth locator allow us to well evaluate the real depth where release the device.
To carry out an appropriate cost analysis to define if Manta is an economically viable alternative to traditional suture-based vascular closure devices for (T)EVAR, further studies are needed. They should consider the pure costs of the devices and above all the additional procedures necessary for the resolution of the related complications, but these data in the literature is limited.
Conclusions
Manta is effective in the hemostasis of the access sites following the completion of (T)EVAR. Compared with the more popular ProGlide, the incidence of intra-operative and post-operative complications is low, although the average size of the sheaths in the Manta group was significantly larger than in the ProGlide group.
Footnotes
Acknowledgements
The authors gratefully acknowledge Edoardo Guarino, Meng, MSc, for the English revision of this manuscript, and Neovasc s.r.l. for their unconditioned support in the open access of this paper.
Authors’ Note
Author Contributions
Concept and design of the work: M.P., E.C., N.T., and E.M. Analysis and interpretation of data: A.P. Drafted the article: M.P., E.C., and E.M. Revised it critically for important intellectual content, approved the version to be published, taken public responsibility for appropriate portions of the content, and acquisition of data: M.P., E.C., U.M.B., A.P., O.S., F.I., V.M., A.E., S.T., A.M.S., C.L., G.L.B., L.C., A.A., I.M., E.C., R.B., N.T., V.S., V.A., S.D.V., P.V., M.M., and E.M.
Data Availability Statement
Raw data were obtained from the all the Italian vascular surgery divisions using Manta and ProGlide, and are readily available for presentation to the referees and the editors of the journal, if requested.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
