Abstract
Purpose:
To demonstrate the feasibility of Octafen technique, a novel endovascular configuration for the treatment of thoracoabdominal aortic aneurysms (TAAA).
Technique:
Two patients with complex TAAA and high surgical risk were treated with Octafen endograft configuration in a hybrid operating room with computed tomography (CT)-fluoroscopy image fusion guidance, using 3D-3D fusion techniques to facilitate procedural success. The procedure is a modification of the previously-described Octopus technique for endovascular repair of TAAA. The main advantage of this technique is the ability to use devices to repair a TAAA with the combination of off-the-shelf and noninvestigational custom-made devices. The devices used are readily available to most practicing vascular surgeons, which provides an alternative treatment in case of limited access to investigational devices, in time-sensitive cases, and in patients with limited functional capacity who cannot undergo open repair. In the modification described herein, we use a combination of standard bifurcated endovascular aneurysm repair (EVAR) devices (Excluder; W.L. Gore & Associates, Flagstaff, Arizona) in combination with a 2-vessel renal fenestrated device (Z-Fen; Cook Medical, Bloomington, Indiana). The article describes a step-by-step approach to this technique to elucidate pitfalls, benefits, and advantages.
Conclusion:
The Octafen technique might offer an alternative option for thoracoabdominal aneurysm treatment circumventing the need for access to custom-made, investigational devices.
Clinical Impact
In this manuscript, we describe a technique for endovascular repair of thoraco-abdominal aortic aneurysms that involves the combination of off-the-shelf and non-investigational, custom-made devices. The ‘Octafen’ technique provides a treatment alternative in case of limited access to investigational devices and can be adjusted according to patient anatomy.
Keywords
Introduction
Endovascular treatment for thoracoabdominal aortic aneurysms (TAAA) has been introduced by Chuter et al 1 and it has been gaining momentum ever since with excellent results. 2 Although custom-made, patient-specific devices are commercially available; their routine clinical use is still hampered by several challenges. Manufacturing times are longer, which makes them unavailable for emergent indications, such as aneurysm rupture. Moreover, commercial availability of such custom-made, fenestrated endografts with more than 2 fenestrations required to treat TAAA is still limited and are available primarily at tertiary care centers under investigational device exemption study.
Off-the-shelf devices offer immediately-available solutions aiming for a “one fits most” approach. The T-branch platform is currently available in Europe but has not yet gained FDA (Food and Drug Administration) approval in the United States. The other off-the-shelf device for TAAA treatment, the Thoracoabdominal Branch Endoprosthesis (TAMBE; W.L. Gore & Associates, Flagstaff, AZ, USA), is still in pivotal phase and not yet commercially available. Although versatile, these devices are not yet commercially available in the United States and do not serve all unique patient anatomic needs.
The Octopus parallel graft configuration for TAAA has been described earlier with good results.3,4 It involves off-label use of FDA-approved, readily-available devices for TAAA exclusion with no requirement for device customization or back-table modification. In this article, we describe a modified Octopus parallel graft technique using a fenestrated endovascular aneurysm repair (EVAR) device called the “Octafen” technique and report early clinical results in treating complex TAAA patients.
Technique
We describe the procedural technique in a 59 year old female patient (Figure 1), presented with a 69 mm type IV TAAA, and past surgical history of EVAR and TEVAR (thoracic endovascular aortic repair) for aneurysmal disease. The degeneration of the pararenal aortic segment had led to a large type Ia endoleak of the abdominal endograft.
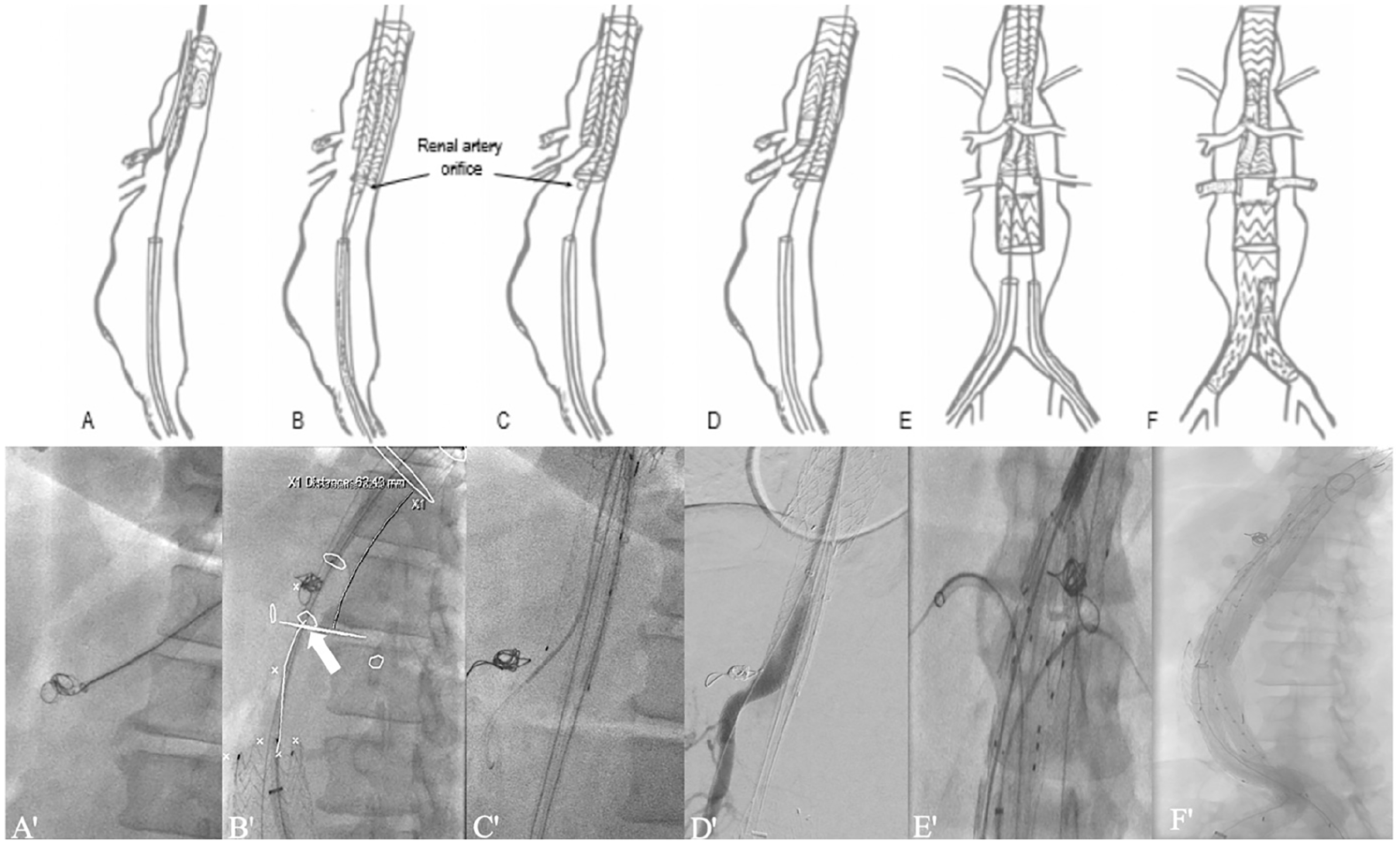
Illustration of the Octafen technique (X) and corresponding fluoroscopic images (X′): The celiac artery is embolized from the axillary access (A, A′). A bifurcated stent-graft is deployed in the thoracic aorta with the ipsilateral gate pointing toward the superior mesenteric artery (SMA) (B, B′). The SMA is cannulated from above and a bell-bottom limb is deployed in the contralateral gate 2 to 3 cm above the highest renal artery plane. Arrow point to the right renal artery (C, C′). The SMA is bridged (D, D′). A custom-made Z FEN device is deployed overlapping with the bell-bottom limb and the renal arteries are bridged (E, E′). Aneurysm exclusion is completed with a bifurcated graft when necessary (F, F′).
The patient was considered as high risk for open thoracoabdominal repair due to significant cardiac comorbidities (Revised Cardiac Risk Index Class III). Aneurysm morphology allowed for complete endovascular treatment with a fenestrated device bridging the aortic segment between the 2 previous endografts. However, given the high surgical risk and increasing size of the aneurysm in the presence of proximal endoleak, it was felt that it would be prudent to treat the patient promptly without additional waiting period for an investigational, custom-made device. We therefore proposed a modified endovascular treatment with the combination of off-the-shelf components and a 2-fenestration custom-made device. Consent for the off-label procedure was obtained and the custom-made device was available within 3 weeks.
Preoperative Planning
The main concern with this case was spinal cord injury (SCI) given the length of aortic coverage because of the previous devices. We considered that the 2 previous aortic interventions conferred a natural staging element to the treatment. Moreover, the subclavian and internal iliac axes were patent. Cerebrospinal fluid drainage was placed as a preoperative measure for SCI prevention. Another consideration was management of the celiac artery (CA). Whenever possible, after assessing the collateral circulation from superior mesenteric artery (SMA), we try to embolize the CA because of potential source of endoleaks.
The 3-dimensional (3D) planning for endovascular TAAA repair was performed using thin-slice (<1 mm), high-resolution multislice computed tomographic angiogram (CTA) covering the entire chest-abdominal-pelvic region (Figure 2). The CT images were reviewed using multiplanar reconstructions for accurate diameter measurements and using semi-automated centerline reconstructions for length measurements. Accurate aortic centerline-based length measurements are crucial to predetermine optimal device length and landing zones. A preliminary assessment of collateral circulation between superior mesenteric and celiac arteries was performed using CTA imaging.
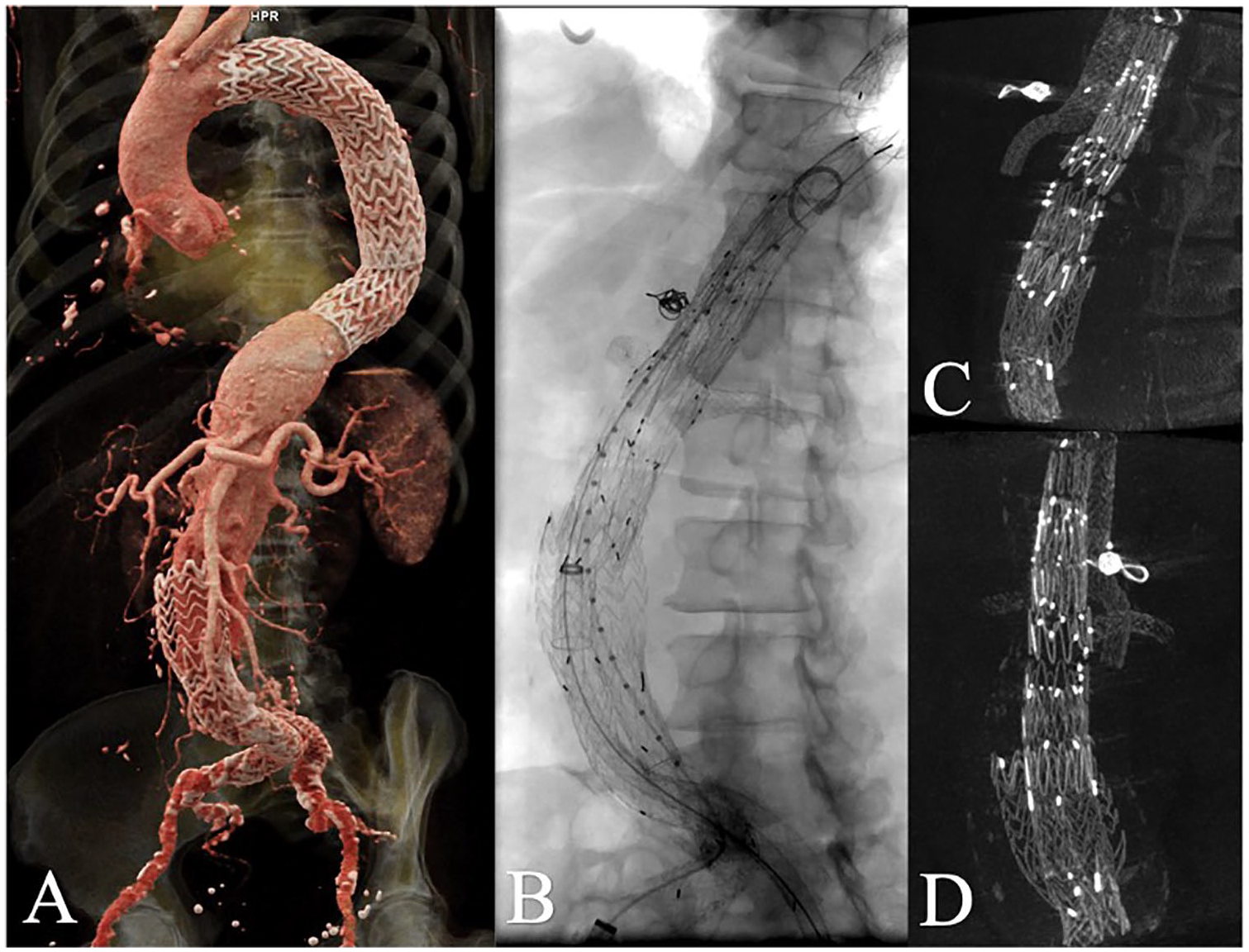
Cinematic 3-dimensional reconstruction of preoperative computed tomographic (CT) angiography illustrating the previous thoracic endovascular aortic repair, endovascular aneurysm repair, and type 1a endoleak (A). Fluoroscopic image of the final endovascular construct and celiac artery coils (B). Lateral (C) and frontal (D) views of the intra-operatory cone-beam CT.
A standard bifurcated EVAR device was selected according to proximal landing zone measurements in the thoracic aorta. The ipsilateral limb was planned to land just proximal to the SMA origin. The distance between the contralateral gate and the highest renal artery determined the length of a bell-bottom EVAR contralateral limb. Specifically, the bell-bottom limb had to land as close to the most proximal renal artery as possible. The bell-bottom limb is oversized by 20% to minimize the risk for type III endoleaks. A fenestrated endograft was planned according to the visceral aortic diameter at the level of the renal arteries according to manufacturer’s instructions. The size of the bell-bottom EVAR limb was selected for appropriate overlap with the size of the Zenith Fenestrated endograft.
Procedure Description
The procedure was performed under general anesthesia, in a hybrid operating room equipped with CT-fluoroscopy image fusion. Cerebrospinal fluid drain was placed to minimize SCI. The patient was positioned supine on the operating table with both arms tucked alongside. Two transverse inguinal incisions were performed to expose the common femoral arteries. The left axillary artery was exposed through a small, transverse, infraclavicular incision, and a 10 mm Dacron conduit is sewn onto it, in an end-to-side fashion, after systemic heparinization (activated clotting time >250 seconds).
The Dacron conduit was accessed with mini-stick needle and sheath. A soft Glidewire (Terumo, Tokyo) was directed down the aorta and exchanged for a stiff Glidewire (Terumo). A long 9F sheath was advanced from the axilla to just above the visceral vessels. Selective angiograms of the SMA and CA were performed to demonstrate collateral flow. An occluding noncompliant balloon was inflated in the CA revealing adequate collateral flow from the SMA. The CA was embolized with 2 Nester coils (Cook Medical, Bloomington, Indiana) (Figure 1A).
Both femoral arteries were accessed with mini-stick needles and sheaths, and after upsizing sheaths and exchanging for Amplatz Super Stiff (Boston Scientific, Natick, Massachusetts) wires, an 18F Dryseal (W.L. Gore & Associates) sheath was placed on the right and a 20F Dryseal sheath was placed on the left. A 35 mm × 14.5 mm × 14 cm Excluder (W.L. Gore & Associates) was advanced from the left femoral access and deployed with the contralateral gate posteriorly and the ipsilateral limb ending proximal to the SMA (Figure 1B). The contralateral gate was cannulated from the right and a 20 mm × 9.5 cm bell-bottom limb was deployed after performing an angiogram to confirm its position above the level of the renal arteries (Figure 1C). The main body was fully deployed. From the axillary access, the ipsilateral limb was selected and the SMA was cannulated. Bridging to the SMA was completed with an 8 mm × 59 mm VBX (W.L. Gore & Associates) stent (Figure 1D). On the angiogram, a type III endoleak was revealed and a further 11 mm × 59 mm VBX stent was deployed in the limb and postdilatated with complete resolution of the endoleak.
From the left femoral access, a Lunderquist Extra-Stiff Guidewire (Cook Medical Inc., Bloomington, Indiana) was positioned through the bell-bottom limb and contralateral gate. The standard configuration, custom-made 2-fenestration Z-Fen device (Cook Medical Inc.) was advanced, orienting the fenestrations based on the fiducial markers for the renal arteries (from prior 3D-3D fusion). The fenestrated device was unsheathed (Figure 1E). A 7F × 90 mm Flexor Ansel (Cook Medical Inc.) sheath was inserted through the Dryseal sheath in the right groin, and through this the right renal artery was selected with a Van Schie 3 (Cook Medical Inc.) catheter and Glidewire (Terumo, Tokyo, Japan). A Quick-Cross support catheter (Philips Healthcare, Cambridge, Massachusetts) allowed exchange to a Rosen wire (Cook Medical Inc.), and the sheath was advanced. The renal artery ostia were electronically marked and overlaid on fluoroscopy using CT-image fusion. The left renal artery was cannulated through a second 7F × 90 cm Flexor Ansel sheath. On the left, renal artery cannulation was very difficult owing to the distance from the fenestration, but eventually, using a TourGuide steerable sheath (Medtronic, Minneapolis, Minnesota), a Rosen guidewire was able to be exchanged into the left renal artery. The restraining wire on the Z-Fen was removed. The left renal was bridged with 2 VBX stents (7 mm × 39 mm, 7 mm × 39 mm), and the right one with 1 VBX (6 mm × 39 mm). The ends were flared with a 10 mm noncompliant balloon (Figure 1F).
From the right femoral access, an Excluder 28.5 mm × 14.5 mm × 16 cm main body was advanced and deployed in the Z-Fen in the standard fashion. The contralateral gate was cannulated from the left and a 16 mm × 13.5 cm limb was deployed (Figure 1F). All overlap points were ballooned with a Coda Balloon (Cook Medical Inc.).
Completion angiogram demonstrated a type III endoleak. Injections proximal and distal to the inflated Coda balloon isolated the problem to the overlap between the Z-Fen and the bell-bottom limb. The SMA branch was fully sealed, and no leak appeared to be coming from the renal stents or fenestrations. A 24 mm × 55 mm Zenith cuff (Cook Medical Inc.) was placed at the proximal end of the Z-Fen and ballooned. The endoleak was reduced but not further treated because it was felt that it would stop with heparinization reversal.
The sheaths were pulled from the axillary artery conduit and both groins. The axillary conduit was oversewn, and the femoral arteries were repaired directly with a running Prolene suture.
The patient was taken to the intensive care unit and extubated on the operative day without neurological complications. The 30 day follow-up CTA demonstrated patency of the SMA and renal arteries with presence of a type II endoleak arising from a phrenic artery with stable aneurysm dimensions (Figure 3). The patient was lost to follow-up.
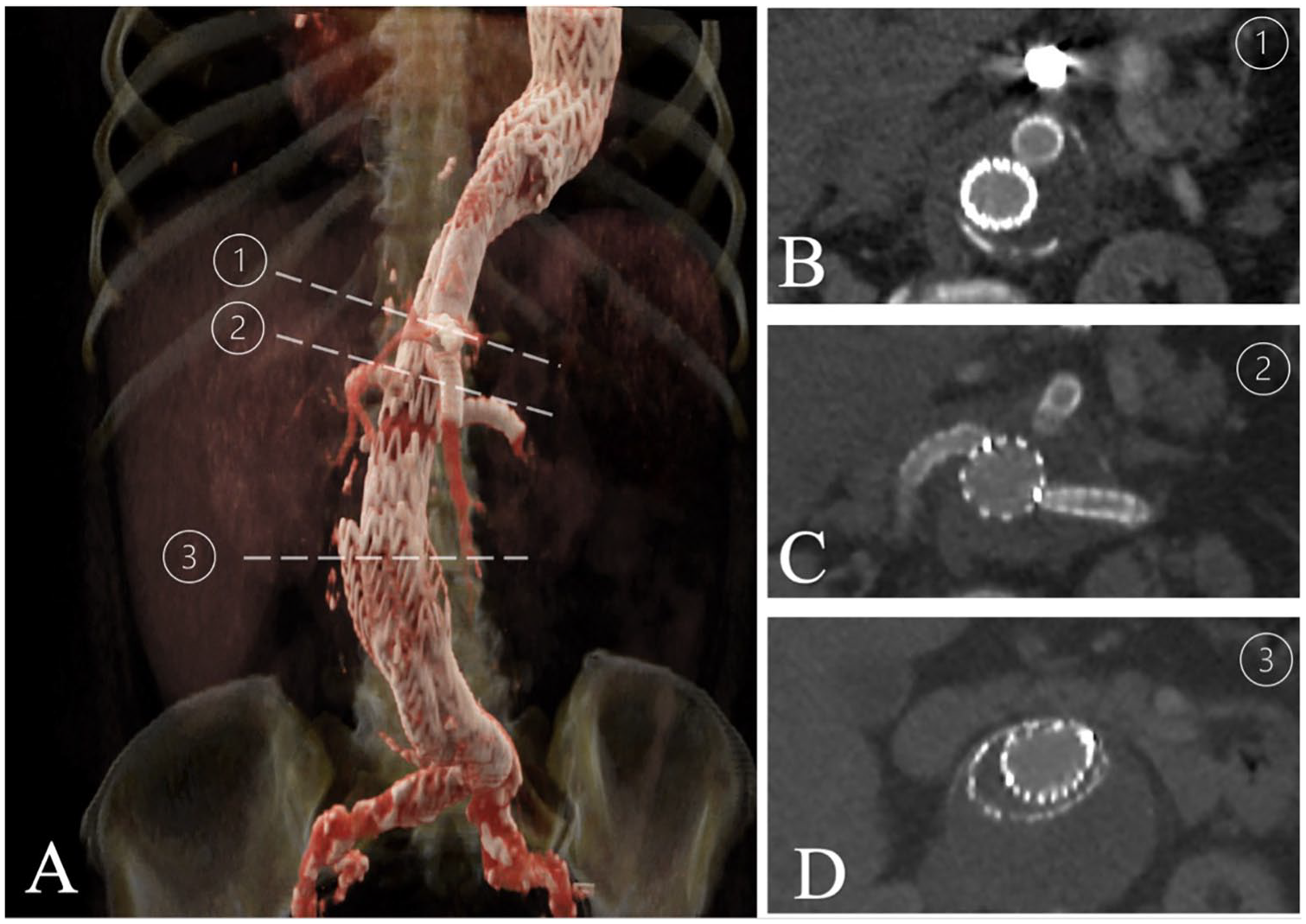
Cinematic rendering of a 3-dimensional reconstruction of the 1 month follow-up computed tomographic angiography (A) and axial views corresponding to planes passing through the superior mesenteric artery and celiac artery coils (B), the renal arteries (C), and the abdominal aorta (D).
Discussion
The Octafen technique allows for TAAA repair with the combination of off-the-shelf and noninvestigational custom-made devices. The devices used are readily available to most practicing vascular surgeons, which provides an alternative treatment in case of limited access to investigational devices, in time-sensitive cases and in patients with limited functional capacity who cannot undergo open repair. Moreover, the technique can be adjusted according to patient anatomy. At our institution, 2 patients have been treated with the Octafen technique (Table 1). To our knowledge, this is the first report to describe the use of the Octopus technique with a Zenith Fenestrated device.
Patient characteristics and results.
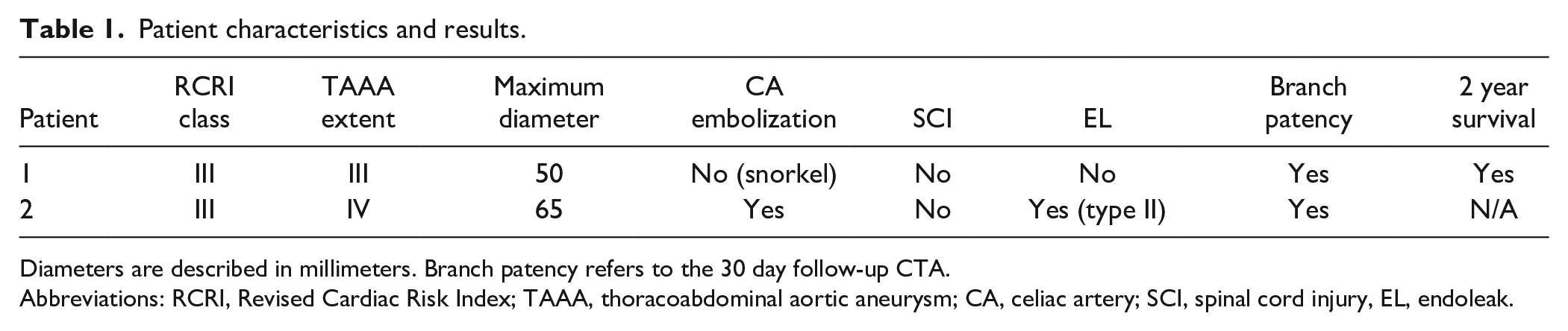
Diameters are described in millimeters. Branch patency refers to the 30 day follow-up CTA.
Abbreviations: RCRI, Revised Cardiac Risk Index; TAAA, thoracoabdominal aortic aneurysm; CA, celiac artery; SCI, spinal cord injury, EL, endoleak.
The Octopus technique has been described by many authors for treating both aneurysmal and post-dissection disease.3 –5 It involves the in-series deployment of 2 standard bifurcated endografts and the extension of bridging stents into the reno-visceral vessels, thereby imitating a branched device with off-the-shelf products. The largest case series was presented by Dua et al 4 who described a cohort of 21 patients who underwent TAAA repair with the Octopus technique. The authors reported a 19% incidence of SCI, 24% acute kidney injury, and 4.8% bowel ischemia. Primary patency at 13.5 months was 93.8% and type I endoleak incidence was 9.5%. The Octafen technique uses a fenestrated endograft for targeting the renal arteries, thus providing the opportunity to treat different anatomies.
In our 2 cases, no incidence of SCI was reported. Multiple authors have demonstrated that the percentage or length of aortic coverage is correlated with SCI and paraplegia.6,7 To effectively exclude the aneurysms with the Octafen technique, the aorta is inevitably covered from Ishimaru zone 2 or 3 up to the aortic bifurcation. However, several techniques can be used to minimize risk of SCI. First, exhaustive preoperative planning allows for accurate assessment of collateral circulation to the spinal cord from the subclavian and hypogastric axes and calculation of SCI risk. 7 A pre-established bundle of neuroprotective strategies such as neuromonitoring, cerebrospinal fluid drainage, hemodynamic parameter optimization is always implemented in our clinical practice.8 –10 If dictated by a high preoperative risk of SCI, the Octafen technique offers the possibility to perform a staged procedure by noncompletion of either the stent bridging or the distal EVAR leg component, thereby intentionally inducing a type Ic or Ib endoleak accordingly. The procedure could be corrected in 2 or more steps, based on clinical outcome, risk of SCI, and aneurysm rupture. Endovascular staging has provided exciting results in SCI prevention. 11
Endoleak insurgence remains one of the main complications after endovascular TAAA repair.12,13 In our experience, gutter endoleaks can be a challenge, especially when snorkeling the celiac trunk. In the original Octopus technique, more parallel grafts are used, which generates an increasing risk of gutter endoleaks. We aim to limit this problem by using a fenestrated device for the renal arteries. Even though some gutter endoleaks can be innocuous, they might lead to an increased number of mid- and late-term re-interventions. Some techniques that we use to limit endoleaks are moderately aggressive oversizing of bridging stents and accurate ballooning of the overlap zones with noncompliant balloons. In our 2 patients, we encountered one type Ia gutter endoleak that was successfully treated with coil embolization.
One of the most common conundrums with endovascular TAAA repair is the choice of bridging stent-grafts. In our series, we have used both balloon and self-expandable stents. We prefer to use balloon-expandable stents (VBX; Gore Medical, Flagstaff, Arizona) in the first intra-arterial segment, especially in the SMA, to obtain more precise and kink-resistant results. Moreover, flared or reverse flared configurations can mitigate size differences between the artery and endograft limb. Balloon-expandable stents are also more ideally suited for branched devices, a concept that we adapted to our off-the-shelf technique for the celiac trunk and SMA. However, Meekel et al demonstrated a 6-fold increase in type Ia endoleaks when using balloon-expandable over self-expandable stents. 14
The Octafen technique can be readily used by expert vascular surgeons for TAAA repair with off-the-shelf devices without benchtop modifications (Table 2). Compared with the original Octopus technique, it offers the possibility to better adapt to challenging patient anatomies, especially in addressing renal arterial anatomy. Our experience has demonstrated good early and mid-term results without bowel ischemia or SCI. Limitations of the technique include the need for extensive aortic coverage due to the proximal bifurcated graft, the high risk of type III endoleaks, and the need for a relatively small proximal landing zone in the thoracic aorta (<36 mm according to most instructions for use [IFU] for abdominal bifurcated grafts). An important feature of the technique is that there are multiple points during the procedure where one can stop, without putting the patient at risk. This is potentially beneficial not only to stage and limit the length of a procedure, but also to limit immediate coverage of all spinal branches, thereby reducing the risk of SCI. 11 Finally, considering the cost for this procedure, the Octafen technique involves 2 off-the-shelf commercially-available EVAR devices plus the additional cost of standard Z-Fen device with 2 fenestrations that may be comparable with a single custom-made device with visceral fenestrations/branches.
Octafen Endovascular Configuration.

Footnotes
Authors’ Note
Abstract accepted for presentation at SVCS (Society for Clinical Vascular Surgery) 48th annual symposium, March 14 to 18, 2020 at Huntington Beach, California.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
