Abstract
Purpose:
In some cases of endovascular thoracoabdominal or juxtarenal aortic aneurysm repair, a thoracic endograft in combination with a fenestrated renovisceral device may be needed in order to create a sufficient proximal landing zone. This study aimed to evaluate the technical aspects and postoperative morbidity of a single- or 2-stage approach.
Methods:
Eighty-seven consecutive patients undergoing thoracic endovascular aortic repair (TEVAR) in combination with elective fenestrated repair (fenestrated endovascular aortic repair [FEVAR]; fenestrated Anaconda device) from 2015 to 2022 were included in this retrospective bicentric study. Underlying pathologies, aortic morphology, technical details, and postoperative morbidity were recorded.
Results:
Single-staged (“1S,” n=61) and 2-staged (“2S,” n=26) interventions were compared. Indications were thoracoabdominal aneurysms (TAAAs) (Crawford I-IV) (n=56, 64%) and juxtarenal aneurysms (n=31, 36%). In 2S, the proportion of TAAA was higher than in 1S (2S: 77%, 1S: 59%; p=0.001). In 2S, the covered length of the descending aorta was longer (1S: 128±60 mm, 2S: 202±64 mm; p=0.003). Temporary aneurysm sack perfusion (TASP) was established in 11 (18%) of 1S and 1 (4%) of 2S patients (p=0.079), as well as cerebrospinal fluid (CSF) drainage catheter in 48 (79%) of 1S and 19 (73%) of 2S. The rate of spinal cord ischemia (SCI) and the severity of SCI were not different in both groups, with a total of 3 cases of persisting paraplegia. The rate of access complications was higher in 2S (n=6, 23%) than in 1S (n=4, 7%; p=0.027). Postoperative 30 day morbidity did not significantly differ in both groups and neither did 30 day mortality (4.6% in 1S vs 3.8% in 2S; p=0.083).
Conclusion:
The combination of TEVAR and FEVAR using a fenestrated endograft is feasible and safe. Aortic morphology does not change significantly after endovascular repair. A single-staged strategy is feasible with excellent results, especially in Crawford IV, Crawford V, or juxtarenal aneurysms. Two-staged repair is recommended in cases with long aortic coverage and a higher American Society of Anesthesiologists (ASA) class. Follow-up data are needed to evaluate the long-term stability of the TEVAR/FEVAR interconnection.
Clinical impact
Our study has revealed the safety and efficacy of the combination of TEVAR and FEVAR in the treatment of TAAAs and juxtarenal aneurysms with compromised supravisceral landing zones. A single-staged concept is not necessary in all cases. Staged procedures may reduce postoperative morbidity in cases with long aortic coverage and higher ASA class.
Keywords
Introduction
During the last decade, fenestrated or branched endovascular repair (FB-EVAR) has developed into a standardized and safe therapeutic concept for the treatment of aortic aneurysms involving the renovisceral segment.1 –5 In thoracoabdominal aneurysms (TAAAs), or juxtarenal aortic aneurysms without a suitable supraceliac segment, the proximal landing zone for the fenestrated graft component is provided by one or more thoracic endovascular aortic repair (TEVAR) grafts; the TEVAR graft is either implanted in the same, single intervention or in a staged, consecutive approach. The intention for delaying FB-EVAR in a multistage concept may be the reduction of postoperative morbidity, including the most devastating complication of spinal cord ischemia (SCI). In previous studies, a reduced rate of SCI was reported for staged endovascular TAAA repairs.6,7 From a technical perspective, the stable interconnection of TEVAR and fenestrated endovascular aortic repair (FEVAR) grafts is paramount, which has been evaluated in vitro. 8 This bicentric study aimed to describe the technical aspects and postoperative morbidity of elective, endovascular TAAA repair using a combination of TEVAR grafts and custom-made fenestrated endovascular repair, in single- and multi-staged interventions.
Materials and Methods
Study Cohort
In this retrospective bicentric study, 87 consecutive cases of FEVAR procedures in combination with TEVAR for thoracoabdominal or juxtrarenal aortic aneurysms with ulcerated or thrombosed supravisceral segments were recorded from 2 tertiary centers during the study period from June 2015 to June 2022. Indications for treatment were aortic aneurysm with a diameter >5.0 cm or 2 times the normal aortic diameter, aneurysm with a history of growth >0.5 cm per year, or saccular aneurysms deemed at significant risk for rupture based upon physician interpretation. Cases requiring surgical debranching, chimney or sandwich interventions, emergency procedures, or urgent interventions for symptomatic aneurysms were excluded. Basic epidemiology, anatomical configuration of the underlying pathology, procedural details, and short-term results (30 days) were assessed. For further statistical analysis, the study cohort was divided into 2 groups of single-staged (“1S,” n=61) and 2-staged (“2S,” n=26) procedures. The study was conducted according to the World Medical Association Declaration of Helsinki and was approved by the local medical ethics committees of both institutions.
Perioperative Monitoring
During case planning, left subclavian arteries (LSAs) were confirmed to be patent, and the status of hypogastric arteries was recorded. The preoperative computed tomography (CT) scan was used to elaborate the custom-made FEVAR stent. In 3 patients, carotido-subclavial bypass was performed prior to TEVAR. Previous aortic interventions and prospective aortic coverage were defined as additional risk factors for SCI. Prophylactic cerebrospinal fluid (CSF) drainage was used in single-staged and the second step of 2-staged procedures. Contradindications for prophylactic CSF drainage were coagulation disorders, previous lumbar spine surgery, or missing consent. Intraoperative motor evoked potential (MEP) monitoring was used according to the local protocols. Postoperative management included manual or automated CSF pressure monitoring and CSF management (LiquoGuard 7, Möller Medical GmbH, Fulda, Germany) and continuous arterial pressure monitoring for at least 24 hours.
Implantation Technique
Vascular access to the femoral arteries was established percutaneously or by transverse cut-down. In selected cases, an additional transaxillary or LSA access was needed. After the implantation of the thoracic graft, FEVAR was performed within the same procedure (single-stage) or as a secondary procedure (2-stage). In selected cases, temporary aneurysm sack perfusion (TASP) was performed via intentionally unconnected fenestration or iliac leg. The FEVAR implantation technique is described in detail elsewhere. 9 All FEVAR procedures were performed using a custom-made fenestrated Anaconda device (Terumo Aortic, Inchinnan, UK).
Aortic Configuration Before and After Implantation
The preoperative thoracoabdominal computed tomography angiographies (CT-As) were analyzed by FEVAR-experienced surgeons using the case planning software EndoSize (Therenva SAS, Rennes, France). Pre-operative and post-operative aortic configurations including thoracoabdominal and juxtarenal aortic diameters and angulation were compared, as described by Squizzato et al. 10
Follow-up
Postoperative follow-up included an early 2-phase thoracoabdominal CT-A to exclude endoleak, stent graft occlusion, migration or dislocation, renal and liver function panel as well as neurophysiologic examination for SCI according to respective protocols. Short-term, postoperative morbidity up to day 30 was recorded using the Clavien-Dindo classification. 11 Specific complications including SCI were reported using the reporting standards of the Society for Vascular Surgery described by Oderich et al. 12 Long-term follow-up was not included in this study.
Statistical Analysis
Epidemiologic, procedural, and postoperative data were collected. Statistical analysis was carried out using SPSS 22 (IBM Corp, Armonk, New York). Continuous data are presented as mean±standard deviation (SD) in case of Gaussian distribution or as median with interquartile range (IQR) in brackets in case of a non-Gaussian distribution. Categorical variables were compared with Fisher’s exact test. The geometrical configuration of treated aortas was evaluated using EndoSize (version 3.1.47, Therenva SAS, Rennes, France).
Results
Anthropometric Data and Pathology
Sixty-one single-staged and 26 2-staged interventions were included. The second stage was performed 94.4±71.8 days after TEVAR. Indications comprised TAAAs (Crawford I-IV) (n=56, 64%) and juxtarenal aneurysms with (n=31, 36%). The proportion of octogenarians in 2S was 19% compared to 10% in 1S; however, the difference was not significant (p=0.292). The mean maximum aortic diameter was 61 (39-94) mm. The indications for TEVAR-FEVAR combinations in 6 (7%) patients with small aneurysms (<50 mm) were either eccentric saccular configurations or large penetrating ulcers combined with small TAAAs. The 2 groups were not different regarding any other epidemiologic parameter that had been recorded during the study. A detailed baseline description is presented in Table 1. In 2S, the portion of TAAAs was higher than in 1S (2S: 77%, 1S: 59%; p=0.001). Patients with 2S had a higher ASA category (ASA 4: 15%) than 1S (ASA 4: 2%; p=0.020) and had a higher rate of prior open aortic surgery in their medical history (2S: 19%, 1S:2%; p=0.008). The TEVAR products were provided by Bolton Medical (80%; Bolton Medical Inc, Sunrise, Florida), Cook Medical (12%; Cook Group, Bloomington, Indiana), and Medtronic (7%; Medtronic, Dublin, Ireland). Balloon expandable covered stents were used as connecting stentgrafts: Advanta V12 (Getinge AB, Gothenburg, Sweden) in 81 cases (95%), others included Gore Viabahn VBX (W. L. Gore & Associates, Inc, Newark, Delaware), BeGraft Peripheral Plus (Bentley InnoMed GmbH, Hechingen, Germany), and the Lifestream stent (Bard Medical, Murray Hill, New Jersey).
Anthropometric Data.
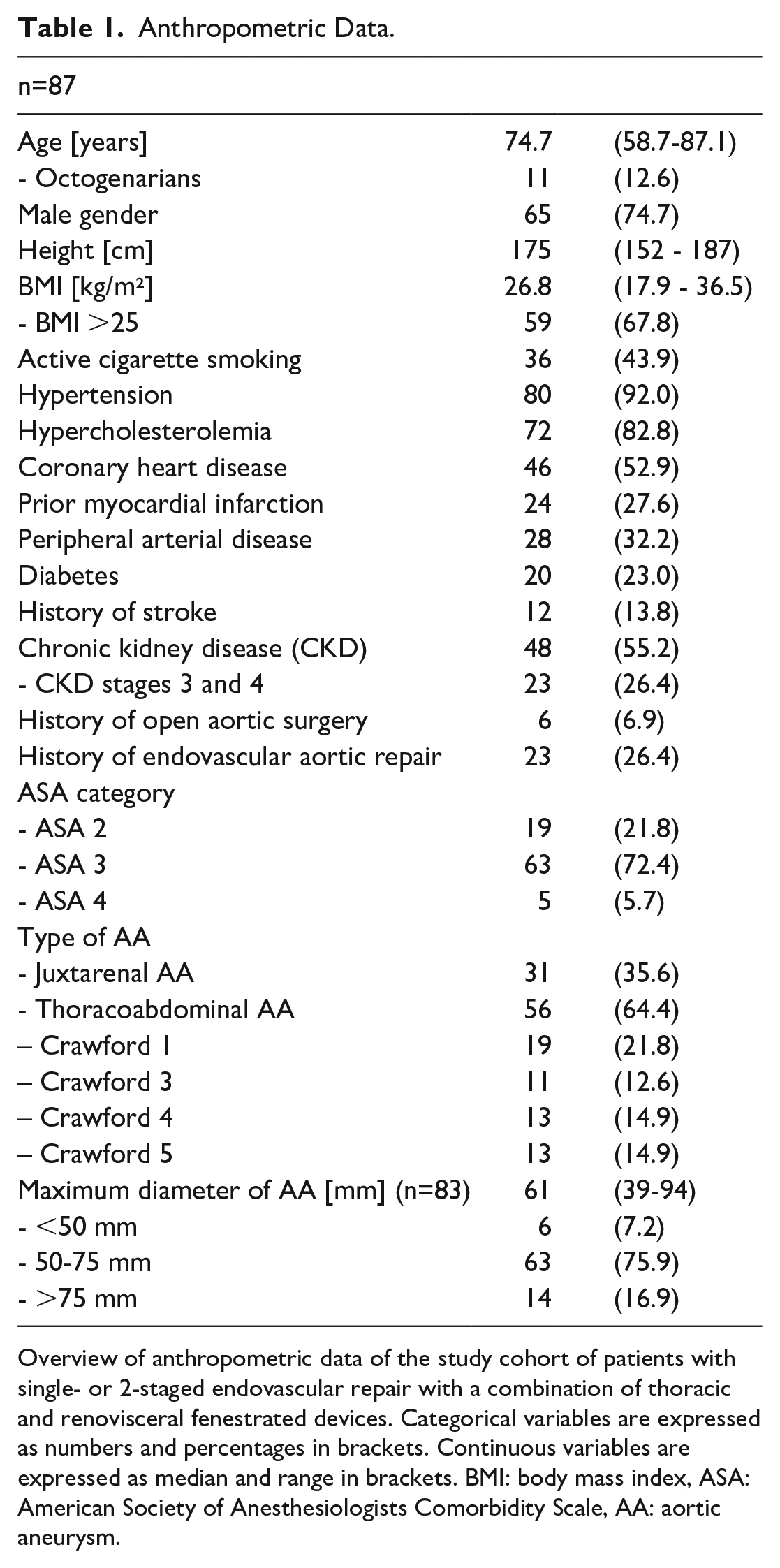
Overview of anthropometric data of the study cohort of patients with single- or 2-staged endovascular repair with a combination of thoracic and renovisceral fenestrated devices. Categorical variables are expressed as numbers and percentages in brackets. Continuous variables are expressed as median and range in brackets. BMI: body mass index, ASA: American Society of Anesthesiologists Comorbidity Scale, AA: aortic aneurysm.
Intraoperative Details
Intraoperative parameters were compared between single- and 2-staged interventions. Antegrade upper extremity access was used for 1S in 20 cases (36%) and in 4 2S cases (15%; p=0.060). In 2S, more main grafts for TEVAR were needed (2S: 2.0±1.1, 1S: 1.4±0.8; p=0.019), and an additional aortic cuff was needed in 31% of 2S cases (1S: 8%, p=0.017), to properly anchor the FEVAR graft. The mean number of fenestrations of the custom-made Anaconda devices was 4.0±0.6. The supravisceral aortic diameter was 41±13 mm in 2S and 33±6 in 1S (p=0.012). In 2S, the covered length of the descending Aorta was longer (1S: 128±60 mm, 2S: 202±64 mm; p=0.003). A preoperative CSF drainage catheter was placed in 79% of 1S and 73% of 2S (p=0.586). Temporary aneurysm sack perfusion was performed in 11 (18%) 1S patients and 1 (4%) 2S patient (p=0.079). A total of 340 renovisceral target vessels were cannulated.
The aortic configuration and its modification after the completion of TEVAR and FEVAR were evaluated. The preoperative aortic angulation was not different in both groups. Postprocedural, thoracoabdominal angulation and the aortic diameter were reduced significantly in both groups (p<0.001). The postoperative infrarenal angle was smaller in 1S (p=0.002) but did not change in 2S (p=0.958). More procedural details are described in Table 2.
Intraoperative and Perioperative Technical Details of TEVAR and FEVAR Procedures.
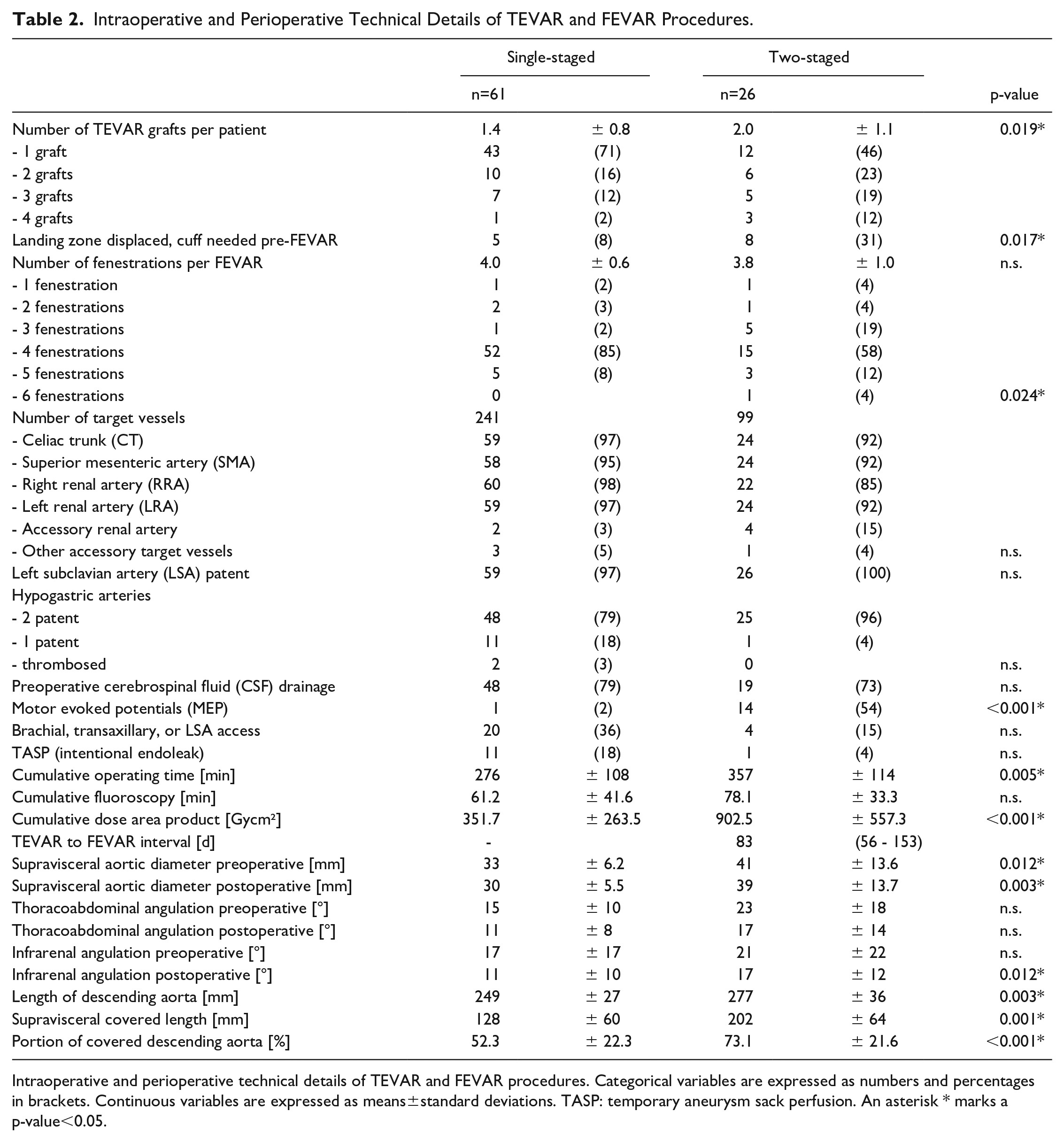
Intraoperative and perioperative technical details of TEVAR and FEVAR procedures. Categorical variables are expressed as numbers and percentages in brackets. Continuous variables are expressed as means±standard deviations. TASP: temporary aneurysm sack perfusion. An asterisk * marks a p-value<0.05.
Postoperative Morbidity
Postoperative general morbidity was not different between single- and 2-staged procedures. Thirty day mortality was 3 (4.9%) in 1S (myocardial infarction, multiorgan failure, retroperitoneal bleeding) and 1 (3.8%) in 2S (myocardial infarction) (p=0.827). Renal function remained stable or returned to the preoperative level within 30 days. Temporary hemodialysis was needed in 1 case. No aneurysm-related morbidity was observed in the TEVAR/FEVAR interval in 2S. The rate of SCI and the severity of SCI were not different in 1S (n=7, 11%) and 2S (n=4, 15%, p=0.727). Spinal cord ischemia symptoms were transient in 4 patients and minimal, non-disabling deficit in the majority of cases. Three patients suffered from paraplegia. Ten of 11 (91%) SCI patients had received a preoperative CSF drain. One CSF drain-associated complication occurred (2.6%); a spinal hematoma had to be evacuated on day 3 after the removal of the CSF drain due to spinal cord compression with paraplegia. The patient recovered with persisting mild motor weakness.
The number of vessel access complications was evaluated per patient, resulting in a higher complication rate in 2S (n=6, 23%) than in 1S (n=4, 7%; p=0.027). Two femoral artery occlusions and 2 hematomas occurred after open surgical access in patients with previous femoropopliteal or iliofemoral bypass surgery. In 340 renovisceral target vessels, there are 1 bleeding complication, 2 occlusions, and 4 non-occluding dissections. Renovisceral target vessel complications were not different in 1S and 2S. No target vessel occlusion by inadvertent TEVAR graft displacement or secondary graft migration in 2S was observed. The postoperative CT-angiogram showed an endoleak (any type) in 26 1S (43%) and 10 2S cases (39%, p=0.978). No type 3 inter-graft endoleak was detected between the TEVAR and FEVAR grafts. The postoperative parameters of the 30 day outcome are presented in Table 3.
Postoperative Morbidity of TEVAR and FEVAR Procedures.
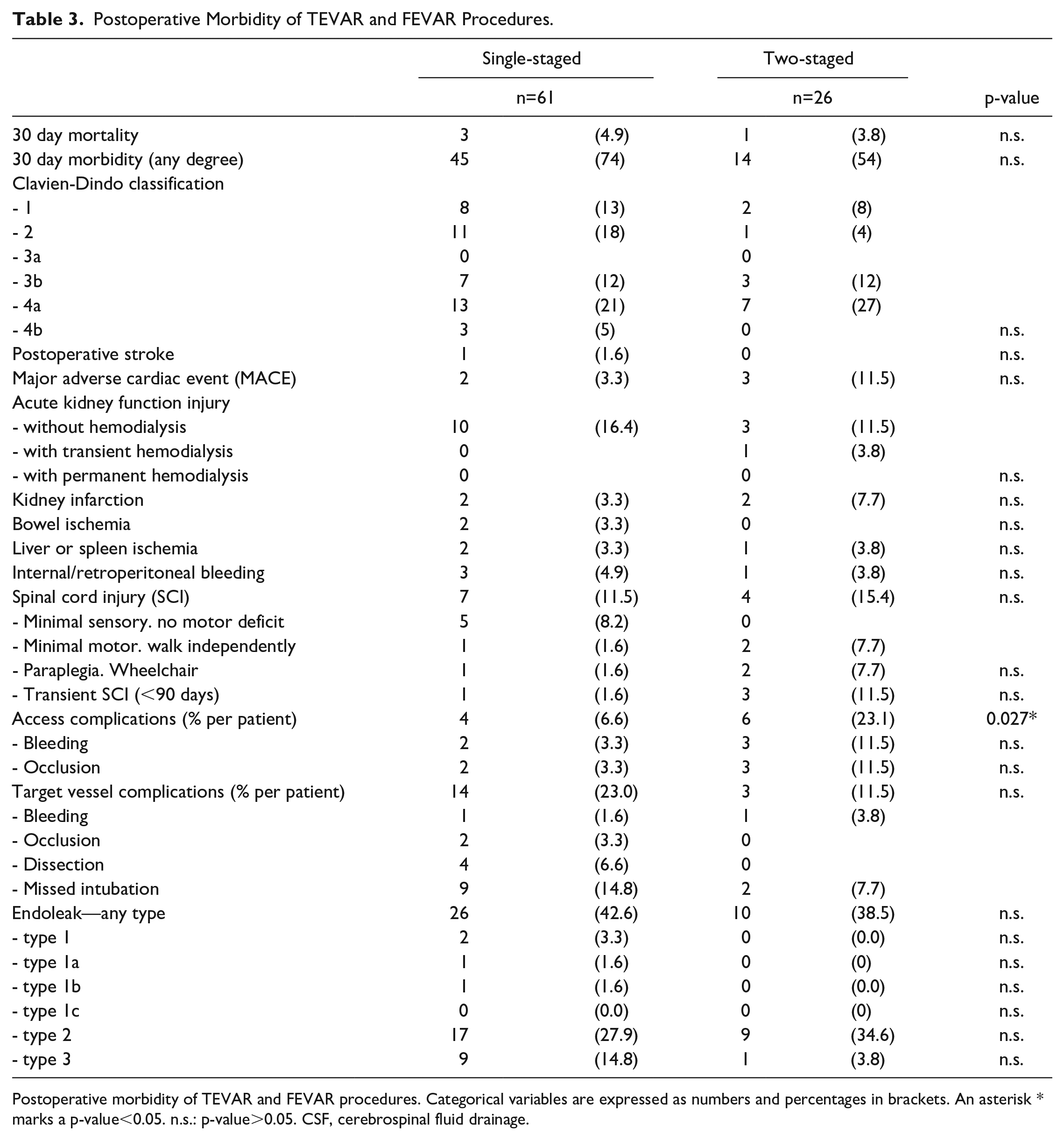
Postoperative morbidity of TEVAR and FEVAR procedures. Categorical variables are expressed as numbers and percentages in brackets. An asterisk * marks a p-value<0.05. n.s.: p-value>0.05. CSF, cerebrospinal fluid drainage.
Discussion
Fenestrated endovascular aortic repair has become a commonly used option for the endovascular treatment of aortic aneurysms involving the renovisceral segment.13 –16 In TAAAs with narrow renovisceral segments, or juxtarenal aneurysms with tortuous, thrombosed, or dilated supravisceral segments, space restrictions do not allow the implantation of a branched device. In these cases, TEVAR is performed prior to FEVAR, to create a proximal landing zone for the fenestrated graft. The combination of grafts demonstrated sufficient connective stability in pull-out bench tests and promising results in a smaller series of patients. 8 In our series, no type 3 endoleak between TEVAR and FEVAR grafts was observed in the short term. This study aimed to investigate technical aspects and postoperative morbidity of single- vs 2-staged procedures combining TEVAR and FEVAR.
The study populations from the 2 centers were homogenous regarding anthropometric parameters. The decision for a single- or 2-staged strategy is based on anatomical features, comorbidities, and patient preferences; but also on the use of standard or customized thoracic products logistics, and the framework of the respective health care system. A more proximal landing zone, resulting in a longer aortic coverage, favored 2-staged procedures (2S) in our study. A higher ASA class, a history of aortic surgery, and octogenarians were more frequent in 2S. As no off-the-shelf branched devices were used in this study, the mean interval of 83 days between TEVAR and FEVAR in 2S reflects the delay in the production of the custom-made FEVAR graft.
Antegrade upper extremity access was used more frequently in the early years of the study period and could mostly be avoided in the later years since steerable sheaths became routinely available. 9 In 2S, more TEVAR grafts were needed per case; the reasons included a longer aortic coverage and a higher probability for the need for distal extension of the thoracic device at the time of the secondary FEVAR implant as a result of foreshortening of the thoracic device in 31% of cases. One hypothesis of the study was that by a long coverage, the aortic configuration would straighten and pose a threat to the graft-graft connection at the thoracolumbar junction. The thoracolumbar, but not the infrarenal aortic angulations were reduced postoperatively. The Anaconda fenestrated device does not have nitinol rings in the middle part or longitudinal wires at all; therefore, it follows the aortic configuration of the renovisceral segment, whereas its proximal part remains anchored in the distal TEVAR graft. No inter-graft type 3 endoleak at the TEVAR/FEVAR junction or graft lacerations by anchoring hooks was observed in our short-term follow-up.
Postoperative general morbidity and mortality were not different between 1S and 2S, and similar to current studies by Verhoeven (7.8%) or Ulskar (4%).17,18 No aneurysm-related events occurred during the staging interval of the 2S group. However, it may be argued that the deliberate delay of therapy in a 2-stage- or a 3-stage strategy of secondary bridging stents or limb extensions may impose the threat of aortic complications in the meantime.
In multivariate analysis, no significant covariates were found among comorbidities, Crawford type, or aneurysm sac diameter in the event of death. Regarding morbidity, the rate of mild complications including Clavien-Dindo type 1 or type 2 was higher in 1S, but not in major complications (Clavien-Dindo 3 and 4). In 1S, the amount of prosthetic material implanted in a single intervention is larger, thus resulting in a more intense systemic response compared to a 2-stage aortic repair. Perioperative major adverse cardiac event (MACE) and stroke rates were in accordance with other studies (2-3.3%).19,20 The vascular access complication per patient rate in our study was higher in 2S than in 1S (p=0.027), but taking into account the former ones being a redo vascular access.
Spinal cord ischemia after FEVAR is considered one of the most devastating complications. Bisdas et al 21 demonstrated that thoracic aortic stenting was the most powerful determinant of SCI; therefore, it can be hypothesized that the combination of TEVAR and FEVAR represents a higher risk of SCI in TAAA patients. The overall rates of SCI in our study were 11% in 1S (n=7) and 15% in 2S (n=4). Three of 4 SCI patients in 2S had a delayed onset and recovered completely within 1 week. Noteworthy, only 3 patients suffered from permanent paraplegia following endovascular repair. Bertoglio et al 22 implemented a 3-staged strategy and reported a similar result in 1S (12%), a lower overall SCI rate in 2S (8%), but a similarly high portion of temporary symptoms and low permanent SCI. This is higher than in FEVAR alone. No SCI occurred among the 12 patients with TASP. Intraoperative MEPs were normal in SCI cases. In our study cohort, the aortic coverage was larger in 2S, but no correlation to SCI was found. In agreement with Gallitto et al, 23 we did not observe an increased incidence of SCI in patients who had previously undergone aortic surgery. Likewise, Kaushik et al 24 observed a lower SCI rate in patients with prior aortic procedures compared to patients without “treated” aortas. This may reflect the benefit of activation of the collateral system that occurs after aortic surgery. This hypothesis has already been confirmed in vivo. 25
Limitations of the study were its retrospective design, different local protocols, and the lack of long-term follow-up. In real life, perfect adherence to standardized protocols is not always possible. This is demonstrated by the fact that despite a CSF drain being deemed mandatory in our protocol for FEVAR/TEVAR combinations, the preoperative CSF drain rate was lower than 80%, due to dual antiplatelet therapy, spine deformities, or patient preference. Owing to a relatively small group of 2-stage interventions, we were not able to identify the contribution of each possible harmful or protective confounding variable. The 1S group contained only a low number of juxtarenal aneurysm, rendering the group comparison underpowered.
Conclusion
The combination of TEVAR and FEVAR using the Anaconda graft is feasible and safe, with acceptable morbidity in single- and 2-staged interventions. Aortic configuration does not change significantly after graft implantation in either way. A single-staged strategy is still feasible with excellent results, especially in Crawford 4, Crawford 5, or juxtarenal aneurysms. Two-staged repair is recommended in cases with long aortic coverage and a higher ASA class, but more TEVAR grafts are needed and the risk of access complications is higher. Follow-up data are needed to evaluate the long-term stability of the TEVAR/FEVAR interconnection.
Supplemental Material
sj-docx-1-jet-10.1177_15266028241255533 – Supplemental material for Technical Aspects and Outcome of Multi-Staged and Single-Staged Thoracoabdominal Fenestrated Endovascular Aortic Repair
Supplemental material, sj-docx-1-jet-10.1177_15266028241255533 for Technical Aspects and Outcome of Multi-Staged and Single-Staged Thoracoabdominal Fenestrated Endovascular Aortic Repair by Jan Paul Frese, Corinna Walter, Jan Carstens, Matthias Bürger, Andreas Greiner, Afshin Assadian, Sebastian Kapahnke and Jürgen Falkensammer in Journal of Endovascular Therapy
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
