Abstract
Purpose:
To describe snare-assisted vessel targeting to selectively overcome a dissection in the iliac bifurcation and gain antegrade access to the hypogastric artery (HA).
Technique:
The technique is demonstrated in a 64-year-old woman with an asymptomatic Crawford type III thoracoabdominal aneurysm. A 2-stage endovascular repair, consisting of a thoracic endovascular aortic repair (TEVAR) and a branched endovascular aortic repair was planned. In the control angiography after TEVAR, a disrupted plaque with consequent dissection in the right iliac bifurcation was detected. The perfusion of the common iliac artery and external iliac artery resulted impaired. The targeting of the right HA through a contralateral antegrade approach failed, whereas an ipsilateral retrograde approach was possible but unsuitable for therapeutic purposes. Using the catheter of the retrograde ipsilateral access, a snare from a contralateral crossover was cached and dragged into the HA, allowing the targeting of the vessels and further endovascular therapy with angioplasty and stenting. Follow-up 8 months postoperatively demonstrated the patency of the stents and well-preserved perfusion in the right iliac bifurcation.
Conclusion:
The snare-dragging technique can be used to gain access to vessels presenting challenging conformations or dissections. This application may be a valuable support for complex endovascular treatment in a variety of patients.
Clinical Impact
The snare-dragging technique can be used to gain access to vessels presenting challenging conformations or dissections. It allows the catheterization to be establish from the easiest and safest approach and then “transferred” from one access to the other. It avoids the risk of repeated loss of catheterization due to unstable and unfavorable working angles, and it saves time and radiation. It permits different material combinations, adapting to the available resources and materials. We believe that the current technique may increase the strategy spectrum available for endovascular therapy and complex endovascular procedures.
Keywords
Introduction
In the growing complexity of endovascular therapies, effective catheterization of vessels plays a central role. Challenging catheterizations increase material costs, operation time, and radiation exposure, and can necessitate additional vascular access or in extreme cases, more invasive therapies and open conversion.
Herein, we describe the use of the snare-dragging technique to deal with a challenging vessel targeting in a dissection of the iliac bifurcation.
Technique
The technique is demonstrated in a 64-year-old woman with an asymptomatic Crawford type III thoracoabdominal aneurysm. A 2-stage endovascular repair, consisting of a thoracic endovascular aortic repair (TEVAR) as stage 1 and a branched endovascular aortic repair (bEVAR) as stage 2 was planned. Past history included aortic valve and aortic root replacement, hypertension, atrial fibrillation and a lung adenocarcinoma treated with lobectomy and chemotherapy resulting in persistent mild dyspnea.
Diagnostic computed tomography angiography (CTA) scans showed a Crawford type III thoracoabdominal aneurysm and patent iliac arteries on both sides. Size and status (ie, tortuosity, plaques and calcifications) of the access vessels were proofed and stated to be suitable for the passage of the 24 French device. The caliber of the common iliac arteries (CIAs) and external iliac arteries (EIAs) was on both sides 10 and 8 mm, respectively. The TEVAR was performed through percutaneous access from both common femoral arteries. A 40 × 100 mm2 thoracic stent graft (E-vita thoracic 3G, JOTEC GmbH, Hechingen, Germany) was implanted through the right common femoral artery; the left common femoral artery was percutaneously accessed for diagnostic purposes.
After deployment of the TEVAR device, the completion angiography demonstrated a disrupted atherosclerotic plaque in the right CIA with a dissection flap covering the right iliac bifurcation, prolonging into the right EIA. The perfusion of the hypogastric artery (HA) and external iliac artery was impaired (Figure 1A).
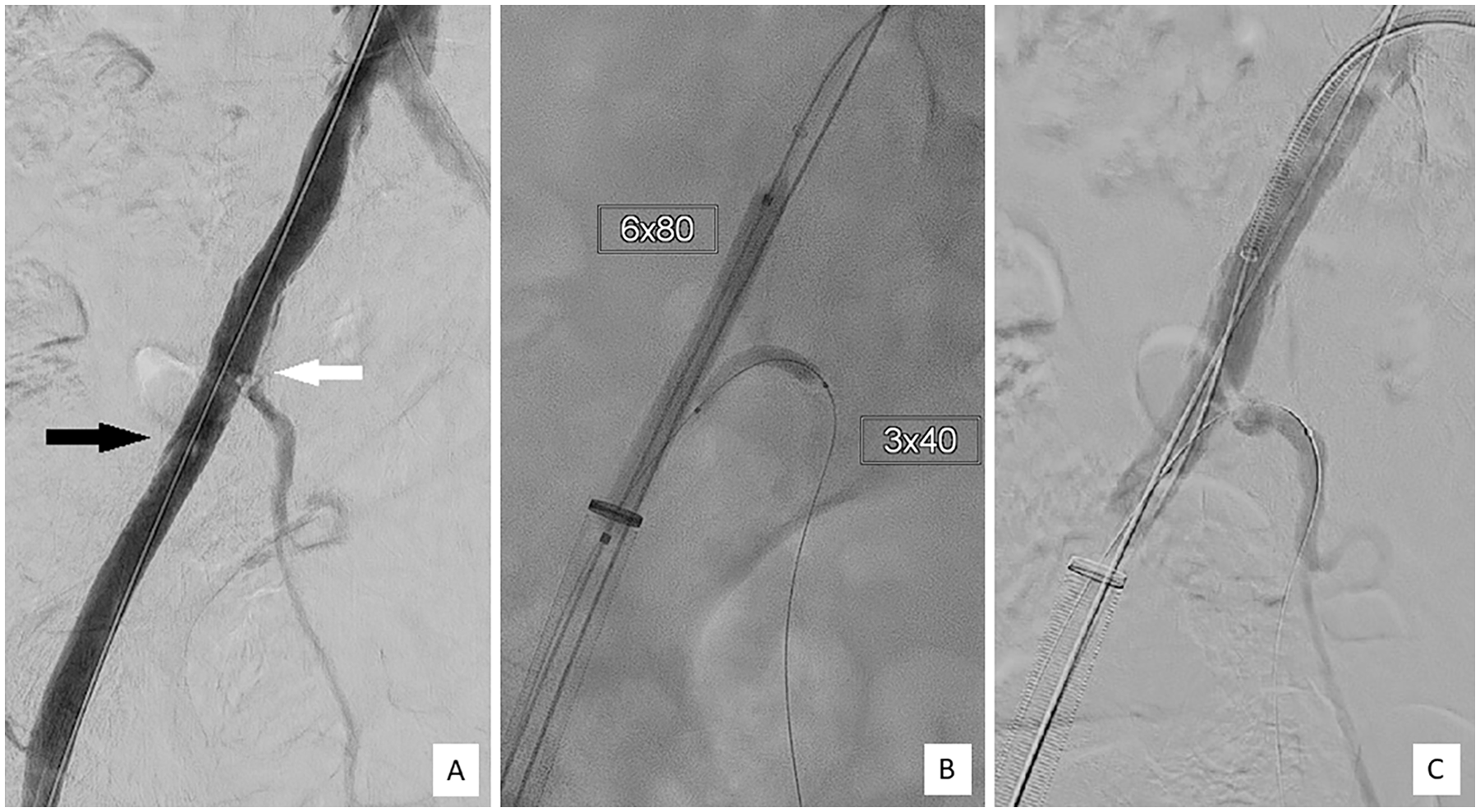
(A) Intraoperative iliac angiogram after implantation of the thoracic endovascular aortic repair (TEVAR) stent graft demonstrating the impaired perfusion of the hypogastric and external iliac artery due to a disrupted atherosclerotic plaque (black arrow) and dissection of the right common iliac artery (white arrow). (B) Ipsilateral retrograde catheterization of the right hypogastric artery (HA), kissing balloon angioplasty. (C) Persistence of the dissection flap and the reduced flow in the right external iliac artery (EIA) and HA.
An antegrade catheterization of the right HA from crossover was unsuccessful, despite use of several catheters, maneuvers, and a steerable sheath. The right HA was cannulated from an ipsilateral retrograde femoral approach. After retrograde catheterization, kissing balloon angioplasty was performed using a 6 × 80 mm2 balloon (Adminal Xtreme, Medtronic, Santa Clara, CA, USA) in the right CIA/EIA and 3 × 40 mm2 balloon (Pacific Plus, Medtronic, Santa Clara, CA, USA) in the HA (Figure 1B). The dissection flap and the reduced flow in the right EIA and HA persisted (Figure 1C). Any effort to gain antegrade access to the HA was hindered because of the conformation of the dissection flap, whereas retrograde access could be smoothly and repeatedly achieved.
From the left contralateral femoral artery, a snare (EN Snare, Merit Medical System, South Jordan, UT, USA) was antegradely delivered in the right CIA; from the ipsilateral femoral approach, a 0.035-inch guidewire (Terumo Medical Corporation, Somerset, NJ) was retrogradely passed through the snare-loop and conveyed to the right HA (Figure 2A). While advancing the 5-French cobra catheter (Glidecath, Terumo, Somerset, New Jersey, USA) over the wire to the HA, the snare-loop was tightened. The snare therefore captured the catheter. The catheter was advanced retrogradely beyond the lesion, steering and dragging the snare into the right HA (Figure 2B). The snare was withdrawn, its delivery catheter was left in the HA, and exchanged with a floppy 0.014-inch wire (Choice PT; Boston Scientific, Marlborough, MA, USA) (Figure 2C). Antegrade kissing balloon angioplasty was then performed twice. The previously used balloons (6 × 80 mm2 for the CIA/EIA and 3 × 40 mm2 for the HA) were maintained inflated for 3 minutes each time. Despite this maneuver, the disrupted atherosclerotic plaque and the dissection flap persisted, maintaining the impairment in the HA of the blood supply. The lesion in the CIA/EIA was therefore treated with a 8 × 60 mm2 self-expanding stent (Everflex, Medtronic, Santa Clara, CA, USA) advanced retrogradely over the wire from the right femoral artery. The contralateral wire in the HA was held in position for bail out parallel stenting. A floppy wire was passed crossing the stent struts into the right HA, a 6 × 80 mm2 balloon advanced, and the coaxial position verified with rotation angiography. The balloon was used to pre-dilate and adapt the stent struts, and the floppy wire was exchanged with a stiffer one, and a 4-French sheath was placed near the ostium of the HA. Under fluoroscopy, a 3 × 40 mm2 balloon expandable stent (Promus, Boston Scientific, Marlborough, MA, USA) was antegradely advanced and implanted between the stent struts and HA. The completion angiogram demonstrated the patency of both iliac stents as well as a good flow in the CIA, EIA, and HA (Figure 3).
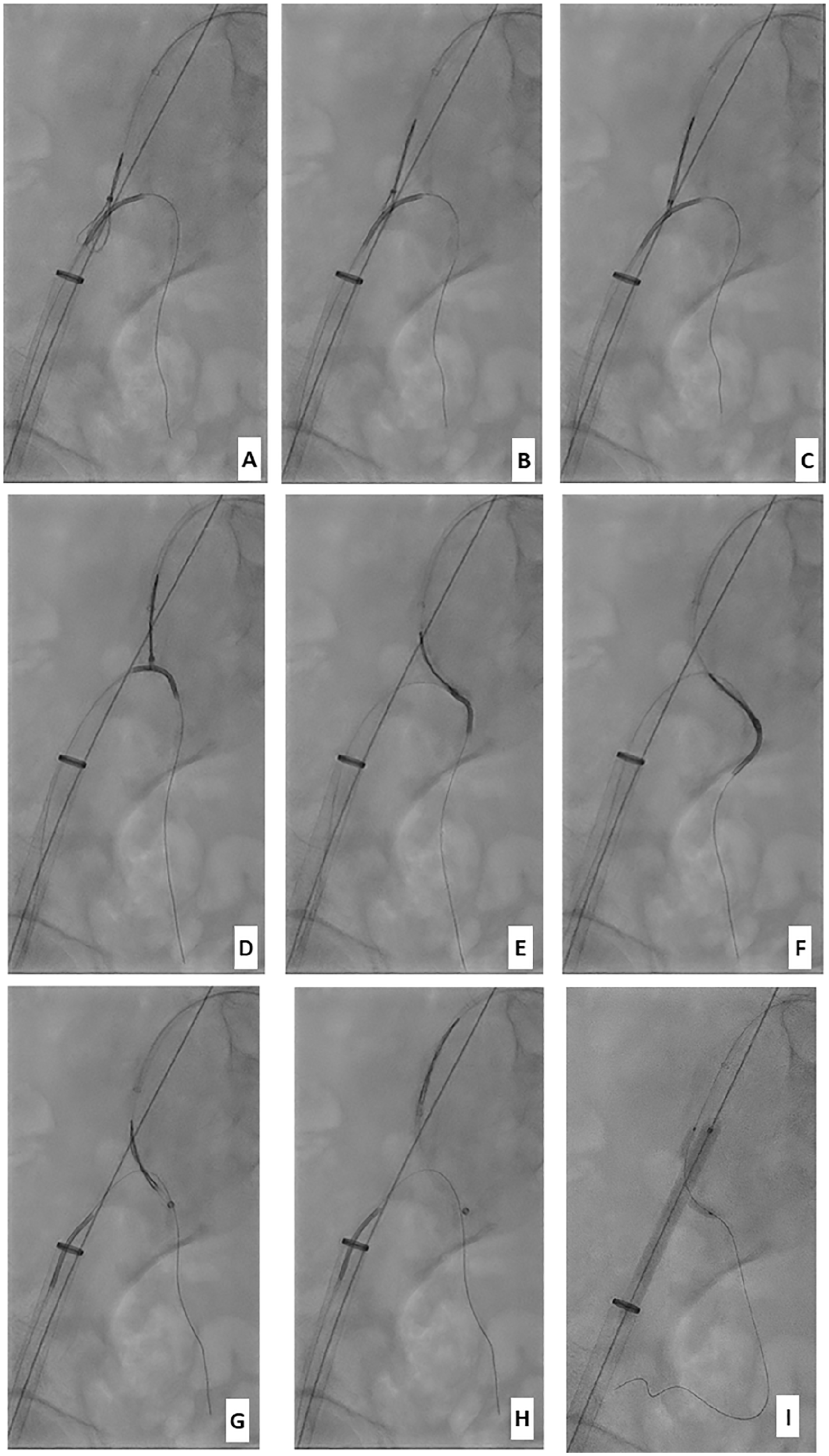
Snare-dragging technique: (A–C) Snare catching the catheter: snare from the left contralateral femoral artery. From the ipsilateral right femoral approach, the 0.035-inch-wire is passed through the snare-loop and conveyed to the right hypogastric artery (HA). (D–F) Snare-dragging: Advancing of the cobra catheter over the wire and tightening the snare-loop. The catheter is advanced beyond the lesion, steering and dragging the snare into the right HA. (G–I) Exchange materials: the snare is withdrawn, and its delivery catheter is left in the HA, and exchanged over the wire with further material.
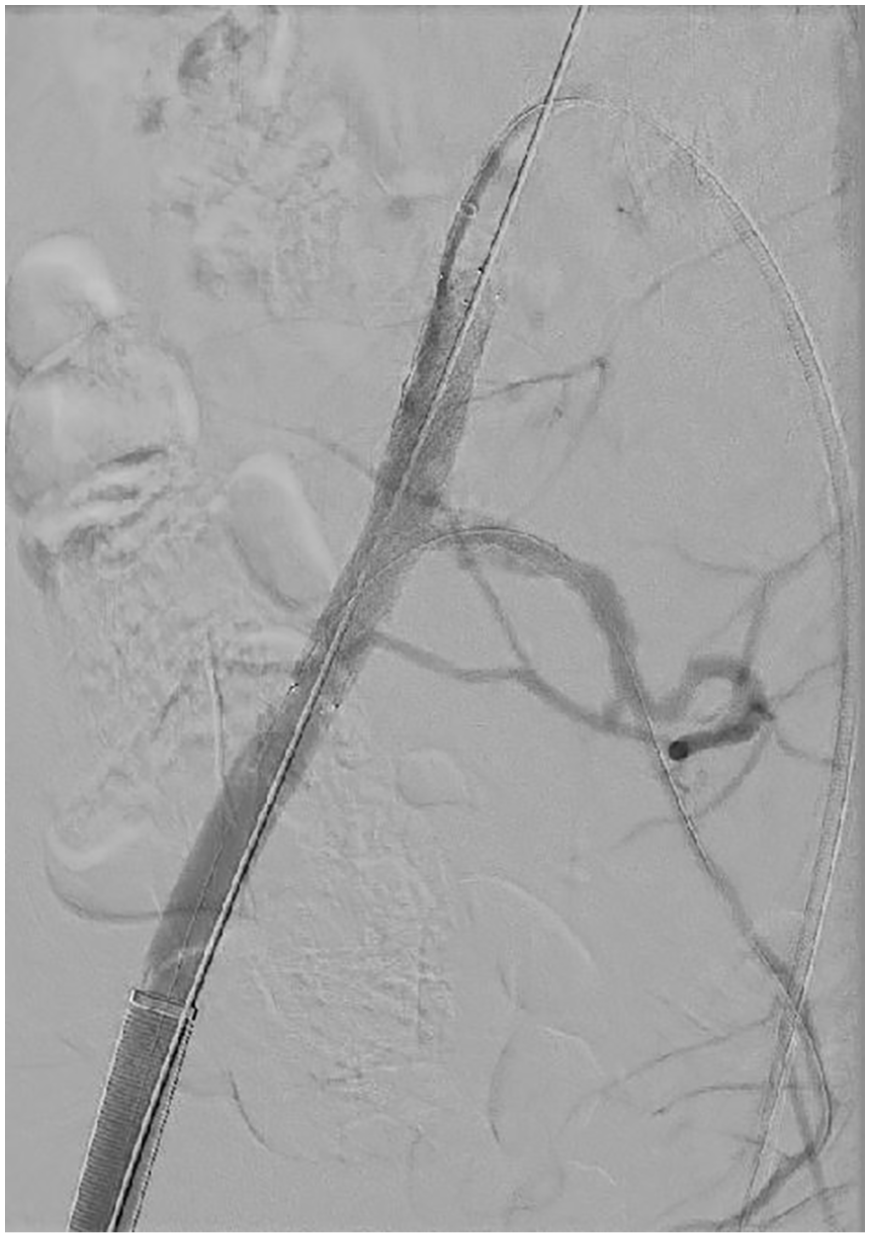
Intraoperative completion angiography after deployment of the stents, demonstrating the patency of the right external and hypogastric arteries.
The patient did not develop any sign of spinal ischemia or malperfusion postoperatively, and CTA 6 days after the procedure showed the patency of the stents and preserved flow in the right iliac bifurcation. The bEVAR implantation was uneventful, and no neurological disorders appeared. Further follow-up CTA at 3 months postoperatively and ultrasound evaluation 8 months postoperatively documented ongoing perfusion of the iliac bifurcation and enduring patency of the stents (Figure 4). Clinically, the patient did not develop any symptoms associable with spinal ischemia or a pelvic malperfusion.
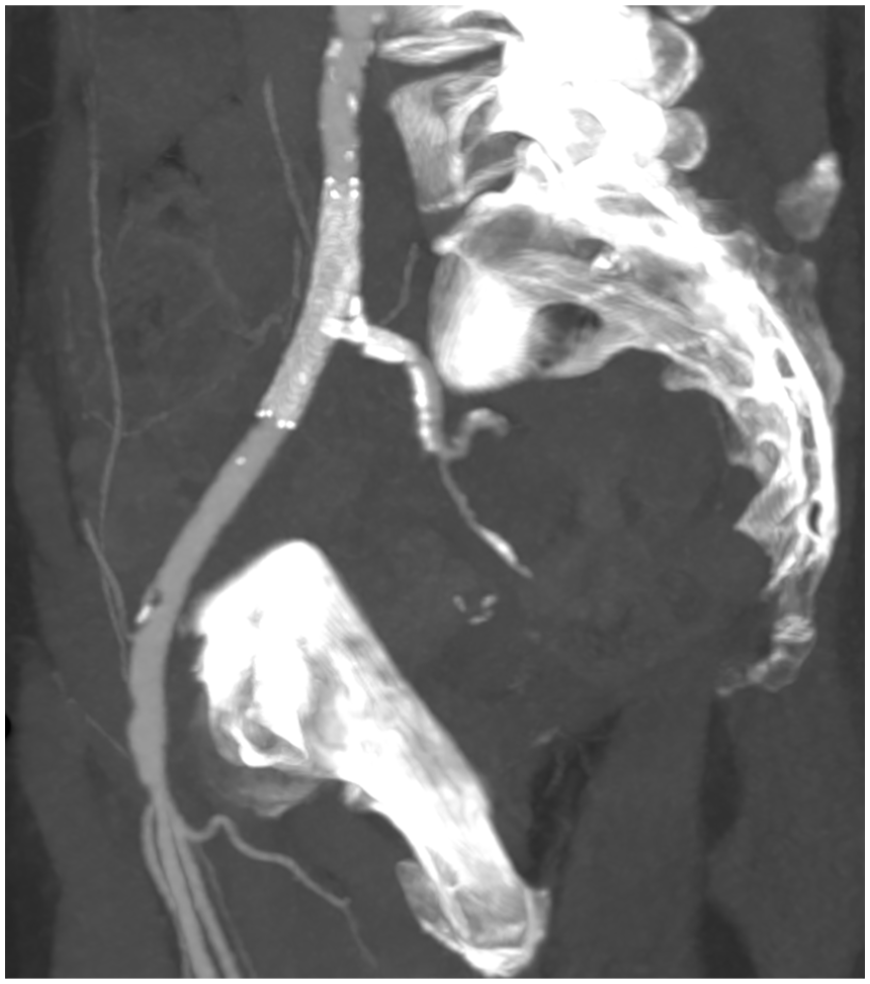
Follow-up computed tomography angiography (CTA) 3 months postoperatively showing patent right external and hypogastric iliac arteries after stenting, CTA reconstruction.
Discussion
Effective catheterization of target vessels plays an essential role in endovascular therapies. Challenging catheterizations increase material costs, operation time, and radiation exposure and can necessitate additional vascular access or in extreme cases, more invasive therapies and open conversion. Different tools have been described to overcome problematic ostium targeting, including a wide range of guidewires, catheters, maneuvers, and steerable sheaths. 1 In the case described here, we used the snare-dragging technique to achieve true lumen catheterization and overcome a dissection in the right iliac bifurcation. Rupture and dissection of the access vessels, in particular of the iliac arteries, represent a severe complication in endovascular procedures. 2 In TEVAR, the risk of iliac dissection and access complication is higher due to the use of a large sheath size (>18 French)3,4 and is described significantly greater in female gender. 5 Injuries of the iliac bifurcation can lead to significant perioperative morbidity and long-term pathology such as claudication, ischemia of the lower extremity, and pelvic underperfusion. Karch et al 6 reported that 32% of buttock claudication is caused by occlusion of the ipsilateral HA following aortomonoiliac grafting, one-third of these occlusions resulting from an intraoperative traumatic iliac dissection. The HA plays also an important role in the spinal collateral network. 7 The risk for spinal cord injury is known to increase with the extent of aortic repair; extrathoracic collaterals, including arteries originating from the iliac bifurcations, have been described by Shijo et al 7 as relevant for the development of a protective auxiliary perfusion. Therefore, the planned extensive endovascular repair with TEVAR and bEVAR with the associated risk of spinal cord ischemia represented a major indication for HA salvage in our case. Crucial in managing iliac bifurcation dissection is the preservation of EIA; however, there is increasing evidence that salvage of the HA should be attempted. 8 Endovascular techniques to preserve HA derive from experiences in treatment of CIA, EIA, and HA aneurysms9,10 and include kissing stents, parallel graft techniques, and iliac branch devices. A hybrid procedure to maintain HA perfusion consisting of direct suture of the stent graft to the CIA with an open extraperitoneal approach was described by Hinchliffe et al 11 in the context of aortoiliac endografting. More invasive options, such as open surgery, can be considered in selected cases.
In the presented case, the iliac vessels had been proofed before TEVAR and stated to be suitable for the access; however, the disruption of the atherosclerotic plaque in the right CIA resulted in a dissection of the right iliac bifurcation with the flap compromising the perfusion of the right EIA und HA. Using the snare-dragging technique, we steered and dragged a snare inserted from the left contralateral femoral artery into the right HA with a 5-French cobra catheter (Figure 2). An alternative to our strategy would have been a stent implantation in the CIA/EIA with deliberate occlusion of the right HA, relying on a compensation of the pelvic perfusion through the left HA, which was patent without relevant stenosis according to the preoperative CTA. However, because of the role of HA in the spinal collateral network, this strategy would have increased the risk of spinal cord injury for the patient after the second stage bEVAR procedure. The option of an open repair was abandoned in favor of the described successful technique.
The snare-dragging technique offers several advantages. It allows the successful catheterization to be “transferred” from one access to the other. It permits navigation to the target vessels, dragging the wire from the preferred access after establishing the catheterization from the easiest and safest approach. It avoids the risk of repeated loss of catheterization due to unstable and unfavorable working angles, and it saves time and radiation. It can be used with different material combinations, adapting to the available resources and materials “off the-shelf” and can be useful in various setups: as dissection, extreme tortuosity of vessel, or territories which cannot be accessed from a distal and retrograde approach, as during bEVAR in HA and visceral arteries.
We believe that the current technique may increase the strategy spectrum available for endovascular therapy and represents a valuable method to navigate endovascular wires to target vessels in complex endovascular procedures.
Conclusion
The snare-dragging technique is an off-the-shelf, feasible, and versatile solution to catheterize vessels with challenging conformations or dissections. This application may be a valuable alternative supporting vessel catheterization and true lumen targeting in selected patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
