Abstract
Purpose:
The purpose of this report is to describe the use of a double-branched custom-made iliac branch device (IBD) for the endovascular repair of an aorto-bi-iliac aneurysm with concomitant bilateral hypogastric aneurysms.
Technique:
A 61-year-old man on peritoneal dialysis underwent a computed tomography (CT) of the infrarenal aorta before planned kidney transplantation. The CT showed an asymptomatic aorto-bi-iliac aneurysm of 54 mm involving the hypogastric artery (HA) bilaterally (right HA 31 mm; left HA 40 mm). The treatment consisted of an endovascular aortic repair (EVAR) and the bilateral implantation of custom-manufactured IBDs with double inner branches to preserve both superior and inferior gluteal arteries. At 1 year follow-up, the patient remains free of symptoms and the postoperative CT showed a successfully excluded aneurysm with patent bridging stent grafts to all HA branches.
Conclusion:
The bilateral implantation of double-branched IBDs is a feasible technique. Preservation of both hypogastric arteries and its branches can be achieved with this technique and therefore decrease the risk of buttock claudication and other ischemic complications.
Keywords
Introduction
Approximately 18% to 40% of all abdominal aortic aneurysms (AAAs) are associated with unilateral aneurysm of the common iliac artery (CIA). In up to 12% of the cases, bilateral CIA aneurysms occur. 1 Concomitant aneurysms of the hypogastric artery (HA) are found in 29% of the cases. 2
In the past, aorto-bi-iliac aneurysms treated by endovascular aortic repair (EVAR) required coil embolization or ostial stent coverage of the HA to extend the distal landing zone of the aortic stent graft to the external iliac artery (EIA). However, the resulting occlusion of the HA carries with itself a considerable risk of buttock claudication, erectile dysfunction, and more pelvic ischemia-related risks. 3 In 36.5% of the cases, the bilateral occlusion of the HA results in buttock claudication. 4 Iliac branch devices (IBDs) were developed as an alternative option to preserve antegrade flow to the HA and its branches. Both unilateral and bilateral implantation of IBDs have been shown to be feasible and safe for the treatment of CIA and HA aneurysms. 5 Furthermore, the implantation of IBD has been safely used for treatment of HA aneurysms as well. 6 In cases of inadequate landing zone at the main stem of the HA, the available off-the-shelf IBDs have to be extended in one of the main branches, that is, superior or inferior gluteal artery (SGA and IGA). Bilateral implantation of an IBD has shown a 7.5% risk of pelvic ischemia-related symptoms. 7 We describe the bilateral use of a double-branched custom-made IBD for the repair of an aorto-bi-iliac aneurysm with concomitant bilateral aneurysm of the HA. The goal is to preserve both SGA and IGA when treating bilateral HAA aneurysms to decrease the risk of ischemic complications.
Technique
A 61-year-old man on peritoneal dialysis underwent a computed tomography angiography (CTA) of the infrarenal aorta before planned kidney transplantation. The CTA coincidentally revealed an AAA of 54 mm, with a concomitant aneurysm of the CIA (right 55 mm, left 47 mm) and of the HA (right 31 mm, left 40 mm).
An endovascular approach was chosen to preserve the functionality of the peritoneal dialysis. As both HA were aneurysmal, there was a lack of distal landing zone and EVAR was challenging. Lacking off-the-shelf options, the planned therapy consisted of an EVAR and the bilateral implantation of custom-made IBDs with a shotgun design and double inner branches to the SGA and IGA (Figure 1).
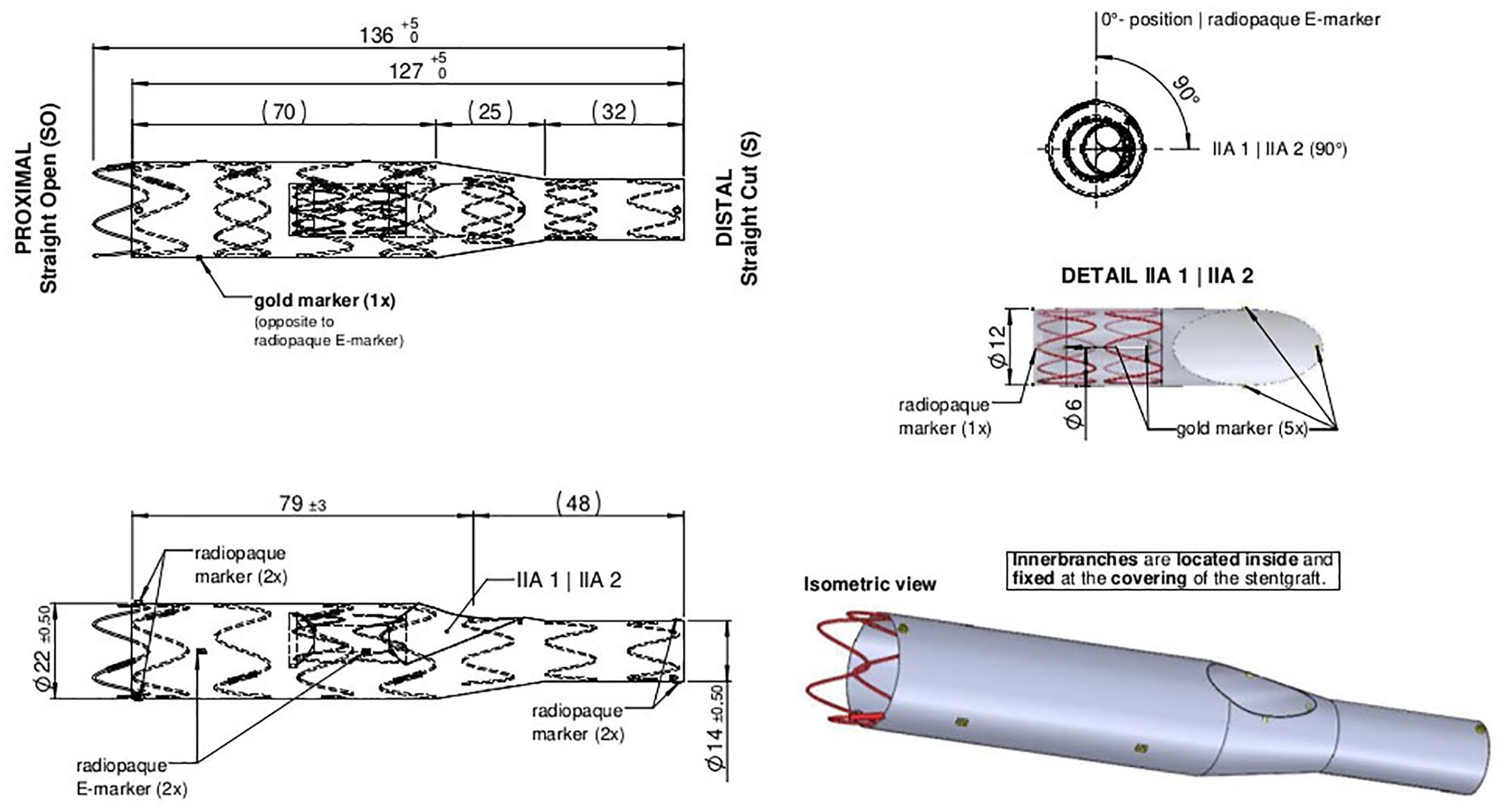
Technical drawing of the custom-made IBD with double inner branches. IIA, internal iliac artery; IBD, iliac branch device.
Bilateral common femoral artery access was obtained using a percutaneous technique with ultrasound guidance. First, the standard EVAR was performed as follows: A self-expanding stent graft (Jotec E-tegra; JOTEC GmbH, Hechingen) was implanted through the right common femoral artery over an Amplatz Super Stiff wire (Boston Scientific, Natick, MA) and was extended to the CIA bilaterally.
The axillary artery was cannulated after axillary cutdown. The custom-made IBD for the left side was advanced through the left iliac axis and deployed to land approximatively 1 cm above the ostium of the HA with the distal end of the inner branches. The first branch of the custom-made IBD was cannulated using a Terumo radiofocus glidewire (Terumo, Somerset, NJ). An 8F, 90-cm long sheath (COOK Medical, Bloomington, IN) was advanced into the branch and the HA. To avoid an endoleak, a small branch of the HA was coiled (Concerto Helix; Medtronic, Plymouth, MN). After identifying the landing zone in the IGA, 2 (7 mm × 79 mm and 7 mm × 59 mm) Viabahn VBX (W. L. Gore & Associates, Newark, DE) were implanted over a Rosen wire (COOK Medical).
The second branch designated for SGA was then cannulated and extended to the SGA with 2 (8 mm × 79 mm and 7 mm × 79 mm) Viabahn VBX (W. L. Gore & Associates). Control angiography showed a migration of the distal bridging stent graft (bSG) to the IGA. After re-cannulation of the IGA and coiling of an additional small branch of the HA (Concerto Helix; Medtronic), the bSG was extended with a 6 × 59 VBX (flared proximal with a 7 mm ballon). Thereafter, the branch was relined with a self-expandable stent Everflex 7 mm × 60 mm stent (Medtronic).
In the same fashion, the custom-made IBD for the right HA was inserted and implanted. Both inner branches were cannulated and extended with bSG of adequate length and diameter in a similar manner as described for the left HA.
After complete deployment of the IBDs, angiographic control detected a type Ib endoleak at the right EIA. The right IBD was extended with an Endurant II limb (Medtronic).
The completion angiography showed no sign of endoleak with a complete exclusion of the aorto-bi-iliac and of the bilateral HA aneurysms (Figure 2). All bSGs to the 4 branches of the HA were patent.
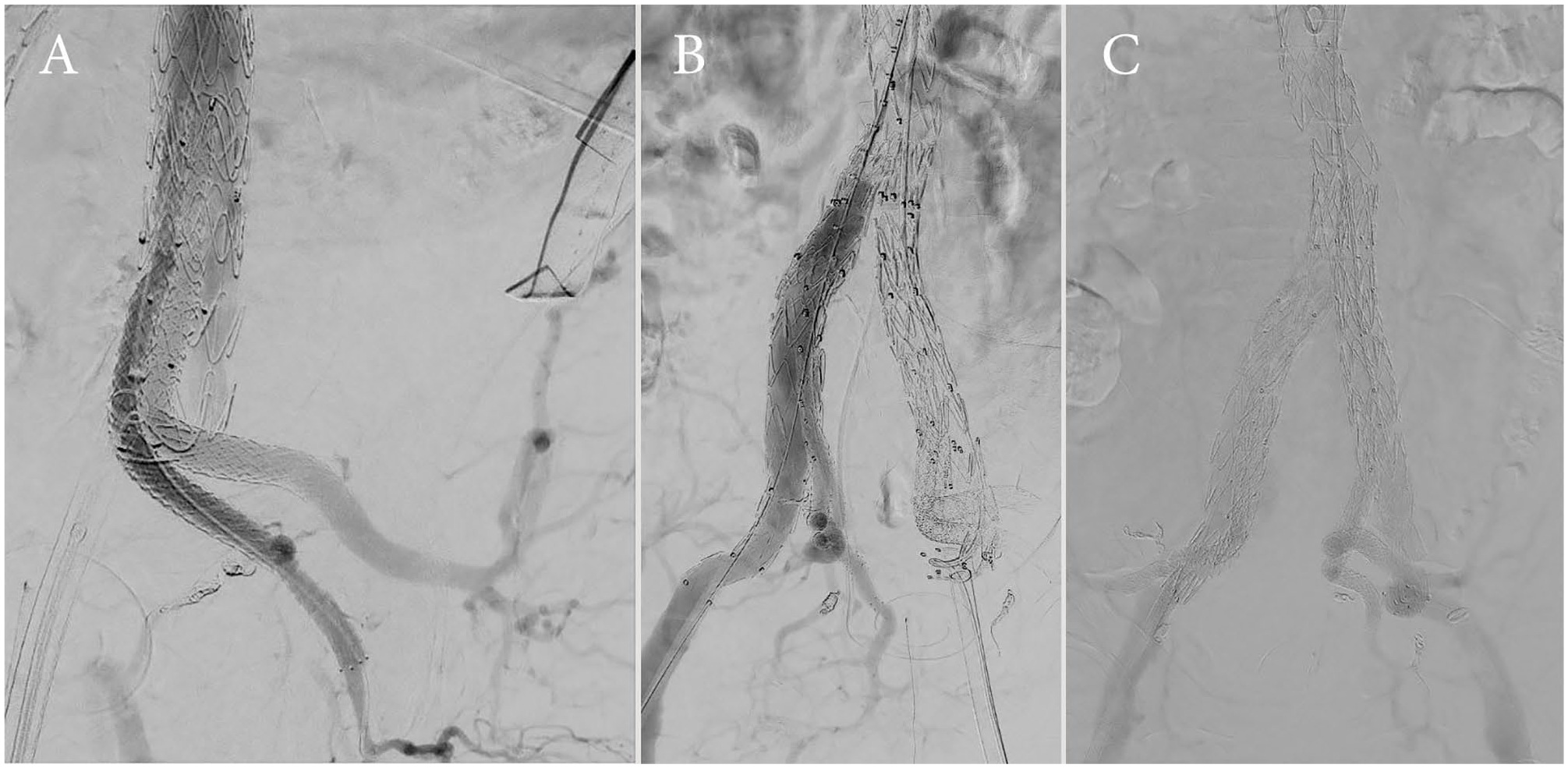
Completion angiography shows successful aneurysm exclusion and patency of both hypogastric branches on the left (A) and the right side (B), as well as no signs of relevant endoleaks on both sides (C).
The total procedure time was 576 minutes, whereas the total fluoroscopy time was 172 minutes with a dose area product of 118 945 mGy*cm². In total, 87 mL of iodinated contrast medium was used during the procedure.
The early postoperative CTA showed an endoleak type II via the inferior mesenteric artery. Successful coil embolization was achieved in a follow-up intervention.
At 1 year the patient remains free of symptoms of leg or pelvic ischemia. The postoperative control CTA showed a successfully excluded aneurysm with patent bSG to all HA branches with a persistent type II endoleak, via lumbar arteries (Figure 3). As the aneurysm sack continued, shrinking the endoleak was considered as benign and left untreated. There were at no time any symptoms of pelvic ischemia.
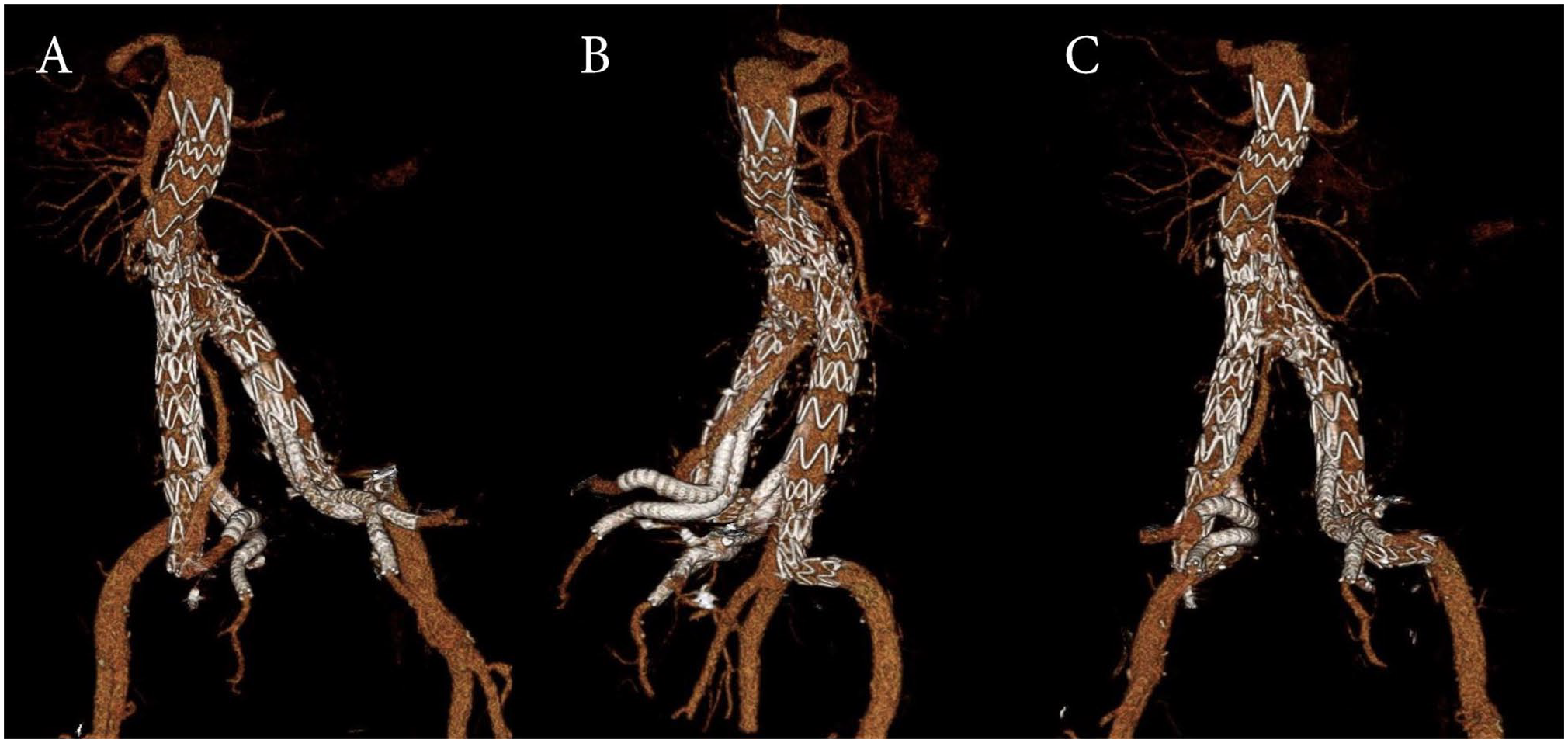
3D reconstruction of the postoperative CT-A visualizes the positioning of both custom-made IBDs. (A) View from the left showing an endoleak type II related to the EVAR procedure. (B) View from the right, and (C) view from posterior. CT, computed tomography; EVAR, endovascular aortic repair; IBDs, iliac branch devices.
Discussion
The occurrence of aneurysms of the CIA and of the HA concomitant to AAA is not uncommon.1,2 Their EVAR can end in sacrificing the HA with a considerable risk of buttock claudication, erectile dysfunction, and bowel ischemia.4,5 Off-the-shelf IBDs have been developed as an alternative option to the coil embolization or ostial stent coverage of the HA to preserve antegrade flow to the HA and its branches. In cases of inadequate landing zone at the main stem of the HA, the embolization of one of the main branches of the HA is mandatory.
We describe the case of a successful treatment of bilateral aneurysms of the HA by the implantation of a custom-made IBD. The present case demonstrates the feasibility of the implantation of a quadruple custom-made IBD as a valuable alternative to the embolization of one of the main branches of the HA. Its shotgun design allowed the preservation of the antegrade flow to both SGA and IGA, resulting in a lack of ischemic symptoms at 1 year postoperatively. The planning criteria were identical to a standard EVAR and IBD. The required ostium diameter of each HA should be 10–12 mm. Furthermore, adequate landing zones in diameter and length of the SGA and IGA are necessary.
Even though this technique is feasible, there are some issues to take into consideration. One of the drawbacks of the described technique is the time it requires. However, in selected cases, bilateral implantation of IBD can be staged.
Furthermore, the cannulation of the second branch of both IBDs revealed to be more challenging and time-consuming when compared with that of the first branch. A possible explanation of this issue could be an external compression of the orifice of the inner branch due to the previously placed parallel bSG. Precannulating the custom-made IBDs, as often the case in fEVARs or in off-the-shelf IBDs, could facilitate more efficient cannulation of the branches and leading to shorter operation time and lesser fluoroscopy time. Furthermore, it may decrease the risks of intraoperative dissections while cannulating.
Extending the endovascular treatment possibilities through improving this technique may give patients with a high risk for open surgery a safer treatment alternative
Conclusions
The bilateral implantation of a double-branched custom-made IBD is a feasible, but technically challenging treatment option for aorto-bi-iliac aneurysms with concomitant aneurysms of the HA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A. Oberhuber has received travel and research funds from JOTEC GmbH (Hechingen, Germany).
