Abstract
Introduction:
Initiating an endovascular aortic program for treatment of complex aortic aneurysms with fenestrated and branched grafts (FB-EVAR) is challenging. Using a Proctor is one option for training and development of the team. However, this approach has not been formally analyzed. The aim of this study was to analyze the learning curve and the effect of the Proctor regarding safety and effectiveness in FB-EVAR.
Methods:
A single-center retrospective cohort study was performed, including all consecutive elective patients submitted to FB-EVAR (including both thoraco-abdominal-TAAA and complex abdominal aortic aneurysms-C-AAA) from 2013 to 2021. Patients were divided into 2 groups, the first operated with the Proctor present and the second without. Primary outcomes were 30-day mortality (safety) and technical and procedure success (efficacy). Secondary outcomes included treatment performance (procedure time, blood loss, contrast, and radiation use), re-interventions, aneurysm shrinking, target vessel patency, 30-day mortality, aneurysm-related mortality, and overall mortality.
Results:
Overall, 105 patients were included in the study, 35 operated with Proctor and 70 operated without. The first 20 patients were operated always with the Proctor, and the remaining were operated with the Proctor selectively. Mean age was 71.8 (±7.3) years and 95 patients were male (90.5%). Overall, 62 (65%) patients had C-AAA or extent IV TAAAs and 43 (35%) had extensive TAAAs. There were no significant differences regarding 30-day mortality (Log Rank=0.99), technical success (p=0.4), or procedure success (p=0.8). Mean surgical time was longer in the non-Proctor group (p=0.005), as well as significant intra-operative blood loss (p=0.042). Contrast use (p=0.5) and radiation (p=0.53) were non-significantly different between groups. There were no significant differences regarding length of stay (p=0.4), major adverse events (p=0.6), target vessel patency (Log Rank=0.97), early (p=0.7) and late endoleaks (0.7), aneurysm shrinking (p=0.6), re-interventions (p=0.2), and overall mortality (Log Rank=0.87).
Conclusion:
In our experience, the use of a Proctor to start and accompany our complex endovascular aortic program for FB-EVAR was both safe and effective and may serve as a template by other countries and centers that aim to developing their programs.
Introduction
Endovascular treatment of thoraco-abdominal and complex abdominal aortic aneurysms is now considered the first-line therapy in most centers.1–3 Initially reserved for patients with prohibitive surgical risk, fenestrated and branched endovascular repair with custom-made devices have allowed for excellent results and treatment has been extended to lower risk patients and for more difficult anatomies.1,3
These procedures are technically complex, as they implicate careful sizing and planning, precise deployment of endografts and stents, and visceral artery manipulation. In addition, high available in-house stock of endovascular material is needed as well as expertise and ability to perform bailout procedures when necessary to achieve technical and treatment success.1–5 Furthermore, they require a high investment from healthcare providers, teamwork, and training and should be performed in experienced and high-volume centers.1,2,5
The last 2 decades have shown a high increase in different centers performing these procedures; however, it is unclear how to start a complex endovascular aortic program and which methods are safe and effective for both patients and healthcare providers. Some centers have opted for formal training of physicians in high-volume experience centers before starting their own program and starting to perform these procedures under the supervision of a highly experienced surgeon, a Proctor. To date, no data have been published on this learning curve accompanied by a Proctor.
In view of a wider experience and expertise in endovascular management of both occlusive and aneurysmal disease, our program was started under the guidance and support from one Proctor (EV—co-author in the manuscript) in 2013. The local surgical team prepared, reviewed, and proposed the endoprosthesis design in all cases, which was reviewed by the Proctor. The aim of this study was to report our current results of fenestrated and branched endovascular repair of thoraco-abdominal and complex abdominal aneurysms, analyzing the learning curve and the effect of the Proctor regarding safety and effectiveness.
Materials and Methods
This study followed the reporting guidelines from the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement for cohort studies. 6
Study Design, Setting and Participants
A single-center retrospective cohort study was performed. The study was conducted from January 2013 to January 2021. All consecutive, elective patients submitted to fenestrated or branched endovascular aortic repair (FEVAR and BEVAR, respectively) for thoraco-abdominal or complex abdominal (para-renal or juxta-renal) aortic aneurysms were included.
Patients admitted with ruptured aortic aneurysms, mycotic aneurysms, connective tissue disease, with aortic arch aneurysms submitted to aortic arch endovascular repair with branched or fenestrated grafts or patients submitted to endovascular repair with parallel grafts (chimneys, periscopes, and sandwich techniques) were excluded from this study.
Our department is one of largest in the country and accommodates a significant part of all referrals to vascular surgery including complex aortic diseases. The complex aortic endovascular repair program started in our center in 2013 under the supervision of a highly experienced Proctor (EV). All fenestrated and branched aortic repairs were performed using the COOK®Medical Zenith® Fenestrated platform. At our center cases are discussed in an Aortic Team composed of vascular surgeons, cardiac surgeons, and dedicated anaesthesiologists. Moreover, cases are also discussed with the intensive care medicine department for postoperative care of patients. Case planning is performed with the COOK® Medical planning center. In the beginning of our learning curve, the Proctor was present in every case. As the team gained experience and autonomy, more cases were performed independently except for selected cases considered more complex in which the Proctor assisted until the current date.
To analyze the learning curve and the outcomes of initiating a complex endovascular aortic program with a Proctor, the patient cohort was divided into 2 groups, the first including all patients treated with the Proctor and the second including all patients treated without the Proctor. Cases were performed in an angiography suite equipped with fusion imaging (Phillips®Azurion). In all cases, bilateral common femoral artery cutdown access was used with purse string sutures placed to allow for rapid reperfusion of the limbs. In BEVAR and selected FEVAR cases, the left axillary artery was used for antegrade catheterization of target vessels.
In all thoraco-abdominal cases, preventive protocol for spinal cord ischemia was implemented, including procedure staging whenever possible, placement of a lumbar cerebrovascular fluid (CSF) drain with a target medullary pressure ≤10 mmHg, target hemoglobin level ≥10 mg/dL, peripheral O2 saturation >95%, mean arterial pressure >90 mmHg, and medullary perfusion pressure ≥80 mmHg. Patients were monitored postoperatively in an intensive care unit at least for 24 hours.
In our center F/BEVAR is the preferred choice for patients with a thoraco-abdominal aneurysm, except for patients with connective tissue disorder, young (<60 years) and very low surgical risk, patients with shaggy aortas or mycotic aortic aneurysms. For patients with complex abdominal aortic aneurysms, FEVAR is preferred for older and higher-risk patients.
Variables, Data Sources/Measurement, Bias, and Study Size
Data were collected prospectively in a prepiloted case report form which was uploaded into a database and maintained regularly. Collected data included patient demographics, risk factors, aneurysm location, and type (thoraco-abdominal extent according to the Safi modified Crawford classification, and complex abdominal as para-renal or juxta-renal), 7 maximum aortic diameter, previous aortic surgery, previous abdominal or thoracic surgery, presentation of the aneurysm, American Society of Anaesthesiology (ASA) risk classification, procedure details (procedure time, radiation and contrast dose, blood loss), techniques used (including number of fenestrations and/or branches), aortic extension repair, patency of target vessels, endoleaks, postoperative complications, intensive care unit length of stay, hospital length of stay, re-interventions, aneurysm shrinking, aneurysm related mortality, 30-day mortality, and overall mortality.
For sub analysis regarding aneurysm extension, aneurysms were divided in 2 groups: extensive thoraco-abdominals—eTAAA (TAAAs I-III and V) and complex abdominal—C-AAA (Juxta and Para-renal aortic aneurysms and extent IV TAAAs). Furthermore, for analysis of the learning curve effect, a time point was established dividing the cohort in 2 groups according to the number of patients treated “autonomously” (ie, first half of the learning curve includes all patients treated until 35 patients had been treated without the Proctor present).
Follow-up was performed with a postoperative computed tomography angiography (CTA) in the first 30 days followed by annually CTAs or every 6 months if any concerns were raised (endoleaks needing monitoring for example). Patients were seen in an outpatient clinic every 6 months with abdominal ultrasound to check for target vessel patency and aneurysm diameter.
Technical success was defined as correct deployment of all aortic devices, catheterization, and stenting of all target vessels, patency of all target vessels and absence of type I or type III endoleaks at completion angiography. Major adverse events were defined as death, myocardial infarction, respiratory failure, renal failure requiring dialysis, bowel ischemia, major stroke, or definitive paraplegia occurring at 30 days. Procedure success was defined as technical success without complications at 30 days, that is, no type I or III endoleaks, no procedure-related adverse events, and no major complications.
Study Outcomes
All outcomes were analyzed separately according to the treatment group (“with Proctor” versus “without Proctor”). In addition, this was further analyzed according to aneurysm location (thoraco-abdominal aortic aneurysm extent I-III or V versus complex abdominal and extent IV thoraco-abdominal aortic aneurysms).
Additional analysis
A further analysis was performed for the primary outcomes comparing 2 time periods, a first when patients were operated exclusively with the Proctor and a second when the patients were operated selectively with the Proctor.
Statistical Analysis
Statistical analysis was carried out using STATA version 16.1 (Statistics/Data analysis, StataCorp LLC, Texas, USA). Continuous variables are presented as mean ± standard deviation when normally distributed and median (interquartile range-IQR) when not. Categorical variables are expressed in numbers (percentage). Students t-test was used when comparing continuous variables and the χ2 test to compare categorical variables, however, when appropriate, the Mann-Whitney or Fisher Exact test were used instead, respectively.
Survival and patency rates were analyzed using Kaplan-Meier graphs and Log Rank test for group comparison. For patency rates, a competing risk regression was performed using mortality as the competing risk. A logistic regression was performed for the primary outcomes to adjust for confounders when comparing groups. Additionally, baseline demographics that were statistically different in univariate analysis were included in the regression model. Results were presented as odds ratio (OR) with 95% confidence interval (CI). All analyses were considered statistically significant if a 2-tailed p-value < 0.05 was observed. All patients provided informed consent before treatment and Institutional Review Board approval was waived for this study.
Results
Demographics and Risk Factors
During the study period, approximately 1200 aneurysms were treated. Of these, 300 were complex (thoraco-abdominal or complex abdominal aortic aneurysms), of which 200 patients were submitted to endovascular or hybrid repair and 100 patients were submitted to open repair. In all, 105 patients fulfilled the inclusion criteria for this study. The remainder endovascular cases that were not included in the study were either hybrid repairs, aortic arch endovascular repairs, patients submitted to parallel graft techniques, or urgent treatment with off-the-shelf devices (Figure 1).
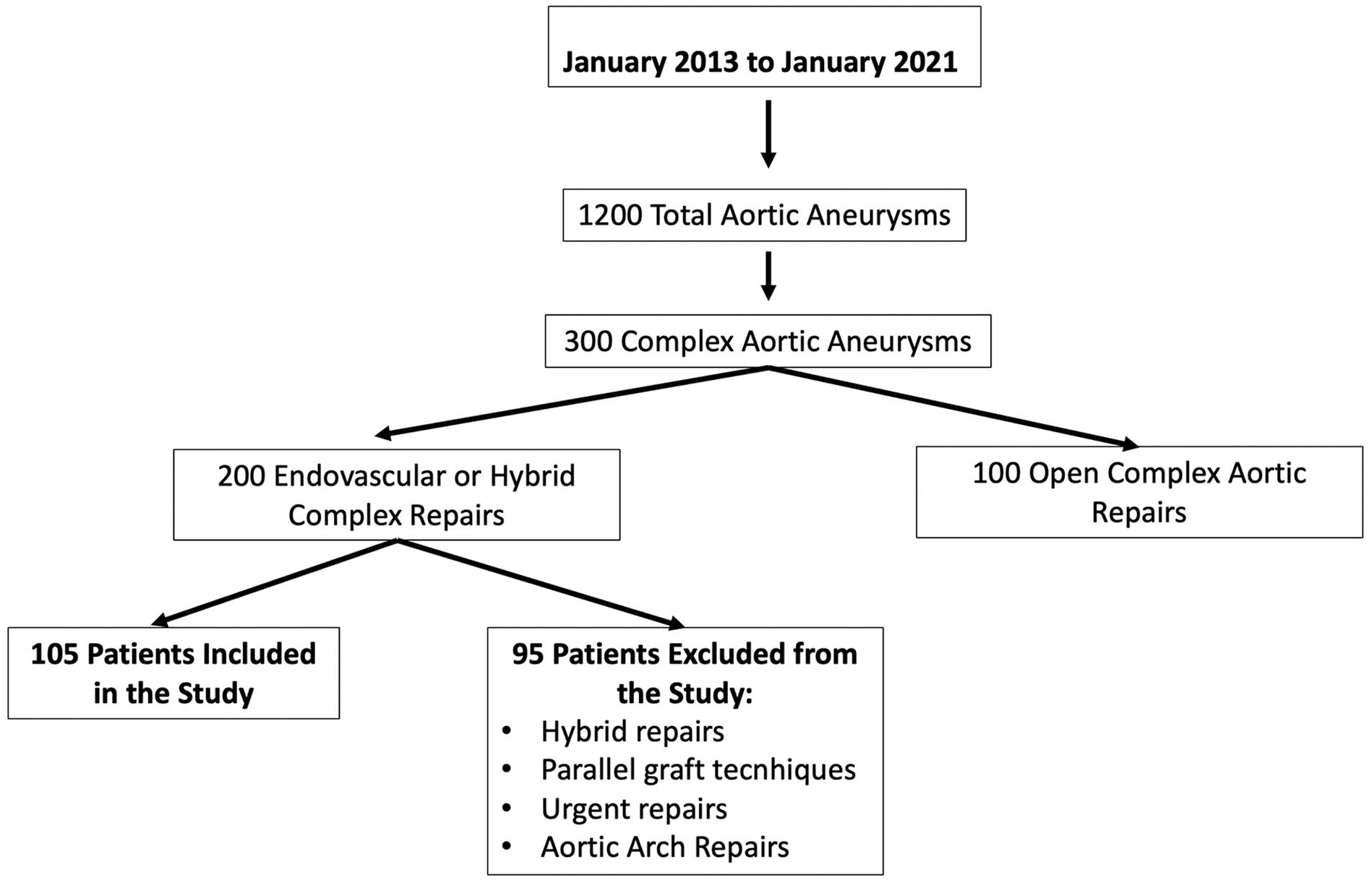
Volume of aneurysms treated during the study period.
The study cohort comprised of 105 elective patients submitted to fenestrated and branched endovascular aortic repair. Thirty-five (33.3%) patients were operated with the Proctor present, while 70 (66.7%) were operated without. The first 20 patients were operated with the Proctor present and thereafter the rate of patients operated without the Proctor increased gradually (Figure 2).
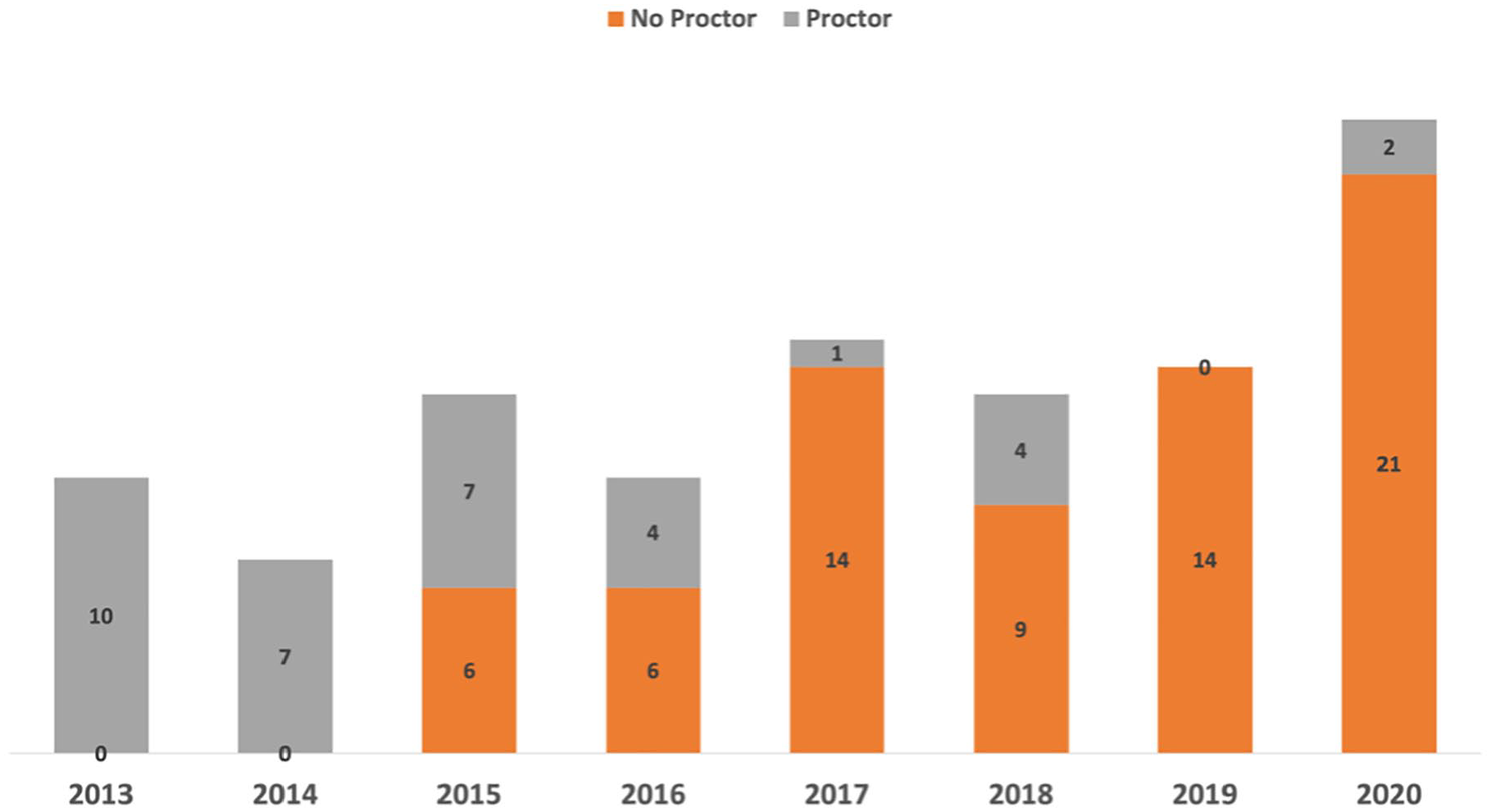
Case volume per year. In gray patients operated with the proctor in orange patients operated without the proctor.
Mean age was 71.8 (±7.3) years and 95 patients were male (90.5%). Patients risk factors are detailed in Table 1. Most patients (96%) were ASA risk III. Overall, there were no significant differences regarding demographics and risk factors, except for valvular heart disease which was more frequent in the Proctor group (22.8% versus 7.1%, p=0.02) and cerebral vascular disease, which was more frequent in the non-Proctor group (18.6% versus 2.9%, p=0.03).
Demographics and Risk Factors.
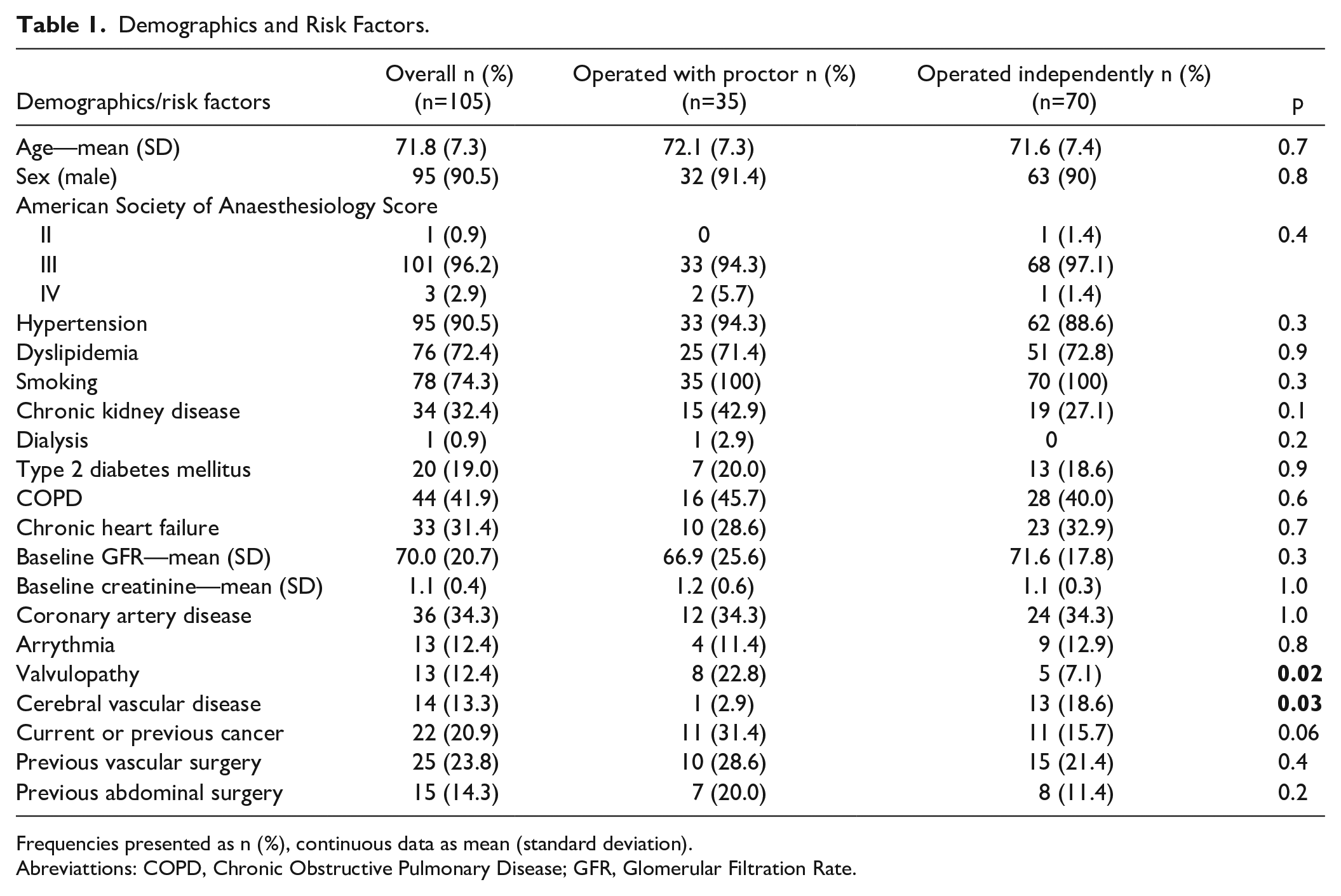
Frequencies presented as n (%), continuous data as mean (standard deviation).
Abreviattions: COPD, Chronic Obstructive Pulmonary Disease; GFR, Glomerular Filtration Rate.
Diagnosis and Procedure Details (Table 2)
Mean aneurysm size was 66.2 mm (SD: 13.3). Overall, 62 (65%) patients had C-AAA (juxta-renal, n=37; para-renal, n=11; extent IV TAAAs, n=13; endoleak 1A post-EVAR, n=1) and 43 (35%) had eTAAAs (extent I, n=4; extent II, n=8; extent III, n=23; extent V, n=8).
Aneurysm and Repair Details.
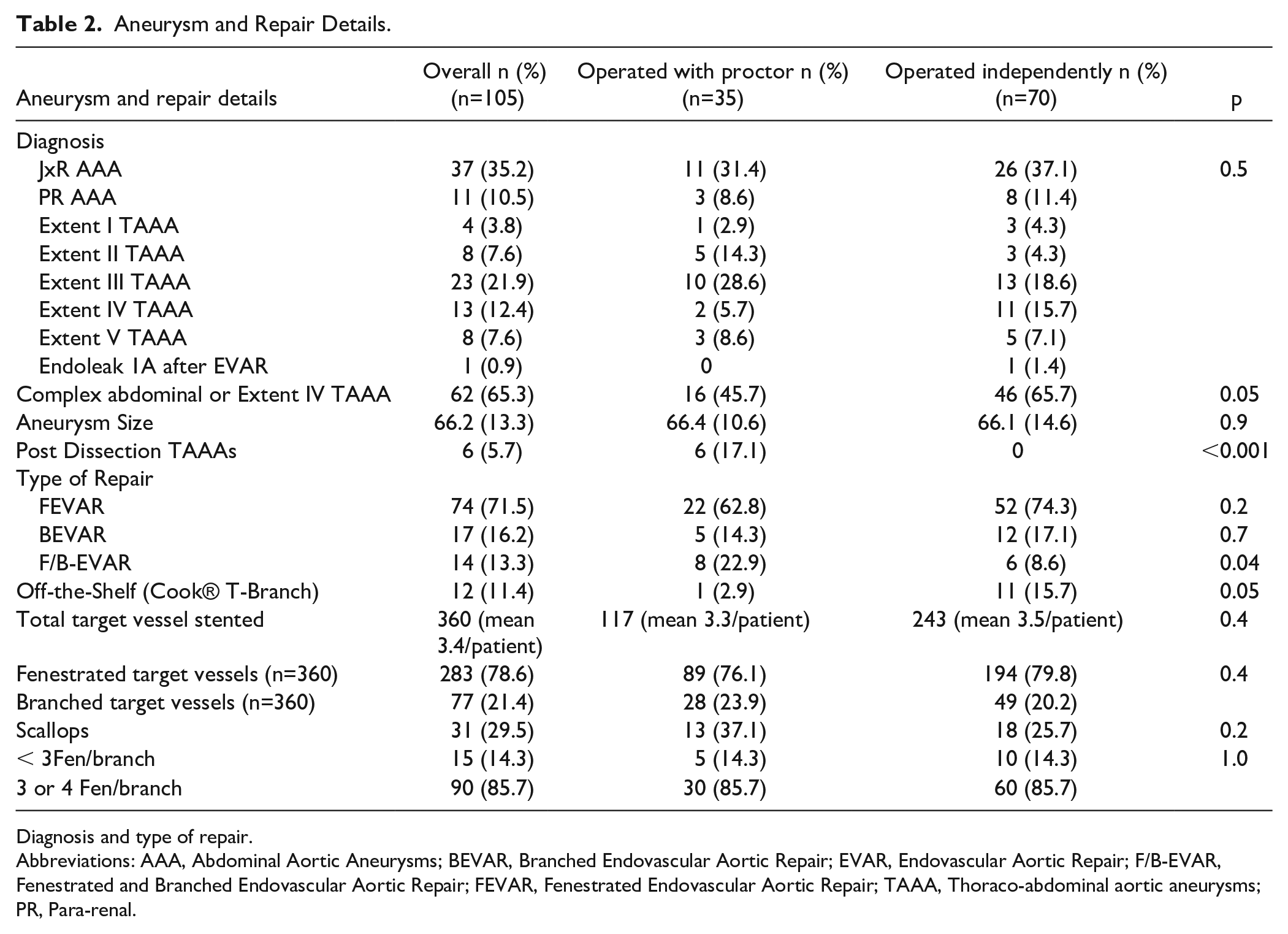
Diagnosis and type of repair.
Abbreviations: AAA, Abdominal Aortic Aneurysms; BEVAR, Branched Endovascular Aortic Repair; EVAR, Endovascular Aortic Repair; F/B-EVAR, Fenestrated and Branched Endovascular Aortic Repair; FEVAR, Fenestrated Endovascular Aortic Repair; TAAA, Thoraco-abdominal aortic aneurysms; PR, Para-renal.
There was no significant difference regarding aneurysm subtype in patients treated with or without the proctor. In the eTAAA group, 6 patients had a postdissection TAAA, and all these patients were treated with the Proctor present (p>0.001). Overall, 360 target vessels were included with either a branch (77) or fenestration (283) (mean included target vessel per patient was 3.4). Additionally, 31 scallops were used. Ninety patients had 3 or more target vessels included with a branch or fenestration, and this was similar in both groups. There was a tendency toward more use of 3 or 4 target vessels repair as the learning curve increased (Figure 3).
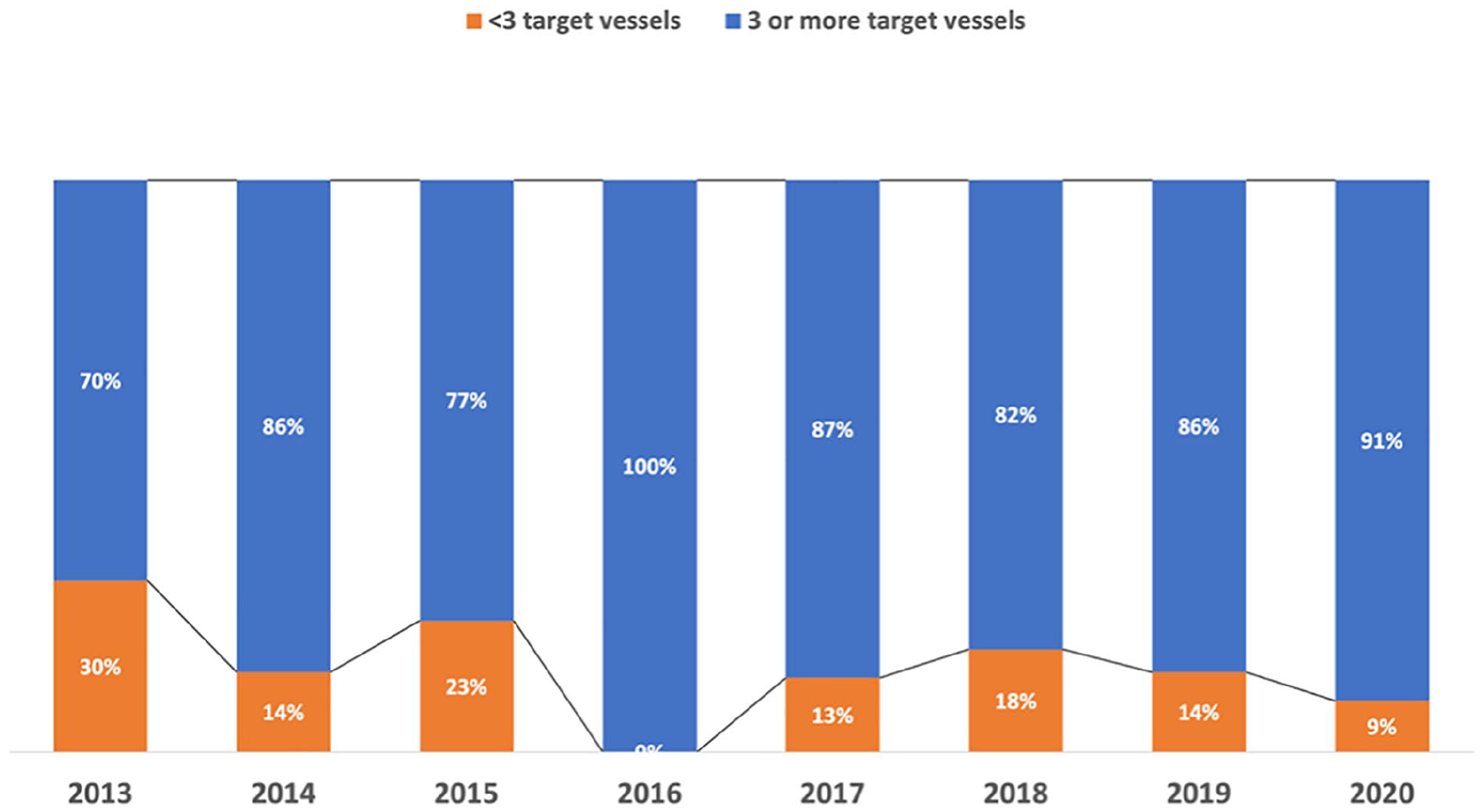
Target vessel incorporation per year of the study.
Regarding repair technique, 74 (71%) patients were submitted to FEVAR, 17 (16.2%) to BEVAR, of which 12 patients with an off-the-shelf Cook® T-Branch, and 14 (13.3%) to both F/B-EVAR. Techniques were similar between both groups except for F/B-EVAR, which was more common in the Proctor group (p=0.04). Furthermore, the Cook® T-Branch was used more commonly in the absence of the Proctor.
Primary Outcomes (Table 3)
Safety—30-day mortality
Overall, 30-day mortality occurred in 3 (2.9%) patients. There was no difference between groups (1 in the Proctor and 2 in the no-Proctor group), Log Rank=0.990. Furthermore, there was no significant difference regarding aneurysm extension (p=0.4) or the learning curve time (p=0.9).
Results.
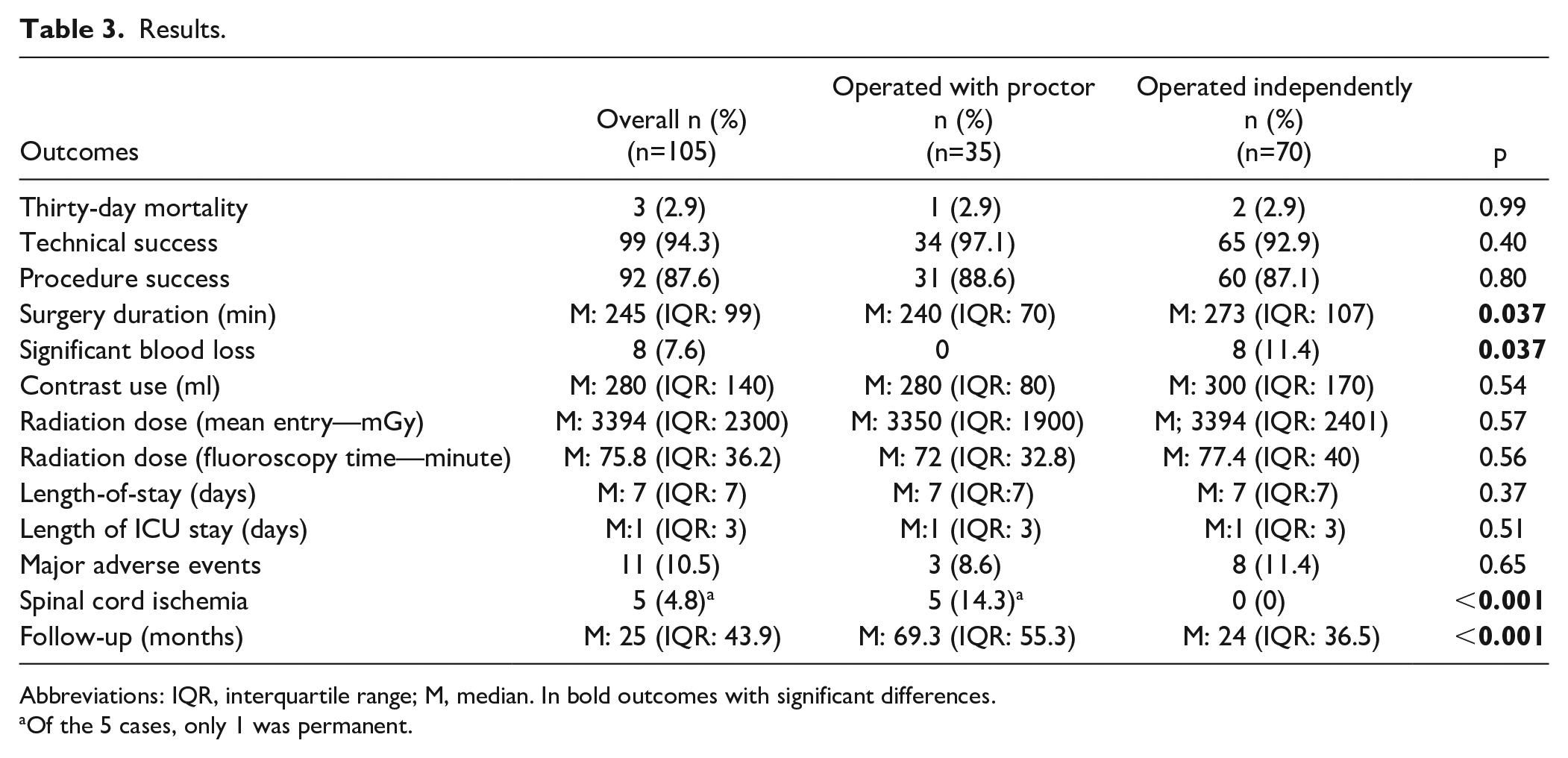
Abbreviations: IQR, interquartile range; M, median. In bold outcomes with significant differences.
Of the 5 cases, only 1 was permanent.
In the non-Proctor group, both patients died due to technical issues. In 1 patient (extent III TAAA), a technical failure occurred due to inability to catheterize the celiac trunk, which was then covered. Stenting of the SMA fenestration was uneventful and control angiography showed a patent gastroduodenal arch. In the postoperative period, the patient developed mesenteric ischemia and multiple organ failure (MOF), was submitted to laparotomy and bowel resection, and eventually died on the fourth day post-op. In the second patient (extent IV TAAA), a femoral artery access occlusion occurred 6 hours after surgery, after a lengthy procedure with >2000 ml blood loss. The patient was immediately re-operated and revascularized but developed MOF and died after 48 hours.
In the Proctor group (extent III TAAA), 1 patient died 8 days after surgery, which was uneventful, due to myocardial infarction.
Efficacy—Technical success
Overall technical success was 94.3%. Technical failures occurred in 3 patients due to inability to catheterize the celiac trunk, in 2 due to intraoperative renal stent occlusions (1 due to crushing, and the other due to renal artery rupture and need for embolization) and in 1 due to inability to catheterize a renal artery.
There was no significant difference regarding technical success in both groups (92.9% non-Proctor and 97.1% in the Proctor group), p=0.4. There was no difference regarding aneurysm extension (p=0.6). All technical failures occurred in patients with 4 target vessels included (5 patients with 4 fenestrated repair and 1 patient with a 4 branched repair). Technical failures occurred more frequently in the late stage of the learning curve (p=0.001).
Efficacy—Procedure success
Overall procedure success was 87.6%, being 88.6% in the Proctor group and 87.1% in the non-Proctor group (p=0.8). There was no significant difference regarding procedure success and the learning curve time (p=0.06) or target vessel incorporation (p=0.1). Procedure success was lower in eTAAAs (79.1%) versus C-AAAs (93.5%), p=0.03.
Secondary Outcomes (Table 3)
Procedure technical differences
Surgery duration
Median surgery duration time was significantly longer in patients treated without the Proctor (non proctor group: 273 minute [IQR: 107] versus proctor group: 240 minute [IQR: 70], p=0.037). This was true even after adjusting for aneurysm extension and target vessel incorporation (mean difference of −53.8 minute (95% CI: −90.7; −17.0), p=0.005).
Blood loss
Significant loss (>1500 ml) occurred in 8 patients. All these patients were treated in the absence of the Proctor (p=0.037).
Contrast use
There was a non-significant difference in the median contrast use between groups (non-proctor group: 300 ml [IQR: 170] versus proctor group: 280 ml [IQR: 80], p=0.54).
Radiation use
There was a non-significant difference regarding radiation use (non-proctor group median entry dose of 3394 mGy [IQR: 2401] versus proctor group 3350 [IQR: 1900], p=0.57), as well as median fluoroscopy time (non-proctor group: 77.4 minute [IQR: 40] versus proctor group: 72 minute [IQR: 32.8], p=0.56).
Length of stay
Overall median length of hospital stay was 7 days (IQR: 7) with a 1 (IQR: 3) median length of ICU stay. There was no difference in both ICU and overall hospital length of stay between groups regarding Proctor (p=0.4 and 0.5, respectively).
Major adverse events
Major adverse events occurred in 10.5% of patients. There was no significant difference regarding the presence of the Proctor (p=0.6). MAE occurred more frequently in more extensive aneurysms (18.6% in eTAAA versus 4.8% in C-AAA), p=0.024. There was no difference regarding number of target vessels incorporated (p=0.7) or the time in the learning curve (p=0.4).
Spinal cord ischemia
Overall, 5 patients developed spinal cord ischemia in the postoperative phase, 4 patients recovered completely, while 1 patient developed a permanent deficit (grade 3B). All these cases occurred in eTAAAs (n=43), resulting in a frequency of 11.6% for SCI, and 2.3% of permanent paraplegia. All these cases occurred in patients treated with the Proctor (p<0.001), and all were performed following the spinal cord ischemia protocol with CSF drainage. There was no difference regarding staging (23 patients, 21.9%) (p=0.7), and aortic extension cover (p=0.5).
Target vessel patency
Overall, 11 target vessel occlusions were observed (7 renal arteries, 3 celiac trunks, and 1 superior mesenteric artery). Three occlusions occurred intra-operatively and 8 during follow-up. One patient developed bilateral renal stent thrombosis with acute renal failure and was submitted to open revascularization with renal-renal bypass using saphenous vein followed by a jump graft to the hepatic artery. In all the other cases, occlusions were observed in the control CTAs and the patients did not present symptoms. The patient with the superior mesenteric artery occlusion had a patent celiac trunk and no complaints of weight loss, intestinal angina, or anorexia.
Overall primary target vessel patency during follow-up was 98.4% (95% CI: 95.7-99.4, SE: 0.8) at 12 months and 97.5% (95% CI: 93.7-99.0, SE: 1.2) at 3 years. Regarding branches patency was 96.6% (95% CI: 86.9-99.1, SE: 2.4) at 12 months and 96.6% (95% CI: 86.9-99.1, SE: 2.4) at 3 years. Fenestration patency was 99.1% (95% CI: 96.5-99.8, SE: 0.6) at 12 months and 97.9% (95% CI: 92.7-99.4, SE:1.3) at 3 years—Supplement Figure 1.
There were no differences regarding target vessel patency between patients operated with and without the Proctor (all target vessels: LongRank=0.97; branches: LongRank=0.51; Fenestrations: LongRank=0.94)—Supplement Figure 1. Adjusting for competing risk of mortality also showed no differences (all target vessels: subhazard ratio 1.19 (95% CI: 0.32-4.47, p=0.79), fenestrations: subhazard ratio 0.95 (95% CI: 0.07-12.23; p=0.97), and branches: subhazard ratio 0.66 (95% CI: 0.15-2.82; p=0.58).
Endoleaks
Endoleaks were classified as immediate (seen on angiography), early (1stCTA) and late (persistent after 1 year on CTA). Immediate endoleaks occurred in 35 patients (33.3%, n=105), occurring in 28.6% of the Proctor group and 35.7% in the non-Proctor group, p=0.5. All of these were type II endoleaks.
Early endoleaks were observed in 26 patients (27.7%, n=94), occurring in 30.3% in the Proctor group and 26.2% in the non-Proctor group (p=0.7). Of these, 3 patients had type I or III: 1 type IIIc from the celiac bridging stent, 1 type Ic from the celiac artery, and 1 type Ic from the SMA. All of these were eTAAAs. The remainder 23 patients had type II endoleaks.
Late endoleaks occurred in 20 patients (29.4%, n=68), occurring in 32.1% of the Proctor group and 27.5% of the non-Proctor group, p=0.7. Of these 7 (10.3%) patients were type I or III, and remainder were type II. Two of which had already known type II endoleaks. Of the 7 patients, 1 developed a type Ib endoleaks after continuous aneurysm growth due to a type II (lumbar and inferior mesenteric artery) endoleak, 2 type IIIa due to thoracic stent disconnection, 2 type Ic due to mesenteric and celiac artery stent pull-out and disconnection and 2 IIIc due a renal and celiac artery stent disconnection from the main graft.
Aneurysm shrinking
Overall successful aneurysm exclusion (no aneurysm growth >=5mm) occurred in 98 patients. Seven patients had an aneurysm growth of >=5mm, 4 of which were successfully re-operated. In the remainder 3 patients, 2 patient refused treatment and 1 patient was deemed unfit for surgery due to endocarditis, heart failure, and suspected aortic infection.
Overall, mean aneurysm shrinking was 5.0 mm (SD: 11.0), with no differences between groups (Proctor group 5.9 mm [SD: 9.0] versus non-Proctor group 4.6 mm [SD:12.0]), p= 0.6. Additionally, 61 patients (58.1%) had an aneurysm shrinking of >5 mm.
Re-interventions
Overall, there were 14 re-interventions in 13 patients. One patient was submitted to an iliac extension to treat a type Ib endoleaks followed by an open surgery with “trap-door” technique to treat a persistent type II endoleaks. Reintervention cases are detailed in Table 4. There was no differences in re-interventions between groups (p=0.2).
Re-Intervention Details.
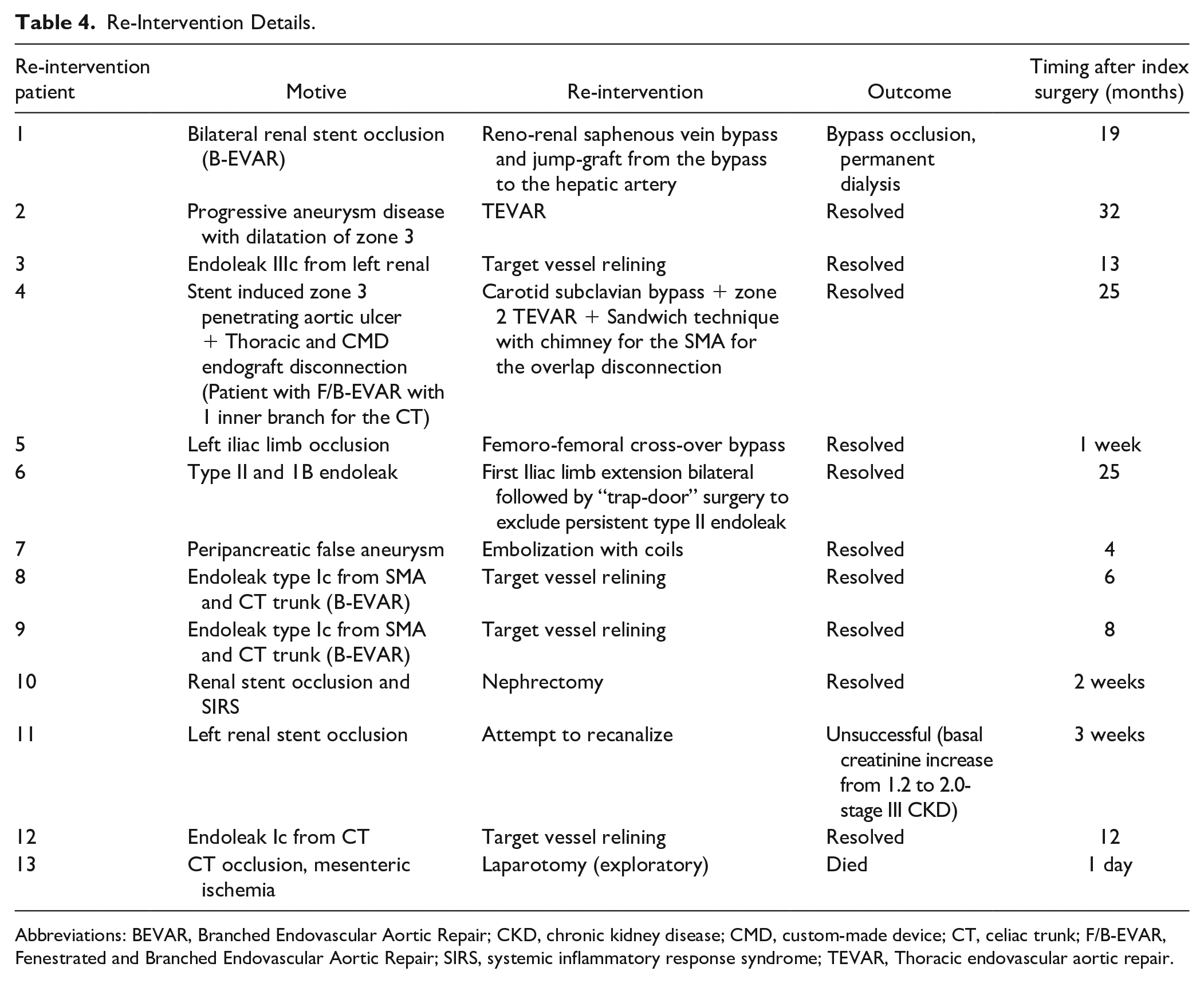
Abbreviations: BEVAR, Branched Endovascular Aortic Repair; CKD, chronic kidney disease; CMD, custom-made device; CT, celiac trunk; F/B-EVAR, Fenestrated and Branched Endovascular Aortic Repair; SIRS, systemic inflammatory response syndrome; TEVAR, Thoracic endovascular aortic repair.
Follow-up
Median follow-up for the entire cohort was 35 months (IQR: 43.9), being 24 (IQR: 36.5) and 69.3 (IQR: 55.3) for the non-Proctor and Proctor groups, respectively (p<0.001).
Full completeness of follow-up (less than 1 year to last CT control) was present in 75 (71.4%) patients, corresponding to an incomplete follow-up of 18 patients (25.7%) in the non-Proctor group versus 12 patients (34.3%) in the Proctor group, p=0.36.
Overall mortality
Overall, 24 patients died during the study period (3 in the first 30 days, and 21 during follow-up). Overall, survival rate was 89% (SE: 3.1) at 1 year and 84.0% (SE: 4.0) at 3 years. There was no significant difference in overall survival between groups (proctor group survival rate 91.2% [SE: 4.9] at 1 year and 84.9% [SE: 6.2] at 3 years versus 87.7% [SE: 4.1] at 1 year and 82.4% [SE: 5.3] at 3 years), log rank=0.87)—Figure 4.
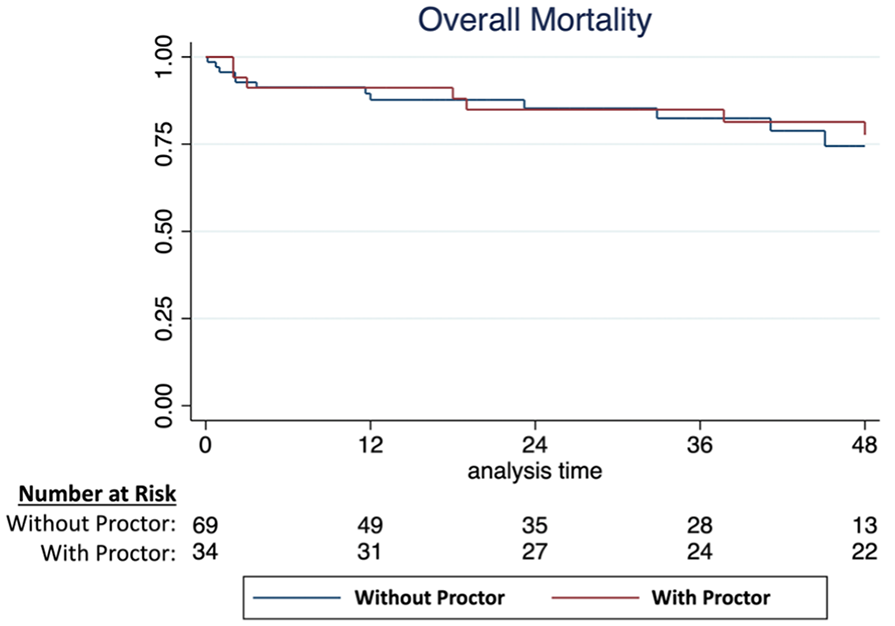
Kaplan-Meyer curve of the overall survival comparing patients treated with and without the proctor.
Of the follow-up deaths, only one case was aneurysm related. This patient developed a gastroduodenal artery false aneurysm, probably from a guide-wire injury, although this was not seen in the control angiogram. He was admitted to the hospital 2 months later, with abdominal pain, a CT-angiogram showed the false aneurysm, and he was submitted to endovascular embolization of the gastroduodenal arch. In the postoperative period, the patient developed acute pancreatitis, multiorgan failure, and died.
Additional analysis
Logistic regressions were performed for the primary outcomes including the variables aneurysm group; 3 or more target vessels; heart valvular disease and cerebrovascular disease. There was no difference in any of the outcomes between patients operated with and without the Proctor: 30-day mortality—OR: 1.76 (95% CI: 0.10-30.48, p=0.70); technical success—OR: 1.18 (95% CI: 0.29-4.69, p=0.82) and procedure success OR: 0.46 (95% CI: 0.12-1.75, p=0.26).
Additionally, we analyzed the primary outcomes comparing 2 periods. The first when cases were done exclusively with the Proctor and the second when cases were performed selectively with the Proctor. All main outcomes were non-significant between both groups (30-day mortality, p=0.44; technical success, p=0.27 and procedure success, p=0.37).
Discussion
We performed a comprehensive analysis of our fenestrated and branched endovascular aortic aneurysm repair results analyzing our learning curve, safety, and effectiveness, looking specifically at the Proctor’s contribution. Overall, 30-day mortality was 2.9%; technical and procedure success were 94.3% and 88.6%, respectively; primary target vessel patency at 1 and 3 years was 96.8% and 89.7%; respectively and 12.4% of patients required re-interventions during follow-up. These are similar results to what has been described in the available literature in high-volume centers.3,5,8,9 Furthermore, these results were similar when comparing patient treatment with and without the Proctor. These findings seem to demonstrate that our learning curve was achieved in a safe manner without putting the patients at risk and being able to provide complex aneurysm repair to these patients. One surprising finding, however, was the lower technical success in the second part of our learning curve, which was unexpected. This is probably due to a selection bias, seeing as experience increased, more complex cases were treated which might have increased technical difficulties. Seeing as all technical failures were due to target vessel catheterization or stenting issues, the more complex the anatomy, the more these complications tend to occur.
Current guidelines recommend that these procedures should be performed in high-volume centers1,2,4 with some authors arguing for a limited number of centers providing this care to increase referral and case-volume. 10 In an elective setting, referral to high-volume and expert centers should always be strived for. However, if referral is not possible, restricting access to these techniques and devices may be a disservice for patients. In these cases, development of expert centers in these necessary locations should be performed to offer the best care and access to patients. In Portugal, at the time our complex endovascular treatment program was started, no other center within the country was able to provide this service. The option to start the program with the Proctor allowed for the development and training of the team in a safe and progressive manner, even with initial smaller numbers, as our results seem to support.
Proctor programs are not uncommon; however, no data have been published regarding the results of such a strategy. In our audit, the progression and description of the program as well as the results achieved may be used as a template by other countries and centers that face a similar reality when aiming at developing these programs.
Obviously, our analysis has some limitations. Selection bias is almost unavoidable. As the team experience increased, more cases were performed independently and the Proctors’ help only requested in the most complex cases, such as postdissection TAAAs. Although we did not find differences in the outcomes when adjusting for aneurysm extension in each group, complete assessment of complexity is challenging and may be biased. However, extensive thoraco-abdominal aneurysms were treated more commonly in the Proctor group thus suggesting reliability of our results.
The only major differences observed between groups were regarding performance details, namely duration of surgery and blood loss, which were both lower in patients treated with the Proctor, which highlights the effect of greater experience and expertise, and the overall benefit of having the Proctor present. More studies are needed to analyze which strategies in developing complex endovascular aortic programs are safe and effective for the benefit of patients. Furthermore, specific conditions apply to different healthcare’s and realities and should be tailored aiming at providing the best care to patients.
Conclusion
The only differences found in the patients treated with and without the Proctor were regarding procedural performance details which highlight the effect of greater experience and expertise, and the overall benefit of having the Proctor present. In our experience, the use of a Proctor to start and accompany our complex endovascular aortic program for FB-EVAR was both safe and effective and may serve as a template by other countries and centers that aim to developing their programs.
Supplemental Material
sj-docx-1-jet-10.1177_15266028221105186 – Supplemental material for The Impact of the Proctor Assistance for a Safe Learning Curve in the Development of a Complex Aortic Endovascular Program
Supplemental material, sj-docx-1-jet-10.1177_15266028221105186 for The Impact of the Proctor Assistance for a Safe Learning Curve in the Development of a Complex Aortic Endovascular Program by Ryan Gouveia e Melo, Ruy Fernandes e Fernandes, Margarida Salvado, António Duarte, Alice Lopes, Eric Verhoeven, José Fernandes e Fernandes and Luís Mendes Pedro in Journal of Endovascular Therapy
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EV (proctor) has received proctoring and speaker fees from COOK® medical as well as royalties.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
