Abstract
In December 2020, 11 months after identifying the first laboratory-confirmed case of COVID-19 in the United States, the U.S. Food and Drug Administration authorized emergency use of two COVID-19 vaccines. To prepare the public for a large-scale vaccination campaign and build confidence in COVID-19 vaccines, the U.S. Centers for Disease Control and Prevention (CDC) funded more than 200 partners and developed a national Vaccinate with Confidence (VwC) COVID-19 framework to support Americans in their decision to get vaccinated. The evolving nature of the pandemic and highly variable confidence in vaccines across populations has resulted in many unique complexities and challenges to reaching universally high vaccination coverage. Here, we describe how 23 professional health associations and national partner organizations, focused solely on building vaccine confidence, operationalized CDC’s VwC COVID-19 framework from February 2021 to March 2022. Capturing how partners deployed and adapted their activities to meet a shifting pandemic landscape, which began with high demand for vaccines that waned over time, is an important first step to understanding how this new strategy was utilized and could be implemented for future surges in COVID-19 cases and other routine immunization efforts. Going forward, evaluation of partner activities should be prioritized to capture learnings and assess VwC program effectiveness.
Background
In 2020, the U.S. government embarked upon the largest vaccination campaign in U.S. history to combat COVID-19. Although vaccines offered the greatest hope of curbing the impact of the COVID-19 pandemic, a STAT-Harris poll conducted in October 2020 indicated that only 58% of Americans intended to receive a COVID-19 vaccine (Silverman, 2020). The WHO identified vaccine hesitancy, the reluctance or refusal to get vaccinated when vaccination services are available, as 1 of the top 10 threats to health worldwide just the year prior (World Health Organization, 2019).
Recognizing the devastating impact that the COVID-19 pandemic was having on the U.S. population and that the most effective tool to mitigate the pandemic was vaccines, including a new type of vaccine, (mRNA vaccines), the U.S. Centers for Disease Control and Prevention’s (CDC) Vaccine Task Force developed a national Vaccinate with Confidence (VwC) COVID-19 Vaccines Framework to boost vaccine confidence. This framework, depicted in Figure 1, focused on reinforcing that the COVID-19 vaccines work, are safe, and are part of a trustworthy medical system. The framework also centered around three primary objectives: (a) build trust in COVID-19 vaccines, (b) empower health care providers to recommend COVID-19 vaccines, and (c) engage communities around vaccine confidence.

CDC’s Vaccinate With Confidence Framework
In 2021, CDC awarded millions of COVID-19 emergency response funds to support the COVID-19 vaccination effort. A very small subset of these funds was awarded to 23 partners to focus exclusively on building vaccine confidence. This commentary describes the work carried out by these 23 professional health associations and national partner organizations, who collectively through their organizations’ memberships, reached nearly one million primary care physicians, pediatricians, obstetricians, gynecologists, pharmacists, nurses, social workers, psychologists, medical students, and local community trusted messengers. Herein, these organizations will be referred to as VwC Partners (see Appendix).
To quickly fund a diverse portfolio of partners with reach to populations at disproportional risk from COVID-19, such as older adults, rural communities, racial and ethnic minoritized groups, and communities with high social vulnerability, CDC capitalized on the breadth of its existing partnerships and funding mechanisms from across the agency. Subsequently, funds were dispersed within weeks instead of the lengthy 10 to 12 months typical for a new federal Notice of Funding Opportunity announcement. This allowed VwC partners to implement COVID-19 vaccine confidence activities in February 2021, less than 2 months after the first COVID-19 vaccine was administered in the United States. Engaging long-standing partners also meant that a shared vision for collaboration already existed, which allowed partners to quickly operationalize vaccine confidence activities. To support VwC partners, CDC’s Vaccine Task Force developed and shared vaccine confidence resources, offered learning sessions, provided technical assistance on a regular basis, and hosted quarterly partner calls to promote peer-to-peer learning and foster strategic collaborations among one another. The Task Force also worked to develop COVID-19 VwC monitoring and evaluating tools, which did not exist at the outset of the pandemic, concurrently with partner implementation and encouraged partners to voluntarily report progress using these tools, which under other circumstances would have been required.
From Framework To Action
With CDC providing a strategic framework, recommendations, and evidence-based strategies for increasing vaccine confidence and demand, partners operationalized this framework and prioritized strategies most relevant for their audiences (CDC, 2022a). Partner implementation consisted of three general overlapping phases following initial partner onboarding: (a) broad communication focus, (b) tailored COVID-19 vaccine approach, and (c) integrated health focus. Figure 2 provides an overarching roadmap of the approaches and activities prioritized by partners from February 2021 to March 2022 to increase vaccine confidence and demand.
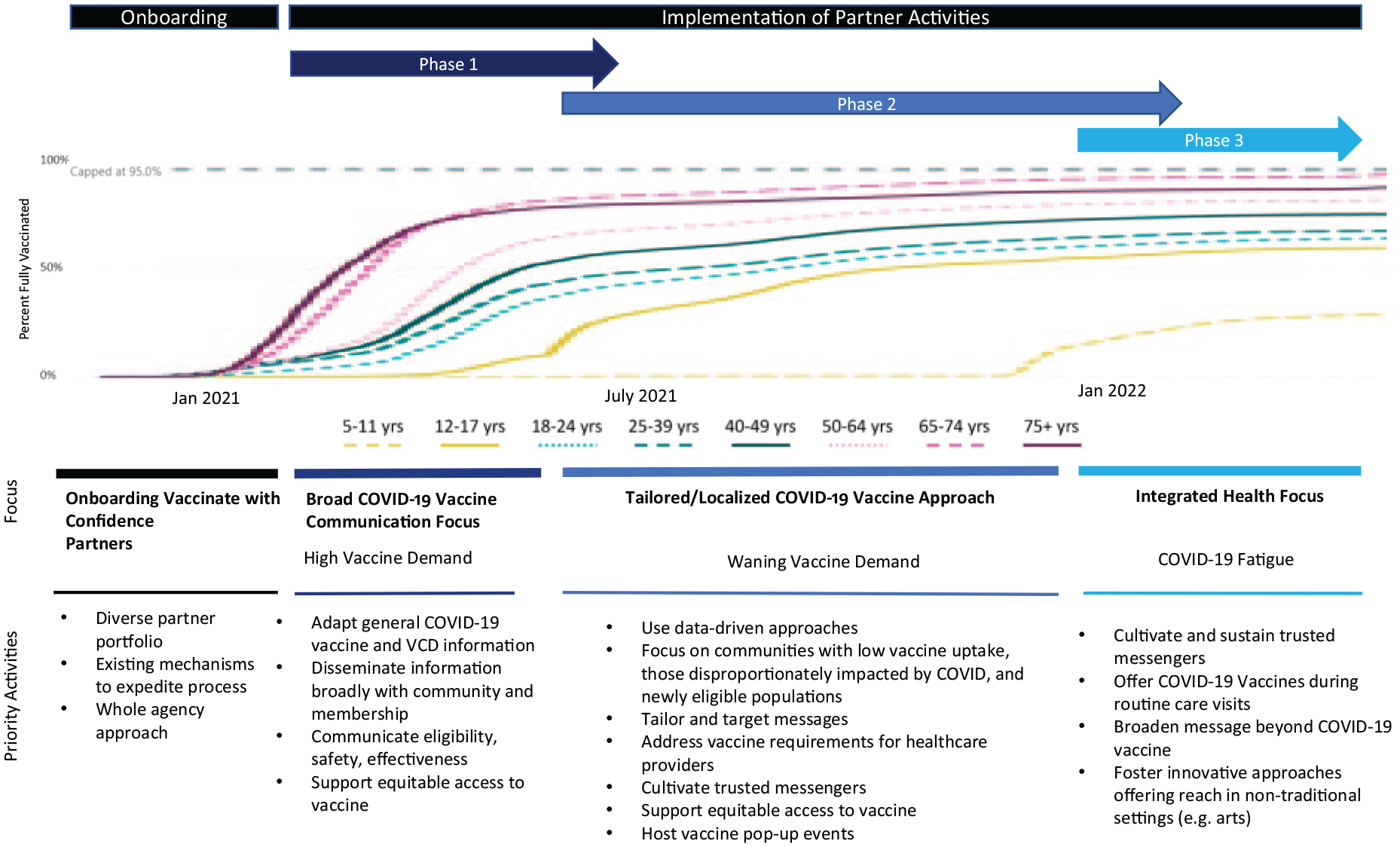
CDC Vaccinate With Confidence Partner Efforts, February 2021–March 2022
Phase 1: Broad Communication Focus (Winter 2021—Summer 2021)
During the first 5 months of COVID-19 vaccine administration, vaccine demand exceeded supply. An incremental rollout was initially deployed based on eligibility criteria established by the Advisory Committee on Immunization Practices (Oliver et al., 2020). By April 19, 2021, when vaccine eligibility was opened to all adults nationwide, half of all adults in the United States had received at least one dose of COVID-19 vaccine, 23 million unvaccinated adults (9%) indicated they planned to get vaccinated right away, and approximately 44 million adults (17%) were considering vaccination (CDC, 2023).
Partner activities during this phase focused on increasing awareness and utilization of vaccine confidence resources and identifying trusted messengers. Partners adapted and shared informational materials with association members frequently via emails, newsletters, social media, webinars, and trainings. Partners also translated materials into more than 40 critical languages and ensured they were culturally and linguistically appropriate for the diverse populations that they reached. Consistent with the strategy in Figure 1, resources focused on (a) benefits of vaccination; (b) generating confidence in the vaccine development and regulatory processes; (c) vaccine safety and effectiveness; (d) clarifying eligibility criteria; and (e) responding to common vaccine misconceptions and frequently asked questions. Professional medical associations also prioritized supporting their members in their decisions to get vaccinated as they were among the first eligible for vaccine receipt and positioned to deliver strong vaccine recommendations. Partners carried out needs assessments and listening sessions to better understand members’ perceptions of COVID-19 vaccination and strengthen members as vaccine champions. Feedback collected in member surveys was shared with CDC and used to refine federal communication products addressing common question and concerns.
Phase 2: Tailored Approach (Summer 2021—Winter 2022)
Despite vaccines being safe and highly effective at preventing severe illness and death, demand for COVID-19 vaccinations and intent to vaccinate began to slow by late spring 2021 (CDC, 2023). Furthermore, vaccine uptake was uneven, particularly between demographic groups, such as by race/ethnicity, metropolitan statistical area, political affiliation, income level, education level, and age group (CDC, 2022b). At this point, mis- and disinformation was also spreading at unprecedented speed and scale, causing confusion that led some Americans to delay or decline COVID-19 vaccination (Kaiser Family Foundation, 2022).
With slowing demand, partners shifted their focus from building vaccine confidence and demand broadly, largely through communication outreach, to using remaining project resources to reach specific groups, such as communities with lower vaccination uptake, communities disproportionately impacted by the pandemic, and newly vaccine-eligible populations (such as caregivers of teens in May and children aged 5–11 years in November 2021). CDC supported caregivers in this effort by collecting and publishing trends in vaccination coverage and intent by socio-demographic characteristics at both the national and jurisdiction levels (CDC, 2023). Partners utilized these public data and conducted their own surveys or listening sessions to better understand who remained unvaccinated, why certain geographic or socio-demographic populations were not getting vaccinated, and how best to reach unvaccinated individuals. Partners then tailored their approaches to strengthen confidence among people who were unvaccinated and to reinforce vaccination as a social norm. Messages sought to reflect audience-specific values, lived experiences, motivators for vaccination, and address vaccine concerns and misinformation. Tailored messages were then disseminated on communication platforms frequented by the intended audience, increasing the likelihood of being seen or heard. Several partners also employed innovative methods, such as utilizing location-based marketing tools and geofencing on mobile devices, which pushed out messages and resources to electronic devices that came within a 1-mile radius around certain free health clinics.
To extend reach, partners developed free downloadable content with editable graphics and curated resource hubs with unique communication content that could be used by others. This included a health Translated Materials Library that grew over time to include >7,600 COVID-19 resources available in more than 100 languages. Partners also fostered community-level partnerships with local pharmacies, health departments, clinics, and community-based organizations to bring vaccine information and vaccination clinics to convenient, trusted community spaces.
Beyond communication outreach, partners also expanded efforts to train trusted messengers, that is, individuals who share similar beliefs and characteristics as their peers and are seen as credible sources of information, to disseminate COVID-19 vaccination messages. Partners identified vaccine champions from communities with low vaccination coverage and provided them with skills-based training on motivational interviewing strategies to have effective one-on-one conversations and respond to misinformation with empathy and facts. These types of tools and trainings allowed trusted messengers to practice communication skills and interviewing techniques thereby increasing both their capacity and confidence to engage in effective vaccine conversations. Trusted messengers then promoted vaccine confidence by making their vaccine decision visible, sharing their testimonial, sharing credible sources of vaccine information, and debunking vaccine misinformation.
Phase 3: Integrated Health Focus (Winter 2022—March 2022)
COVID-19 vaccination remains essential to mitigate the impacts of future COVID-19 variants and surges in cases. However, by January 2022, vaccination progress largely stalled among eligible adults and children with approximately 66% of the American population fully vaccinated (CDC, 2023). Most Americans believed the worst of COVID-19 was over by winter 2022 (Sparks et al., 2022) and partners subsequently found it increasingly difficult to leverage the same activities in impactful ways. Although partners continued to utilize trusted messengers, they also prioritized new and innovative approaches to ensure that both vaccine messengers and the public remained engaged.
To keep trusted messengers engaged, partners acknowledged the tremendous mental and physical toll of the COVID-19 pandemic on health care workers. Through podcasts, listening sessions, and interactive webinars, partners offered strategies for health care providers on how to prevent and manage burnout, foster resilience, and sustain morale. This included topics such as how to navigate negative encounters with patients, overcome frustration, and provide supportive environments for members to connect with each other for psychosocial support. To mitigate COVID-19 fatigue among the public, partners expanded their communication mediums, messengers, and messages (Sparks et al., 2022). Partners shifted communication from a disease-centric focus to integrated health messages using a bottom-up approach that promoted COVID-19 vaccination as a part of well-being and healthy choices. Partners reported that by including COVID-19 activities as a part of school, routine immunization, or community wellness events, they were able to reach greater audiences than through COVID-19 vaccination events alone. Non-traditional CDC-funded partners, such as arts organizations, museums, and libraries also increasingly implemented activities during this phase. They delivered fact-based information in memorable ways through stories, song, dance, theater, comic books, and quilting (CDC, 2021).
Conclusion
COVID-19 vaccines are estimated to have prevented over 2 million deaths and 17 million hospitalizations in the United States between December 2021 and March 2022 (Schneider et al., 2022). With 34% of the U.S. population not fully vaccinated by March 2022, and far fewer up to date on their COVID-19 vaccinations, substantial work remains to prepare for and mitigate the effects of potential new variants and surges in COVID-19 cases. Partners operationalized the VwC Framework by implementing evidence-based strategies tailored to meet the nuanced and ever-changing needs and interests of their particular audiences through each phase of the COVID-19 vaccination program (CDC, 2022a). Going forward, evaluation of partner activities should be prioritized to capture learnings and identify their effectiveness in empowering individuals to feel confident in their choice to get vaccinated for COVID-19. New learnings from the COVID-19 vaccination program should also be considered and extended to strengthen confidence in other vaccine-preventable diseases.
