Abstract
Significance:
Melanin is no longer best understood as a passive UV filter. It is emerging as a programmable, redox-active pigment system that converts environmental energy and oxidative stress into lasting genomic and immunological consequences, placing pigment metabolism at the center of melanoma biology.
Recent Advances:
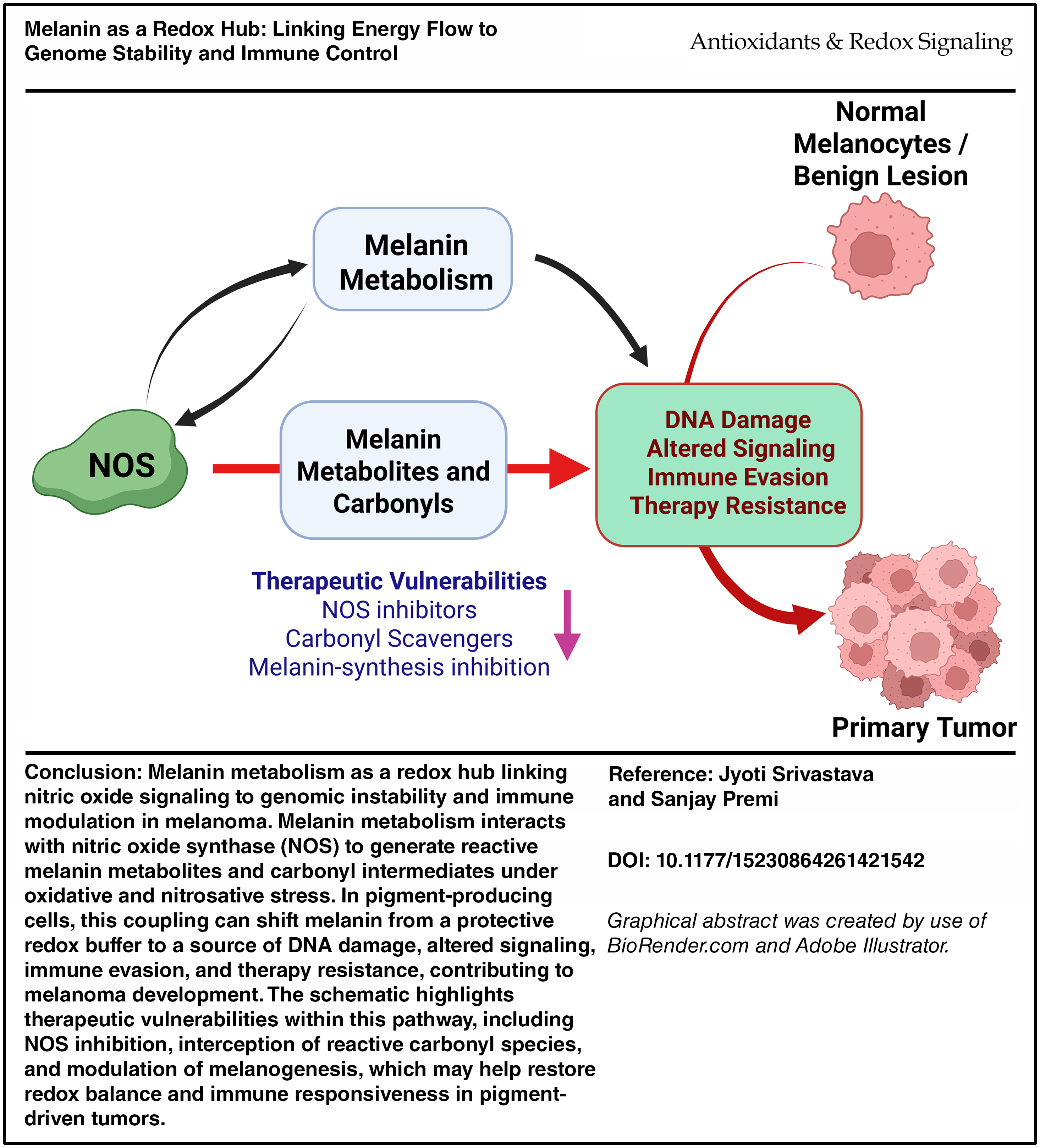
Oxidative melanogenesis activates nitric-oxide synthase (NOS), generating reactive nitrogen species that oxidize melanin fragments into electronically excited triplet carbonyls. These intermediates drive delayed cyclobutane pyrimidine dimers (dCPDs) hours after ultraviolet exposure, establishing a pigment-powered window of “dark mutagenesis.” In melanoma, this pigment-NOS-chemiexcitation axis forms a lineage-specific redox circuit linking oxidative metabolism to mutation density, nitrosylation signaling, and immune evasion. Genome-wide mapping further indicates that chemiexcitation concentrates damage within open, transcriptionally active chromatin.
Critical Issues:
Key gaps remain in defining how pigment oxidation state, NOS flux, and subcellular organization coordinate chemiexcitation and nitrosylation in vivo, and how these processes reshape DNA repair and interferon programs across melanoma states. Parallels in neuromelanin, the retinal pigment epithelium, and mucosal pigment systems are provocative but require direct mechanistic validation.
Future Directions:
Targeting pigment chemistry itself, through NOS inhibition, denitrosylation, and triplet-state quenching, offers a compelling translational strategy to suppress dCPDs, rebalance redox signaling, and restore immune function. Coupling these interventions to quantitative biomarkers of pigment oxidation, nitrosylation networks, and chemiexcitation activity may enable patient stratification and rational therapeutic sequencing, transforming melanin from a risk modifier into an actionable redox interface.
Graphical Abstract
Keywords
Get full access to this article
View all access options for this article.
