Abstract
Aims:
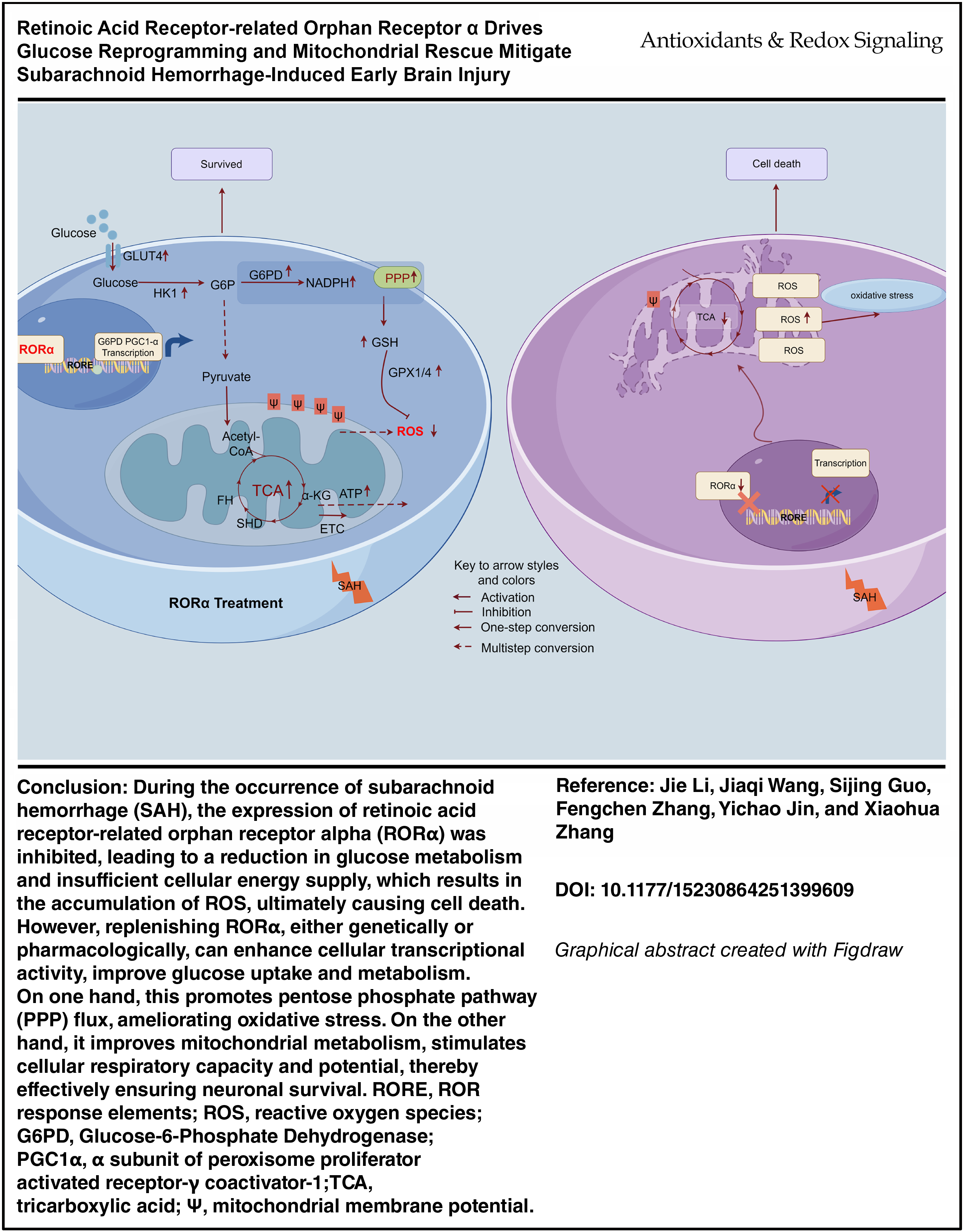
Subarachnoid hemorrhage (SAH) is a devastating cerebrovascular event characterized by early brain injury (EBI) within 72 h that is driven by oxidative stress, mitochondrial dysfunction, and metabolic collapse. The retinoic acid receptor-related orphan receptor alpha (RORα) is a nuclear receptor implicated in metabolic and inflammatory regulation, but it has not been studied in SAH. We aimed to determine whether RORα confers neuroprotection after SAH and to elucidate its underlying mechanisms.
Methods and Results:
We used mouse SAH models and primary cortical neurons to assess the RORα expression, functional outcomes, and metabolic changes. The RORα expression was markedly reduced post-SAH. Genetic knockdown or deficiency (staggerer mice) exacerbated neuronal apoptosis, neuroinflammation, and behavioral deficits. Conversely, pharmacological activation with SR1078 significantly improved neurological scores, preserved neuronal morphology, and reduced oxidative stress. RORα overexpression or SR1078 treatment enhanced neuronal viability in vitro under hemoglobin-induced stress. Transcriptomic and epigenomic profiling revealed that RORα directly regulated glucose-6-phosphate dehydrogenase and α subunit of peroxisome proliferator activated receptor-γ coactivator-1. This promoted pentose phosphate pathway flux and mitochondrial biogenesis. A metabolic flux analysis confirmed increased nicotinamide adenine dinucleotide phosphate hydrogen and glutathione synthesis, reduced reactive oxygen species accumulation, and an improved oxygen consumption rate and spare respiratory capacity. All of these results indicated a shift toward oxidative phosphorylation and enhanced bioenergetics.
Innovation and Conclusion:
We are the first to demonstrate that RORα activation reprogrammed neuronal glucose metabolism and strengthened antioxidant defenses to mitigate SAH-induced EBI. The targeting of RORα could represent a promising therapeutic strategy for stroke-related metabolic failure and oxidative stress. Future work should explore the translational potential in clinical settings. Antioxid. Redox Signal. 44, 275–291.
Graphical Abstract
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
