Abstract
Aims:
Obesity remains a major global health issue, with the increasing focus on the incretin hormone glucagon-like peptide-1 (GLP-1) and its receptor (GLP-1R) for therapeutic strategies. D-allulose is predicted to modulate GLP-1R via mechanisms linked to endoplasmic reticulum stress and reactive oxygen species (ROS) pathways, positively influencing GLP-1R stability and functionality. This study investigates the potential of D-allulose as a therapeutic and preventive agent against obesity. It focuses on the impact of D-allulose on adipocyte differentiation and obesity in high-fat diet (HFD)-administered and GLP-1R knockout (KO) mice over 12 weeks.
Results:
D-allulose effectively regulated adipocyte differentiation by inhibiting the NADP+/NADPH-ROS-inositol-requiring enzyme 1α (IRE1α)-regulated IRE1-dependent decay (RIDD) axis, resulting in controlled decay of GLP-1R, a newly identified RIDD target. Furthermore, in vivo studies revealed that D-allulose administration significantly regulated body weight and other obesity parameters in HFD-fed mice. However, these effects were not observed in GLP-1R KO mice, suggesting that the antiobesity effects of D-allulose rely on the presence of GLP-1R.
Innovation and Conclusion:
This study highlights the efficacy of D-allulose in controlling obesity through mechanisms dependent on GLP-1R, suggesting its potential as an effective treatment for obesity with normal GLP-1R function. Antioxid. Redox Signal. 43, 819–832.
Introduction
Oxidative stress and endoplasmic reticulum (ER) stress have been implicated in metabolic disorders, including insulin resistance, obesity, and type II diabetes (Ghemrawi et al., 2018). Recently, obesity has been recognized as a chronic and recurrent disease and has seen a threefold increase in prevalence over the last 40 years. In addition, increased oxidative stress in accumulated fat is vital to the pathogenic mechanism of obesity-associated metabolic syndrome (Furukawa et al., 2004). In obese animals, mature adipocytes produce higher levels of reactive oxygen species (ROS) than other tissues, including the liver, skeletal muscle, and aorta. Thus, mature adipocytes are major sources of ROS, accelerating the differentiation of adjacent preadipocytes (Lee et al., 2009; Liu et al., 2012). Furthermore, xanthine oxidoreductase regulates adipogenesis and functions downstream of CCAAT/enhancer-binding protein (C/EBP) and upstream of peroxisome proliferator-activated receptor γ (PPARγ), the main transcription factors for adipogenesis (Cheung et al., 2007). From a molecular perspective of adipocyte differentiation, ROS is directly or indirectly related to ER stress, characterized by the accumulation of unfolded or misfolded proteins. Here, the ER transmembrane kinase/endoribonuclease and inositol-requiring enzyme 1α (IRE1α) are critical among other ER stress signaling pathways. Upon ER stress, IRE1α undergoes autophosphorylation and oligomerization, activating its endoribonuclease activity, which splices the mRNA encoding the transcription factor X-box-binding protein 1 (XBP1). The spliced form of XBP-1 (XBP1s) translocates to the nucleus and upregulates genes involved in protein folding and degradation (Sha et al., 2009). XBP1s also upregulates C/EBPα and enhances PPARγ expression during adipogenesis (Cho et al., 2018). Furthermore, redox imbalance-related IRE1α sulfonation-regulated IRE1-dependent decay (RIDD) signaling appears to play a critical role in adipocyte differentiation (Riaz et al., 2020). The RIDD mechanism involves the selective degradation of mRNAs by IRE1α, which can influence cell fate under stress conditions. Depending on the RIDD target, the pathological meaning of ER stress varies (Chen and Brandizzi, 2013). Therefore, considering the importance of the pathological mechanisms of ROS and ER stress in obesity, it is essential to identify compounds that can simultaneously regulate ROS and ER stress signaling while being involved in obesity metabolism.
Innovation
The observations of this study present strong evidence supporting a preclinical rationale and molecular basis to investigate the possible therapeutic uses of oral D-allulose as a simple, effective, and safe intervention for obesity and related metabolic syndromes. The investigation emphasizes D-allulose’s potential as an ideal therapeutic candidate drug for future clinical applications in metabolic health management, especially in combating obesity, insulin resistance, and other associated conditions.
D-allulose is a low-calorie C-3 epimer of D-fructose found naturally in vegetables and fruits and exhibits 70% of the sweetness of sucrose (Xia et al., 2021). Interestingly, recent investigations suggest that D-allulose suppresses adipocyte differentiation and lipid accumulation by modulating adipogenic transcription factors (Lee et al., 2021). In addition, D-allulose reduces intra-abdominal fat accumulation and subdues blood glucose levels during glucose loading (Franchi et al., 2021). Most previous studies conducted with animal models demonstrated the beneficial effects of D-allulose on body weight, fat mass, and energy intake (Han et al., 2018; Hossain et al., 2015). In a short-term high-fat diet (HFD) animal model, D-allulose was shown to enhance glucagon-like peptide-1 (GLP-1) release, acting via vagal afferents to restrict feeding and hyperglycemia where pharmacological GLP-1R blockade and genetic inactivation of GLP-1R were applied (Iwasaki et al., 2018). Chronic HFD conditions involve ROS and its related ER stress mechanism to a greater extent than short-term feeding conditions (Zeeshan et al., 2016). However, recently focused GLP-1R agonists have been suggested to have some limitations due to GLP-1 resistance, especially in chronic/severe metabolic disorders (Zheng et al., 2024). Therefore, the GLP-1 receptor, an N-linked glycosylated protein, needs further investigation under chronic HFD-induced ER stress conditions with D-allulose. D-allulose is hypothesized to regulate GLP-1R through mechanisms involving ER stress and ROS pathways, potentially enhancing GLP-1R stability and function, which can be crucial in chronic metabolic conditions. In this study, liquid and powder forms of D-allulose were applied to 3T3-L1 preadipocytes and HFD conditions in GLP-1R knockout (KO) mice to determine the significance of GLP-1R in D-allulose’s protective mechanism against adipocyte differentiation and obesity.
Results
D-allulose regulates NADP+/NADPH-ROS-ER stress signaling and its associated adipocyte differentiation
First, NADP+/NADPH ratio and NADPH oxidase (NOX) activity were measured as NADPH-based redox imbalance because ROS signalings are intrinsically linked to obesity and adipocyte differentiation (Xiao et al., 2018). In D-allulose-treated condition, the NADP+/NADPH ratio (Fig. 1A), NOX activity (Fig. 1B), dihydroethidium (DHE) fluorescence (Fig. 1C), and lipid peroxidation-representing malondialdehyde (MDA) concentration (Fig. 1D) were significantly inhibited in each adipocyte differentiation culture on days 5, 7, and 10. However, on the 12th day of culture, no regulatory effect of D-allulose was observed. This loss of regulatory effect may be due to the onset of irreversible ER stress and pathological states associated with excessive differentiation (Varghese and Ali, 2021). Next, the NADPH imbalance-based ER stress signaling, IRE1α phosphorylation, and sulfonation were examined. In addition, D-allulose inhibited IRE1α phosphorylation and XBP-1 splicing (Fig. 1E) and IRE1α sulfonation (Fig. 1F) during the adipocyte differentiation period except for the 12th day. Interestingly, IRE1α downstream signaling, CUGCAG sequence-based RNA decay, called RIDD, was confirmed under differentiation condition. In contrast, the decay was inhibited in the D-allulose-treated condition (Supplementary Fig. S1A, Fig. 1G). Moreover, GLP-1R, a recently focused obesity and diabetes target molecule, was identified as the RIDD target gene through RNA decay experiments using a mutant (Fig. 1H). Furthermore, the treatment of 4μ8C, an inhibitor of IRE1α RNase activity, abrogated the decay of GLP-1R mRNA (Supplementary Fig. S1B). This observation demonstrates GLP-1R mRNA as an IRE1α-RIDD target, validating its association with the pathway. Consistent with these observations, the GLP-1R mRNA was decayed in differentiated adipocytes, whereas the decay was controlled in the presence of D-allulose (Fig. 1I). However, the decay pattern was not regulated in the D-allulose-treated condition at later stages, especially after 12 days. Consistently, adipocyte differentiation was regulated by D-allulose during the first 10 days, but not at the later stage, day 12 (Fig. 1J).
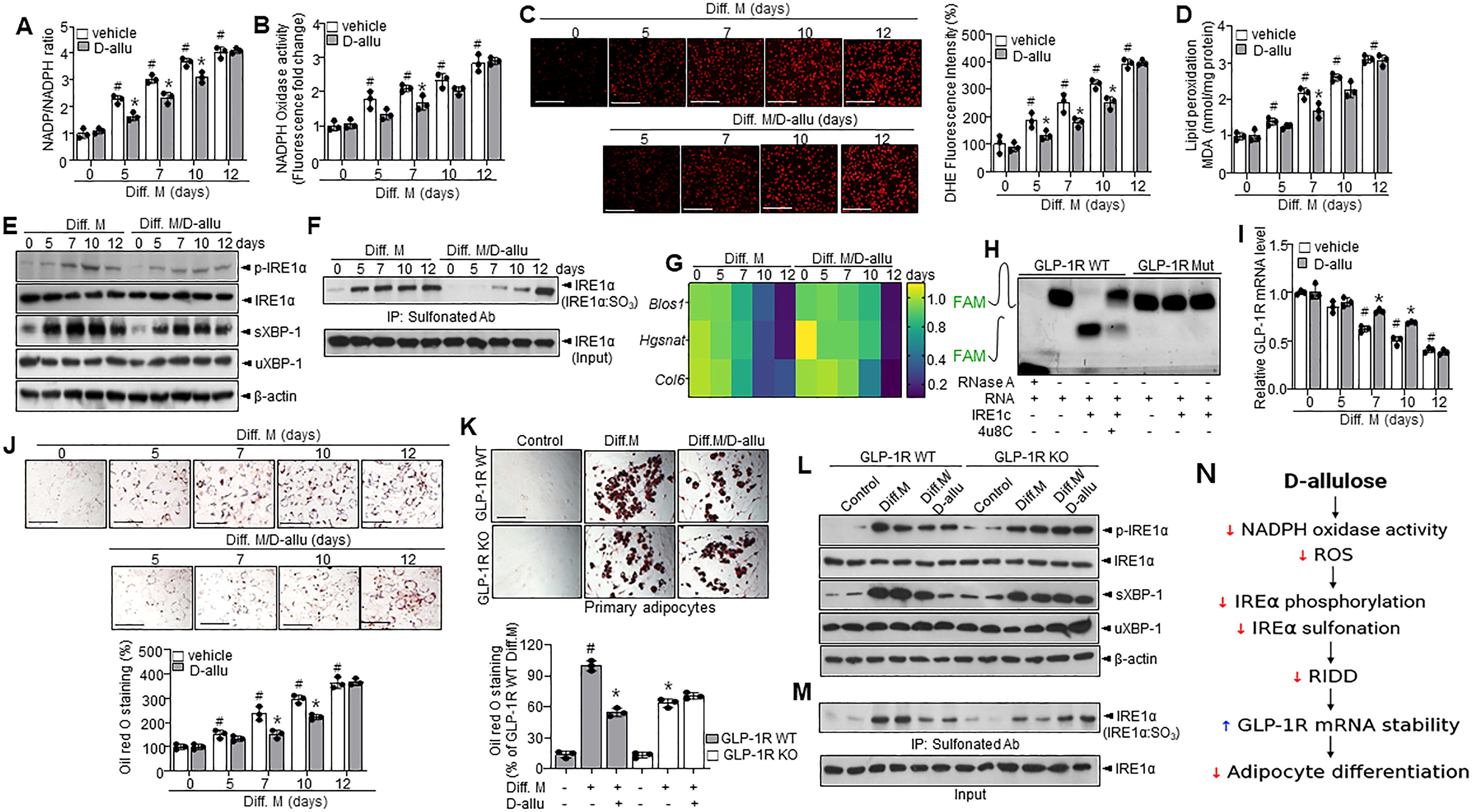
To confirm the role of GLP-1R in the inhibitory effect of D-allulose on adipocyte differentiation, D-allulose was applied to primary adipocytes derived from GLP-1R wild-type (WT) and KO mice. The regulatory effect of D-allulose on adipocyte differentiation was not observed in GLP-1R KO adipocytes (Fig. 1K and Supplementary Fig. S1C). Moreover, the inhibitory effect of D-allulose on adipocyte differentiation was attenuated in the presence of GLP-1R antagonist and enhanced in the presence of GLP-1R agonist (Supplementary Fig. S2A–D), indicating that the antidifferentiation effect of D-allulose is dependent on the presence of GLP-1R. NAC alone also inhibited adipocyte differentiation; however, co-treatment with D-allulose did not lead to an additive effect (Supplementary Fig. S3A, B).
In addition, phosphorylation of IRE1α and subsequent splicing of XBP-1 were decreased in D-allulose-treated differentiated adipocytes of GLP-1R WT mice compared with those of GLP-1R WT mice without treatment (Fig. 1L). Sulfonation of IRE1α was also decreased in D-allulose-treated differentiated adipocytes of GLP-1R WT mice compared with differentiated GLP-1R WT mice without treatment (Fig. 1M). In contrast, this regulatory effect of D-allulose was not observed in differentiated KO mice. Similar results were observed in bone marrow mesenchymal stem cells (BM-MSCs), as the inhibitory effect of D-allulose on differentiation was also absent under GLP-1R knockout conditions (Supplementary Fig. S4A, B). A time-course analysis following actinomycin D revealed that GLP-1R mRNA stability was decreased in adipocyte differentiation, whereas GLP-1R mRNA decay was regulated in D-allulose treatment (Supplementary Fig. S5A, B). Figure 1N illustrates the overall mechanism of D-allulose-mediated inhibition of adipocyte differentiation via ROS-IRE1α-GLP-1R signaling.
The involvement of the GLP-1R in the regulation of obesity by D-allulose
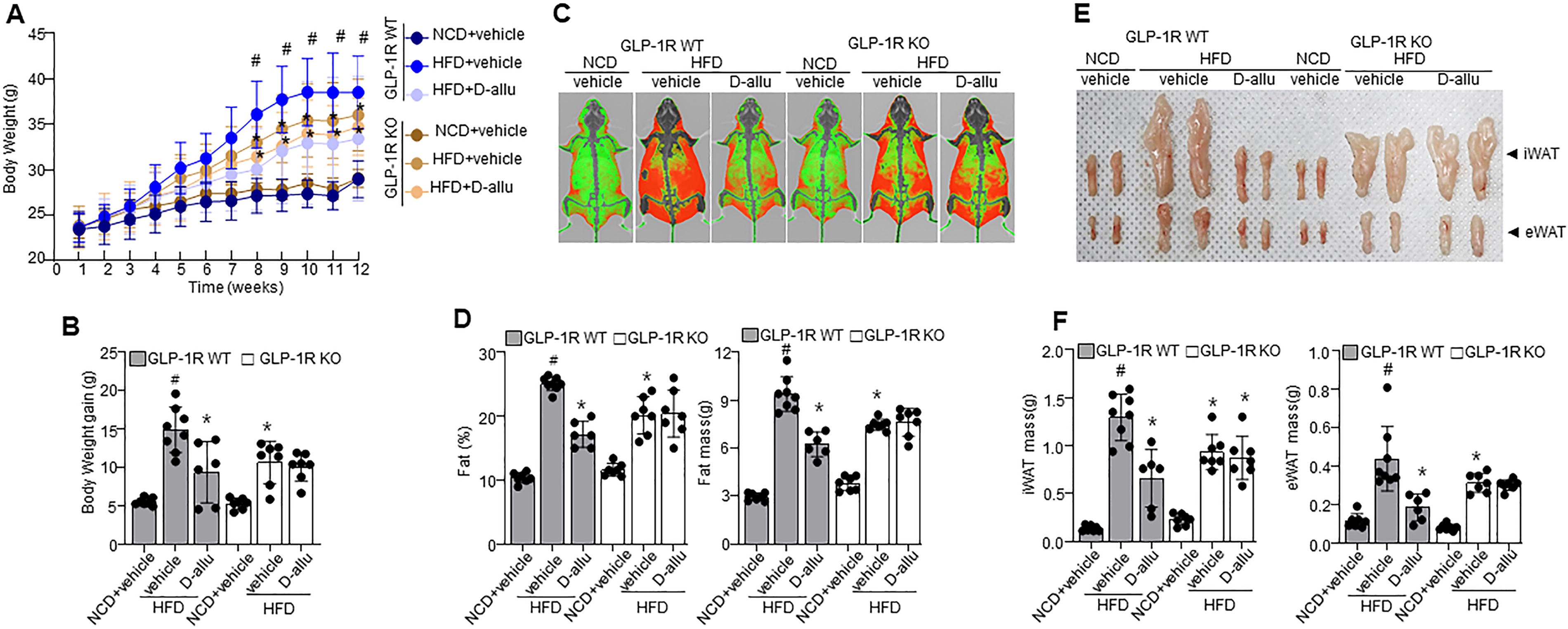
The study examined the effects of D-allulose on body weight and its dependence on GLP-1R. D-allulose administration significantly suppressed the increase in body weight in the HFD-fed GLP-1R WT mice (Fig. 2A). GLP-1R KO HFD mice showed increased body weight less than that in the GLP-1R WT HFD mice. However, D-allulose administration did not affect the weight gain of GLP-1R KO mice (Fig. 2B, Supplementary Fig. S6A). Dual energy X-ray absorptiometry (DEXA) scan demonstrates a reduction in orbital fat volume (%) and mass (g) in GLP-1R WT HFD mice supplemented with D-allulose (Fig. 2C,D). Similarly, inguinal white adipose tissue (iWAT) and epididymal WAT (eWAT) mass were reduced in GLP-1R WT HFD mice supplemented with D-allulose (Fig. 2E,F), indicating their regulatory effects. These observations correlate with the body weight observations. There was no difference in food intake between HFD-fed GLP-1R WT and HFD-fed GLP-1R KO mice (Supplementary Fig. S6B). Next, the serum concentrations of GLP-1 in WT and KO mice were compared. GLP-1 was increased considerably in HFD-fed GLP-1R WT and KO rodents, whereas D-allulose did not induce any significant difference (Table 1). Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were substantially elevated in HFD-fed GLP-1R WT mice, whereas D-allulose significantly regulated these enzyme levels. However, D-allulose did not alter the enzyme activity in the HFD-fed GLP-1R KO mice. Similarly, triglyceride (TG), total cholesterol (T-Chol), low-density lipoprotein cholesterol (LDL-Chol), and their associated endocrine factors, adiponectin and leptin, were significantly influenced by D-allulose in GLP-1R WT mice. At the same time, profiles were not affected in the D-allulose-administered HFD-fed GLP-1R KO mice. Furthermore, the OGTT and ITT analyses showed a consistent pattern. Here, the D-allulose-induced regulatory effect in GLP-1R WT HFD mice was greater than in GLP-1R KO HFD mice, indicating the GLP-1R dependence of the D-allulose effect on glucose and insulin tolerance (Supplementary Fig. S6C, D). However, there was no difference in diabetic parameters such as insulin, HbA1c, and fasting glucose. All the enzymes and biochemical analysis observations are shown in Table 1.
Biochemical and Metabolic Parameters in Mice Fed a Normal Chow Diet or High-Fat Diet with or Without D-allulose Treatment. Measurements Include Circulating GLP-1, Hepatic Enzymes (AST, ALT), Lipid Profiles (TG, T-CHOL, LDL-CHOL), Adipokines (Adiponectin, Leptin), Insulin, HbA1c, and Fasting Glucose Levels
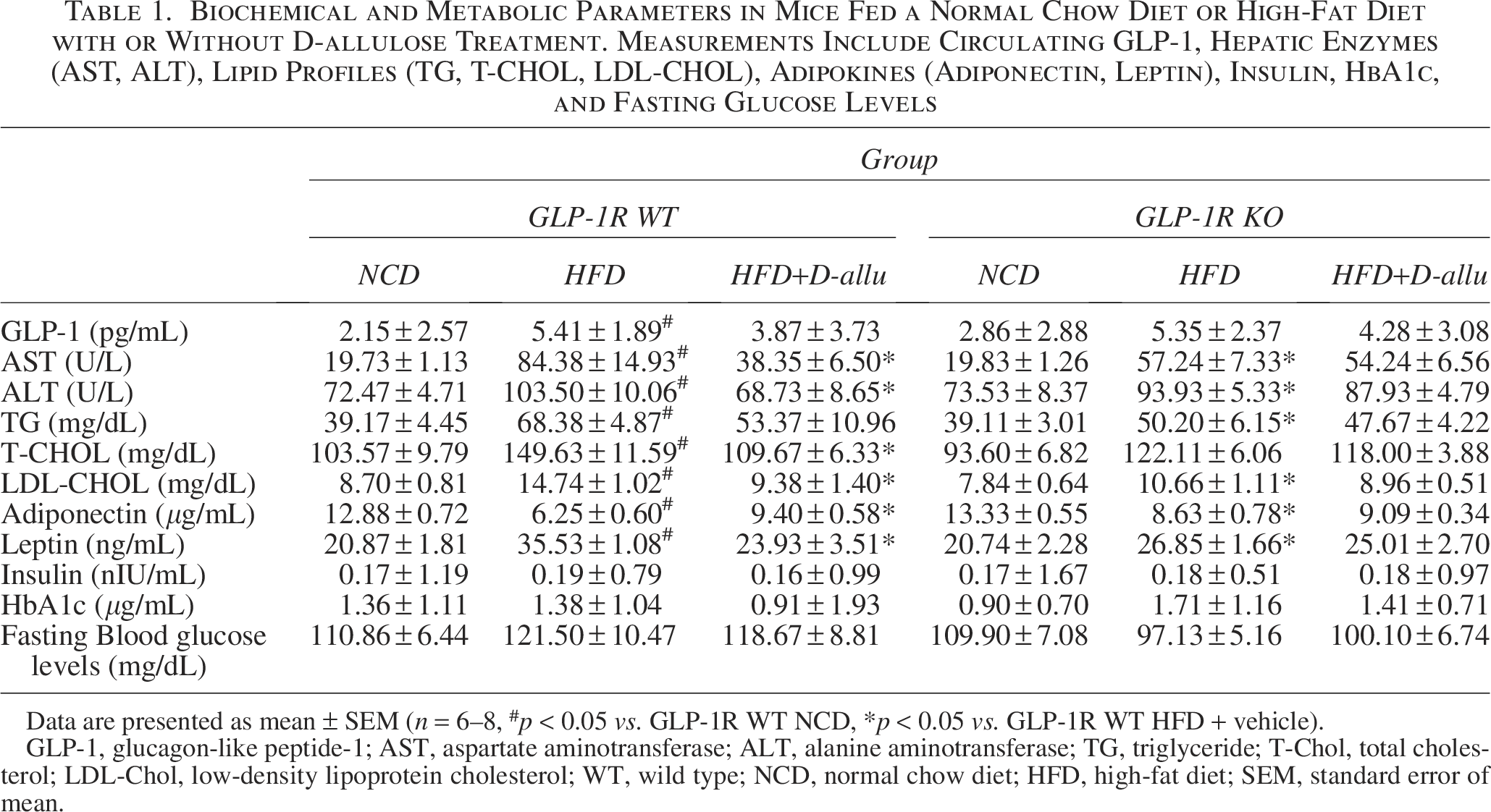
Data are presented as mean ± SEM (n = 6–8, #p < 0.05 vs. GLP-1R WT NCD, *p < 0.05 vs. GLP-1R WT HFD + vehicle).
GLP-1, glucagon-like peptide-1; AST, aspartate aminotransferase; ALT, alanine aminotransferase; TG, triglyceride; T-Chol, total cholesterol; LDL-Chol, low-density lipoprotein cholesterol; WT, wild type; NCD, normal chow diet; HFD, high-fat diet; SEM, standard error of mean.
D-allulose regulates RIDD/GLP-1R in white adipocytes and triggers GLP-1R-AMP-activated protein kinase in brown adipocytes
The mechanism underlying the antiobesity effect of D-allulose, specifically its influence on adipogenesis, requires further investigation. Therefore, this study first determined whether D-allulose inhibits adipocyte differentiation and assessed whether its regulating effect depends on GLP-1R. D-allulose significantly reduced adipocyte size in GLP-1R HFD WT mice compared with GLP-1R HFD KO mice (Fig. 3A). To investigate further the molecular mechanism, mRNA and protein expression levels of key adipogenic transcription factors were analyzed. As shown in Figure 3B, C, D-allulose treatment decreased the expression of sterol regulatory element-binding protein (SREBP)1, fatty acid synthase (FAS), C/EBPα, and PPARγ in HFD-fed WT mice, while no changes were observed in GLP-1R KO mice, confirming that the inhibitory effect of D-allulose on adipogenesis is GLP-1R dependent.
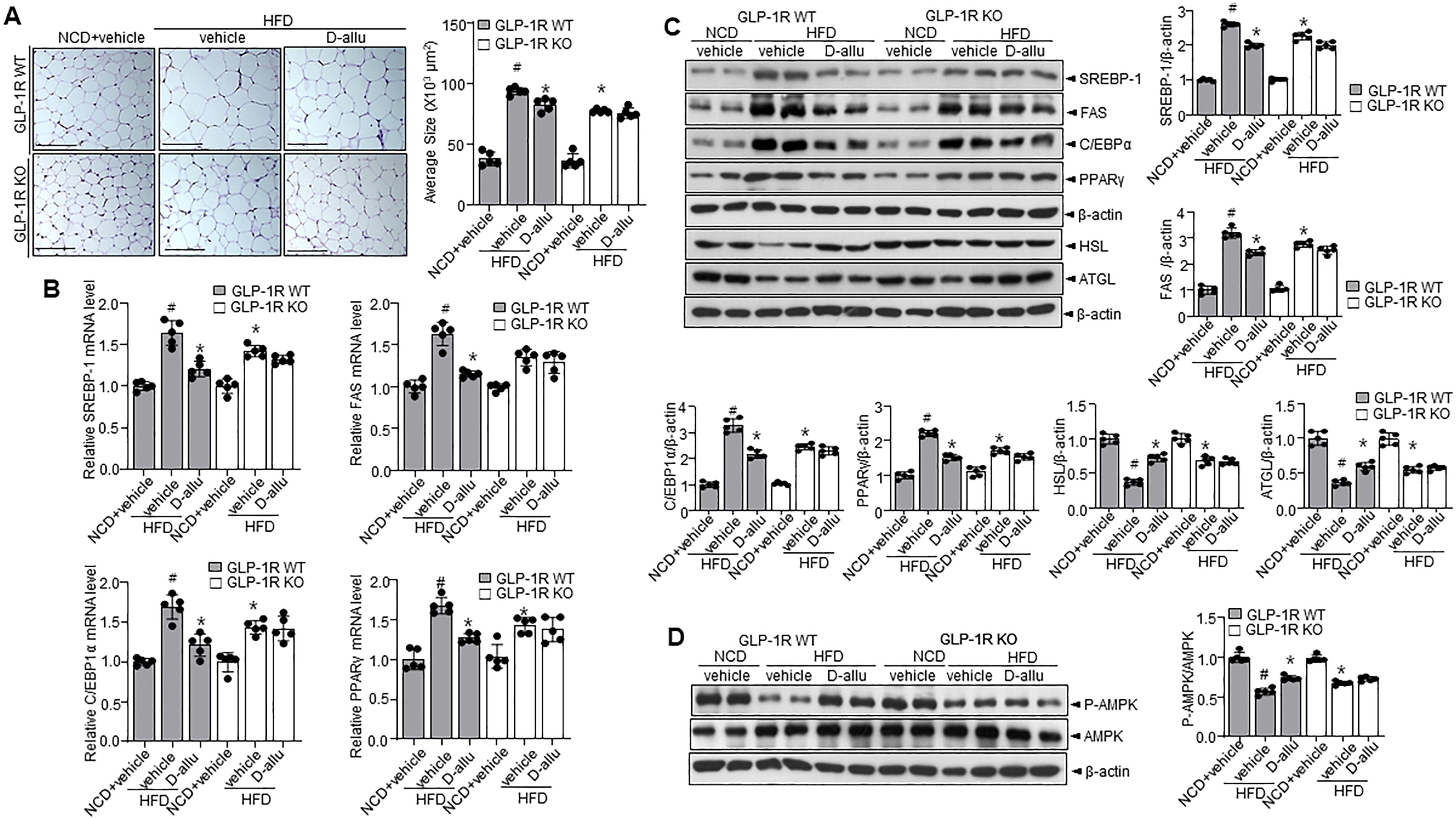
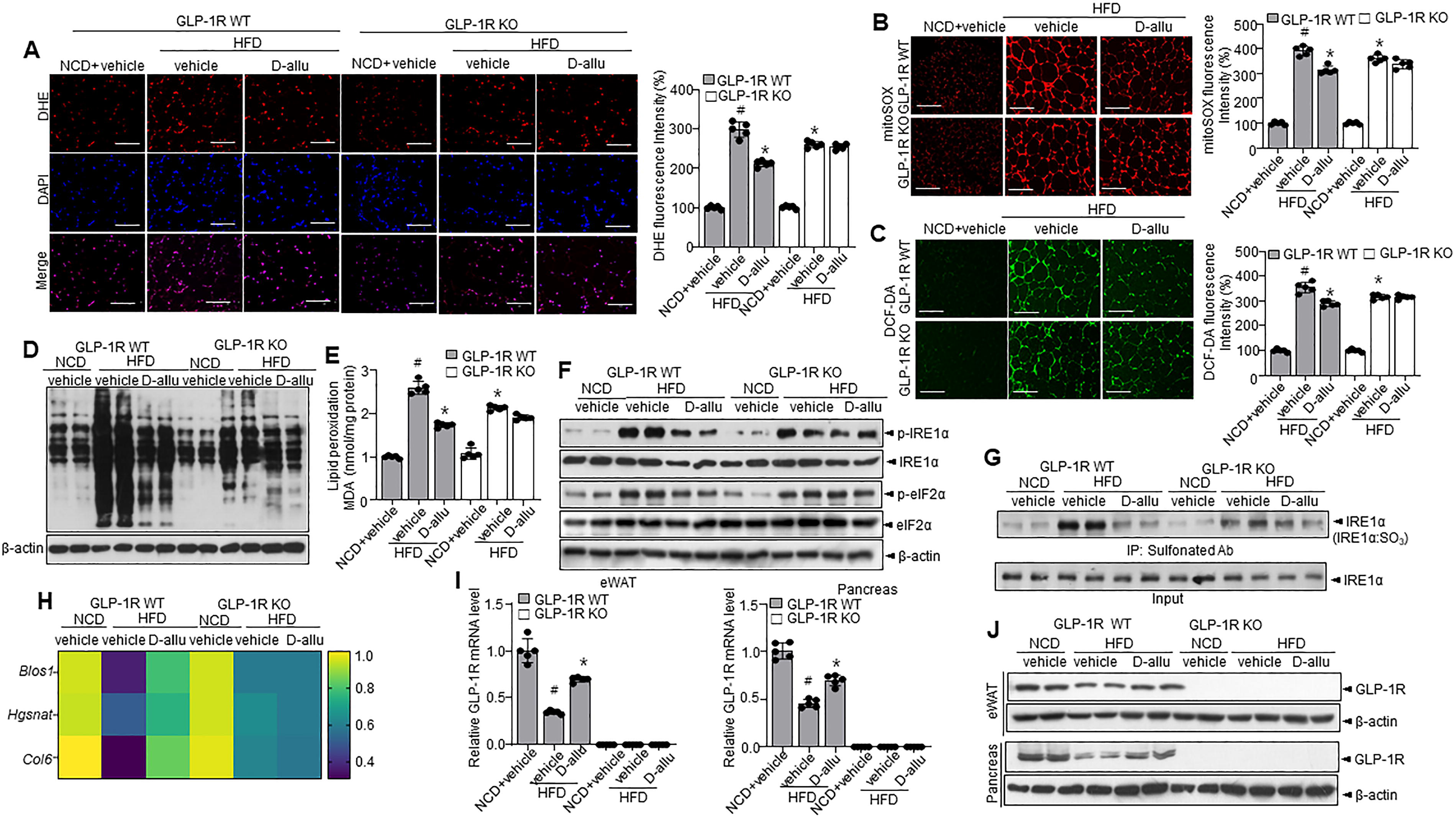
In addition, HSL and ATGL expression were assessed. The HSL and ATGL expression levels were increased in GLP-1R WT mice treated with D-allulose, whereas no significant changes were observed in GLP-1R KO mice. Next, AMP-activated protein kinase (AMPK) phosphorylation–sirtuin 1 (SIRT1) expressions associated with D-allulose were confirmed. Expectedly, D-allulose increased the phosphorylation of AMPK in the GLP-1R HFD-WT mice while not affecting the AMPK state in the GLP-1R HFD-KO mice (Fig. 3D). In addition, a cold tolerance test was performed to evaluate adaptive thermogenesis in D-allulose-administered GLP-1R HFD-WT and KO mice. Acute cold exposure caused a significant upregulation of body temperature in D-allulose-administered GLP-1R HFD-WT mice. In contrast, no difference was observed in GLP-1R HFD-KO mice (Supplementary Fig. S7A). D-allulose increased the expression of UCP-1, a representative thermogenesis protein, in GLP-1R HFD-WT mice but not in GLP-1R HFD-KO mice (Supplementary Fig. S7B–E). Multiple investigations indicate that ROS and related ER stress are necessary for adipogenesis and insulin resistance (Masschelin et al., 2019). Thus, ROS was measured via DHE staining. Expectedly, D-allulose reduced ROS accumulation in GLP-1R HFD-WT mice, but not in GLP-1R HFD-KO mice (Fig. 4A). Similar results were observed with MitoSOX and 2’,7’-dichlorofluorescein diacetate (DCF-DA) staining (Fig. 4B,C). Consistent with these observations, OxyBlot assay revealed a regulatory effect of D-allulose in GLP-1R HFD-WT mice (Fig. 4D). Similarly, the lipid peroxidation assay demonstrated a regulatory effect of D-allulose in GLP-1R HFD-WT mice but not in GLP-1R HFD-KO mice (Fig. 4E). Next, ER stress response, including the phosphorylation of IRE1α and eIF2α and its subsequent XBP-1 splicing, was decreased in D-allulose-administered GLP-1R HFD-WT mice, compared with vehicle-administered GLP-1R HFD-WT mice (Fig. 4F). The post-translational modification (PTM), sulfonation of IRE1α, was decreased in D-allulose-administered GLP-1R HFD-WT mice than the vehicle-administered GLP-1R HFD-WT mice (Fig. 4G). A similar expression pattern was observed with IRE1α-RIDD target genes like Blos1, Hgsnat, and Col6 (Fig. 4H). Notably, GLP-1R, a RIDD target gene, was also decayed in GLP-1R HFD-WT mice, while it was recovered with D-allulose supplementation (Fig. 4I). Similar patterns were observed with respect to GLP-1R protein expressions in eWAT and pancreas from D-allulose-administered GLP-1R WT and KO mice (Fig. 4J). D-allulose supplementation increased the expression of SIRT1, a recently identified RIDD target (Lee et al., 2022) in GLP-1R HFD-WT mice. In contrast, the D-allulose regulatory effect was not observed in the KO mice (Supplementary Fig. S8A, B).
D-allulose shows antiobesity effect in different formulations
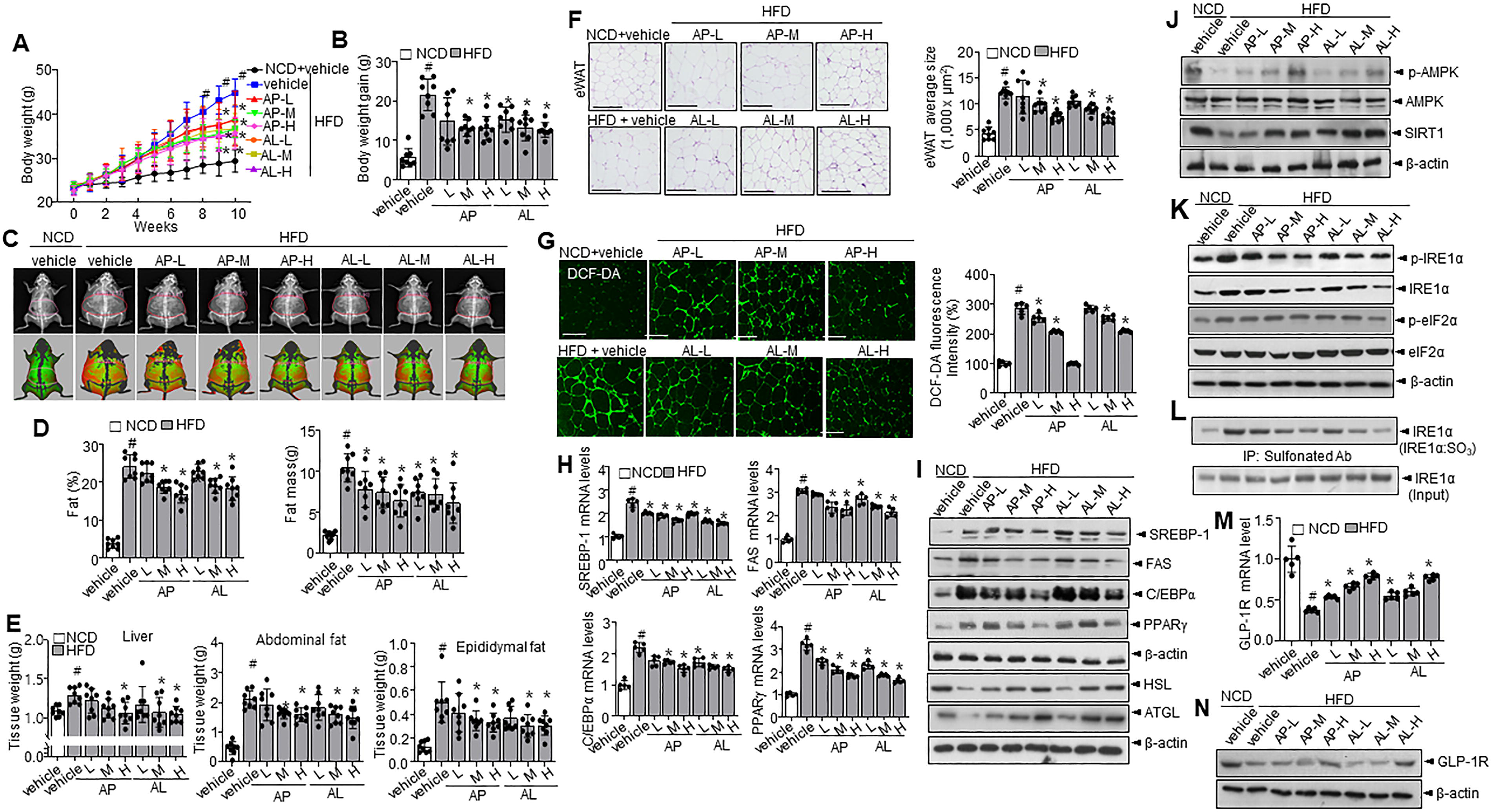
To determine the dose-dependent effect of D-allulose and the effect of different formulations of D-allulose (liquid and powder), HFD mice were administered with powder and liquid formulations of D-allulose. Expectedly, body weight, weight gain, and the weight of specific organs, including liver, abnormal fat, and epidermal fat, decreased significantly in a dose-dependent manner in both the D-allulose formulations (Fig. 5A,B). DEXA scan observations support these observations, where different forms of D-allulose effectively reduced the fat and fat mass (Fig. 5C–E). Furthermore, hepatic enzymes like AST and ALT were decreased in the presence of D-allulose powder and liquid formulations (Supplementary Table S1). Similarly, lipid profiles, including TG, total cholesterol, and LDL, were significantly decreased in both D-allulose formulations. In addition, leptin levels decreased, and adiponectin levels increased in mice administered both D-allulose formulations. However, fasting glucose levels and OGTT analysis showed no difference between groups (data not shown). Since D-allulose inhibited HFD-induced obesity, we further investigated the effect of D-allulose on the morphology of different adipose depots. Hematoxylin and eosin (H&E) staining showed that adipocyte size in the eWAT of HFD-fed mice was notably larger than in the normal chow diet (NCD) mice, as indicated by increased adipocyte area in the eWAT, whereas administration of both D-allulose forms reduced the increased size in a dose-dependent manner (Fig. 5F). ROS was measured via DCF-DA staining. Expectedly, D-allulose reduced ROS accumulation in HFD-fed mice (Fig. 5G). Moreover, D-allulose specifically affected the expression of lipid metabolism-related genes and proteins, including SREBP1, FAS, C/EBPα, PPARγ, HSL, ATGL, p-AMPK, and SIRT1, in eWAT (Fig. 5H–J). In addition, D-allulose regulated ER stress response proteins, including sulfonation of IRE1α, p-IRE1α, and p-eIF2α (Fig. 5K,L). GLP-1R mRNA and protein expression were decreased in the HFD condition, whereas the expression was recovered with D-allulose administration (Fig. 5M,N). GLP-1 was increased in the HFD condition, but dose-dependently decreased with administration of different formulations of D-allulose (Supplementary Table S1). Food intake was observed to be similar in all the groups (Supplementary Fig. S9A). In BAT analysis, the adipocyte size in BAT was increased in HFD condition, whereas different formulations of D-allulose restricted the size of adipocytes (Supplementary Fig. S9B). Furthermore, thermogenesis-browning associated genes and proteins like PGC1α and UCP-1 were highly increased in the D-allulose-administered condition (Supplementary Fig. S9C–E). In the BAT, the D-allulose supplementation recovered the decreased phosphorylation of AMPK and expression of SIRT1, inhibiting the acetylation of PGC-1α in a dose-dependent manner (Supplementary Fig. S9F, G), indicating that AMPK-SIRT1-PGC-1α axis is enhanced by the D-allulose irrespective of its form. We also examined GLP-1R activation by assessing the phosphorylations of ERK and CREB, which are downstream signaling molecules indicative of GLP-1R activation (Oh and Jun, 2017) (Supplementary Fig. S10A,B). D-allulose treatment was associated with increased phosphorylation of these proteins, suggesting a potential involvement of D-allulose in GLP-1R signaling pathways.
Discussion
The critical roles of ROS and ER stress in the pathophysiology of obesity make them ideal targets for developing prospective obesity management therapies. This study demonstrated that HFD-induced ROS production was mediated by the imbalance between NADP+/NADPH and upregulation of IRE1α sulfonation, a master ER stress response signal, and associated RIDD-GLP-1R mRNA degradation could be controlled by D-allulose treatment. In GLP-1R KO or decayed condition, D-allulose displays little effect against adipocyte differentiation and obesity-associated metabolic markers, suggesting that D-allulose has a controlling effect against adipocyte differentiation and its associated lipid dysmetabolism and obesity, at least under GLP-1R-maintained state.
The study observations strongly suggest that D-allulose regulates adipocyte differentiation by preserving the NADP+/NADPH balance and stabilizing the IRE1α-RIDD/GLP-1R mRNA axis.
Interestingly, the failure of D-allulose to suppress oxidative stress, lipid peroxidation, and adipogenesis at day 12 can be attributed to the extensive incubation period. In addition, significant ROS accumulation and irreversible IRE1α sulfonation develop by day 12 (Fig. 1C,D,F), leading to increased RIDD activity and subsequent GLP-1R mRNA decay (Fig. 1G–I). Consequently, the beneficial effects of D-allulose are not observed, as GLP-1R is not functioning optimally, indicating that D-allulose requires a functional GLP-1R to exert its effects against adipocyte differentiation. In addition, it highlights the limitations of D-allulose, as its effectiveness is diminished under highly stressful conditions. To confirm the role of GLP-1R in D-allulose’s inhibitory effect on adipocyte differentiation, we applied D-allulose to primary adipocytes from GLP-1R WT and KO mice. The regulatory role of D-allulose on differentiation was not observed in the GLP-1R KO primary adipocytes (Fig. 1K), and the role against adipocyte differentiation was also inhibited in the presence of GLP-1R antagonist 1 (Supplementary Fig. S2A,B), indicating that the regulatory role of D-allulose against adipocyte differentiation is dependent on the presence of GLP-1R.
Spliced XBP-1 and RIDD activity suggest the shift in the stress response, determining the differentiation process. The phosphorylation and sulfonation of IRE1α unfolded during the XBP-1 to RNA decay or simultaneous process of XBP-1 splicing and RIDD. IRE1α dimerization and oligomerization state are considered a switch for adaptation to alarm signaling or from phosphorylation to sulfenylation–sulfonation (Abo and Weerapana, 2019). The study observations indicated that sulfonation followed or coincided with phosphorylation (Fig. 1E,F), which could be correlated with ROS accumulation and consequent adipocyte differentiation (Fig. 1C,D,J,K). Similarly, previous investigations proposed a ROS-based adipocyte differentiation theory in which aging and high-calorie diet-induced obesity are discussed (Kawai et al., 2021). ROS in adipocytes can contribute to insulin resistance, inflammation, and metabolic dysfunction, which are key factors in obesity-related disorders. The antioxidant defenses in different tissues, such as the liver and kidneys, might have developed more due to their roles in detoxification and metabolism, potentially explaining why adipose tissue might be more vulnerable to ROS-induced damage in obesity (Furukawa et al., 2004; Jensen, 2006). In this study, ROS-linked IRE-1α PTM, sulfonation-RIDD/GLP-1R mRNA, and complete loss of GLP-1R mRNA were observed during the late differentiation period (Fig. 1F–I). The late stage of adipocyte differentiation was unaffected by D-allulose (Fig. 1J), indicating that the D-allulose effect might be maintained in GLP working state, but not in a GLP-resistant state. Furthermore, the decayed expression of GLP-1R over an extended period of 12 days under D-allulose clearly suggests that the beneficial effect of D-allulose is dependent on the expression of GLP-1R. Consistently, the GLP-1R KO primary adipocytes and BM-MSC differentiation process showed a similar pattern (Fig. 1K, Supplementary Fig. S4A,B). Besides, the inhibition of GLP-1R through GLP-1R antagonist (Supplementary Fig. S2A,B) abolished the regulatory effect of D-allulose on adipocyte differentiation at the final stage of adipocyte differentiation, 12 days. However, D-allulose showed a regulatory effect throughout the differentiation process, indicating that its beneficial effects are dependent on the expression of GLP-1R. In addition, the experiment with actinomycin D suggests that the regulatory effect of D-allulose depends on the expression and stability of GLP-1R mRNA rather than its transcription (Supplementary Fig. S5A,B).
In this study, body weight observations indicate the antiobesity effect of D-allulose in GLP-1R WT mice, whereas the antiobesity effect of D-allulose appears ineffective in GLP-1R KO mice (Fig. 2A,B). The secretion of GLP-1 was highly increased in the HFD mice, whereas D-allulose controlled the secretion, and levels of GLP-1 were similar to GLP-1R KO mice (Table 1). In contrast, oral administration of D-allulose was reported to induce GLP-1 release, activate vagal afferents, reduce food intake, and promote glucose tolerance via enhanced insulin secretion and action in the 1-week HFD model (Iwasaki et al., 2018). Moreover, GLP-1-resistant conditions in HFD-induced obesity suggest that substantial GLP-1 release does not affect the interaction between its receptor and downstream signaling. This suggests that controlling GLP-1R by D-allulose is absolutely vital in reducing the resistance and preserving the sensitivity of the receptor, consequently guaranteeing its metabolic advantages in chronic conditions.
Proteins associated with lipid synthesis pathways, including SREBP1, PPARγ, and C/EBP1α, were downregulated by D-allulose, along with activation of the AMPK pathway (Fig. 3B–D). These findings confirm the role of D-allulose in suppressing adipocyte differentiation via GLP-1R-dependent regulation of key metabolic transcription factors. Furthermore, while this study primarily focuses on adipocyte differentiation, an increase in HSL and ATGL expression was observed in GLP-1R WT mice treated with D-allulose, suggesting a potential link between GLP-1R stability and lipolysis regulation. However, this effect was not observed in GLP-1R KO mice, indicating that further studies are needed to determine whether D-allulose modulates lipolysis through GLP-1R-dependent mechanisms. Consistent with these observations, recently reported pathways such as AMPK-SIRT1 and IRE1α-RIDD/SIRT1 have been implicated in HFD-induced obesity regulation (Lee et al., 2022). The study further confirms that ROS-mediated ER stress, IRE1α sulfonation, and RIDD activity serve as metabolic switches regulating GLP-1R mRNA decay (Fig. 4). These in vivo findings are supported by in vitro results demonstrating IRE1α sulfonation and subsequent GLP-1R mRNA degradation (Fig. 1). Interestingly, the antiobesity and metabolic regulatory effects of D-allulose were absent in GLP-1R KO mice, aligning with observations from primary adipocyte differentiation and BM-MSC differentiation models (Fig. 1K, Supplementary Fig. S4A,B). In addition, the absence of an additive effect when co-treating NAC with D-allulose suggests that D-allulose primarily regulates GLP-1R stability through an ROS-associated ER stress mechanism, particularly via IRE1α sulfonation and RIDD. This supports the idea that D-allulose acts through a central ROS regulatory mechanism rather than through general antioxidant effects (Supplementary Fig. S3A,B). Previous reports suggest that D-allulose is an attractive candidate for preventing and ameliorating obesity and diabetes (Hossain et al., 2015). However, the findings of this study specifically demonstrate that D-allulose’s effects are largely dependent on GLP-1R stability and function, suggesting that its role in obesity management may be less effective in GLP-1R-impaired states.
In addition, the observations indicate that D-allulose supplementation can be a helpful supplement in prediabetes or preobesity individuals with a normal GLP-1R function. Despite D-allulose improving glucose and insulin tolerance via GLP-1R, there were no significant changes in long-term diabetic markers such as insulin, HbA1c, and fasting glucose over the 12 weeks. This outcome may be attributed to the short-term effects of D-allulose, which appear insufficient to influence long-term markers. In addition, other metabolic pathways and compensatory mechanisms might counteract the benefits observed in short-term experiments.
The GLP-1R knockout model of the study primarily focused on adipose tissue, with interpretations derived from adipose tissue data. However, GLP-1R is weakly expressed in adipose tissue compared with other tissues like pancreatic beta cells, the duodenal part of the intestine, and the heart. Therefore, it is important to consider the broader context of GLP-1R involvement in obesity, including its role in pancreatic islets, liver, and muscle. For instance, insulin secretion, a key component in managing obesity and diabetes, relies on GLP-1R signaling in pancreatic beta cells. In the liver, GLP-1R impacts insulin sensitivity and glucose uptake in muscle tissue, in addition to glucose synthesis and lipid metabolism. Collectively, these organs play a role in the manifestation of the obesity phenotype seen in GLP-1R KO models. Thus, a clear understanding of GLP-1R function in each tissue helps better understand the effect of D-allulose and its therapeutic potential.
In this study, different forms of D-allulose demonstrated similar regulating effects against HFD-induced obesity and its related adipocyte metabolism. Specifically, D-allulose in liquid or powder form significantly restored the decreased expression of GLP-1R mRNA (Fig. 5M,N) and demonstrated a similar regulatory effect on adipocyte metabolism and obesity. Furthermore, GLP-1 receptor agonists like GLP-1 RA and DPP-IV inhibitors have some clinical limitations. Generally, a high dose of GLP-1RAs may be needed for weight loss in obesity because it is more effective at reducing food intake and body weight (Nauck et al., 2021). However, 100 pM of GLP-1 RA is detected following therapeutic subcutaneous administration of exendin 4, a GLP-1 receptor agonist (Fehmann et al., 1995). Besides, DPP-IV inhibitors can increase GLP-1 concentration, but these agents are considered weight neutral, suggesting that elevation of endogenous incretin levels is insufficient to promote weight loss. Indeed, DPP-IV inhibitors such as sitagliptin increase glucose-stimulated insulin secretion but do not induce significant weight loss, unlike GLP-1RA (Razavi et al., 2022). These investigations strongly suggest that the antidiabetic and obesity roles of GLP-1 require a higher blood concentration of GLP-1 than its physiological concentration. Notably, GLP-1R binding potential in the obese pancreas was observed to be reduced by 75% compared with lean animals (Malbert et al., 2020). This observation can explain that a lower GLP-1R density accounts for the diminished pharmacological effect of GLP-1R agonists in patients with type II diabetes and obesity. Thus, the normal functioning of GLP-1R is essential to suppress the progression of diabetes and obesity. Moreover, GLP-1R KO conditions potentially influence the vital parameters. In this study, body weight, adipose tissue mass, and associated endocrine factors, including cholesterol, LDL, and leptin, were significantly lower in the HFD GLP-1R KO mice than in the HFD GLP-1R WT mice (Fig. 2, Table 1). It has been reported that cellular lipid accumulation correlates with tissue insulin resistance, suggesting that a decrease in hepatic lipid accumulation in GLP-1R KO mice could positively influence insulin action (Ayala et al., 2010). This study also suggests that the reduced lipid accumulation may overcome any deleterious effect of disrupting GLP-1R expression on hepatic insulin action in HFD-feeding mice.
In conclusion, D-allulose inhibits ROS-associated ER stress signaling and IRE1α-RIDD/GLP-1R mRNA expression and regulates body weight gain and its associated metabolic factors in endogenously GLP-1R-maintained conditions but not in GLP-1R mRNA decayed or knockout state. The GLP-1R downstream pathway exerts its effects against adipogenesis through several mechanisms, including activating the AMPK pathway, which enhances lipid metabolism and reduces lipogenesis. GLP-1R activation also leads to increased insulin sensitivity and the suppression of pro-adipogenic transcription factors such as PPARγ and C/EBPα. In a “GLP-1R mRNA-fully decayed state,” the expression of GLP-1R mRNA is significantly reduced or absent, impairing signaling through these pathways and resulting in diminished effects on adipocyte differentiation and lipid metabolism. Interestingly, the study findings reveal that D-allulose demonstrated comparable efficiency against obesity, regardless of its formulation, exhibiting equivalent performance in both liquid and powder forms. Collectively, the study strongly suggests that D-allulose effectively regulates weight gain in mild or moderately obese conditions with normal GLP-1R function.
Materials and Methods
Chemicals
GLP-1R antagonist 1 was purchased from MedChemExpress LLC (NJ, USA). Dulbecco’s modified Eagle’s medium (DMEM), penicillin–streptomycin, and fetal bovine serum (FBS) were obtained from Gibco BRL (Grand Island, NY, USA). Insulin, 3-isobutyl-1-methylxanthine (IBMX), and dexamethasone were purchased from Sigma Aldrich (St. Louis, MO, USA). NADP/NADPH ratio assay kit was purchased from Abcam (ab65349, Cambridge, UK). DHE was purchased from Thermo Fisher Scientific (Waltham, MA, USA).
Cell culture and differentiation of 3T3-L1 preadipocytes
3T3-L1 preadipocyte cells were purchased from the American Type Culture Collection (Manassas, VA, USA) and were maintained in DMEM containing 10% FBS. Cells were seeded in DMEM containing 10% FBS and penicillin/streptomycin mixture and maintained up to the contact inhibition stage for 2 days. Differentiation of 3T3-L1 was stimulated by adding induction media containing 10% FBS along with 0.5 mM IBMX, 0.5 µM dexamethasone, and 5 µg/mL insulin in the presence or absence of D-allulose. After 48 h of differentiation induction, DMEM supplemented with 10% FBS and 5 µg/mL insulin was replaced for the next 3 days with or without D-allulose. Later, cells were nourished daily with DMEM supplemented with 10% FBS until 12 days in the presence or absence of D-allulose.
Oil red O staining
Oil red O (ORO) staining was performed as described earlier (Ramirez-Zacarias et al., 1992). Briefly, the 3T3-L1 cells and liver sections were rinsed, fixed, and stained with ORO solution at room temperature. The residual dye was then removed from the tissue sections/cells using 60% isopropanol, and images were acquired using a Nikon microscope (Nikon Co., Ltd., Tokyo, Japan).
Measurement of MDA
Cell lysate was mixed with 40 mM Tris-HCl buffer and centrifuged at 12,000× g for 10 min at 4°C to obtain supernatant. MDA levels were assessed with OxiTec™ TBARS (Lipid peroxidation) assay kit (#BO-TBR-200, BIOMAX Inc., Gyeonggi-do, Republic of Korea) by following manufacturer guidelines.
Isolation and differentiation of primary adipocytes
Epididymal fat pads were harvested from male mice following euthanasia and sterilization of the abdominal region. The fat pads were excised, minced, and digested with collagenase type I (1–2 mg/mL) at 37°C for 30 min to isolate the stromal vascular fraction (SVF). After filtering and centrifugation at 300–500 g, the SVF pellet was resuspended in DMEM with 10% FBS and 1% penicillin–streptomycin and cultured at 37°C with 5% CO2 until cells reached 80%–90% confluence. For adipogenic differentiation, cells were treated with a differentiation medium containing 10 µg/mL insulin, 1 µM dexamethasone, and 0.5 mM IBMX for 2–3 days. Later, differentiating cells were maintained in a medium with 10 µg/mL insulin and refreshed every 2–3 days. After 5–7 days, differentiation was confirmed by ORO staining to visualize lipid droplets.
Immunoblotting
Immunoblotting was conducted following standard protocols. Briefly, separated protein samples were transferred onto the PVDF membrane. Following the transfer, membranes were blocked with skimmed milk and incubated with specific antibodies. Then the membrane is then rinsed and incubated with secondary antibodies. The following antibodies were used in the study: AMPK (#2532), phosphorylation of AMP-activated kinase (p-AMPK, P-2535), IRE1α (#3294), CHOP (#2895), ATGL (#2138), p-ACC (#11818), ACC (#3676), HSL (#4107), p-eIF2α (#9721), eIF2α (#9277), and sXBP-1 (#40435 were purchased from Cell Signaling, MA, USA. uXBP-1(#sc-8015), GLP-1R (#sc-390774), SREBP-1c, #sc-365513), peroxisome proliferator-activated receptor α (PPARα, #SC-398394), PPARγ (#sc-7273), C/EBPα (#sc-166258), FAS (#sc-48357), SIRT1 (#sc-74465), GRP78 (#sc-13539), and β-actin (#sc-47778) were purchased from Santa Cruz Biotechnology, whereas p-IRE1α antibody (#PA1-16927) was purchased from Invitrogen, Waltham, MA, USA.
IRE1α sulfonation
Sulfonation was performed as described previously (Kim et al., 2021). The total WAT lysates (∼500 μg) were prepared using a lysis buffer (Cell signaling) comprising protease and phosphatase inhibitor cocktail (Sigma). To detect sulfonation of IRE1α, immunoprecipitation was performed using anti-cysteine-sulfonate (ab176487, Abcam) derived from lysates, followed by application of the anti-IRE1α antibody (3294, Cell signaling). Protein A/G Sepharose beads (Sigma) were added and incubated for an additional 1 h. Immunoprecipitates were washed with phosphate-buffered saline (PBS)-T buffer or PBS five times before being resolved by SDS-PAGE and were immunoblotted with the indicated antibodies.
Quantitative real-time polymerase chain reaction
Quantitative real-time polymerase chain reaction (qPCR) was conducted using standard methods. In brief, total RNA was extracted from tissues using TRIzol (#15596026, Invitrogen, Waltham, MA, USA). Reverse transcription was performed using oligo dT primers with 2 µg of RNA. SYBR™ Green PCR Master Mix (#4309155, Applied Biosystems, Foster City, CA, USA) was used to measure mRNA expression of the target genes, and PCR was conducted on an ABI PRISM 7500 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA). The comparative cycle threshold method was used for quantification. Primer sequences used for qPCR are listed in Supplementary Table S1.
Animal experiment: The application of GLP-1R KO mice
Seven-week-old GLP-1R WT and KO C57BL6/J mice were obtained from JACKSON LAB, Bar Harbor, ME, USA. Mice were fed with a NCD or HFD with vehicle or 5.16 g/kg D-allulose (Samyang Ltd, Seongnam, Kyungki do, Korea) once daily for 12 weeks by oral gavage. The body weight of the experimental animals was measured once a week for 12 weeks. Daily food intake was determined by subtracting the residual food from a weighed aliquot and dividing it by the number of mice within the cage. All the animals were anesthetized and euthanized at the end of the 12th week. Mice were euthanized following an intraperitoneal injection of 100 mg/kg ketamine (#VINB-KET0-7021, Henry Schein Animal Health, Dublin, OH, USA) with 10 mg/kg xylazine hydrochloride supplement (#X1251-1G, Sigma-Aldrich). Blood samples were collected from the truncal vein. The adipose tissue sections and liver sections were used for immunoblotting, qPCR, and histological analysis. All dissected tissues were immediately frozen in liquid nitrogen and stored at −80°C until further analysis. Housing and handling of animals were done as suggested in the Jeonbuk National University Hospital animal care and use committee guidelines, which comply with ARRIVE guidelines. The approval number assigned to this study is JBUH-IACUC-2020–23. Blinding and randomization were applied where appropriate; however, genetic group allocation (e.g., knockout vs. wild-type) could not be blinded or randomized due to facility regulations and genotype labeling requirements.
In vitro cleavage assay
Recombinant Human IRE1α (465–977) proteins (IRE1c) were purchased from Sino Biological Inc. (Beijing, China). 5′-Carboxyfluorescein (FAM)- and 3′-Black Hole Quencher (BHQ)-labeled GLP-1R-WT single stem-loop mini-substrate (5′FAM-GGCCUCUGCAGUGCC-3′BHQ) (Itzhak et al., 2014) and 5′-FAM and 3′-BHQ labeled GLP-1R mutant single stem-loop mini-substrate (5′FAM-GGCCUCUCCAGUGCC-3′BHQ) were purchased from Bioneer (Daejeon, Republic of Korea). RNA cleavage reactions were conducted at 30°C for 30 min in cleavage buffer containing 20 mM HEPES (pH 7.4), 70 mM NaCl, 2 mM ATP, 2 mM MgCl2, 5 mM DTT, and 5% glycerol as described before (Karagoz et al., 2019). IRE1c was used with 0.5 μg for the reaction. A measure of 10 μM 4u8C was preincubated for 10 min on ice to inhibit IRE1c. The reaction was quenched by adding urea to a final concentration of 4 M, the cleavage products were resolved by 15% TBE-Urea PAGE, and the gels were stained with a SYBR-gold nucleic acid gel stain solution. The band intensity was quantified.
Animal experiment: The application of D-allulose with different formulations, liquid, and powder
To evaluate the potential effectiveness and practical applications of D-allulose in obesity management, both liquid and powder formulations were tested. Liquid formulations are often associated with enhanced bioavailability and faster absorption, whereas powder forms are favored for their longer shelf life and convenience in handling and administration (Chen et al., 2022). These considerations guided the evaluation of D-allulose’s effects in different formulations. To apply different formulations of D-allulose, 7-week-old male mice were fed with HFD or a NCD for 10 weeks. All mice were randomly divided into eight groups (n = 8/group): normal control (NCD; water), negative control (HFD; water), three test groups [HFD; allulose powder (AP)-low, AP-medium, and AP-high], and three test groups [HFD; allulose liquid (AL)-low, AL-medium, and AL-high]. D-Allulose was administered at doses of 1.02 g/kg (low), 3.07 g/kg (medium), or 5.16 g/kg (high) to the appropriate test groups for 10 weeks. Powdered D-allulose was mixed with water to make a 10 mL/kg stock concentration before preparing the experimental dosage. Test products (water and D-allulose) were administered daily via oral gavage. Housing and handling of animals were done as suggested in the Jeonbuk National University Hospital animal care and use committee guidelines. The approval number assigned to this study is JBUH-IACUC-2020–5.
Serum assay
The levels of TG, T-Chol, LDL-Chol, ALT, and AST in serum were measured with biochemical analyzer (Hitachi-7020, Hitachi Medical, Tokyo, Japan). Serum leptin and adiponectin levels were evaluated with a commercially available ELISA Kit (R & D Systems, Minneapolis, MN, USA). Next, HbA1C was measured with a commercially available ELISA kit (MBS2024955, MyBioSource, San Diego, CA, USA).
Tissue collection, weight, and histological analysis
Mice were dissected to obtain abdominal subcutaneous fat (abdominal subWAT), eWAT, iWAT, and liver tissues. Immediately after dissection, collected tissues were weighed and processed for further analysis. For adipocyte and tissue morphology analysis, eWAT and liver samples were fixed in 10% formalin, embedded in paraffin, sectioned at 5 μm, and stained with H&E solution. In addition, ROS levels in tissue sections were assessed using DHE, MitoSOX, and DCF-DA staining to evaluate oxidative stress. These staining protocols were conducted on separate sections of eWAT tissues. Following staining, ROS fluorescence images were also captured with the same inverted confocal microscope under identical imaging settings to ensure comparability.
Adipocyte size measurement
The size of adipocytes was measured using the method described previously (Lee et al., 2023). Briefly, fixed adipose tissues were sectioned at 5 µm and stained with H&E. Microphotographs were acquired with BX51 microscope (Olympus, Tokyo, Japan) equipped with a DP70 camera. All the acquired images were processed with Image-Pro Plus (Bioimager, Maryland, USA). To determine the differences in adipocyte measurement, the area and diameter of 100 versus 50 adipocytes from identical samples were compared. This comparison yielded no significant difference. Hence, 50 adipocytes that did not overlap were used for measurements and calculations to maintain uniformity.
DEXA scan
DEXA scan was carried out as described earlier (Yeu et al., 2019). The fat and fat mass percentages were measured using a cone-beam flat panel detector DEXA (iNSiGHT VET DXA, Daegu, Korea) according to the manufacturer’s instructions. The fat mass was determined by subtracting lean body mass from the total body mass. The lean body mass was deduced from the total body weight to calculate fat mass. In color-composition images, fat tissues are indicated in red, whereas lean tissue is indicated in green. The whole-body scan was utilized to identify the ROI (region of interest) for assessing abdominal obesity. The proportion of abdominal fat was calculated using the formula abdominal fat (DEXA)/total fat (DEXA) ×100.
Statistical analyses
Statistical analyses were performed using GraphPad Prism 10 software (La Jolla, CA, USA). Data are presented as the mean ± standard deviation. For comparisons among more than two groups, one-way analysis of variance was conducted, followed by Tukey’s post hoc test. A p value of <0.05 was considered statistically significant. Although no statistical methods were used to predetermine sample size, the sample sizes employed were comparable to those used in previously published studies (Huang et al., 2022; Wang et al., 2021). An electronic laboratory notebook was not utilized in this study.
Authors’ Contributions
G.-H.L. and H.-Y.L. planned the experiments, conducted the studies, analyzed the data, and was primarily responsible for writing the article. Y.J.L., J.-h.K., and S.-Y.R. conducted the studies and analyzed the data. M.J.C., S.Y.P., S.S., H.L., and Y.S. reviewed and edited the article. J.K. and H.-J.C. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Author Disclosure Statement
The authors declare that this research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. All authors read and approved the final article.
Funding Information
This work was supported by Samyang Corp. This research was supported by the National Research Foundation of Korea (NRF), Republic of Korea (NRF-2021R1C1C2007371, RS-2024–00335573).
