Abstract
Objective:
To assess feasibility and safety of a decision support system (AI-DSS) that provides algorithm-generated insulin dosing recommendations directly to individuals with type 1 diabetes (T1D) managed with multiple daily injections (MDI).
Methods:
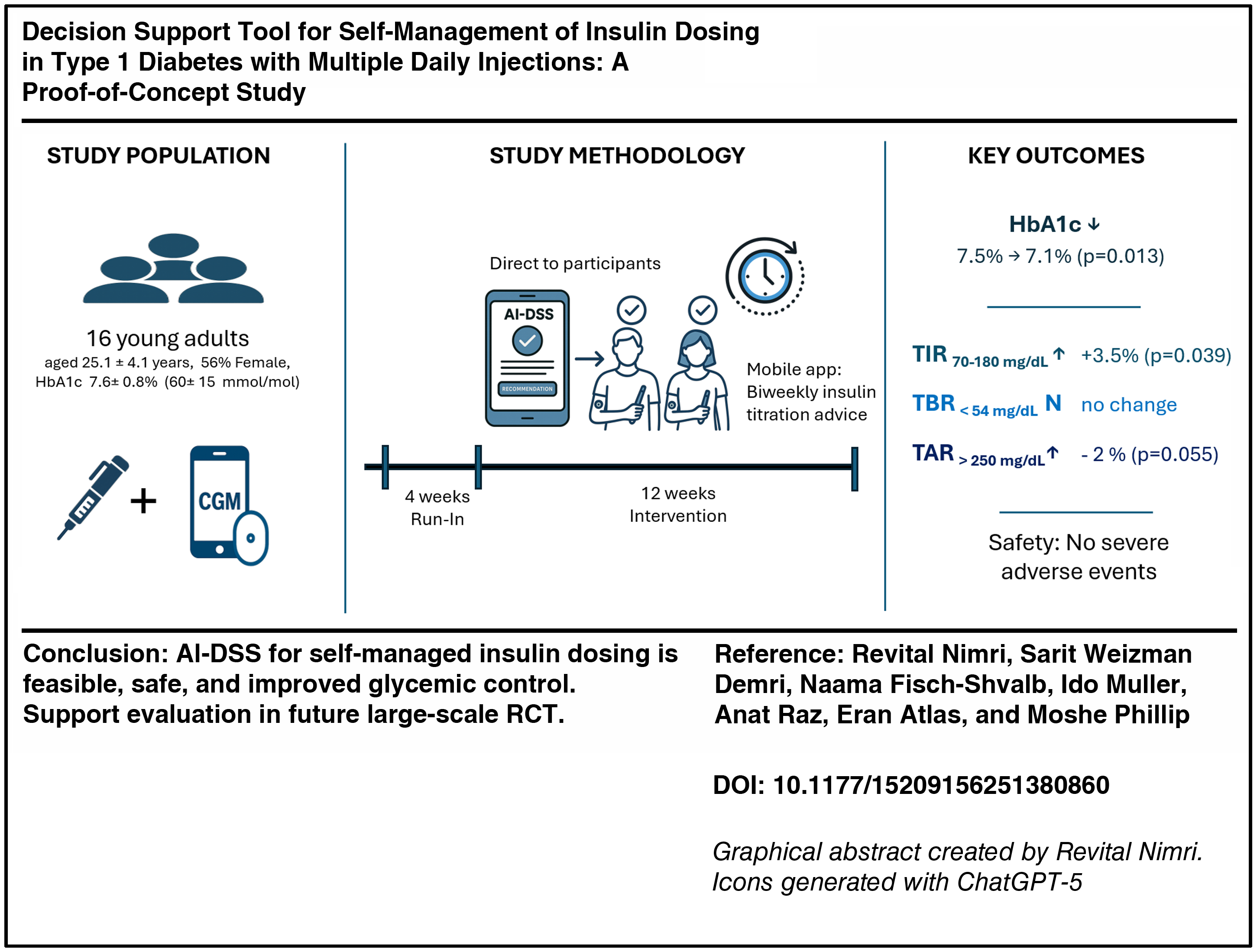
This single-arm, prospective proof-of-concept study included individuals with T1D managed with MDI and continuous glucose monitoring (CGM). Participants underwent a 4-week run-in period followed by a 12-week intervention phase, during which every two weeks algorithm-generated insulin titration recommendations were provided via a mobile application. CGM metrics were compared between the last 2 weeks of the run-in (baseline) and the last 2 weeks of the intervention periods. Primary safety outcomes included percent time <54 mg/dL and >250 mg/dL. Secondary outcomes included changes in HbA1c and time in range (TIR, 70–180 mg/dL).
Results:
The study cohort included 16 young adults (mean age 25.1 ± 4.1 years; 56% female, mean HbA1c 7.6% ± 0.8%) who completed the study. Median HbA1c significantly decreased from 7.5% (IQR: 7.1, 8) to 7.1% (IQR: 6.5, 7.3), from start to end of study (P = 0.013). TIR significantly improved by 3.5% ± 7.3% (P = 0.039). Time <54 mg/dL remained unchanged (0.9% ± 0.86% vs. 1.12% ± 1.11%; P = 0.191), with a trend toward reduced time >250 mg/dL (14.3% ± 10.71% vs. 12.32% ± 10.91%; P = 0.055). No severe adverse events were reported.
Conclusion:
Decision support tool for self-managed insulin dosing in individuals with T1D using MDI was feasible, safe, and improved glycemic control, supporting further evaluation in large-scale randomized trials.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
