Abstract

The pivotal studies in MiniMed 670G Hybrid Closed Loop (HCL) system1,2 included participants experienced with insulin pump therapy, assuming that the success of HCL systems depends on prior use of technology. However, there is no evidence for the superior glycemic control achieved by patients with prior experience with advanced technologies over those on multiple daily injections (MDI). For this end, the following standardized protocol to initiate HCL system in individuals on MDI was created: two days, HCL system assessment; five days, HCL system training (two-hour sessions in five consecutive days with groups of three to five participants); three days, Manual Mode use of HCL system, cumulating in ten days from MDI to Auto Mode activation.
The aim of the study was to evaluate this ten-day initiation protocol for MiniMed 670G HCL system in people with diabetes (PWD) type 1 on MDI in achieving desirable glycemic control.
An open-label single-arm, single-center study was performed in 30 children (age 10.24 ± 2.6 years; duration of diabetes 2.8 ± 1.7 years) on MDI following the structured protocol for ten days and followed for 84 days. The participants used the sensor for a median of 92% of the time and spent a median of 89% in Auto Mode. HbA1c decreased from 8.2% ± 1.4% (66 ± 15.3 mmol/mol) at baseline, to 6.7% ± 0.5% (50 ± 5.5 mmol/mol) at the end of the study (P = .017) and sensor glucose levels decreased from 193 ± 41 mg/dL to 142 ± 12 mg/dL (P = .001), accordingly.
Time in range (TIR; 70-180 mg/dL) continuously improved over time from 46.9% ± 18.5% at baseline, reaching a plateau after one month, to 75.6% ± 7.1% in the third month of Auto Mode (as shown in Figure 1). This was achieved without any severe hypoglycemia or diabetic ketoacidosis.
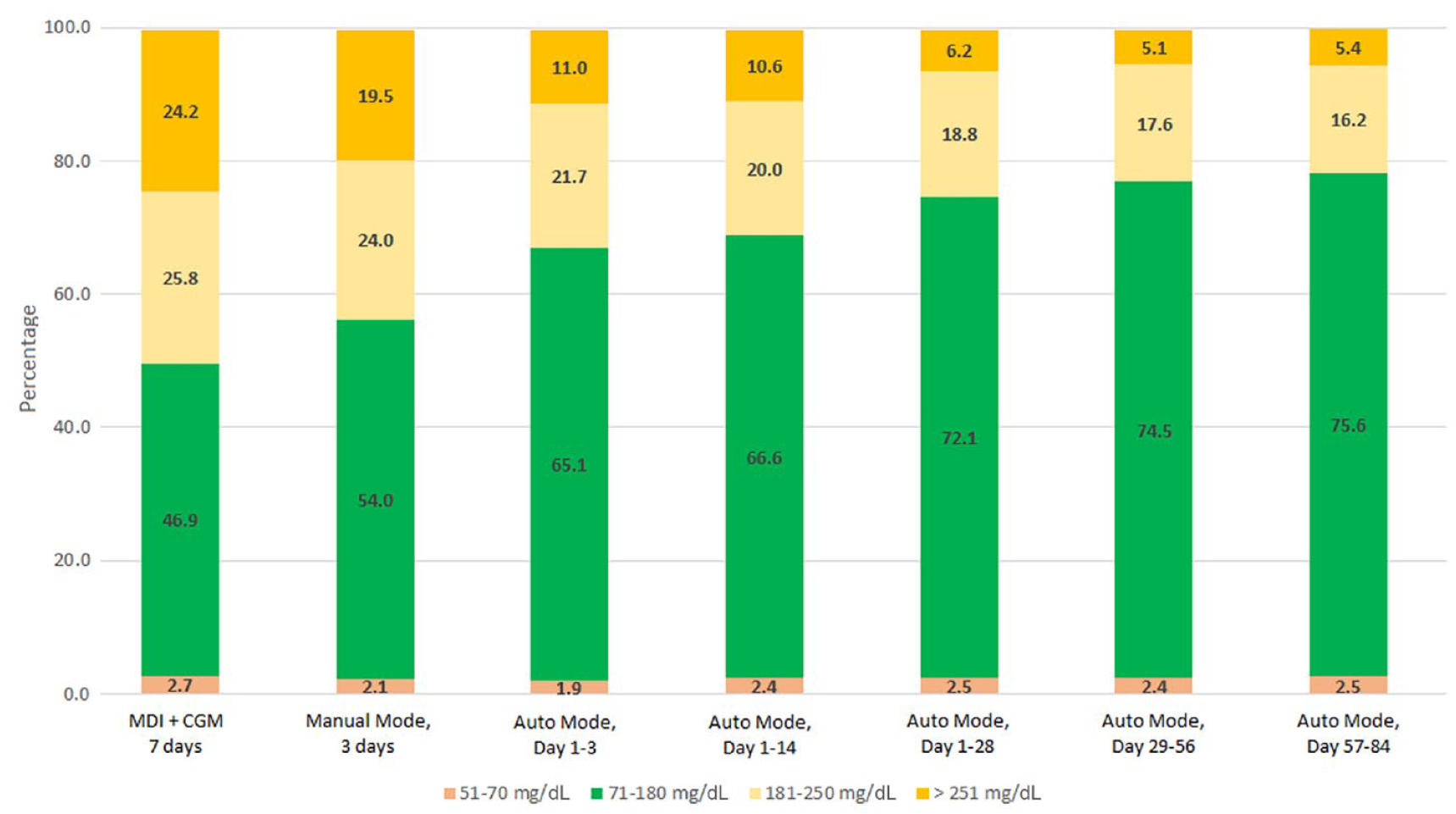
Time in ranges at baseline, during Manual Mode and Auto Mode periods.
Insulin-to-carbohydrate ratio (ICR) was initially made more aggressive regardless of the meal period from a median of 15 g/unit at the beginning to 12 g/unit at the end of the study (P = .002). Total daily insulin of 0.8 ± 0.3 U/(kg/d) at baseline increased to 0.9 ± 0.2 U/(kg/d) at the end of the study (P = .020).
The reductions in HbA1c and in the sensor glucose in our study are greater than those previously reported in children 1 and adolescents, and similar to adults. 2
The median TIR (70-180 mg/dL) achieved in Auto Mode was 74.8% ± 7.1%, which is similar to the previously reported medians of 73.5% 3 and 74.9% (2-19) in adults, but significantly higher than 68.8% in adolescents 2 and 64.6% in children. 1 The clinical outcomes were most probably driven by the high sensor and auto mode use and possible specific initiation protocol.
The total number of Auto Mode exits significantly decreased from 8.4 ± 1.8 in the first two weeks to 4.2 ± 0.9 events per week in the third month after enabling Auto Mode (P = .016).
Modifying ICR by increasing the meal bolus dose by almost 20% during the first month of Auto Mode use is similar to previously reported findings. 4
We conclude that PWD type 1 on MDI therapy can successfully initiate the HCL system, using a short structured ten-day protocol. Further investigation on more varied population and ages should be performed to confirm these findings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
