Abstract
In the pediatric population, insulin pump therapy, or CSII, is often considered the gold standard for intensive diabetes management. Insulin pump technology offers families and caregivers many beneficial features including a calculator for insulin dosing and the ability to review diabetes management data to provide data-driven diabetes management. However, for those who find CSII challenging or choose to use multiple daily injections (MDI) there is an option that offers similar features called the Smart Insulin Pen (SIP). Even though SIP technology provides a safe and data-driven diabetes self-management tool for the pediatric population using MDI, there is limited pediatric specific literature. This article will describe current options, data-driven diabetes management, benefits, challenges and clinical use of SIP technology in the pediatric population.
Background
Children make up over 200,000 of approximately 7 million Americans using insulin. The vast majority have type 1 diabetes (T1D) and need intensive insulin therapy and education of caregivers.1-3 The remaining are a subset of children with type 2 diabetes (T2D), many which require insulin.1,3
The Gold Standard: Insulin Pump Therapy
For many pediatric clinicians, CSII is the optimal insulin delivery choice.1,16,19 Insulin is infused through a catheter or needle inserted into subcutaneous tissue for up to 3 days.8,16,19 For some children (eg, toddlers, teenagers) eating occurs frequently and users can administer insulin as often as needed without additional injections. Recommendations for insulin doses, calculated by the pump, are based on
There are additional benefits, and challenges, to CSII. Benefits include: small dosing increments (0.025 units), advanced capabilities (temporary basals, extended boluses), and hybrid closed-loop options (pump with Continuous Glucose Monitoring (CGM) resulting in automatic insulin adjustments).1,24 Challenges include: accessibility, lack of long-acting insulin injection, time required for education, troubleshooting, technology reliance, and wearing a device.1,20
The Next Best Thing: Smart Insulin Pen
The ability for CSII to: 1. calculate
The InPen, launched in 2017 by
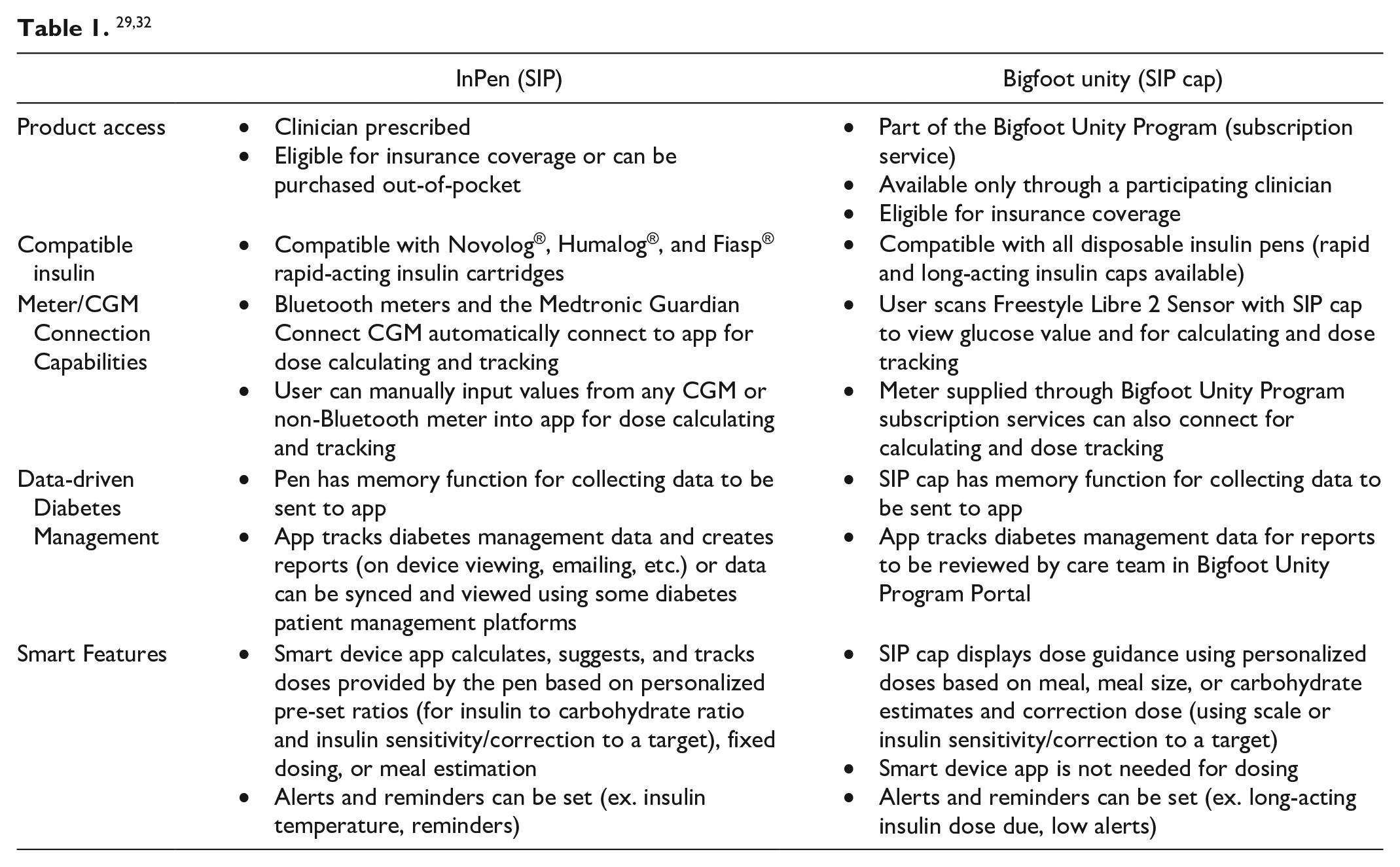
Literature on SIP use, even in adults is limited. This
SIP Data-Driven Diabetes Management
The importance of data-driven diabetes management has drastically increased due to telemedicine.2,42-47 The SIP can assist with effective remote review of data captured and potential for pattern management.2,25,27-29,31,33,38,41
Improved patient satisfaction, QoL, and time-in-range are all indicators of data-driven diabetes management.2,9,10,38,41,46,48 Literature on SIP technology demonstrates adults have less missed boluses,
34
positive user experiences
28
and improved clinical outcomes at lower cost
39
An SIP can also assist
Benefits of SIP in Pediatrics
When starting a new technology, the entire team must be willing, for it to be successful. Knowledge on the benefits of SIP technology for children will help encourage inclusion of SIPs in the options provided to families. 22
Parents and patients, especially at diagnosis, can be nervous and stressed.1,4,8,44 The SIP can help decrease these feelings, especially for those struggling with calculations, keeping logs, remembering doses, and understanding the concept of IOB. Although patients must ultimately still learn these important concepts, the SIP may help minimize mistakes and increase self-efficacy. For example, misunderstanding IOB can result in insulin stacking, which could lead to more frequent, and fear of, hypoglycemia. Parents of children using SIP, may also feel more comfortable when they are making dosing decisions without them, when with friends or at school.
As opposed to CSII, which involves hours of education, SIP education requires less time to introduce. Specifically, InPen, has proven to be
If a patient using CSII wants a “pump break” SIP technology may make this easier. This break may be for, a summer at the beach, for others, a social event. Having an SIP will provide these patients the bolus calculator, preprogrammed settings, and features they have become accustomed to using CSII.
Another benefit of InPen is, 2 pens can be connected to one app. This allows for one to be at home and another at school (or other location). There is also a memory function within the pen. If the pen is separated from the app, information is collected, and transferred to the app when reconnected. 33 This allows caregivers to get a more complete and accurate report of what happened while separated from the child.
Additional pediatric specific benefits exist. Given that children may not be as attentive to daily diabetes tasks, important alerts and alarms can be set. Insulin temperature tracking is particularly beneficial for children who may be less thoughtful about where they keep their pen. The colors,
Challenges in Pediatrics
Although benefits are clear, pediatric patients using SIP technology do not always use it as prescribed. Research must focus on challenges and ways to encourage long-term use.
Out-of-pocket costs due to variable coverage remains a barrier. While there may be options to buy, cost will be too high for some. 33 Another major barrier may be the lack of smart devices. Even if parents have a smartphone, children may not. At times the child will be without the parent and therefore without the app’s beneficial features. Options allowing users, who lack access to a smartphone and that decrease out-of-pocket costs, will help broaden use.
Another obstacle, as with any technology, is user dependence. If the device or app is lost or malfunctioning, the user will need to perform calculations manually. Frequent reminders and ongoing education about managing device failure is critical.
Parents are currently unable to remotely monitor their child’s SIP use. The app is available on one device per pen and information cannot be seen in real-time by a follower. InPen reports must be
Challenges of the InPen include: the need to
Clinical Use of SIP Technology in Pediatrics
The majority of pediatric patients under the authors’ care at an urban academic institution are considered for SIP prescription. At diagnosis, all patients and families must learn essential concepts and show proficiency in calculating and understanding insulin dosing, however once established, or unable to be, an SIP can be prescribed. This way, families are given a tool to assist in tracking and calculating doses for optimal diabetes management early in diagnosis. Clinicians are able to review data to complement handwritten logs or data not being documented. Although many newly diagnosed children are quickly transitioned to CSII, and SIP use is short lived, it may be an easier transition from one technology to another. In addition, the SIP and app will remain available to them if they experience a pump failure or want a “pump break.”
When a child is struggling with or refusing CSII, a compromise can be SIP technology, often satisfying caregivers and patients. Our experience however has been the majority of these patients are adolescents who may be experiencing diabetes burnout or who have not fully accepted their diagnosis. After prescribing an SIP for this cohort, we have found most do not choose to use the technology fully. Perhaps patients resistant to CSII are also resistant to other technology or devices. Patients report wanting a decrease in the burden of diabetes management: do less not more, and so SIP technology may not entirely satisfy this. 49
Patients who also may benefit from SIP technology are those who have demonstrated difficulty following protocols to recognize and address CSII site failure and have presented with recurrent DKA.
Conclusion
SIP technology provides users with diabetes technology features most needed for optimal diabetes management.
Footnotes
Abbreviations
(HbA1c) Hemoglobin A1c, (MDI) Multiple Daily Injections, (SIP) Smart Insulin Pen, (T1D) type 1 diabetes, (T2D) type 2 diabetes, (QoL) Quality of Life, (CSII) Continuous Subcutaneous Insulin Infusion, Diabetes Ketoacidosis (DKA)
Author(s) contributions
All authors accept responsibility for all aspects of the work.
Conception of the work: JI
Drafting and revising: JI, VW, MPG
Final approval of the version: JI, VW, MPG
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
