Abstract
This single-arm handling study assessed whether the reusable DuraTouch® pen injector is easy to use and easy to learn how to use in adults with type 1 diabetes (T1D) or type 2 diabetes (T2D) with or without prior pen injector experience. Following a 15-min training session, participants were instructed to independently administer a simulated DuraTouch injection. Of the 100 included participants (91% with T2D, 9% with T1D), 50% had prior pen injector experience. Pen injector-naive participants required more training time than pen injector-experienced participants (median time, 11 vs. 9 min, respectively). Overall, 85% of participants found DuraTouch very easy or extremely easy to use (using the Diabetes Pen Experience Measure), and 87% found it easy or very easy to learn how to use (using the Injection Device Experience and Acceptability questionnaire). DuraTouch may be a viable pen injector alternative for individuals with diabetes, irrespective of prior pen injector experience.
Introduction
Pen injectors were developed for individuals with diabetes to overcome the challenges of the vial and syringe.1–3 NovoPen®, the first pen injector, was introduced in 1985 for the administration of insulin. Compared with conventional vials and syringes, pen injectors are considered to be more accurate, simple to use, convenient, portable, efficient, and less painful; they have also been associated with reduced overall health care costs, driven predominantly by a reduction in hypoglycemic episodes. As rated by individuals with type 1 or type 2 diabetes (T1D or T2D) and health care professionals, the most valuable features of pen injectors are the ease of use; the delivery of fast, accurate, and painless injections; and the reliability of delivering the required dose in one injection.4–10
Pen injectors may be either single-use (disposable) or multi-use (reusable); disposable pen injectors are prefilled with the medication and discarded once depleted, whereas reusable pen injectors are designed for use with replaceable insulin cartridges.2,3 Recent data from England indicated that most injected insulin is prescribed using disposable pen injectors (72.5% of total insulin prescriptions between January 2021 and December 2023). 11 As the prevalence of pen injector use increases, the environmental costs of disposable pen injectors and their packaging (use of materials and waste) present a growing global challenge.12–14 Several ongoing initiatives aim to reduce the amount of diabetes technology waste, particularly that associated with pen injectors (e.g., Novo Nordisk’s PenCycle recycling program, The Diabetes Technology Society Green Diabetes Initiative, the Sustainability in Quality Improvement project, ReMed, and the SoloStar Reborn project).15–19
DuraTouch® (Novo Nordisk, Bagsværd, Denmark) is a multiple variable dose, needle-based pen injector designed for administration of insulin (Supplementary Fig. S1). It has no dose button extension owing to the mechanical spring-based injection system, which can be reused to deliver variable insulin doses (maximum dose 80 units of U100 insulins). The pen is compatible with replaceable Penfill® cartridges (Novo Nordisk; prefilled insulin cartridges for use in a reusable pen) and single-use pen needles. Except for the ability to replace cartridges, DuraTouch performance and handling characteristics are similar to those of FlexTouch®, a prefilled insulin pen injector (Novo Nordisk).
Education, training, and ongoing support are key for the optimal use of pen injectors. 20 This study assessed whether individuals new to DuraTouch found the DuraTouch pen injector easy to use and easy to learn how to use following training in a simulated-use environment.
Materials and Methods
This single-arm handling study was performed at a single site in Boston, Massachusetts, USA. The study design is illustrated in Supplementary Fig. S2. The study included adults (aged ≥18 years) with T1D or T2D with or without pen injector experience. Full inclusion and exclusion criteria are shown in Supplementary Table S1. “Pen injector naive” was defined as never having administered an injection (to self or others), not having administered more than one injection in the past 3–12 months, or not having administered an injection to self or others in the past 3 years. “Pen injector experienced” was defined as self-administration of an injection using a pen injector or vial/syringe daily for at least the past week, twice a week for at least the past 3 weeks, weekly for at least the past 1.5 months, once every other week for at least the past 3 months, or monthly for at least the past 6 months.
All participants provided written informed consent before study entry and were compensated according to standard practice. All participants attended a 60-min session designed to evaluate the ease of use of DuraTouch and the ease of learning how to use DuraTouch. Observers could view the sessions through a one-way mirror from an adjacent room or via a video link.
Sessions included up to 15 min for training and were recorded. Actual training time for each participant was logged. Training was conducted by a certified diabetes educator on a 1:1 basis, during which participants were provided with an overview of the pen injector, needle, and cartridge, a demonstration of a “complete” injection, and an opportunity to practice one or more simulated injections into an injection pad.
Per the test plan, the demonstration of a “complete” injection during training was required for progression to the simulated test injection. Eight handling steps contributed to a “complete” injection (Table 1). If any of the eight steps were not completed, the injection was considered to be “incomplete.” Owing to the simulated nature of this test, priming, defined as releasing a stream of medication before the injection, did not contribute to a complete injection.
Participant Performance Results
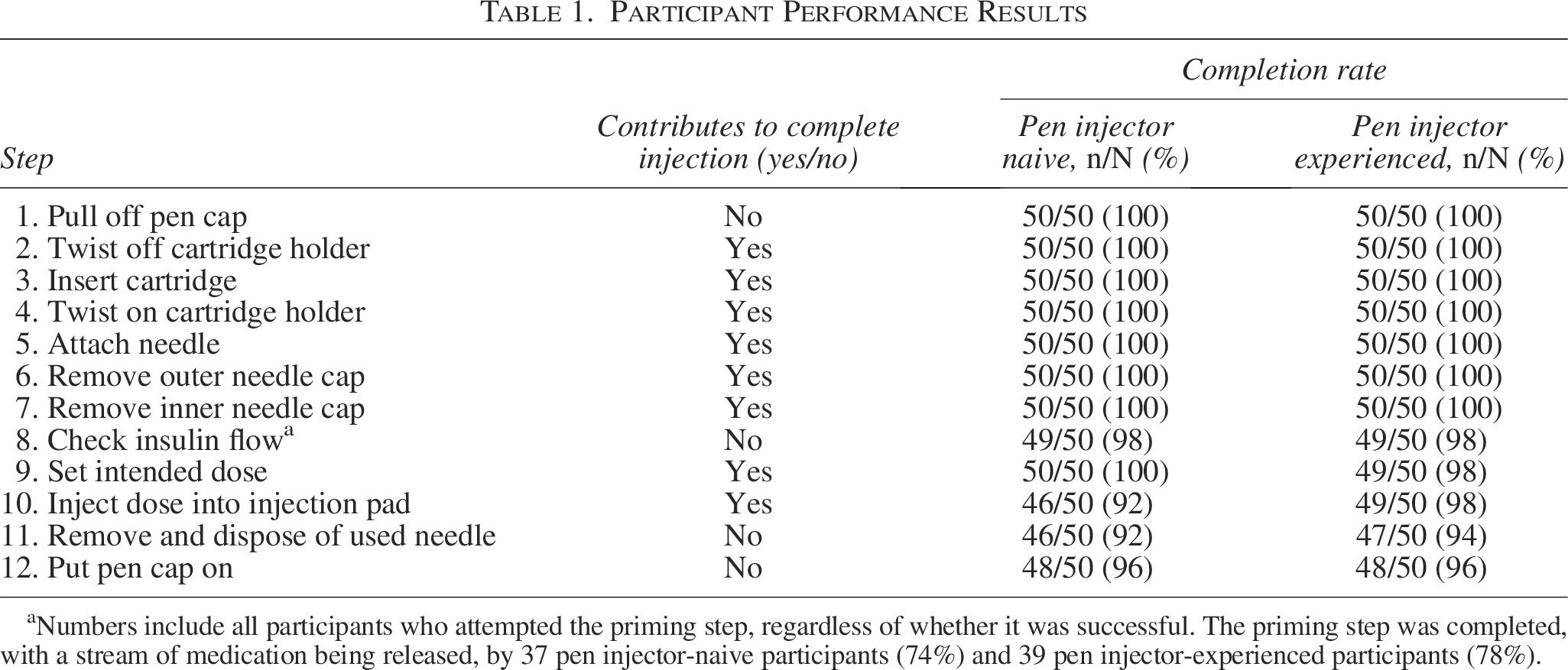
Numbers include all participants who attempted the priming step, regardless of whether it was successful. The priming step was completed, with a stream of medication being released, by 37 pen injector-naive participants (74%) and 39 pen injector-experienced participants (78%).
Following the training session, participants were directed to independently complete one simulated test injection into an injection pad mounted on their body in the location where they would administer the injection. Participants were then asked to complete the Diabetes Pen Experience Measure (DPEM) patient-reported outcome (PRO) measure 21 and one question from the Injection Device Experience and Acceptability (IDEA) 22 questionnaire about their experience with DuraTouch. The DPEM PRO measure, consisting of seven items, is designed to assess a participant’s experience with a pen injector, specifically in terms of ease of use, convenience, and satisfaction. 21 Scores were collected for each item individually, and a DPEM total score (calculated by summing the score for the seven items and transforming it onto a 100-point scale) was used to assess overall pen experience. Question 2 from the IDEA questionnaire assesses the ease of learning how to use a pen injector. The DPEM and IDEA PRO measures were provided on a five-point Likert scale. Both PRO measures were completed regardless of whether the simulated injection was evaluated as complete or incomplete.
The test plan, participant screeners, data processing, and consent forms were reviewed and approved by Allendale Institutional Review Board, Old Lyme, Connecticut, USA.
Study end points were described using descriptive statistics and included training time from start to end of training and the overall number of complete injections during testing.
Results
Participants
Although 109 individuals were initially recruited to the study, nine were disqualified, resulting in an analysis group of 100 participants for the simulated test. Participant flow and reasons for disqualification are presented in Supplementary Fig. S3. The 100 participants who proceeded to the simulated test had a mean (range) age of 62 (28–80) years, were equally split between male and female, and represented a broad range of education levels (Supplementary Table S2). Most participants (91%) had T2D, and 50% had previous experience with a pen injector (n = 41 with T2D, n = 9 with T1D). Participant demographics were broadly comparable between the analysis set and disqualified cohort, although eight of the nine participants who were disqualified had no previous pen injector experience. Test plan deviations were reported for three participants; all deviations were deemed not to have affected the quality of the data (Supplementary Table S3).
Test performance
On average, pen injector-naive participants required more training time than pen injector-experienced participants as follows: median (range) training time was 11 (4–20) min and 9 (5–15) min, respectively (Supplementary Fig. S4).
During the test session, 94 complete injections were recorded, with 46/50 pen injector-naive participants (92%) and 48/50 pen injector-experienced participants (96%) completing simulated test injections. The proportions of participants completing each of the injection steps are shown in Table 1. Six incomplete injections were recorded as follows: four by pen injector-naive participants and two by pen injector-experienced participants. One participant selected an incorrect dose amount. Two participants released the dose button before the full 20 units had been administered. Two participants pressed the needle into the hard “skin” of the injection pad with excessive force, and when the needle hit the hard plastic, the flow of medication was inhibited. One participant attempted to administer the injection with the pen cap still on the pen injector.
Priming attempts were completed by 37 pen injector-naive participants (74%) and 39 pen injector-experienced participants (78%), whereas failed or incomplete priming attempts were reported for 10 (20%) and 8 (16%) participants, respectively.
PRO measures
DPEM results showed that 85% of all participants found DuraTouch very easy or extremely easy to use, irrespective of previous experience (Fig. 1, Supplementary Table S4). Most participants were very confident or extremely confident that they were using DuraTouch correctly (81%) and that the device was delivering a correct full dose (85%). In general, compared with pen injector-experienced participants, pen injector-naive participants were less confident that they were using DuraTouch correctly and that the injection device was delivering a correct full dose (Supplementary Table S4). Overall, 83% of all participants found DuraTouch very or extremely convenient to use and 78% of participants were very or extremely satisfied with the device. Similar percentages for overall user convenience were observed between pen injector-naive and pen injector-experienced participants, whereas overall user satisfaction was slightly lower in pen injector-experienced participants (Fig. 1). Mean total DPEM scores were similar for pen injector-naive and pen injector-experienced participants (78.4 and 81.4, respectively).
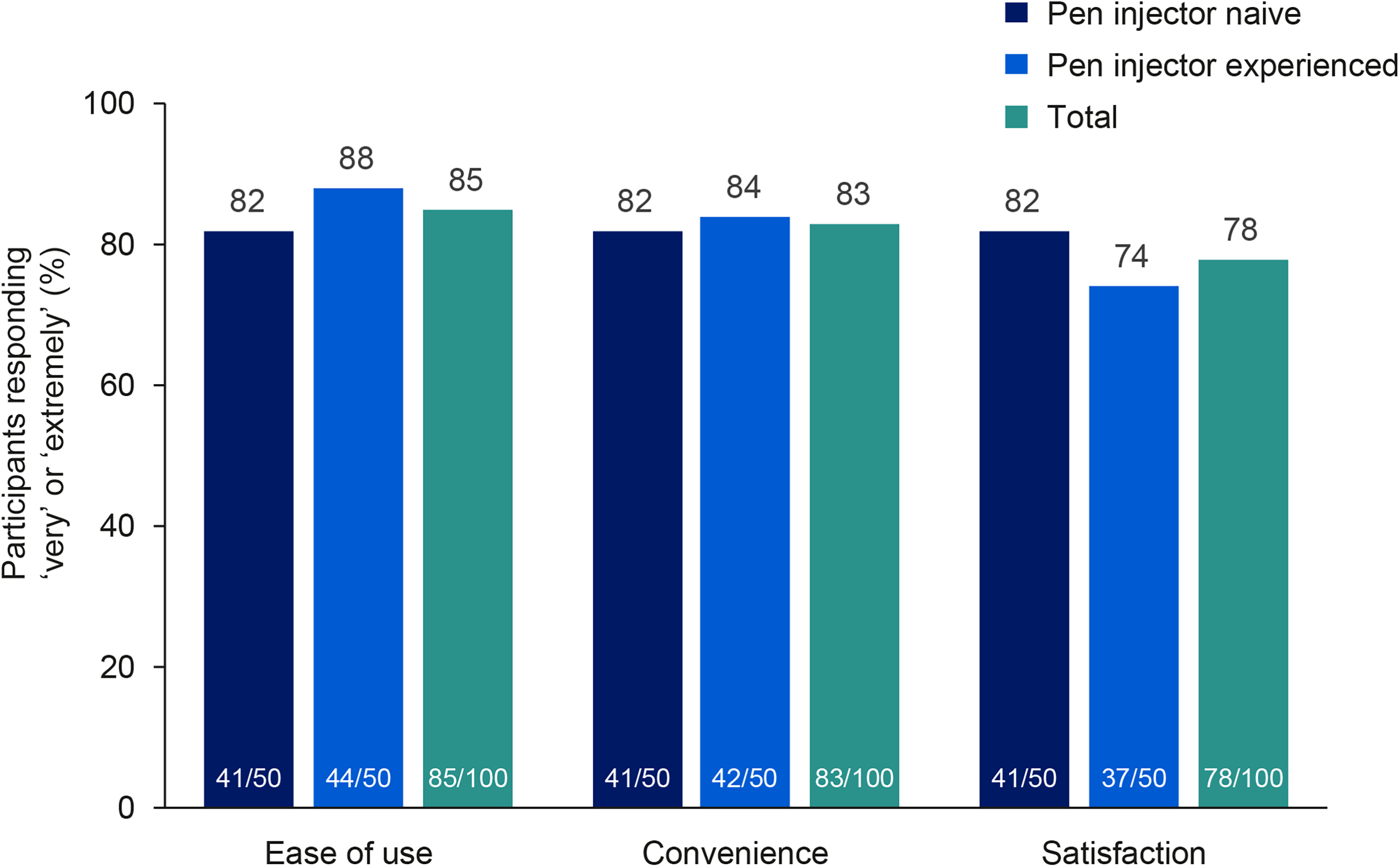
Ease of use of DuraTouch and overall convenience and satisfaction with DuraTouch in participants with diabetes with and without previous pen injector experience. All participants (n = 100) completed the DPEM PRO measure. Outcomes were reported on a five-point Likert scale ranging from “not at all easy/convenient/satisfied” to “extremely easy/convenient/satisfied.” Figure shows the proportion of participants choosing “very easy/convenient/satisfied” or “extremely easy/convenient/satisfied.” “Pen injector naive” was defined as never having administered an injection (to self or others), not having administered more than one injection in the past 3–12 months, or not having administered an injection to self or others in the past 3 years. “Pen injector experienced” was defined as self-administration of an injection using a pen injector or vial/syringe daily for at least the past week, twice a week for at least the past 3 weeks, weekly for at least the past 1.5 months, once every other week for at least the past 3 months, or monthly for at least the past 6 months. DPEM, Diabetes Pen Experience Measure; PRO, patient-reported outcome.
Data from question 2 from the IDEA questionnaire showed that 87% of all participants found it easy or very easy to learn how to use DuraTouch irrespective of previous experience as follows: 44/50 (88%) in the pen injector-naive group and 43/50 (86%) in the pen injector-experienced group.
Discussion
In this study, participants with diabetes received training by a certified diabetes educator to independently administer DuraTouch injections into an injection pad. Given the time pressures on clinicians and their teams, short training times are desirable. This study showed that a 15-min training session was sufficient for most participants to learn how to perform a DuraTouch injection successfully. The training time for DuraTouch was similar to that reported with NovoPen 3 and NovoPen 4 (both Novo Nordisk) in pen injector-naive participants (11.5 min and 9.9 min, respectively). 23
In the simulated test, 94 complete injections were recorded with 74% of pen injector-naive and 78% of pen injector-experienced participants completing the priming step. Most participants found DuraTouch very easy or extremely easy to use and to learn how to use, irrespective of previous pen injector experience. Most participants also found it very easy or extremely easy to prepare the pen injector and were very or extremely confident that the device had delivered the correct dose. Ease of use and confidence in delivering the correct dose are important factors in overall treatment satisfaction for people who require insulin treatment. 4
As recognized in clinical practice, the choice of pen injector needs to be tailored to each individual’s dexterity, possible cognitive or psychosocial impairment, or other physical limitations. 20 DuraTouch shares a similar spring-based injection mechanism and no dose button extension to that of FlexTouch, which has been shown to be easier to use and easier to learn how to use compared with other insulin pens (e.g., KwikPen®, SoloStar®).24,25 Reusable pens, such as NovoPen 4, have also showed high user satisfaction and confidence in dose delivery, in part, due to ergonomic and dose display improvements, making them easy to learn and use effectively.26,27 Our initial findings suggest that DuraTouch may be a viable alternative for individuals accustomed to using devices like FlexTouch or NovoPen 4, but further investigation is warranted. Moreover, DuraTouch may offer other benefits in terms of sustainability and accessibility owing to being reusable and not disposable, unlike FlexTouch.
An individual with diabetes using approximately 40 units of insulin/day may use about 50 prefilled plastic pens/year. 2 The environmental impact of switching from disposable to reusable pens should therefore not be underestimated. There is an urgent need to tackle the growing problem of medical device waste, specifically concerning prefilled insulin pens, and a gradual progression toward reusable pens, such as DuraTouch, may help to address this global need.12–14
Limitations of the study included the absence of a comparative control group, heterogeneity across participants in pen injector experience, and the use of a simulated setting (vs. real-use setting). Although the cohort distribution was heavily skewed toward T2D, the relative proportions of T1D and T2D were representative of real-world prevalence rates.28,29 Finally, training was limited to 15 min; however, longer times may have been allowed, at the trainer’s discretion.
In conclusion, after a short training session, most participants found the DuraTouch pen injector easy or extremely easy to use and to learn how to use, irrespective of previous pen injector experience.
Authors’ Contributions
All authors participated in developing the study concept and design and in collecting the data. All authors were involved in the analysis and interpretation of data, participated in preparing the article, and approved the final article for submission.
Footnotes
Author Disclosure Statement
S.A., A.S.W., B.B., J.C., A.-M.B.J., B.G.K., S.K., N.K.R., G.T.-B., and T.S. are employees of Novo Nordisk and hold shares. At the time this research was conducted, E.R. was an employee of PA Consulting.
Funding Information
This study was funded by Novo Nordisk and was performed by PA Consulting of Boston, Massachusetts, USA. Medical writing support was provided by Tove Anderson, PhD, of Oxford PharmaGenesis, Oxford, UK, funded by Novo Nordisk.
Data Sharing Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
