Abstract
Background and Aims:
The use of continuous glucose monitoring (CGM) has become standard practice in people with type 1 diabetes. The evaluation of user satisfaction is crucial. The Glucose Monitoring Experiences questionnaire (GME-Q) includes 23 items with a 5-point Likert scale to produce a total satisfaction score and three subscale scores. The study aimed to translate the GME-Q from English into Spanish and to validate its use in Spanish-speaking CGM users with type 1 diabetes.
Methods:
The linguistic translation and validation process of the GME-Q was established. T1D CGM users were asked to complete the produced Spanish version of the GME-Q and interviewed about difficulties or misunderstandings. Total satisfaction, effectiveness, convenience and intrusiveness subscales and internal consistency reliability were computed.
Results:
Forward and backward translations and cognitive debriefing produced a final version of the GME-Q in Spanish. Ninety-eight subjects with type 1 diabetes were selected (age: 40 ± 12 years, 63% females, Hb1c: 7.2 ± 0.9% (55 ± 10 mmol/l), pump users: 78%, CGM use: 3.7 ± 2.6 years). The completion rate was 99% and the Cronbach’s alpha coefficient was 0.8. The total satisfaction score was 3.9 ± 0.4 (effectiveness: 4.1 ± 0.6, convenience: 3.8 ± 0.6, intrusiveness: 2.2 ± 0.7).
Conclusion:
The GME-Q was translated into Spanish and validated for Spanish-speaking CGM users with type 1 diabetes.
Keywords
Introduction
The use of continuous glucose monitoring (CGM), both real-time and intermittently scanned CGM, has widely spread in people with type 1 diabetes (T1D) in the past decade, either as stand-alone systems or as a component of sensor-augmented pumps (SAP) and closed-loop (CL) systems.1–7
With the increasing utilization of technology, it has become clear the importance of taking into account the user satisfaction as a key factor for long-term use and optimized outcomes. Patient-reported outcomes instruments are being increasingly used in trials and clinical settings.8–12
Regarding CGM, an objective analysis of user experiences and satisfaction with CGM is desirable, for research purposes and also for clinical practice evaluation. Speight et al13,14 developed a novel measure, the Glucose Monitoring Experiences Questionnaire (GME-Q). The GME-Q includes 23 items with a 5-point Likert scale, from “strongly disagree” to “strongly agree,” with statements about their current monitoring device. These items relate to three domains, convenience, intrusiveness, and effectiveness, with a final question referring to overall satisfaction. The negatively formulated questions have to be reversed for computation. The total satisfaction score and each subscale score ranges from 1 to 5, with higher scores meaning a higher satisfaction level, except for the intrusiveness domain, where higher scores reflect a lower satisfaction level. Additionally, three open-ended questions ask the subjects to highlight the three best and worst aspects of their glucose monitoring devices, as well as the improvements or changes they would like to be implemented.
The GME-Q is intended to be used in SMBG (self-monitoring of blood glucose) and CGM users, and it has been tested in SMBG users,13,14 but it has not previously been validated in CGM users. Several CGM devices are available, and more will be available in the near future. These devices have different characteristics, such as calibration, interface, sensor life, size, options for body placement, visibility, and ease of use. It would be useful to be able to use this tool to compare satisfaction in users of different CGM devices.
The GME-Q is only available in English. It is important to be able to use this tool in different populations, so translated and culturally validated versions are desirable. A protocol for translation and validation is available, 15 including a forward translation, a backward translation, and cognitive interviews with questionnaire users.
The study aimed to translate the GME-Q into Spanish and to validate its use for native Spanish-speaking users of different CGM devices.
Material and Methods
The author Speight et al. 13 of the GME-Q was contacted and the process of translation and linguistic validation was transferred to the distributor of the questionnaire, the Mapi Research Trust organization. 16 License to translate and validate the GME-Q was obtained and a translation agreement was signed. The established methodology for translation and linguistic validation was agreed on.
People with T1D, who had been using different CGM devices for at least two months, were asked to complete the Spanish version of the GME-Q and to highlight any possible misunderstandings. The most recent 14 days of their CGM systems were downloaded and the following variables were recorded: glucose management indicator, time in range 70–180 mg/dl (%), time < 70 mg/dl (%), time > 180 mg/dl (%), sensor use (%), average sensor glucose (mg/dl), standard deviation of sensor glucose (mg/dl) and coefficient of variation of sensor glucose (%).
Total satisfaction and the three subscales were computed. For this calculation, the sum of scores divided by the number of items completed was used. The effectiveness subscale comprised nine items (items 7, 13, 14, 16, and 18 to 22), the convenience subscale seven items (items 1 to 6 and 17) and the intrusiveness subscale comprised six items (items 8 to 12 and 15). All the items scored from 1 to 5. Higher scores related to a more positive glucose monitoring experience, except for intrusiveness items, where higher scores relate to a less positive experience. Items 2, 5, and 17 (part of the convenience domain) and all the intrusiveness domain items were negatively worded. These negatively worded items were reversed before the calculation of the convenience score and the total satisfaction score, except for the intrusiveness items, which were reversed only for the calculation of the total satisfaction score.
The protocol was approved by the local Clinical Research Ethics Committee and followed the Declaration of Helsinki principles. All the subjects signed written informed consent forms to participate in the study.
Data analysis was conducted using SPSS statistics software v22. Results are presented as mean ± standard deviation values or percentages. The Pearson test was used for the analysis of correlation. Cronbach’s alpha coefficient was used to assess internal consistency reliability.
Results
The standard recognized methodology of translation and linguistic validation was followed. The established protocol includes four steps: a forward translation (step 1), a backward translation (step 2), cognitive debriefing (step 3), and proofreading (step 4). 15 The translation and validation process included the title, introduction to the questionnaire, identification of the CGM device, response choices, the 23 items with multiple-choice responses, the three open-ended questions and the final remarks of the questionnaire.
The forward translation (step 1) from the source language (English) to the target language (Spanish) was performed by two independent translators, native Spanish speakers and bilingual in English, with no medical background. A local coordinator compared both translations. Both translations were identical or almost identical in 70% of the content. Only minor issues were found when comparing both translations, in particular different wording but with similar meaning in 8 items. A significant difference in meaning was found in the translation of item 1, and a related infinitive form of the verb was used. The author of the original GME-Q did not need to be contacted. Reconciliation between both translations was easily achieved and the two translations were merged into a single one.
Afterwards, a backward translation (step 2) of the translation produced in step 1 was performed, from Spanish into English. This translation was conducted by a native English translator, bilingual in Spanish, with no contact with the original GME-Q. The local coordinator compared the translation produced in this step to the original GME-Q. Identical or almost identical translations comprised 60% of the content. Different wording but with similar meaning was found in 12 items. Again, a significant difference was found in the translation of item 1 and an agreement was reached. After this process, a second version of the Spanish GME-Q was created.
For cognitive debriefing (step 3), 98 subjects with T1D, consecutively attending their diabetes clinic appointments, were selected. They were asked to complete the Spanish GME-Q produced in step 2 and they were interviewed and asked about any difficulties or misunderstandings. These interviews were performed in person or by telephone when the appointments were part of the virtual clinic and were conducted by a diabetes physician or nurse. None of the patients who were asked to participate refused to complete the questionnaire. Patient characteristics are shown in Table 1. The CGM devices used by these patients were Guardian®: 69% (n = 68), Dexcom G6®: 19% (n = 19), FreeStyle Libre®: 11% (n = 11). These devices were used as stand-alone CGM or as part of a SAP or a hybrid CL system.
Patient Characteristics.
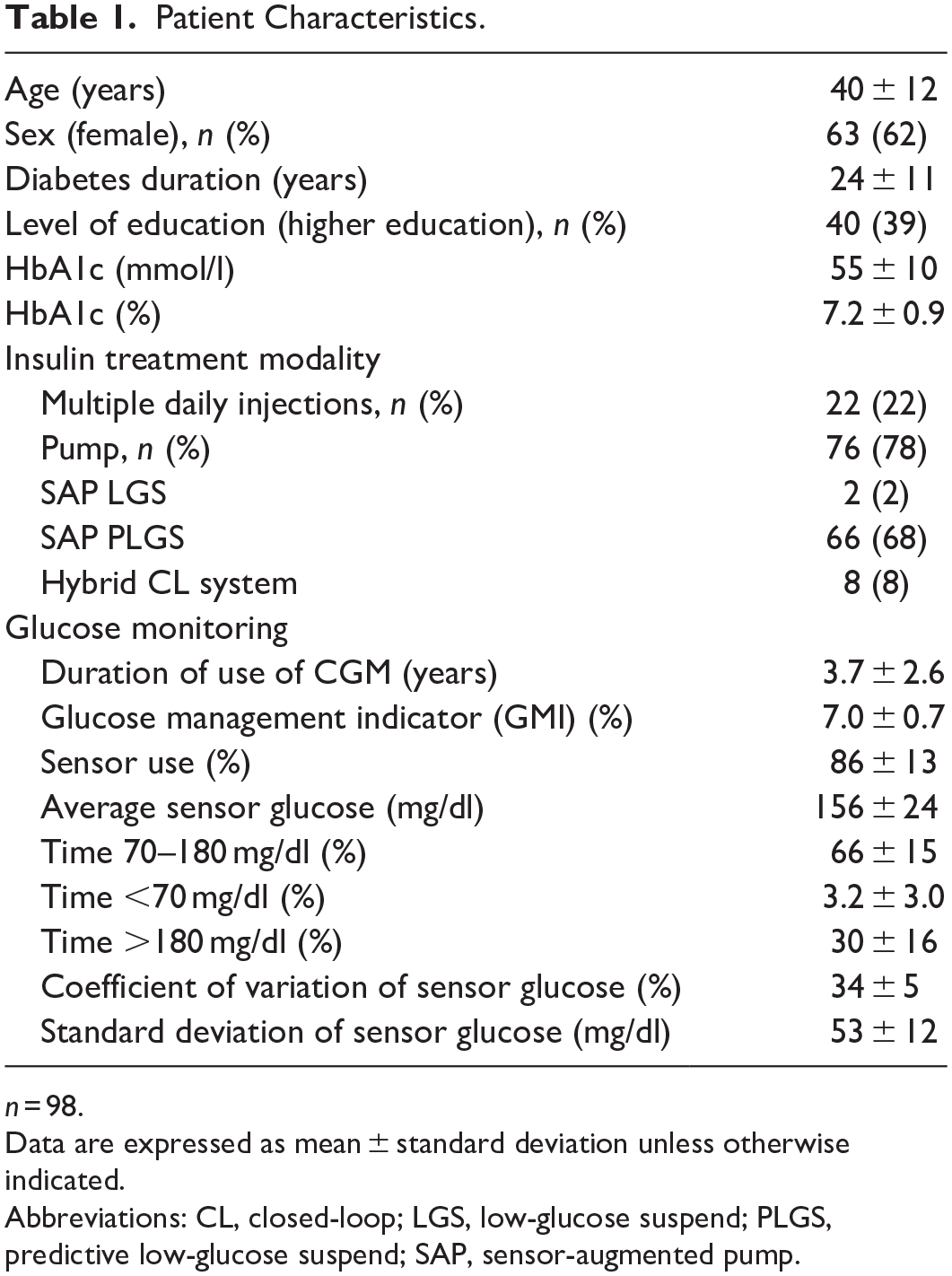
n = 98.
Data are expressed as mean ± standard deviation unless otherwise indicated.
Abbreviations: CL, closed-loop; LGS, low-glucose suspend; PLGS, predictive low-glucose suspend; SAP, sensor-augmented pump.
The general satisfaction GME-Q scores and the calculated subscales scores are summarized in Table 2. There was a significant correlation between the total satisfaction score and each subscale score (effectiveness: r = 0.790, convenience: r = 0.727, intrusiveness: r = −0.609; all p < 0.001). The Cronbach’s alpha coefficient was 0.8, which reflects a satisfactory internal consistency reliability. 17 When analyzed independently for each subscale, the Cronbach’s alpha coefficient was 0.7 for convenience, 0.9 for effectiveness, and 0.7 for intrusiveness. The Cronbach’s alpha coefficient was 0.8 for pump users and for patients on multiple daily injections. When analyzing separately the group of subjects using different CGM devices, the internal consistency reliability was 0.9 for Dexcom® users, 0.8 for Guardian® users and 0.7 for FreeStyle Libre® users.
Glucose Monitoring Experiences Questionnaire (GME-Q) Scores.
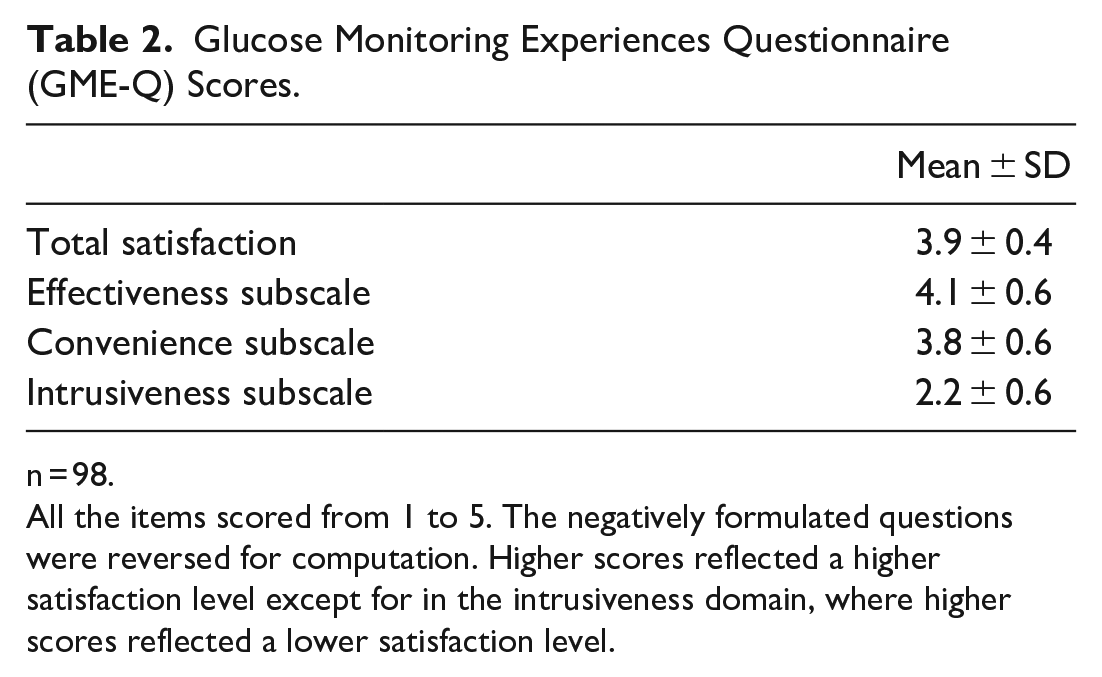
n = 98.
All the items scored from 1 to 5. The negatively formulated questions were reversed for computation. Higher scores reflected a higher satisfaction level except for in the intrusiveness domain, where higher scores reflected a lower satisfaction level.
Five additional T1D subjects, age: 37 ± 6 years, users of the implantable sensor Eversense® for 1.6 ± 0.3 years, had also been interviewed. The reliability of the questionnaire was extremely low in this group of subjects, Cronbach’s alpha coefficient < 0.4, for total satisfaction and the three subscales. These five subjects were excluded from the analysis.
Ninety percent of the subjects answered all the questions and the completion rate was 99%. Only 20 out of 2254 answers were missing; seven people missed one or two questions and one person skipped nine questions. Three people reported some difficulties understanding the questionnaire; specifically, they found the multiple-choice responses confusing. Additionally, one person suggested an additional point, related to his satisfaction with his CGM device, that was not addressed in the questionnaire.
When analyzing the answers to the open-ended questions, in which the subjects were asked about the advantages and disadvantages of their CGM devices, 88% of the subjects named at least one advantage, the most common advantages being ones related to the feeling of safety and to a better glycemic control. Regarding the disadvantages of their CGM devices, 66% of the subjects named at least one disadvantage, the most common being ones related to the inaccuracy, loss of connectivity and loosening of the tapes. Ten pump users included in their answers some disadvantages related to their insulin infusion systems and closed-loop algorithms and not exclusively related to their CGM devices, i.e., “the pump is too big,” “the insulin delivered by the system is not enough to correct my high blood sugars,” “it should infuse glucagon.” The last open-ended question, asking about possible improvements of the glucose monitoring devices, was answered by 55% of the subjects, and the most common suggestions referred to increasing the sensor life, improving the adhesion to the skin and reducing the visibility of the devices.
Finally, proofreading (step 4) was performed and duplication in one word was detected and deleted. This final editing of the questionnaire produced the validated final version of the GME-Q.
The validated final version of the GME-Q was transferred to Mapi Research Trust organization, along with a written report of each step of the process, outlining the translation issues discussed item by item and how the final decisions were made. The process of translation was completed and approved. The Spanish version of the GME-Q is available by request to the eProvide website. 16
Discussion
In our study, we present the Spanish translation and linguistic validation of the GME-Q, which is the first and only validated, as reported by the only distributor of the questionnaire. 16
The internal consistency reliability was satisfactory, 17 with a Cronbach’s alpha coefficient of 0.8, close to the Cronbach’s alpha coefficient of 0.9 in the original GME-Q. Also, the questionnaire was easily understood by Spanish speakers and the high completion rate, 99%, was considered proof of its acceptability.
The original GME-Q had been validated in SMBG users; however, its use has been applied to CGM users. 14 In our study, the validation process in T1D individuals using CGM allows extending the use of the questionnaire to this population.
Other measures have been developed in the recent years to evaluate satisfaction in CGM users. The CGM Satisfaction scale (CGM-SAT) and the Glucose Monitoring Survey (GMS) are reliable measures to evaluate patient-reported CGM outcomes, 18 but they have not been validated in Spanish. The System Usability Scale (SUS) 19 has been used in CL studies to evaluate satisfaction with the system, but it does not address specific aspects related to CGM and, as far as we are aware, no validated Spanish translation is available. The Technology Acceptance Scale (TAS) is a Control-IQ technology-specific questionnaire to assess experiences with this technology.20,21 The Diabetes Impact and Devices Satisfaction (DIDS) scale evaluate user experience concerning their insulin delivery device. 22 In sum, to the best of our knowledge, this Spanish version of the GME-Q is the only validated questionnaire available to Spanish-speaking users to specifically evaluate satisfaction with CGM devices.
Our study has some limitations. The group of subjects selected to complete the questionnaire had an acceptable glycemic control and good adherence to sensor use, and the results could have been different in a different population. Similarly, a high percentage of our study subjects had a higher level of education, therefore it is not possible to extend the results to a more heterogeneously educated population. Also, the small number of subjects included using each commercial CGM device was low, so it was not possible to make specific conclusions regarding each device. Further research is needed regarding satisfaction with CGM devices in subjects using implantable sensors instead of transcutaneous sensors and separately in individuals on multiple daily insulin injections and pump users, with and without automation of the insulin infusion.
In summary, a Spanish version of the Glucose Monitoring Experiences Questionnaire (GME-Q) was produced and it was shown to be culturally relevant, conceptually equivalent to the original questionnaire and easily understood by native Spanish- speaking CGM users. This is the first validated tool, available to native Spanish speakers, to specifically assess satisfaction with a glucose monitoring system in people with diabetes.
A preliminary analysis of these data has been submitted for presentation in the Advanced Technologies & Treatments for Diabetes conference (ATTD 2021).
Footnotes
Abbreviations
CGM, Continuous Glucose Monitoring; CL, Closed-Loop; GME-Q, Glucose Monitoring Experiences Questionnaire; SAP, Sensor-Augmented Pumps; T1D, Type 1 Diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
