Abstract
Objective
This retrospective study in individuals with chronic respiratory diseases (CRDs) compared changes in quality of life, anxiety and depressive symptoms, and exercise capacity after a home pulmonary rehabilitation (PR) programme, based on the care manager’s professional background.
Methods
PR team was composed of nurses (Gr1, n = 989 patients), physiotherapists (Gr2, n = 466), one dietician and one sociomedical beautician (Gr3, n = 794) and one respiratory physician (Gr4, n = 201) who supervised one home session per week during 8 weeks. Quality of life, anxiety and depressive symptoms, dyspnea and exercise capacity were assessed at the beginning and end of PR, and at 12 months after the end of PR.
Results
2450 participants were included (females: 41.6%, age: 64.1 ± 13.0 years, FEV1: 50.7 ± 25.2% of predicted, people with COPD: 51%, with ILD: 23%, with asthma: 10%). Gr 1, Gr 3 and Gr 4 improved all the assessments at short- and long-term (p < 0.01). Improvement in the Visual Simplified Respiratory Questionnaire in Gr2 was not maintained at long-term. According to the linear mixed models, changes over time were not statistically different between groups.
Conclusion
A personalised PR programme delivered by one care manager, led to similar short-term benefits regardless of the care manager’s initial professional background.
Keywords
Introduction
The benefits of pulmonary rehabilitation (PR) in improving health-related quality of life, exercise capacity and functional capacity and reducing dyspnea are well established in individuals with chronic obstructive pulmonary disease (COPD). 1 New evidence supports extending PR to individuals with interstitial lung disease (ILD), asthma, pulmonary hypertension or lung cancer. 2 PR is defined as an integrated, person-centred intervention comprising three core elements: education, self-management strategies and exercise training —aimed at promoting long-term adherence to health-enhancing behaviours. 3 PR teams typically involve various health professionals, including physiotherapists, nurses, dieticians and respiratory physicians. 4 Most adopt a multidisciplinary model (where each discipline focuses solely on its specific role) or, at best, an interdisciplinary model (where team members share information about their work with the patient). 5 However, in other fields such as pain management, 6 this siloed approach has been shown to be less effective than transdisciplinary models, in which all team members share a comprehensive understanding of each discipline’s contribution to patient care. 7 Similarly, a pediatric rehabilitation study 8 in children with developmental disabilities reported greater participation in team meeting, more holistic goals, and fewer miscommunications under a transdisciplinary model compared with a multidisciplinary one. Whether these benefits translate to PR remains to be confirmed in randomised controlled trials.
Home-based PR programmes are feasible and achieve short-term outcomes comparable to those of traditional centre-based programmes. 9 Given the well documented barriers such as travel, limited access, and work-related constraints, home-based interventions appear more convenient and offer greater flexibility, allowing better integration into participants’ daily routines. 10 In this context, over the past decade we developed an 8-weeks PR programme in which a single healthcare professional— the care manager—delivers all PR components face-to-face at the patient’s home. 11 In this transdisciplinary model, patient allocation was not random or based on the care manager’s professional background (e.g., nurse, physiotherapist, dietitian), but on a defined geographical perimeter around the care manager’s residence. This system covers a wide area while minimising travel distances for the mobile team. Overall, this face-to-face home-based PR model effectively improved short- and long-term outcomes (up to 12 months) in patients with COPD, ILD or asthma.11,12 However, it remains unclear whether these outcomes depend on the care manager’s professional background. In the context of strained healthcare systems, enabling healthcare professionals to deliver a broader range of care may improve efficiency and accessibility, and recent French legislation recommends assigning each patient a designated healthcare referent.
This real-life retrospective study conducted in individuals with CRDs aimed to compare short- (end of PR) and long-term (12 months post-PR) changes in health-related quality of life, dyspnea, anxiety and depressive symptoms, and exercise capacity, based on the initial professional background of the care manager. As all care managers developed transdisciplinary knowledge and skills through a standardised training programme, our primary hypothesis was that effectiveness would not differ across care manager groups.
Methods
Study design and participants
This retrospective pre-post study was conducted using prospectively collected data from January 2010 to December 2021. Data were entered and stored in a secure electronic medical record system (Care Itou), specifically developed for the home-based care facility. Individuals were referred to the home-based programme (FormAction Santé, Pérenchies, France) by both hospital-based and private practice respiratory physicians, who provided a prescription along with relevant medical information, and were responsible for documented the diagnosis of a CRD. Whenever feasible, physicians offered patients the option of a centre-based or home-based programme, and the final choice was made according to programme availability, geographical accessibility, and patient preference. Following receipt of the referral, the PR mobile team contacted each patient to schedule the initial home visit and initiate the programme. However, given that 299 physicians have referred patients since 2010, we cannot guarantee that the option of programme modality was consistently offered to all patients. Prescribers confirmed the absence of contraindications to exercise, with cardiopulmonary exercise testing performed at their discretion, as it is not required for PR enrolment in France. Exclusion criteria included unstable cardiovascular disease, dementia or poorly controlled psychiatric illness, neurological sequelae or bone and joint diseases preventing training. Participants lacking a 12-months follow-up in their protocol—such as those with lung cancer or those enrolled in a second PR programme within the same year—were excluded retrospectively. Approval for the observational research protocol was obtained from the CEPRO (French Language Society of Pulmonology) Research Ethics Committee under successive authorisations since 2010, the most recent renewal being CEPRO 2021-054. These approvals permitted the retrospective analysis of prospectively collected routine clinical data. All participants provided written informed consent prior to initiating PR.
Home-based PR programme
Participants enrolled in a supervised, home-based programme consisting of a 90-minute face-to-face supervised session once per week for 8 weeks, including individually prescribed self-management education and exercise training. They were also instructed to engage in personalised physical activity and adhere to a self-management plan for the remainder of the week and throughout the 1-year follow-up, during which no visit or supervised maintenance was provided by the PR team.
The home-based PR programme is detailed in the Supplemental Materials. Briefly, education and self-management interventions were adapted to respond to the individual’s goals. The core educational topics included pathophysiology of lung disease and comorbidities, medication and its use, prevention and recognition of exacerbations and allergic factors, indoor air pollution, physical activity, breathing techniques and stress management. The training programme followed the guidelines for exercise prescription in CRDs. 13 Each participant received home endurance training equipment (bicycle, stepper or mini bike) during the 8-weeks programme, tailored to their needs, preferences, and home environment constraints. They were encouraged to exercise for 30–45 minutes, (performed in 10-min intervals or shorter, depending on individual capacity), 5 times per week. Endurance training was completed with upper and lower limb muscle strengthening exercises. For participants with severe deconditioning, the programme began with daily sessions of self-administered quadriceps electrostimulation, with endurance training introduced as soon as feasible.
Care manager training and role
PR team consisted of a respiratory physician, a physiotherapist, six nurses, two kinesiologists, a dietician and a sociomedical beautician. All team members completed the same 40-h standardised therapeutic education training programme, delivered by a licensed instructor. This training, including theoretical and practical components such as case analysis, is required for transdisciplinary PR models and promotes a holistic approach, covering main educational topics and teaching motivational communication, assessment of disease acceptance, and consideration of patients’ personality traits to enhance the therapeutic alliance.
New care managers worked alongside experienced colleagues during a 2-month mentorship period. This shadowing allowed them to observe the delivery of PR, without independently providing care. To support transdisciplinary practice, weekly four-hour team meetings were held to discuss care plans and provide ongoing training. As a result, each care manager was qualified to deliver all components of the PR programme in the patient’s home. Care managers communicated with families and respiratory physicians by providing reports at the start and end of PR, and again 12 months post PR. Care manager training and role are detailed in the Supplemental Materials.
Data collection
Lung function, medication and comorbidity data were obtained from medical records provided by the respiratory physician. All assessments were conducted at the participant’s home, at baseline (M0), post PR (M2), and 14 months after baseline (M14), completing 1 year of follow-up. Participants completing both the 2-month PR programme and the M14 assessment were defined as PR completers. Prospectively collected data were stored in a secure electronic medical record system (Care Itou), developed for the home-based care facility. For retrospective analysis, data were extracted into a spreadsheet via secure, password-protected access by an authorised user. As a retrospective study conducted over 11 years of real-life practice, no sample size was calculated, and no single assessment was designated as a primary outcome. Health related quality of life was assessed using the Visual Simplified Respiratory Questionnaire (VSRQ) from 2010 to 2016 (8 items rated 0-10, total score 0–80; higher is better) 14 and then the COPD Assessment Test (CAT) from 2017 to 2021 (8 items, total score 0-40, lower is better). The change in assessment tool was made based on a clinical decision. Anxiety and depressive symptoms were assessed with the Hospital Anxiety and Depression (HAD) scale (14 items, each subscore 0-21; lower is better) 15 ; and dyspnea by the mMRC breathlessness. Exercise capacity was evaluated with the 6-minute stepper test (6MST) as previously described. 11 In individuals with COPD, the minimal clinically important difference (MCID) of the VSRQ, CAT, HAD and 6MST is considered to be a change of 3.4, 2 and 1.5 points, and 40 strokes, respectively.14,16,17 The proportion of participants reaching the MCID for each outcome was calculated for each care manager group. These thresholds were applied to all participants, as they have been primarily established in COPD populations.
Statistical analyses
Data were analysed using the SAS software version 9.4 (SAS Institute, Cary, NC) and significance threshold was considered at 0.05. Quantitative variables are expressed as means (standard deviation, SD) in the case of normal distribution or medians (interquartile range, IQR) otherwise. Normality of distributions was assessed using histograms and Kolmogorov Smirnov test. Between groups baseline characteristics were compared using Chi-square test for categorical variables and using analysis of variance (ANOVA) or Kruskal-Wallis test for quantitative variables.
Changes in the variables at M2 and M14 (compared to M0) between groups were compared using separate linear mixed regression models, with time, group, time*group and time*baseline value as fixed effects, and patient*sample and patient as random effects to account for correlation between repeated measures. Normality of model residuals was verified graphically. Because of a deviation in normality of residuals for the mMRC scale, a non-parametric analysis of covariance model (ANCOVA) was used. These analyses were repeated with adjustment on unbalanced confounding factors between groups (geographic area, sex, airflow obstruction, COPD prevalence, number of comorbidities, 6MST at baseline). All analyses were repeated after handling missing values and confounding factors by multiple imputation procedure. Missing data were imputed under the missing at random assumption using a regression switching approach (m = 20 imputations) with predictive mean matching method for continuous variables and logistic regression (binary, ordinal, or polynomial) for qualitative variables. The imputation procedure was performed using all main baseline characteristics and outcomes, and estimates obtained in the different imputed data sets were combined using the Rubin’s rules.
Results
A total of 2734 individuals with CRDs were contacted by the PR team: 78 (2.8%) were not enrolled in PR after the learning needs assessment visit (lack of motivation, n = 45; hospitalisation, n = 8; lung transplantation, n = 3; death, n = 22). Retrospectively, 57 participants (2.1%) were excluded for enrolling in a second PR programme within the same year, and 149 (5.6%) with active cancer treatment.
Baseline characteristics of the participants according to the care managers groups.

Abbreviations. BMI, body mass index; COPD, chronic obstructive pulmonary disease; ILD, interstitial lung disease; OSAS, obstructive sleep apnoea syndrome; OHS, obesity hypoventilation syndrome; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; LTOT, long term oxygen therapy; NIV, non-invasive ventilation; CPAP, continuous positive airway pressure.
aOthers CRDs included: chronic bronchitis, cystic fibrosis, aspergillosis, sarcoidosis and silicosis.
bcollected from the medical record provided by the respiratory specialist.
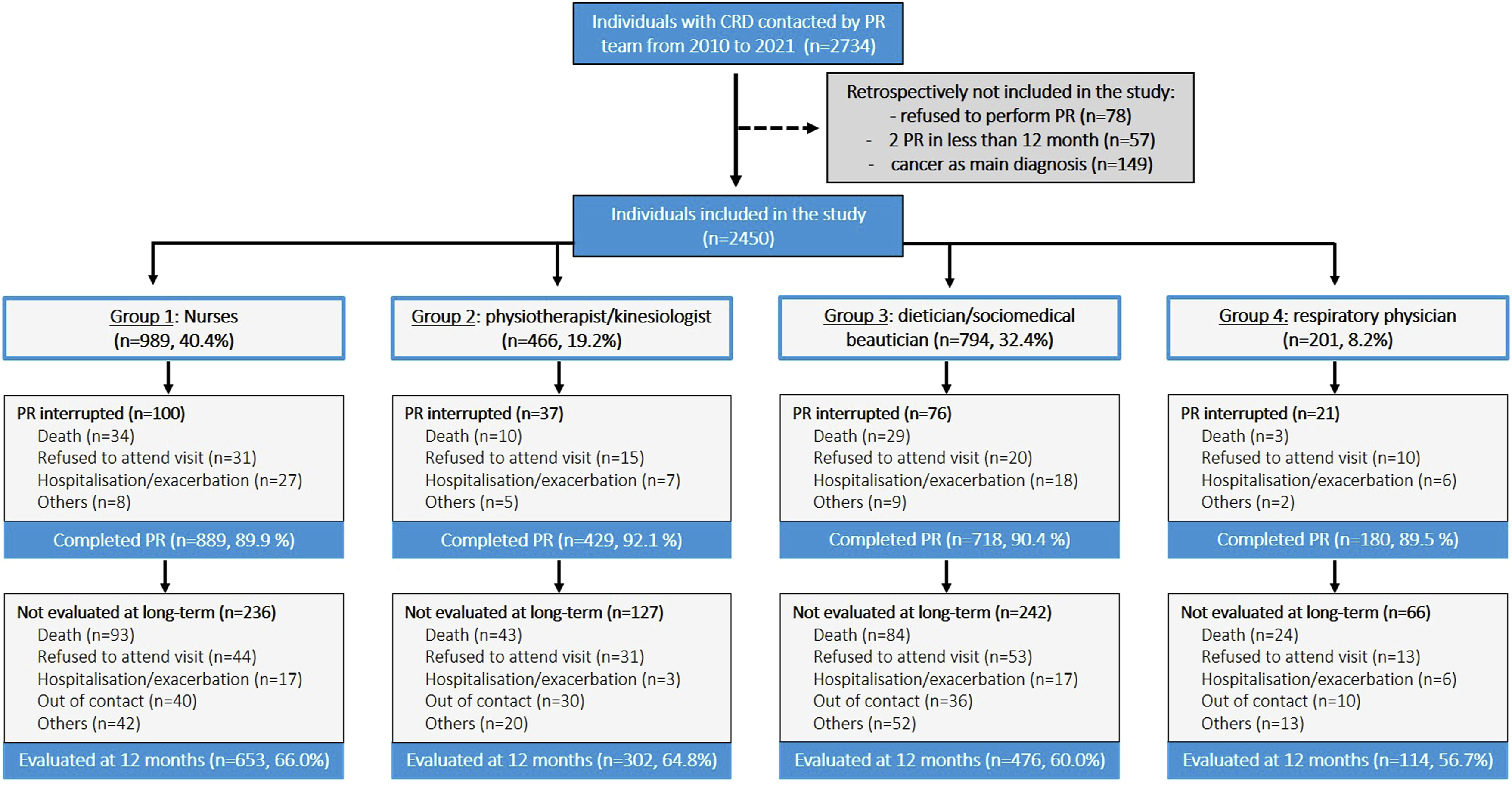
Flow chart of study participants according to care manager groups. Abbreviations. CRD, chronic respiratory disease; PR, pulmonary rehabilitation.
Baseline characteristics and assessments
Baseline assessments score according to the care managers groups.

Abbreviations. CAT, COPD assessment test; VSRQ, visual simplified respiratory questionnaire; HAD, hospital anxiety and depression scale; mMRC, modified medical research council scale; 6MST, 6-minute stepper test.
Follow ups and dropouts
Of the 2450 participants, 234 (9.6%) did not complete PR and 671 (27.4%) missed the 1-year follow-up (Figure 1). Gr 1 and Gr 2 had more individuals assessed at 1 year than Gr 4 (
Changes at the end of the programme
Delta of the assessments changes at short (M2) and long-term (M14) after PR according to care manager group (per protocol analysis).

Abbreviations. CAT, COPD assessment test; VSRQ, visual simplified respiratory questionnaire; HAD, Hospital Anxiety and Depression scale; 6MST, 6-minute stepper test.
The minimal clinically important difference of the CAT, VSRQ, HAD and 6MST is considered to be a change of 2, 3.4 and 1.5 points (subscore), and 40 strokes, respectively.
Changes at 12-months after the end of the programme
Only one outcome—VSRQ—was no longer improved at M14 in Gr 2 compared to baseline (mean change = + 1.9 [−2.9 to 6.7] points,
Discussion
The main finding of this real-life study is that health-related quality of life, anxiety and depressive symptoms, dyspnea, and exercise capacity improved similarly across care manager groups by the end of the programme. In other words, with intensive initial training specific to PR (2 months) and ongoing weekly in-service training (4 h/week), healthcare professionals—regardless of their original discipline—were able to deliver an effective face-to-face, home-based programme for individuals with CRDs. This home transdisciplinary model provides an alternative to conventional centre-based multidisciplinary PR by reaching patients with various CRDs and comorbidities in urban, suburban, and rural settings who are unable or unwilling to attend facility-based programmes.
In line with recent international guidelines,2,18 the home-based programme included not only individuals with COPD, 11 but also those with ILD, 12 asthma, 19 obstructive sleep apnea 20 and more recently, COVID-19, 21 involving over 2500 individuals with CRDs over 11 years. Due to the real-world design, 40% of participants were cared for by nurses and less than 20% by physiotherapists—reflecting the recruitment of six nurses, compared to three kinesiologists and one physiotherapist between 2010 and 2021. Baseline characteristics differed between groups, including sex, geographical location, primary diagnosis, comorbidities and airflow obstruction. Gr 4 comprised only one male respiratory physician, who occasionally cared for male patients when the originally assigned (mostly female) care managers felt unsafe continuing visits (e.g., due to sexist remarks or unsafe neighborhoods). Group 3 managed more rural patients, as allocation was based on care managers’ residence. These factors introduced allocation bias. Nonetheless, patients across groups showed similar short-term improvements, and results remained consistent after adjusting for baseline differences.
A substantial body of evidence supports the efficacy of home-based PR.5,9,22 However, comparisons with existing studies are limited, as most protocols rely on remote delivery (e.g., telephone or group videoconference).23–25 The closest model, conducted in Australia for individuals with COPD, involves an initial supervised home visit followed by weekly phone calls over 7 weeks. 24 These calls included exercise guidance and personalised education, delivered by a physiotherapist trained in motivational interviewing. Although home-based PR was equivalent to center-based PR for improving health-related quality of life and exercise capacity, long-term benefits were observed to decrease in both PR modalities. 24 In the present study, at long-term follow-up, only the physiotherapist group showed no sustained improvement in VSRQ and had the least maintenance of 6MST performance. This was unexpected, as physiotherapists typically lead exercise training in PR programmes. 1 Long-term benefits depend heavily on motivational communication and self-management strategies, 26 which may be underemphasised in the initial training of physiotherapists in France. In addition, this group had the least prior professional experience, as their employment was among their first positions, whereas care managers in other groups were older and had more extensive patient-care backgrounds. Together with missing long-term data, these factors may have contributed to the reduced maintenance of benefits observed in this group.
The case manager concept—typically held by a nurse or physiotherapist in the context of pulmonary rehabilitation—has primarily been developed to provide education and support self-management during the PR programme. 27 More recent studies have extended their role to support physical activity maintenance post-PR and coordinate care with community healthcare providers.28,29 Case managers act as key resources, maintaining regular contact with patients, their families, and respiratory physicians, while also facilitating communication within the interdisciplinary PR team. However, this role differs from the care manager model developed in the present study, as case managers primarily coordinate rather than directly deliver the PR programme. However, telephone follow-up by case managers represents a cost-effective approach to patient education and monitoring 30 and can promote the maintenance of physical activity and self-management strategies following PR.28,29 These two models are complementary and represent approaches that warrant further investigation for their potential to improve accessibility and maintain long-term outcomes, in conjunction with standard centre-based programmes.
Strengths and limitations
Although a large number of individuals with CRDs were included, the monocentric, non-randomised design and absence of a control group—where participants would have been managed by different healthcare professionals—limit the applicability of the findings. This design led to differences in group allocation and baseline characteristics, which were addressed through adjusted and imputed statistical analyses. The real-life personalised design of the study and absence of a physical activity diary precluded any conclusion on educational and physical training adherence or fidelity. Over 11 years of practice, no major events (e.g., death or cardiac arrest) were reported. Minor adverse events such as hypoglycemia, arrhythmia, vagal discomfort, and muscle soreness did occur and were recorded in the electronic medical record system. However, this data cannot yet be extracted and may not have been consistently recorded across all 2450 participants. All assessments were conducted at the patient’s home by the same care manager delivering the programme, which may have introduced minimal bias. Exceptionally, a different care manager performed the 12-months assessment (e.g., if the original care manager left the team or was on extended leave). Linear mixed models were first applied to individuals with complete data at all time points (baseline, post-PR, and 12-months follow-up). This approach reduced the sample size, particularly in Group 4 and for the 6MST, where only 31.8% had complete data. Multiple imputation was used to address this, but the high proportion of missing data remains a major limitation. For all these reasons, this study should be complemented by robustly designed randomised, controlled and multicentric trials.
Implementing the care manager model within conventional multidisciplinary centre-based PR may be challenging due to legal and scope-of-practice limitations. Healthcare professionals may be accustomed operating in silos: “my job, my role and responsibilities”. 7 In a transdisciplinary model, however, traditional boundaries must be crossed: each team member contributes their expertise while also allowing others to apply that knowledge in practice. Effective implementation requires dedicated training, and programme leadership must be willing to invest in professionals who may not generate revenue during the early phase of their onboarding. However, in France at least, roles are now expanding—nurses, physiotherapists, and pharmacists can prescribe treatments such as nicotine substitutes—suggesting that similar approaches could benefit PR programmes.
Conclusion
This real-life study of over 2500 individuals with chronic respiratory disease showed that a personalised home-based PR programme delivered by one care manager, led to similar short-term benefits in health-related quality of life, dyspnoea, anxiety and depressive symptoms and exercise capacity, regardless of the care manager’s initial professional background. Home-based transdisciplinary PR could complement conventional centre-based programmes, offering a promising approach to enhance accessibility and support long-term outcomes, which should be further investigated.
Supplemental material
Supplemental material - Does care managers’ initial professional background affect the outcomes of pulmonary rehabilitation? A retrospective cohort study of 2450 individuals with chronic respiratory diseases
Supplemental material for Does care managers’ initial professional background affect the outcomes of pulmonary rehabilitation? A retrospective cohort study of 2450 individuals with chronic respiratory diseases by Sarah Gephine, Olivier Le Rouzic, Emeline Cailliau, Cécile Chenivesse and Jean-Marie Grosbois in Chronic Respiratory Disease
Footnotes
Acknowledgments
We would like to thank the patients who participated in this research and the home-based PR team (Aurore Benitende, Geneviéve Bressy, Annie Chaud, Sophie Duriez, Gaetan Evrard, Géraldine Gabet, Mathieu Grosbois, Audrey Krol, Marjorie Lambinet, Anna Lorthioy, Maite Mabille, Valentine Opsomer, Camille Paluch, Christine Pruvost, Lucas Sion, Aurore Taccoen, Gaelle Tywoniuk, Florence Urbain, Sandrine Vanexem, Laura Verstrynge, and Virginie Wauquier) for their role in delivering the interventions and help with data collection.
Ethical considerations
Approval for the observational research protocol was granted by the CEPRO (French Language Society of Pulmonology) Research Ethics Committee (2021-054), which also approved the retrospective analysis.
Consent to participate
All participants provided written informed consent prior to the start of the study.
Author contributions
SG and JMG: conceptualisation, execution, acquisition of data, formal analysis and interpretation, writing the original draft. EC: formal analysis. CC, OLR: writing – review and editing. All authors gave final approval of the version to be published.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The delivery of the home-based PR programme was financially supported Adair, Aeris Santé, Elivie, France Oxygène, Homeperf, LVL, Médopale, NorOx, Santélys, Santéo, SOS Oxygène, Sysmed, VentilHome, VitalAire and ARS Hauts-de-France. The funders played no role in the design, conduct or reporting of this study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SG and EC have nothing to disclose. OLR reports personal fees and non-financial support unrelated to the submitted work from AstraZeneca, Boehringer Ingelheim, Chiesi, CSL Behring, GlaxoSmithKline, MSD France, Vertex and Vitalaire. OLR is principal investigator in studies for Vertex and CSL Behring. CC reports personal fees and non-financial support unrelated to the submitted work from ALK-Abello, AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKlein, MEDA Pharma, Medexact, Novartis, Pierre Fabre, Pfizer, Roche, Sanofi, Santélys, and TEVA. JMG reports personal fees and non-financial support unrelated to the submitted work from AstraZeneca, Boehringer Ingelheim, Chiesi, CSL Behring, GlaxoSmithKlein, Menarini.
Data Availability Statement
De-identified individual data are available on reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
