Abstract
Introduction
Current research mainly examines the link between sarcopenia and obstructive spirometry, with limited studies on preserved ratio impaired lung function (PRISm). This study aims to investigate the association between skeletal muscle index (SMI) and lung function indicators in populations with different characteristics, with particular emphasis on the PRISm population.
Methods
Data were utilized from the National Health and Nutrition Examination Survey (NHANES) 2011-2012 cycle. The participants were categorized into three groups: obstructive spirometry, PRISm, and normal spirometry. The SMI was calculated by dividing appendicular skeletal muscle mass by body mass index. Multiple linear regression analysis was employed to explore the relationship between the SMI and lung function indicators, including forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC).
Results
Among the 2513 participants aged 20–59 years, 2096 had normal spirometry, 186 were in the obstructive spirometry group, and 231 were in the PRISm group. The SMI was significantly associated with lung function indicators after adujustment: in the obstructive spirometry group, per 0.1 unit increase in SMI was linked to an increase in FEV1 of 0.214 L (95% CI: 0.115-0.313, p < 0.001) and in FVC of 0.339 L (95% CI: 0.213-0.465, p < 0.001); in the PRISm group, per 0.1 unit increase in SMI was associated with an increase in FEV1 of 0.184 L (95% CI: 0.149-0.219, p < 0.001) and in FVC of 0.222 L (95% CI: 0.169-0.274, p < 0.001); and in the normal spirometry group, per 0.1 unit increase in SMI was linked to an increase in FEV1 of 0.240 L (95% CI: 0.222-0.257, p < 0.001) and in FVC of 0.316 L (95% CI: 0.294-0.338, p < 0.001).
Conclusion
Our study newly identifies a significant positive association between SMI and lung function in the PRISm group, a relationship also consistently observed in obstructive and normal spirometry groups.
Keywords
Introduction
Sarcopenia is characterized by the progressive loss of muscle mass and function, predominantly affecting the elderly but also starting in mid-life, and is linked to serious health problems such as disability, functional impairment, and increased mortality. 1 Skeletal muscle mass index (SMI), which is obtained through dual-energy X-ray absorptiometry (DXA), calculated from appendicular skeletal muscle mass (ASM) adjusted for body mass index (BMI), has been proposed for the assessment of sarcopenia. 2 Low SMI has been recognized as an independent prognostic factor for skeletal muscle wasting.3–6 Furthermore, SMI plays a significant role in evaluating prognosis across a range of diseases, encompassing cardiovascular, digestive, and respiratory conditions.7–9
Chronic obstructive pulmonary disease (COPD), a chronic respiratory disorder, is pathologically characterized by persistent respiratory symptoms, airflow limitation, and progressive decline in lung function. 10 The Global Initiative for Chronic Obstructive Lung Disease (GOLD) defines and stages COPD based on lung function parameters, including forced vital capacity (FVC) and forced expiratory volume in 1 second (FEV1), using spirometry. 11 Preserved Ratio Impaired Spirometry (PRISm) is a distinct spirometric phenotype characterized by an FEV1/FVC ratio greater than 0.70 and an FEV1 less than 80% of the predicted value. 12 Although these patients do not meet the criteria for COPD, they experience significant respiratory symptoms and may progress to GOLD-defined COPD. 13 While previous epidemiological studies have often grouped it under a “restrictive spirometric pattern”, 14 PRISm is not equivalent to true restriction, which requires reduced total lung capacity confirmed by lung volume measurement.13,14 Its recognition in the 2023 GOLD report as a pre-COPD stage highlights its clinical importance, particularly since such individuals have often been overlooked in prior studies.15,16 It is reported that the prevalence of COPD in the US adult population was 13.1%-14.3%, while the prevalence of PRISm was 9.6%–-10.2%. 17
Sarcopenia is a prevalent and significant comorbidity across chronic respiratory diseases. In patients with obstructive conditions like COPD, its prevalence is considerably higher (15.5%–34%) than in the general population.18,19 It adversely influences the clinical course by increasing exacerbation risk, accelerating functional decline, and reducing exercise tolerance and quality of life.20–23 Furthermore, it independently predicts increased mortality and imposes a greater economic burden on the healthcare system. 24 Similarly, in restrictive lung diseases such as idiopathic pulmonary fibrosis, sarcopenia is highly prevalent (approximately 30%) and constitutes an independent risk factor for mortality, increasing the risk of death by nearly 88%. 25 Current researches primarily focus on the relationship between sarcopenia and obstructive spirometry,21,26 with relatively few studies exploring the association between sarcopenia and lung function in patients with PRISm. Therefore, this study aims to investigate the association between SMI and lung function indicators in populations with different characteristics, with particular emphasis on the PRISm population.
Methods
Study population
The National Health and Nutrition Examination Survey (NHANES) utilizes a rigorous, stratified, multistage probability sampling method to ensure a representative sample of the U.S. population. Administered by the National Center for Health Statistics (NCHS), NHANES provides a comprehensive overview of the health and nutritional status of a diverse group of noninstitutionalized civilians in the United States through physical examinations, laboratory tests, and dietary interviews. The NCHS Ethics Review Board granted ethical approval for NHANES, and all participants provided written informed consent before participation.
Our analysis focused on data from the NHANES 2011-2012 cycle. We applied specific inclusion and exclusion criteria to the NHANES 2011-2012 dataset, which initially included 9756 individuals. Following an initial exclusion of individuals who failed to complete lung function testing (n = 2261) and DXA testing (n = 1803), we retained 5692 individuals. Further exclusions were made for participants with missing lung function values (n = 529), missing DXA data (n = 921), and those under the age of 20 years (n = 1729). A total of 2513 individuals aged 20-59 years were deemed eligible for inclusion in our study, categorized into three groups: obstructive spirometry (n = 186), PRISm (n = 231), and normal spirometry (n = 2096). In this study, data on PIR were unavailable for 155 individuals, smoking status was missing for 424 individuals, and drinking status was absent for 141 individuals. The flowchart for inclusion and exclusion is shown in Figure 1. Flowchart for participant selection.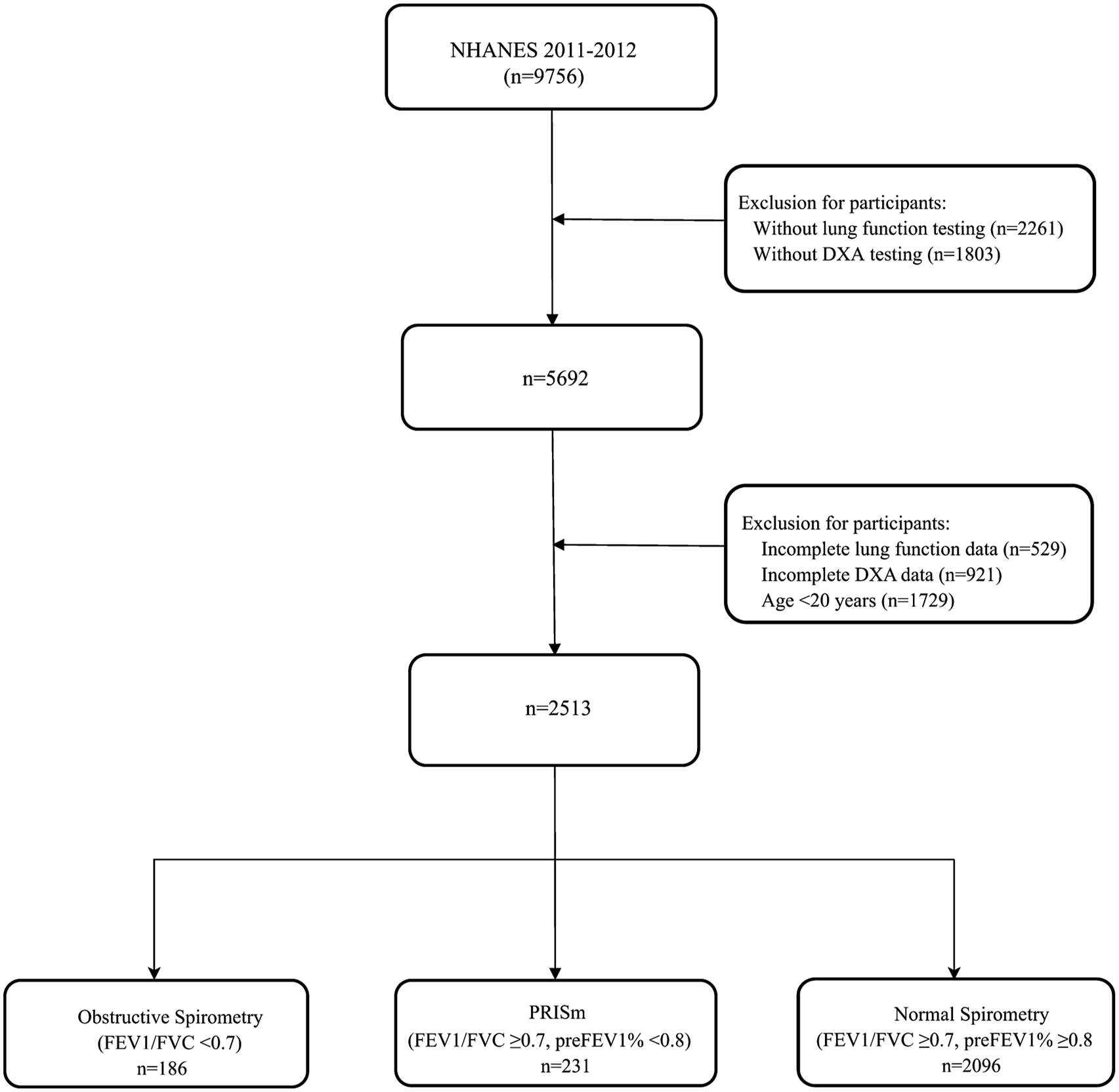
Definition of obstructive spirometry, PRISm, and normal spirometry
The spirometry procedures adhered to the recommendations of the American Thoracic Society (ATS), and the National Institute for Occupational Safety and Health (NIOSH) Division of Respiratory Disease Studies served as the NHANES Spirometry training and quality control consultant. Technicians received formal training and completed the NIOSH-approved spirometry course. Spirometry data were reviewed by expert reviewers at the NIOSH quality control center, ensuring internal data consistency. A detailed description of the process can be found at https://wwwn.cdc.gov/Nchs/Data/Nhanes/Public/2011/DataFiles/SPX_G.htm. In our study, eligible participants aged 20-59 years underwent a baseline spirometry examination, collecting lung function indices, including measured values for FEV1 and FVC. The predicted values for FEV1 and FVC were calculated based on race-neutral equations. 27 The groups are now defined as 12 : (1) Participants with normal spirometry, defined as FEV1/FVC ≥ 0.7 and FEV1 ≥ 80% predicted 28 ; (2) Participants with PRISm, defined as FEV1/FVC ≥ 0.7, FEV1% < 80%predicted12,17,29; and (3) Participants with obstructive spirometry, defined as FEV1/FVC < 0.7. 30
Definition of skeletal muscle index
DXA is a widely recognized gold standard technique for assessing body composition, including parameters such as muscle mass and fat mass. In the NHANES 2011-2012 cycle, DXA data were processed and analyzed in accordance with stringent standard procedures. Whole-body scans were conducted using the Hologic Discovery model A densitometers (Hologic, Inc., Bedford, Massachusetts) with software version Apex 3.2, and the results were reviewed by experts to ensure accuracy and consistency. 31 In our study, muscle mass was assessed using the standardized protocol of the Foundation for the National Institutes of Health (FNIH), which calculates the ASM to BMI ratio as a reliable muscle mass indicator. 32 To quantify muscle mass, ASM was derived by summing the lean mass of the arms and legs, excluding bone mineral content, as measured by DXA. The SMI was calculated by dividing ASM by the BMI, as informed by recent studies.32–36
Covariates
The present study included the following covariates: sociodemographic variables such as age, gender, race, education level, marital status, and poverty income ratio (PIR); lifestyle variables including BMI, smoking status, and drinking status; and the comorbidity of asthma, which was identified through the interview question, “Have you ever been told you have asthma?”.
Statistical analysis
This study utilized the recommended weighting scheme from the NHANES database for data analysis. The participants were categorized into three spirometric groups: obstructive spirometry, PRISm, and normal spirometry. Continuous variables were presented as means ± standard deviations (SD) and assessed for differences using survey-weighted linear regression, with P-values reported. Categorical variables were expressed as survey-weighted percentages, with differences evaluated using survey-weighted Chi-square tests. The correlation between SMI and lung function was analyzed using Pearson’s correlation coefficient. Multiple linear regression models were constructed to examine the relationship between SMI and lung function indicators (FEV1, FVC) across different spirometric groups. We fitted three models, one unadjusted and two adjusted for different factors. Model 1 was adjusted for age, gender, race, educational attainment, marital status, and PIR. Model 2 incorporated all adjustments from Model 1 and further accounted for BMI, drinking status, smoking status, physical activity, and asthma. Missing values in categorical covariates (PIR, smoking status, and drinking status) were modeled as a distinct group. Additionally, we utilized predicted values of the lung function indicators (FEV1, FVC) to replicate the analysis. Sensitivity analyses were performed to examine the association between SMI and lung function when SMI was calculated using ASM/height. 2 To assess the stability of our results across different groups, subgroup analyses were conducted to assess the association between SMI and lung function, stratified by age groups (20-39 years vs 40-59 years), gender, and asthma status. We performed interaction analyses to further investigate whether this association is different across adults with normal spirometry, obstructive spirometry, and PRISm. All analyses were conducted using R version 4.2.3, with P-value of <0.05 considered statistically significant.
Results
Baseline characteristics of the study participants.
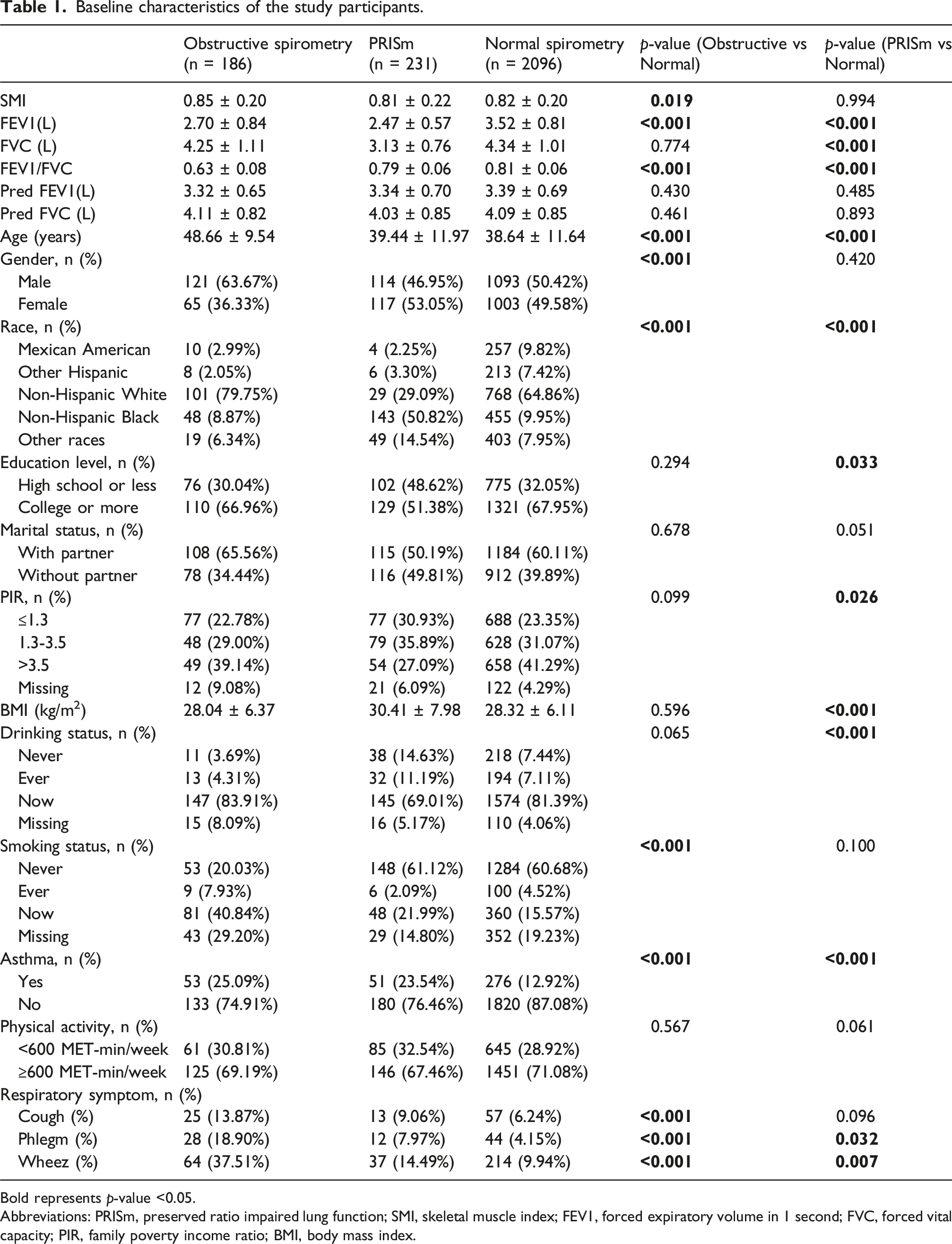
Bold represents p-value <0.05.
Abbreviations: PRISm, preserved ratio impaired lung function; SMI, skeletal muscle index; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PIR, family poverty income ratio; BMI, body mass index.
The scatterplot showed the correlation analyses between SMI and lung function (Supplemental Figures 1–4). The SMI was moderately positively associated with FEV1 in three groups (r = 0.579, 0.798, and 0.757, respectively; all p < 0.001, Supplemental Figure 1). Besides, the SMI was moderately positively associated with FVC in three groups (r = 0.643, 0.767, and 0.754, respectively; all p < 0.001, Supplemental Figure 2). The SMI had a highly positive correlation with the pred FEV1 (r = 0.830, 0.854, and 0.870, respectively; all p < 0.001, Supplemental Figure 3). Besides, the SMI was highly positively associated with pred FVC in populations with different characteristics (r = 0.844, 0.870, and 0.855, respectively; all p < 0.001, Supplemental Figure 4).
Association between SMI and lung function (measured value) across different groups in linear regression models.

Model 1: Adjusted for age, gender, race, education level, marital status, and PIR.
Model 2: Adjusted for Model 1 plus BMI, drinking status, smoking status, physical activity and asthma.
Note: SMI changes corresponding to the effect values in the regression models were as follows: for each 0.1-unit increase in SMI.
Abbreviations: PRISm, preserved ratio impaired lung function; SMI, skeletal muscle index; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PIR, family poverty income ratio; BMI, body mass index; CI, confidence interval.
Association between SMI and lung function (predicted value) across different groups in linear regression models.

Model 1: Adjusted for age, gender, race, education level, marital status, and PIR.
Model 2: Adjusted for Model 1 plus BMI, drinking status, smoking status, physical activity and asthma.
Note: SMI changes corresponding to the effect values in the regression models were as follows: for each 0.1-unit increase in SMI.
Abbreviations: PRISm, preserved ratio impaired lung function; SMI, skeletal muscle index; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; PIR, family poverty income ratio; BMI, body mass index; CI, confidence interval.
The association between SMI (ASM divided by height 2 ) and lung function across different groups in linear regression models can be seen in Supplemental Table 1. In the obstructive spirometry group, SMI was significantly positively associated with FEV1, FVC, pred FEV1, and pred FVC (all p < 0.01), but after further adjustment, these associations were not significant (all p > 0.05). In the PRISm group, SMI was significantly positively associated with FEV1, FVC, pred FEV1, and pred FVC in unadjusted models (all p < 0.001), and these associations were not statistically significant in adjusted models (all p > 0.05). In the normal spirometry group, SMI maintained a significant positive association with all four lung function indicators (FEV1, FVC, pred FEV1, pred FVC) regardless of whether confounding factors were adjusted (all p < 0.001).
In subgroup analyses, participants were stratified by age (20-39 vs 40–59 years), gender, and asthma status. Positive associations between SMI and lung function parameters were consistently observed across all subgroups (Supplemental Tables 2-4). Among the 40–59 years group, the SMI demonstrated a significant positive association with all lung function parameters (FEV1, FVC, pred FEV1, pred FVC, all p < 0.001). Among the 20–39 years group, a similar positive association was observed; however, the association between SMI and FEV1 within the obstructive spirometry category did not reach statistical significance (p = 0.199). In males, the SMI demonstrated a significant positive association with all lung function parameters (FEV1, FVC, pred FEV1, pred FVC), with all P-values being less than 0.001. In females, a similar positive association was observed; however, the association between SMI and FEV1 within the obstructive spirometry category did not reach statistical significance (p = 0.068). Among participants without asthma, the SMI demonstrated a significant positive association with all lung function parameters (FEV1, FVC, pred FEV1, pred FVC, all p < 0.001). Among participants with asthma, a similar positive association was observed; however, the association between SMI and FEV1 within the obstructive spirometry category did not reach statistical significance (p = 0.069).
To further investigate whether this association is different across adults with obstructive spirometry, PRISm, and normal spirometry, we tested for interactions. The interaction analysis showed that the association between SMI and lung function was significantly different across different lung function subgroups (p for interaction <0.05, Supplemental Figure 5).
Discussion
Our study demonstrated that there was a significant positive association between SMI and lung function (FEV1, FVC) in individuals with PRISm. This positive association remained robust across obstructive and normal spirometry groups. To our knowledge, this is the first study to investigate the associations between SMI and multidimensional lung function parameters across different spirometric groups.
In our study, we included a relatively younger population (20–59 years), in which the prevalence of PRISm was 9.19%, while obstructive spirometry was observed in 7.4% of the participants, lower than the prevalence of the U.S. population aged 20–79 years 17 Most COPD studies have included populations with mean ages older than 60 years 37 The concept of “early COPD” highlights the intricate nature of disease progression and the potential advantages of early intervention. 38 Evidence strongly supports studying younger COPD patients, as early intervention can alter the disease’s course, emphasizing the need for research on early COPD.39–41 A significant innovation of our study lies in its focus on the PRISm group, a population that has been relatively understudied in previous research, whereas prior research predominantly focused on COPD cohorts or individuals with normal spirometry.21,26,42,43 PRISm, defined as a pre-clinical abnormal spirometric pattern, was associated with a significantly increased risk for mortality and poor cardiovascular and respiratory outcomes compared to normal spirometry. 12 In addition, PRISm patients typically exhibit more respiratory symptoms, more comorbidities, greater health burden, and poorer quality of life. 44 Our findings provide new insights into the relationship between muscle mass and lung function in the PRISm group, which may help inform future research and clinical practice.
In the obstructive spirometry group, our unadjusted model demonstrated that a 0.1 unit increase in SMI was associated with an increase in FEV1 of 0.274 L (274 mL), and this association remained significant at 0.214 L (214 mL) in our adjusted model (Model 2). For FEV1 in patients with obstructive spirometry, such as COPD, some studies have proposed that the minimal clinically important difference (MCID) in FEV1 is approximately 100 mL, 45 while others have suggested a range between 0.18 L and 0.32 L.46,47 The unadjusted model demonstrated that a 0.1 unit increase in SMI was associated with an increase in FVC of 0.400 L (400 mL), and this association remained significant at 0.339 L (339 mL) in our adjusted model (Model 2). The MCID in FVC is approximately 200 mL. 48 This effect size indicates that the observed relationship is not only statistically significant but also potentially clinically meaningful. The MCID in FEV1, FVC among PRISM has not yet been determined, and more research is needed. Our findings align with those observed in studies conducted in other countries.21,26,42,43 In a study involving 185 Mexican patients with COPD aged >40 years, analysis revealed a significant positive association between the SMI and indicators of lung function. 21 In the Rotterdam study, involving 5082 participants, chronic airway disease in older adults was found to be associated with a reduction in ASM. 26 In Korean adults, SMI demonstrated a positive and significant association with both FVC and FEV1. 42 Similarly, in the elderly long-term care population in Japan, SMI has been positively linked to lung function, particularly in terms of respiratory muscle strength. 43
According to the guidelines established by the FNIH, the SMI values utilized in this study were calculated by dividing ASM by the BMI. 32 Concurrently, several studies have been published concerning the definition of SMI adjusted for BMI.32–36 BMI serves as an effective indicator of the proportional relationship between an individual’s weight and height, thereby offering a straightforward and widely applicable method for adjusting muscle mass. 32 Another way to define SMI is to use ASM/height 2 as the criterion. 49 In our study, sensitivity analysis showed that the positive association between SMI and lung function was consistent when ASM/height 2 was used to calculate SMI.
Our research identified a consistent association between SMI and pulmonary function (FEV1, FVC) across spirometric groups, suggesting that SMI may reflect the functional status of the respiratory muscles. Declines in SMI are likely associated with reduced respiratory muscle strength, directly impairing lung function, a pathway supported by evidence from aging and sarcopenia models. 50 In populations with COPD and PRISm, systemic inflammation and oxidative stress may modify this relationship. In COPD, inflammatory mediators (e.g., TNF-α and interleukins) and oxidative stress promote muscle protein degradation and mitochondrial dysfunction, exacerbating sarcopenia and respiratory muscle impairment. 51 In contrast, in individuals with normal spirometry, the association may be more directly linked to age-related declines in muscle mass and function, absent significant inflammatory drivers. This may partially explain the variations in the relationship between SMI and lung function across the different spirometric groups. Therapeutic strategies targeting oxidative stress and inflammation in COPD have shown promise in mitigating muscle dysfunction. For instance, dietary inclusion of antioxidants such as polyphenols has been explored as a potential treatment to reduce oxidative damage and improve muscle function. 52 Exercise is another intervention that has been shown to enhance mitochondrial function and reduce oxidative stress, thereby improving muscle strength and endurance in COPD patients. 53 Notably, muscle training, particularly resistance exercise, is instrumental in optimizing muscle function. It significantly contributes to the enhancement of both the strength and endurance of respiratory muscles, thereby improving respiratory efficiency and alleviating symptoms of dyspnea. 54
There were several limitations in our study. Firstly, this study was conducted during the NHANES 2011-2012 cycle due to the availability of data on SMI and lung function assessment, which were not accessible in more recent cycles. Secondly, the SMI values were calibrated in accordance with BMI following the FNIH guidelines in our study, and may consequently differ from those derived using alternative diagnostic criteria. 32 Thirdly, DXA accuracy can be affected by device calibration, operator skill, and data processing. We followed the NHANES Body Composition Procedures Manual, which enforces strict quality control for accurate measurements. Fourthly, we utilized the SMI index to represent muscle mass status, without incorporating measures of muscle strength and physical performance. Consequently, our analysis may not fully capture the relationship between a comprehensive diagnosis of sarcopenia and lung function. Fifthly, we included participants with complete data on SMI and lung function among American adults, restricting our analysis to individuals aged 20–59 years. This selection criterion may introduce bias; therefore, it is recommended that future studies incorporate older adults (aged 60 years and above) or non-US populations for further validation.
In conclusion, we found a significant positive association between SMI and lung function (FEV1, FVC) in individuals with PRISm. This positive association persisted consistently across both obstructive and normal spirometry groups. Consequently, the evaluation and intervention focusing on muscle mass should be integrated into the management strategy for patients across the entire spectrum of lung function.
Supplemental Material
Supplemental Material - Association between skeletal muscle index and lung function in populations with different characteristics
Supplemental Material for Association between skeletal muscle index and lung function in populations with different characteristics by Xian-xin Xia, Xin-xin Xue, Li Xiong, Ya-jun Chen, Hao Chen, Fang He, Qi Li in Chronic Respiratory Disease.
Footnotes
Acknowledgements
The authors would like to acknowledge and thank the participants for their valuable contributions during the data collection phase of the NHANES project.
Ethical consideration
Ethical approval was not required for this study as it utilized publicly available data from the National Health and Nutrition Examination Survey (NHANES).
Consent to participate
The study protocol was approved by the NHANES Institutional Review Board, and was performed in accordance with the Declaration of Helsinki, with all NHANES participants providing signed informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The NHANES database offers free access to the information utilized in this investigation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
