Abstract
Background
Chronic respiratory diseases frequently cause a decrease in physical activity and lower exercise tolerance. Eccentric Cycling Training (ECT) offers a potential rehabilitation strategy. However, its effectiveness when combined with Helmet Ventilation (HV) is not fully established.
Methods
This nonblinded randomized controlled trial (RCT) investigated the effects of ECT with or without HV in individuals with chronic respiratory diseases. Participants were randomly assigned to the ECT, ECT-HV, or control group and underwent graded exercise testing. The ECT sessions spanned 9 weeks with gradually increasing intensity. The primary outcome was peak oxygen consumption (VO2peak). Secondary outcomes included functional tests and symptom scores.
Results
Forty-four patients with chronic respiratory diseases were enrolled and divided among the ECT (n = 13), ECT-HV (n = 15), and control (n = 13) groups. After 9 weeks, significant improvements in the 6-min walk test (6MWT) distance, energy expenditure index during the 6MWT, and timed up and go test were observed in both the ECT and ECT-HV groups compared to the control group. Reductions in initially high Borg leg fatigue were noted over time in these groups. VO2peak at follow-up was 17.6 ± 2.9 ml/min/kg in the control group, 17.2 ± 4.5 ml/min/kg in the ECT group, and 18.2 ± 3.9 ml/min/kg in the ECT-HV group, with no significant differences among groups. No significant differences in achieving the targeted exercise intensity were found between the ECT and ECT-HV groups.
Conclusions
Over a 9-week period, eccentric cycling training improved functional capacity and mobility in patients with chronic respiratory diseases, although no significant changes were observed in peak oxygen consumption or overall cardiopulmonary fitness.
Keywords
Introduction
Exercise training is a vital component of pulmonary rehabilitation, with substantial evidence supporting its efficacy as a therapeutic intervention for managing chronic obstructive pulmonary disease (COPD). 1 Endurance training enhances cardiopulmonary exercise capacity and peripheral muscle function in patients with COPD.2,3 Conventional concentric exercises may be intolerable for some elderly individuals 4 or those with chronic diseases because of exercise intolerance.5,6
Eccentric exercise, compared to concentric exercise at similar workloads, is associated with lower oxygen consumption (VO2), 7 a lower exercise heart rate, and a reduced perception of effort. 8 The similar VO2 between both exercise types implies that eccentric exercise yields higher power output than concentric exercise. 9 The benefits of eccentric cycling training (ECT) have also been observed in patients with chronic heart failure 5 and elderly individuals. 10 The distinct physiological and mechanical features of eccentric contractions, characterized by higher forces and lower energy requirements,11,12 have sparked growing interest, particularly in rehabilitation.4,13
For individuals struggling to reach the desired exercise intensity due to respiratory conditions, 14 noninvasive ventilation (NIV) has been proposed as an adjunct in exercise programs. However, its implementation is complex and labor-intensive, making it feasible only in specialized units and for experienced patients. 2 Studies report dropout rates of 7.1% to 28% in exercise training supplemented with NIV.15,16 Helmet ventilation (HV), a type of NIV interface, delivers gas flow through a transparent plastic hood originally designed for hyperbaric oxygen therapy. 17 Compared with face masks, helmets form a seal around the neck with a soft collar and provide better comfort, fewer air leaks, and greater seal integrity. These features may enhance patient tolerance and adherence during exercise training.18,19 HV provides continuous positive pressure support, facilitating respiratory assistance, improving oxygenation and gas exchange, and reducing the work of breathing.20,21
Although numerous studies have examined eccentric or traditional exercise with NIV in patients with chronic respiratory disease, research combining these approaches is lacking. Therefore, this study investigated the impact of ECT and concurrent HV in this patient population. We hypothesized that both ECT and ECT-HV would lead to significant improvements in VO2peak, functional outcomes compared to the control group, and that ECT-HV would produce greater improvements than ECT alone.
Methods
Study design
This nonblinded randomized controlled trial was conducted at the Pulmonary Rehabilitation Center of Fu Jen Catholic University in Northern Taiwan from April 2023 to February 2024.
Ethics
We conducted the trial in accordance with good clinical practice guidelines and the Declaration of Helsinki. The study was approved by the Institutional Review Board of the Fu Jen Catholic University Hospital, New Taipei City, Taiwan (FJUH111193), and registered with ClinicalTrials.gov (NCT05760352, 02/27/2023). Written Informed consent was obtained from all individual participants included in the study.
Population
This study included eligible participants who were outpatients with chronic respiratory diseases requiring a pulmonary rehabilitation program. The inclusion criteria were age between 20 and 80 years, a diagnosis of chronic respiratory diseases, and pulmonary rehabilitation for more than 3 months. Chronic respiratory diseases included bronchiectasis, chronic bronchiolitis, emphysema, and/or COPD. The classification of disease severity also encompassed the “pre-COPD” stage, as defined by the 2023 Global Initiative for Chronic Obstructive Lung Disease report. 1 Patients with tracheostomy, those requiring long-term oxygen therapy or home ventilation, those with severe left heart failure (NYHA III–IV), those with neuromuscular diseases, those who had experienced acute exacerbations of COPD within the past 3 months, and those unable to cooperate with the cardiopulmonary exercise test were excluded from the study.
Randomization
Participants meeting the inclusion criteria underwent randomization, assigned to one of three groups with a 1:1:1 allocation ratio: the intervention group (ECT), the ECT combined with HV (ECT-HV) group, or the control group. The randomization sequence was generated by KY Chao using block randomization with a fixed block size of six, for a total of 10 blocks, via the website https://www.sealedenvelope.com. Allocation was concealed using sequentially numbered, sealed, opaque envelopes to ensure allocation concealment. The envelopes were administered by a research assistant who was not involved in participant recruitment or outcome assessment. Blinding was not feasible due to the nature of the intervention.
Protocol
All participants in the three groups underwent a graded exercise test (GET) to assess their maximum exercise capacity, both within 1 week before and after the 9-weeks pulmonary rehabilitation program.
Graded exercise test
To define the maximal workload (Wmax), a graded exercise test (GET) was conducted using a stationary bicycle ergometer (Cortical Cpet, LODE B.V, Groningen, Netherlands). The participants were fitted with a respiratory mask to monitor VO2, minute ventilation (VE), and carbon dioxide production (VCO2); these parameters were measured by using a cardiopulmonary measurement device (MasterScreen CPX, CareFusion Corp., Hoechberg, Germany). The oxygen uptake efficiency slope (OUES) and the VE-VCO2 slope were calculated from these parameters. The VO2peak was defined in accordance with the guidelines for cardiopulmonary exercise testing recommended by the American Thoracic Society and American College of Chest Physicians Statement. 22 Baseline parameters were initially recorded for 5 min. Subsequently, they began pedaling at 0 W of resistance and 60 r/m for a 2-min warm-up. The workload was then increased every 3 min by 30 W until a load of 120 W was reached. Subsequently, the workload was increased by 20 W every 3 min until the participant reached their maximum exercise intensity.
Eccentric cycling training
The participants in both the ECT-HV and ECT groups engaged in exercise training by using a stationary bicycle ergometer (ECCR-Re100, eHuntSun Health Tech, Taichung, Taiwan) twice a week over 9 consecutive weeks. The exercise training routine commenced with a 5-min warm-up at 60% of the Wmax, followed by 30 min at the target exercise intensity, concluding with a 5-min cool-down at 60% of the Wmax, resulting in a 40-min session. The participants maintained a pedal cadence between 60 and 70 rpm. To minimize adverse exercise-induced responses, the first week of training served as a familiarization phase with two adaptive training sessions. The target exercise intensity was set progressively increasing it by 10% for each subsequent session until reaching an intensity of 160% of the Wmax by the sixth week. This intensity level was maintained for the remaining sessions in the seventh to ninth weeks (Figure 1). In case of an emergency, the training was halted when the participant exhibited symptoms or signs consistent with the criteria outlined in the American College of Sports Medicine guidelines.
23
Study flowchart illustrating the progression of eccentric cycling training (ECT) intensity over each week. Each week and the flow of participants through the trial. One participant from the control group and two from the ECT-helmet ventilation (ECT-HV) group withdrew after providing informed consent but before undergoing the baseline graded exercise test. An additional control group participant voluntarily withdrew during the intervention period, and another was hospitalized due to an acute exacerbation of COPD. One participant in the ECT group was hospitalized for pneumonia. In the ECT-HV group, one participant withdrew due to a traffic accident that affected lower-limb mobility, based on the clinical assessment of the principal investigator. LTOT: long-term oxygen therapy, NYHA: New York Heart Association; AECOPD: acute exacerbation of chronic respiratory pulmonary disease; HV: helmet ventilation; Wmax: the maximal workload of the first graded exercise test; W: week; S: session.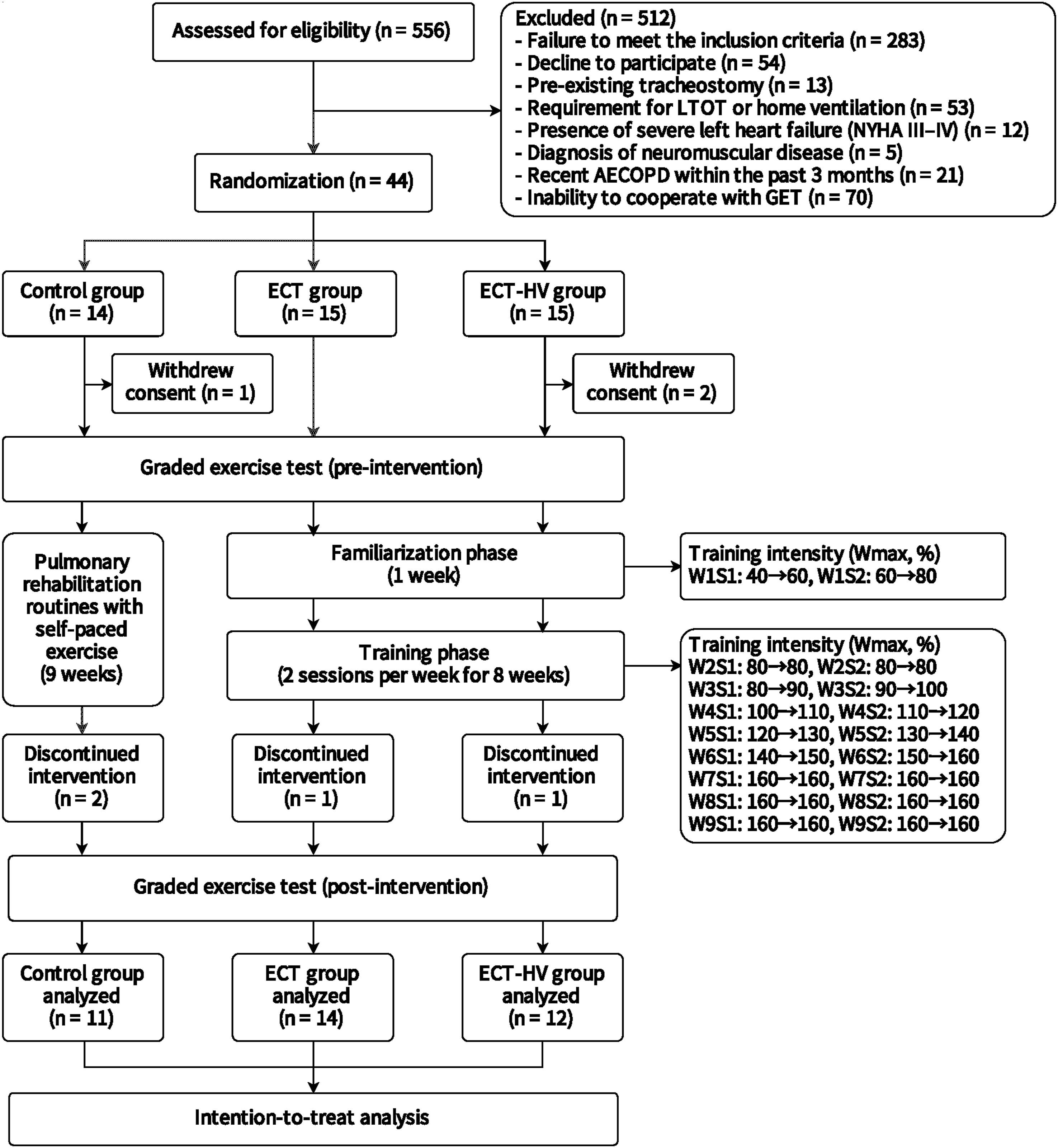
Helmet ventilation
The participants in the ECT-HV group received a combination of ECT and HV. This involved the use of an intensive care ventilator (Servo-i, Maquet, Getinge group Critical Care, Solna, Sweden) operating in the noninvasive ventilation-pressure support mode. The initial settings for the ventilator included a pressure support (PS) level of 14 cm H2O, positive end-expiratory pressure (PEEP) of 6 cm H2O, and a fraction of inspired oxygen (FiO2) of 0.21. The participants were instructed to report any discomfort during the training, and the respiratory therapist made real-time adjustments to the ventilator to address respiratory issues, as needed. To minimize patient–ventilator asynchrony, a double-limb circuit was used, the inspiratory rise time was set to 50 ms, and the ventilator off-cycling threshold was adjusted to 50% of the peak inspiratory flow rate.24,25 The selection of the head hood (StarMED CaStar-R, Intersurgical, Wokingham, Berkshire, UK) was based on the participant’s neck circumference.
Pulmnoary rehabilitation routines
All the participants underwent pulmonary rehabilitation routines, which encompassed thoracic expansion, airway clearance, and exercise training. In contrast to the ECT and ECT-HV groups, the control group participated in self-paced exercise by using a concentric cycling training (CCT) ergometer, with the autonomy to determine their exercise intensity based on personal preference.
Measurement
The primary focus of this study was to examine the change in VO2peak following the 9-weeks intervention. Secondary outcomes included physiological parameters, spirometry results, hemodynamics data, physical mobility, and functional activity.
Physical mobility and functional activity were evaluated using the timed up and go (TUG) test and the 6-min walk test (6MWT). Throughout the 6MWT, heart rate (HR) and oxygen saturation (SpO2) were continuously recorded using a wrist-worn pulse oximeter (WristOx2, Nonin Medical, Plymouth, MN, USA) equipped with Bluetooth capability. The energy expenditure index (EEI) was calculated by dividing the 6MWT HRmean by the walking speed. In this study, a higher EEI indicated poor walking economy. 26
Lung function was assessed using a spirometer (MicroLab 3500, CareFusion, Kent, San Diego, CA, USA). To monitor transcutaneous partial pressure of carbon dioxide (PtcCO2), a PtcCO2 monitor (TCM4, Radiometer, Medical AsP, Brønshøj, Denmark) equipped with an electrochemical transducer was employed. The measurement site on the upper-left side of the chest was cleaned with an alcohol pad, followed by the application of two to three drops of contact gel before securing the sensor in place to ensure stable and accurate readings. Hemodynamic monitoring was conducted using a noninvasive electrical cardiometry-based system (ICON, Osypka Medical, Berlin, Germany). This system employed an array of four surface electrocardiography electrodes strategically placed on the left side of the neck and the lower thorax, positioned approximately at the level of the xiphoid process. These data were collected on a weekly basis from weeks 2 to 9.
The participants’ self-reported scores for modified Borg dyspnea (Borg-D) and leg fatigue (Borg-L) scale ranging from 0 to 10, where 0 indicated “none” and 10 denoted “the worst.” For Borg-D at rest, modified-Medical Research Council (mMRC), 27 and COPD assessment test (CAT), 28 participants were requested to evaluate their dyspnea at rest both before and after the 9-weeks intervention. For Borg-D and Borg-L assessments, the participants were instructed to rate their dyspnea and leg fatigue immediately after each ECT session.
Statistical analysis
Statistical analysis was conducted using SPSS version 25.0 (IBM, Chicago, IL, USA), with a significance level set at p < .05. The normality of the distribution of the variables under examination was assessed using the Shapiro–Wilk test. All analyses were performed on an intention-to-treat basis. Categorical variables were assessed using the chi-squared test and are presented as frequencies and percentages. Continuous variables are presented as means ± standard deviations. For intragroup comparisons, the paired t test was applied to variables with a parametric distribution. Intergroup comparisons were conducted using one-way analysis of variance, followed by Tukey’s post hoc analysis for individual variances to correct for multiple comparison tests. To further investigate and compare the effects of the two alternative training approaches (ECT and ECT-HV) over 9 weeks. To further investigate and compare the effects of ECT and ECT-HV on PtcCO2, Borg-D, and Borg-L from week 2 to week 9, we employed a generalized estimating equation approach to account for the within-subject correlation of repeated measures, accommodate unbalanced data, and provide unbiased population-averaged estimates.
Results
Baseline characteristics of study participants before enrollment.

Data are presented as means ± SDs or numbers and percentages. BMI: body mass index; FEV1: forced expiratory volume in the first second; FVC: forced vital capacity; FRV: functional residual volume; RV: residual volume; TLC: total lung capacity; GOLD: Global Initiative for Chronic Obstructive Lung Disease; HR: heart rate; SpO2: peripheral oxygen saturation; sBP: systolic blood pressure; dBP: diastolic blood pressure; MAP: mean arterial pressure.
Cardiopulmonary responses to the graded exercise test and secondary outcomes.

Data are presented as means ± SDs.
aData are derived from the 6-min walk test.
VO2: oxygen consumption; OUES: oxygen uptake efficiency slope; VE: minute volume; VCO2: carbon dioxide production; HR: heart rate; 6MWD: 6-min walk distance; EEI: energy expenditure index; TUG: Time Up and Go; FEV1: forced expiratory volume in the first second; FVC: forced vital capacity; Borg-D: Borg dyspnea; mMRC: modified-Medical Research Council; CAT: chronic obstructive pulmonary disease assessment test; SV: stroke volume; CO: cardiac output; TFC: thoracic fluid content; ICON: index of contractility; FTC: correct flow time; SVV: stroke volume variation; SVR: systemic vascular resistance; SVRI: systemic vascular resistance index; STR: systolic time ratio; PEP: pre-ejection period; LVET: left ventricular ejection time.
*p < .05, **p < .01, for one-way ANOVA conducted postintervention among the three groups.
†p < .05, ††p < .01, †††p < .001 for intragroup analysis (preintervention vs postintervention).
Following the 9-weeks intervention, both the exercise training groups exhibited significant improvements in the 6-min walk distance (6MWD) and EEI (p < .05 and p < .01; Table 2). In intragroup comparisons, the changes in 6MWD and EEI on the 6MWT at the end of the 9-week period were clinically significant in the ECT (6MWD: 64.2 ± 58.3 m; mean change: 16.3%, p < .01; EEI: −0.19 ± 0.21 beat/m walked; mean change: −12.7%, p < .01) and the ECT-HV groups (6MWD: 69.3 ± 57.7 m; mean change: 19.2%, p < .01; EEI: −0.22 ± 0.14 beat/m walked, mean change: −14% p < .001) compared with the initial results (Figure 2). However, no significant difference in the improvement of 6MWD and EEI was observed between the ECT and ECT-HV groups. The control group exhibited no change in the 6MWT after the 9-weeks intervention (Figure 2). Percentage change variation over the 9-weeks intervention period in 6-mintue walk distance (6MWD), energy expenditure index (EEI), and timed up and go (TUG) test duration among the control, eccentric cycling training (ECT), and ECT-helmet ventilation (ECT-HV) groups. *p < .05, **p < .01, ***p < .001.
At the end of the 9-week period, a significant reduction in the TUG test duration was noted in the ECT and ECT-HV groups (p < .05; Table 2). Significant improvements in TUG variation were observed in the ECT (−1.71 ± 1.11 s; mean change: −16.7%, p < .001) and ECT-HV groups (−1.9 ± 1.58 s; mean change: −17.3%, p < .01; Supplementary Table S1), with significantly greater reductions in the TUG in both the groups compared with the control group (p < .01 and .001, respectively). However, no significant difference was observed in the improvement in 6MWD and EEI between the ECT and ECT-HV groups. The control group exhibited no change in the 6MWT after the 9-weeks intervention (Figure 2).
A significant difference in PtcCO2 was observed between the ECT and ECT-HV groups at week 5 (54.4 ± 10.9 vs 69.1 ± 24.8 mmHg) and week 7 (55.3 ± 17.8 vs 69.5 ± 18.3 mmHg), with p < .05 for both comparisons. No significant differences were found at other time points throughout the training phase. Similarly, Borg-D scores showed significant between-group differences at week 4 (1.6 ± 1.5 vs 2.1 ± 1.3) and week 6 (1.4 ± 1.4 vs 1.8 ± 1.4) (p < .05 for both). In addition, the ECT-HV group exhibited a notably higher Borg-D score at week 2 compared to subsequent weeks. All longitudinal comparisons were corrected for multiple testing (Figure 3). A Although statistical differences were identified, the observed elevations in PtcCO2 in the ECT-HV group remained within physiologically acceptable ranges, and the magnitude of change was not considered clinically meaningful. Changes in transcutaneous partial pressure of carbon dioxide (PtcCO2), Borg dyspnea (Borg-D) scores, and Borg leg fatigue (Borg-L) scores from weeks 2 to 9 (training phase) in the eccentric cycling training (ECT), and ECT-helmet ventilation (ECT-HV) groups. *p < .05 for the interaction between groups. Changes between time points were analyzed using GEE. †p < .05, ††p < .01, †††p < .001 for the different time points in the ECT group versus week 2. ‡p < .05, ‡‡p < .01, ‡‡‡p < .001 for the different time points in the ECT-HV group versus week 2. #p < .05 for ECT versus ECT-HV groups at the same week.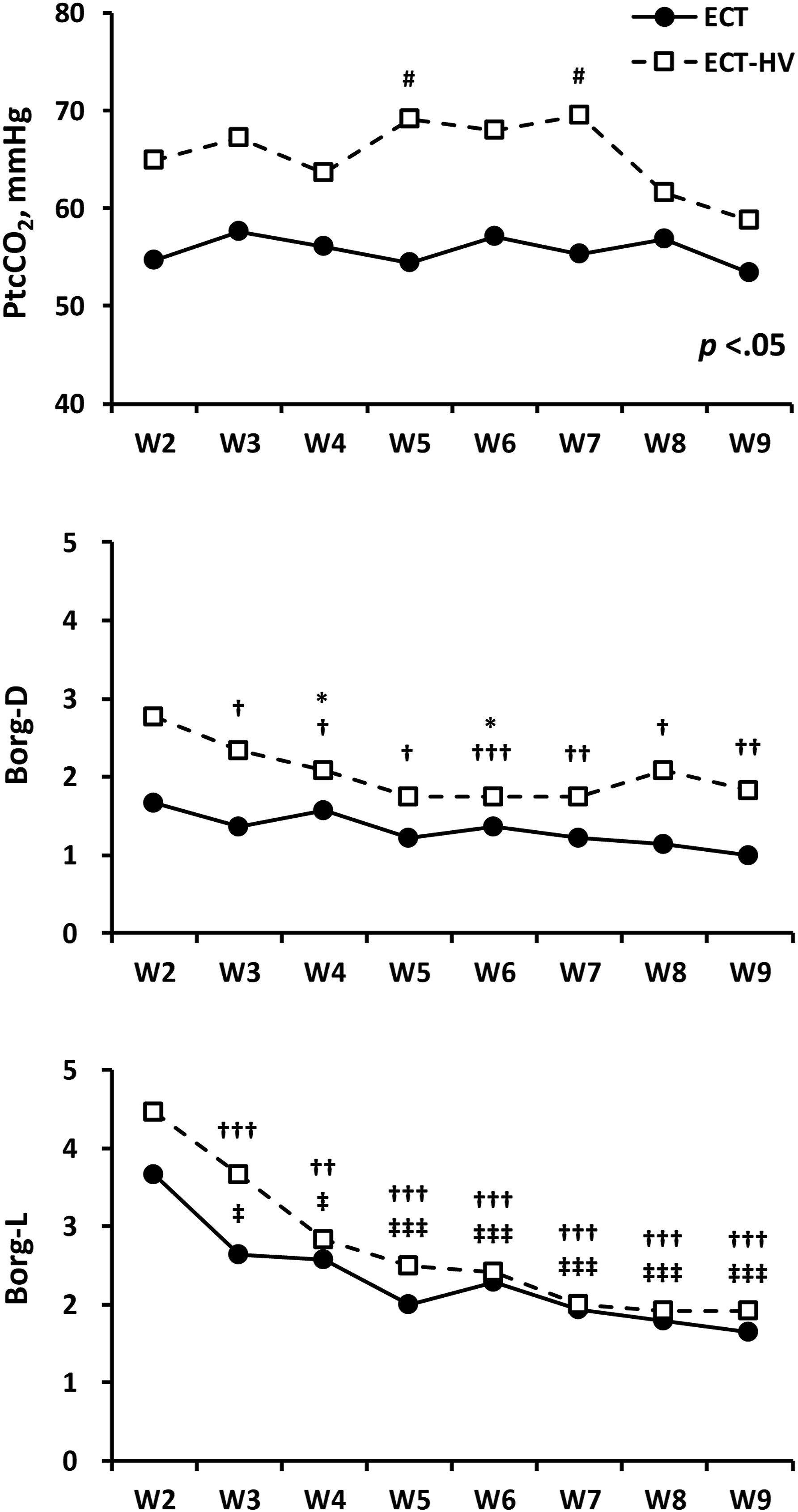
Ventilation parameters during eccentric cycling training with helmet ventilation.
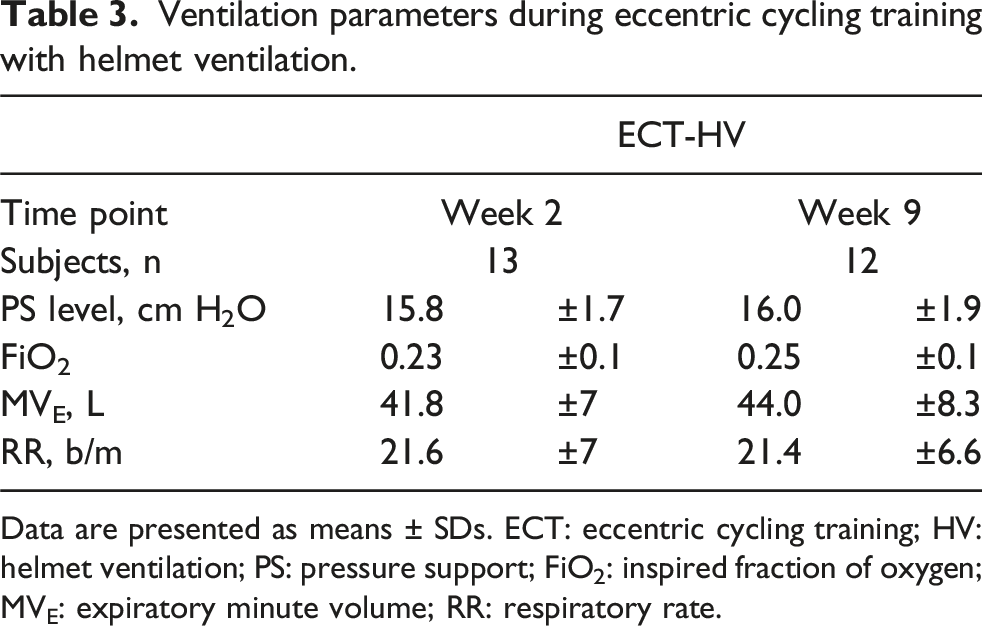
Data are presented as means ± SDs. ECT: eccentric cycling training; HV: helmet ventilation; PS: pressure support; FiO2: inspired fraction of oxygen; MVE: expiratory minute volume; RR: respiratory rate.
Discussion
Despite no significant changes in maximal cardiopulmonary exercise performance, our study demonstrated that a 9-weeks ECT program significantly enhanced physical performance, functional activity, and mobility in patients with chronic respiratory diseases, regardless of HV use. VO2peak was selected as the primary outcome because it is widely recognized as the gold standard for evaluating maximal aerobic capacity. It provides a comprehensive assessment of cardiovascular, pulmonary, and muscular function during graded exercise testing. This measure has been extensively used in cardiopulmonary rehabilitation studies as a reliable indicator of central and peripheral adaptations resulting from exercise training. Although VO2peak did not demonstrate a statistically significant improvement in our study, other physical performance indicators such as the 6MWD showed clinically meaningful gains, with statistical power reaching 0.8. However, the improvement in functional performance should be interpreted with caution, given the potential effects of supervision and lack of blinding.
Studies have emphasized that ECT induces lower cardiopulmonary responses than traditional concentric exercise, both in healthy individuals and those with heart failure.29,30 Moreover, evidence supporting the safety and feasibility of ECT in patients with chronic respiratory disease is available.31,32 Specifically, in patients with severe COPD, ECT provided greater improvements in lower limb muscular strength compared with CCT, with lower sensations of dyspnea and leg fatigue during exercise at equivalent relative heart intensities. 31 However, CCT is associated with increased exertional cardiac power output compared with ECT. 33
Inostroza et al. compared ECT with CCT in patients with moderate COPD, demonstrating that ECT elicited significantly higher workloads, lower heart rate and dyspnea, and yielded superior improvements in functional performance, including the 6MWT and TUG. 34 Rodríguez and Rodríguez-Jaime published two systematic reviews on eccentric exercise in patients with COPD.35,36 These studies suggest that eccentric cycling improves physical fitness, quality of life, and functional performance with less cardiorespiratory strain than concentric modalities. Notably, improvements were observed in isometric and isokinetic quadriceps strength and 6MWT performance, while aerobic capacity showed no consistent changes, which aligns with our results. Despite encouraging outcomes, limited studies and protocol variability hinder the development of standardized guidelines. Further high-quality trials are needed to confirm efficacy and inform clinical practice.
Our recent study in sedentary healthy men without cardiopulmonary symptoms showed that CCT led to greater improvements in cardiac output, VO2, and work rate at peak performance than ECT, likely due to the higher cardiopulmonary demand associated with CCT. Nevertheless, both CCT and ECT effectively improved aerobic efficiency by enhancing muscular hemodynamic responses and erythrocyte rheology, as well as increasing antioxidant capacity against oxidative stress from intense exercise. 37 These findings suggest that eccentric cycling primarily induces peripheral rather than central adaptations. The improvement in 6MWD and TUG, despite unchanged VO2peak, may therefore reflect enhanced muscular efficiency, neuromuscular coordination, and movement economy, rather than a true increase in maximal aerobic capacity. In chronic respiratory disease, peripheral muscle dysfunction often limits functional performance before maximal cardiopulmonary capacity is reached; thus, training-induced gains in muscular strength and endurance can translate into better mobility and submaximal exercise tolerance even without significant VO2peak elevation. Therefore, ECT presents as a promising exercise modality for patients with chronic respiratory disease who often experience exercise intolerance due to muscular weakness and dyspnea, particularly among elderly individuals with cardiopulmonary symptoms.30,31
An interesting finding in this study is the decrease in work rate at peak performance in the control group. The key distinction between the ECT and ECT-HV groups compared with the control group lies in the approach to exercise training. Studies have suggested that supervised exercise programs are associated with higher aerobic capacity and greater exercise tolerance than unsupervised programs. 38 In this context, the ECT and ECT-HV groups received supervised exercise, whereas the control group followed a self-paced program, which can be considered unsupervised. This difference in supervision may contribute to the lower work rate observed in the control group.
In this study, the ECT group exhibited a mean change in 6WMD of 64.2 m from baseline, whereas the ECT-HV group exhibited a change of 69.3 m, both surpassing the minimum clinically important difference (MCID) of 25–35 m. 39 Additionally, the mean change in the TUG test duration from baseline was −1.71 s in the ECT group and −1.9 s in the ECT-HV group, exceeding the MCID of 0.9–1.4 s. 40
To the best of our knowledge, this study is the first investigation into the incorporation of HV into an exercise training program. Consistent with our previous findings that revealed no improvement in short-term self-paced exercise performance among patients with stable mild-to-moderate COPD, 41 HV did not yield additional benefits for exercise training in patients with chronic respiratory disease or mild-to-moderate COPD in this study. HV is characterized by features such as a large internal volume and high compliance of the polyvinyl chloride hood, potentially leading to CO2 rebreathing and patient–ventilator asynchrony, particularly with delayed triggering. 42 Moreover, due to these characteristics, the exhaled tidal volume (VT) cannot be reliably measured during HV. 21 An in vitro study demonstrated a dead space (VD) of up to 10 L during HV, indicating that almost half of the VT consists of VD. 43 During exercise, patients with COPD experience a limited increase in VE, typically restricted to between 30 and 40 L/min. 44 The baseline average VE at peak performance in our study was 47.9 L/min across all groups. HV was not originally designed for exercise training; thus, in patients with less severe chronic respiratory diseases, HV may not be beneficial due to the higher VE required during exercise training. Further evidence is necessary to determine whether patients with greater disease severity may derive benefits from HV.
Consistent with our earlier observations, no participant in our study reported claustrophobia or dropped out due to interface-related issues. 41 However, a potential concern was the risk of CO2 retention associated with helmet ventilation. In our findings, the ECT-HV group demonstrated a tendency toward higher transcutaneous CO2 levels compared to the ECT group. Specifically, from week 2 to week 9, PtcCO2 values in the ECT-HV group ranged from 59 to 70 mmHg, while those in the ECT group ranged from 53 to 58 mmHg. Although this difference did not reach statistical significance, it may suggest reduced CO2 clearance or rebreathing during helmet use, warranting further investigation into the clinical relevance of these elevations. This may also explain the higher Borg-D scores observed in the ECT-HV group at the beginning of training. As documented in prior research, individuals commonly experience fatigue in major thigh muscles during the initial stages of training. 45 Nonetheless, this fatigue typically diminishes after several sessions, corroborating our study’s findings.
The study has several limitations that should be acknowledged. First, the small sample size from a single institution may limit the generalizability of the findings to broader populations. Moreover, since most participants demonstrated relatively preserved lung function, suggesting a mild or pre-COPD disease profile, the findings should not be extrapolated to patients with moderate or severe respiratory disease. Second, the implications of maintenance and long-term follow-up remain unclear, necessitating further research. Third, the ventilation settings of the helmet ventilator may not have been optimal for patients with less severe chronic respiratory disease. Moreover, exhaled tidal volume could not be measured during training, and ventilator adjustments relied solely on vital signs, patient-reported dyspnea, and the expertise of respiratory therapists. Fourth, although some studies have reported data within the helmet,46,47 measuring end-tidal CO2 during HV remains challenging, and practical detection methods are lacking. Lastly, as this trial could not be blinded, there is a risk of performance and detection bias.
Conclusions
While combining HV with exercise training showed no added benefits, our study highlights the potential of a 9-weeks ECT program to improve functional activity and mobility in patients with chronic respiratory diseases. Despite no significant improvements in cardiopulmonary responses, ECT was associated with favorable changes in 6MWD and TUG. However, part of these improvements may be attributable to supervised training and the unblinded design. Future studies should incorporate long-term follow-up and investigate biochemical and inflammatory biomarkers to further elucidate the underlying mechanisms.
Supplemental Material
Supplemental Material - Eccentric cycling for chronic respiratory disease: Does helmet ventilation add value? A randomized controlled trial
Supplemental Material for Eccentric cycling for chronic respiratory disease: Does helmet ventilation add value? A randomized controlled trial by Ke-Yun Chao, Wei-Lun Liu, Hui-Wen Hsu, Hsin-Cheng Chen, Ai Yin Lim, Jong-Shyan Wang in Chronic Respiratory Disease
Footnotes
Acknowledgments
This manuscript was edited by Wallace Academic Editing.
Author Contributions
KYC, HCC, and JSW conceived and designed the study. HWH and HCC performed the experimental work and collected the data. KYC, WLL, and AYL contributed to data analysis. KYC, WLL, and JSW wrote the paper. All the authors revised the manuscript for important intellectual content and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received funding from Fu Jen Catholic University Hospital (PL-202108001-T), the National Science and Technology Council, Taiwan (112-2314-B-182-025-MY3), and the Keelung Chang Gung Medical Research Program (CMRPD1N0041). These funders were not involved in the study design; the collection, analysis, and interpretation of data; the writing of the report; or the decision to submit the article for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial registration
ClinicalTrial.gov NCT05760352 (02/27/2023).
IRB number
FJUH111193.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
