Abstract
Objectives
To determine the feasibility and acceptability of home-based pulmonary rehabilitation (HBPR) for individuals with idiopathic pulmonary fibrosis (IPF).
Methods
In this single-arm feasibility trial, individuals with IPF were recruited from Delhi, India, to a 6-weeks HBPR programme using a paper-based manual. Primary outcomes were feasibility (classified by ≥60% of eligible patients recruited and ≥70% of recruited patients completing the follow-up assessment) and intervention acceptability (semi-structured interviews).
Results
Out of 42 screened, 36 individuals were eligible (86% of screened), and 30 were recruited (83% of eligible, 71% of screened; 60 ± 13 years, 53% female), with 25 completing their follow-up assessment (83% of recruited). HBPR was generally well-accepted, with qualitative themes including: ‘facilitators and barriers to HBPR’ (family support and flexibility of home environment were facilitators whereas lack of supervision and inability to follow a routine were barriers), ‘perceived changes from taking part in HBPR’ (improved exercise capacity, breathlessness, and independency), and ‘how to improve HBPR in the future’ (translating the manual into various languages, and incorporating into a more hybrid approach).
Conclusion
HBPR using a paper-based manual was feasible and acceptable, potentially suitable for improving the uptake and completion of PR for individuals with IPF in Delhi, India.
Keywords
Introduction
The global adjusted prevalence of idiopathic pulmonary fibrosis (IPF) is estimated to be between 0.33 and 4.51 cases per 10,000 people. 1 In India, IPF has an estimated national prevalence of 5.8–11.6 per 100,000. 2 IPF is characterised by a progressive decline in lung function, associated worsening of dyspnoea, and has a poor prognosis.3,4 Furthermore, IPF is economically burdensome 5 and is associated with reduced health-related quality of life (HRQoL), especially affecting domains of physical health.6,7
Evidence-based guidelines 8 have provided strong recommendations from moderate quality evidence for the participation of adults with interstitial lung diseases (ILD) in pulmonary rehabilitation (PR) programmes to improve HRQoL and functional capacity, relying on evidence generated in high income countries. These guidelines have identified the need for further research to determine the optimal model of PR in this population (e.g., centre-based, home-based, telerehabilitation). The availability of PR in low-and middle-income countries (LMICs), including India, remains limited 9 and alternative models may be attractive to providers and participants.10,11 Barriers to conventional, centre-based PR in LMICs, such as limited access, lack of infrastructure, family dependency, costs of travel, and household and/or employment responsibilities10,12,13 emphasize the need to develop a home-based PR (HBPR) model to address these barriers, and optimize uptake, compliance, and completion of PR in India.
A preference for the development of a paper-based manual, over an online or digital approach, to facilitate HBPR for individuals with IPF in Delhi, India has previously been identified as many reported not being digitally literate. 10 Studies utilising this paper-based mode of PR for individuals with COPD in the UK (“Self-monitoring Programme of Activity Coping and Education for COPD: SPACEforCOPD©”) have found promising results relating to improved HRQoL, increased exercise capacity, and reduced dyspnoea.14,15
Due to both population and cultural differences between the UK and India, there is a need to develop and test a contextually-appropriate manual for individuals with IPF in Delhi which may improve the uptake to, and completion of, PR in this population. While initial findings in this area are promising, 16 it is important to determine the feasibility and acceptability of PR, which conforms to guidelines on duration, 17 before assessing clinical effectiveness.
This trial aimed to assess the feasibility and acceptability of a culturally-tailored, manual-based HBPR for individuals with IPF in Delhi, India.
Methods
Trial design
A single-arm mixed-methods feasibility trial was conducted. The trial was approved by the ethics review committees of Metro Hospitals & Heart Institute, Noida, India (ref: 62/MERB/2021; 10th February 2022) and University of Leicester, UK (ref: 31989; 13th March 2022), as part of the National Institute for Health and Care Research (NIHR) Global Health Research Group for Pulmonary Rehabilitation (Global RECHARGE) project. 18
This trial was registered with the International Standard Randomized Controlled Trial Number (ref: ISRCTN14831771) and has been written following the Consolidated Standards of Reporting Trials (CONSORT) checklist for feasibility trials (Supplemental material A), 19 the Template for Intervention Description and Replication (TIDieR) checklist (Supplemental material B), 20 and the COnsolidated criteria for REporting Qualitative research (COREQ) checklist (Supplemental material C). 21 All participants provided written informed consent.
Setting
The trial was conducted in the Metro Centre of Respiratory Diseases (MCRD), Noida, Delhi, India. Recruitment occurred between 31st October 2022 to 5th January 2023.
Participants
The inclusion criteria for this trial were adults aged ≥18 years, with a confirmed diagnosis of IPF (according to ATS/ERS guidelines), 22 who were willing to provide informed consent. As the HBPR manual was written in English language, participants had to be able to understand basic English (defined as A1 or A2 level of the Common European Framework of Reference [CEFR] for Languages). 23 Exclusion criteria were adults with comorbidities such as severe or unstable cardiovascular, other chronic conditions or locomotor difficulties that precluded exercise, malignant disease, or other serious illness which interfered with participation in the trial. Those ineligible for the trial were recorded on a trial screening log.
Recruitment
Patients were referred by a pulmonologist at MCRD from outpatient clinics, or identified for the trial from medical records by a physiotherapist. The proposed sample size for the trial was 30. No formal sample size calculation was performed due to the feasibility nature of this trial; however, our proposed sample size was in line with previous feasibility trials.24,25
Intervention
Our 6-weeks HBPR programme utilised a paper-based manual entitled “Self-monitoring Programme of Activity Coping and Education (SPACE) for ILD” (Figure S1) developed by researchers at the University of Leicester and MCRD, Delhi with expertise in pulmonary rehabilitation, and adapted from the ‘SPACEforCOPD©’ manual developed in the UK.14,15 Suggestions reported by individuals with IPF and caregivers regarding HBPR development in Delhi, India were incorporated during the development of this manual. 10 This involved adding sections relating to goal setting, advice for carers, and contextually-appropriate case studies and dietary advice.
The manual contained the core elements of evidence-based PR (i.e., a programme of exercises and health education).26,27 The exercise programme consisted of individually prescribed walking, with diaries provided to monitor adherence and progression (Figure S2). Values achieved during walking tests at baseline along with walking goals (Figure S2) were added to the manual by the healthcare professional and participant at the end of the baseline assessment. Participants were instructed to walk at home around the same speed as during the baseline endurance shuttle walk test (ESWT). Participants were advised to perform resistance exercises of both upper and lower muscle groups (Figure S3) 3 times/week. Exercises were tailored for a low-resource environment by incorporating low-cost or self-made equipment (i.e., water bottles instead of dumbbells).
Self-management skills were promoted through goal-setting strategies, coping planning, and case studies. The manual included an action plan for exacerbation management. The researchers contacted the participants after 1 week of the intervention to determine their adherence and challenges related to HBPR. Following this period, participants were asked to contact healthcare workers (HCW; physiotherapists) if they experienced any further challenges or had any queries. Further details are provided in Supplemental material D.
Data collection and management
Data were entered into the Research Electronic Data Capture (REDCap) web-based database.28,29 REDCap automated checks and 100% manual data checking were completed prior to locking the dataset for analysis. Interview data were collated in Microsoft Excel with quotes from transcripts, lists of codes, subthemes and themes.
Primary outcome: Feasibility
Primary feasibility outcomes were recruitment and completion based on a predetermined traffic light system where green indicated the feasibility of the trial using the set methodology, amber indicated the need for modifications in the methodology, and red indicated the non-feasibility of the trial. The following thresholds were used: (i) recruitment (green ≥60%; amber 25%–59%; red <25% of eligible patients who were recruited) and (ii) completion (green ≥70%; amber 50%–69%; red <50% of those recruited who attended the discharge assessment).
Acceptability
The acceptability of the intervention among participants was assessed by semi-structured interviews (interview schedule provided in Supplemental material E). Interviews were conducted by a physiotherapist (HH; female; MPT) with previous experience in qualitative data collection and analysis. Further support was provided by researchers at University of Leicester, UK. The purpose of the interview was explained to participants i.e., to understand their experiences of HBPR. Interviews were conducted face-to-face at MCRD or via telephone if in-person visits were not possible, and varied between English, Hindi, or combination of the two languages based on participant preference. No relationship between interviewer and participants were established prior to trial commencement. Field notes were taken during the interviews.
All interviews were audio recorded, transcribed verbatim and then translated into English to facilitate the analysis process. Translated quotes were checked against original transcripts by the interviewer (HH) to ensure meaning was maintained.
Secondary outcomes
Data were collected in-person at MCRD and in accordance with the recommended minimum dataset for trials of PR in LMIC. 18 Details of outcome measures and minimal clinically important differences (MCIDs) can be found in Supplemental material F. In brief, these included HRQoL (EuroQol Five Dimensions Five Levels [EQ-5D-5 L] 30 and King’s Brief Interstitial Lung Disease [KBILD]), 31 symptoms, (Medical Research Council [MRC], 32 COPD Assessment Test [CAT], 33 and Clinical COPD Questionnaire [CCQ]), 34 psychological wellbeing (Hospital Anxiety and Depression scale [HADS], 35 and physical capacity (Incremental and Endurance Shuttle Walk Tests [ISWT; ESWT],36,37 and 5x sit-to-stand). 38
Data analysis
Quantitative analysis
Data for baseline and follow-up time points were presented as descriptive statistics. Changes in secondary outcomes were benchmarked against previously published MCIDs (Supplemental material F).
Qualitative analysis
Template thematic analysis 39 via the codebook approach was used to analyse qualitative data. Theories were generated via grounded theory and inductive reasoning. A six-phase process was conducted. 40 (i) “Data familiarisation”: transcripts examined in detail by three researchers (HH, OA, JM); (ii) “Preliminary coding”: codes generated through an inductive approach and recorded in a codebook by one researcher (HH), (iii) “Generating initial themes”: patterns and relationship within and across the codes were discussed with three researchers (MO, JM, AB) and one researcher (HH) developed initial themes; (iv) “Developing and reviewing themes”: initial themes were discussed by four researchers (HH, AB, JM, MO) to ensure they told the story of the data; (v) “Refining, defining, and naming themes”: a detailed analysis of each theme was performed by four researchers (HH, AB, JM, MO) to decide on informative theme names and descriptions; (vi) “Writing up”: generated themes and illustrative quotes were contextualized, compared and contrasted to existing literature and the local context by one researcher (HH), with support from all authors.
Results
Feasibility: Participant screening and recruitment
Overall, 42 patients were screened of which 36 (86%) were eligible and invited to a baseline assessment (Figure 1). Reasons for ineligibility included contraindications for the walking tests (n = 5) and being unable to read English (n = 1). 30 participants (83% of those eligible) were recruited (Figure 1), with the main reason for declining participation being personal commitments (n = 3). CONSORT diagram of the flow of participants through the trial.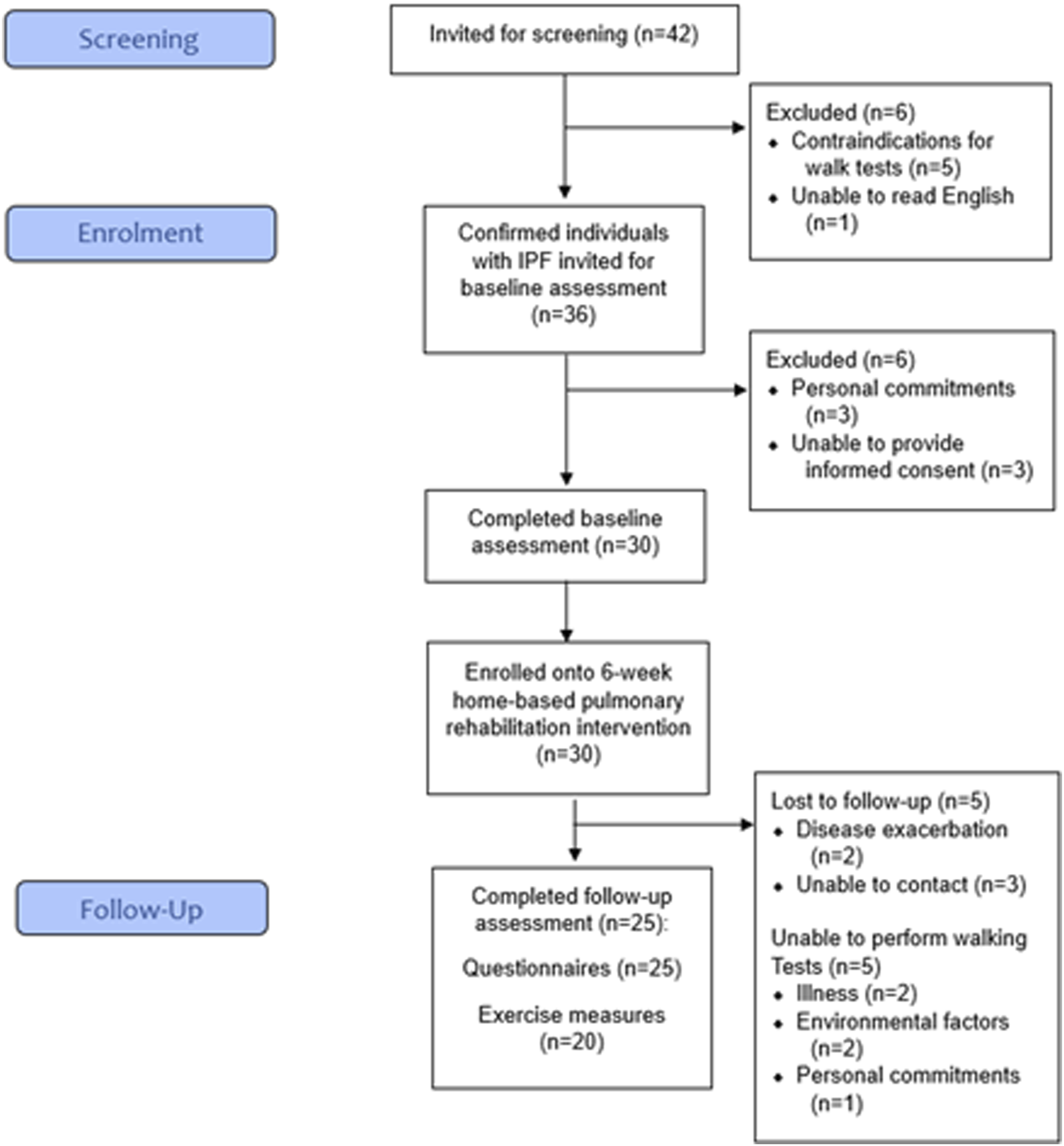
Baseline characteristics
Baseline participant characteristics.

Note: Data presented as mean (SD) unless otherwise stated.
Feasibility: Participant completion
Out of the 30 recruited participants, 25 participants (83%) completed their follow-up assessment (Figure 1). Five participants (17%) withdrew due to disease exacerbation (n = 2, 7%) or being uncontactable (n = 3, 10%). Of the 25 completers, five participants (20%) completed their follow-up visit by telephone (rather than face-to-face) due to illness (n = 2, 8%), environmental factors (n = 2, 8%) or personal commitments (n = 1, 4%). The physical measures (i.e., ISWT, ESWT, and 5x sit-to-stand) at follow-up were therefore missing for these participants.
Acceptability
Qualitative themes and illustrative quotes.

Theme 1 - Facilitators and barriers to HBPR
The main advantage of HBPR described by participants was that it overcame the barriers related to centre-based PR. There were no time restrictions to do exercise at home, and reduced the stress burden of travel. Participants described family members actively supporting their exercise programme by exercising together, reading instructions from the manual, and correcting them as needed.
Some individuals faced challenges in completing the HBPR programme. Due to certain domestic circumstances, such as guest arrival or household work, it was difficult to find the time and follow a strict routine at home. Time flexibility and lack of supervision were among the reasons for reduced discipline and punctuality; therefore, some participants could not adhere to the program as intended.
Theme 2 - Perceived changes from taking part in HBPR
Participants were satisfied with the increase in walking duration and exercise repetitions, and reported improvements in their functional abilities after completing the program. Furthermore, participants felt that their clinical symptoms were reduced and their independency in daily activities, such as washing, dressing, walking, and travelling had been enhanced, which improved their mental wellbeing and mood.
Belief in the value of exercise and self-management for their IPF was reported, with some participants wanting to recommend this intervention to other individuals with the condition.
Theme 3 - How to improve HBPR in the future
Some participants felt that with minor alterations the HBPR programme would be suitable for a broader patient group. For example, translating the manual into the national language (Hindi). A hybrid model of both centre-based and HBPR was also recommended, especially for those who require a greater level of supervision to build self-confidence, understand exercise techniques, and follow an exercise routine.
Secondary outcomes
Pre- and post-intervention values, and change scores for secondary outcomes.
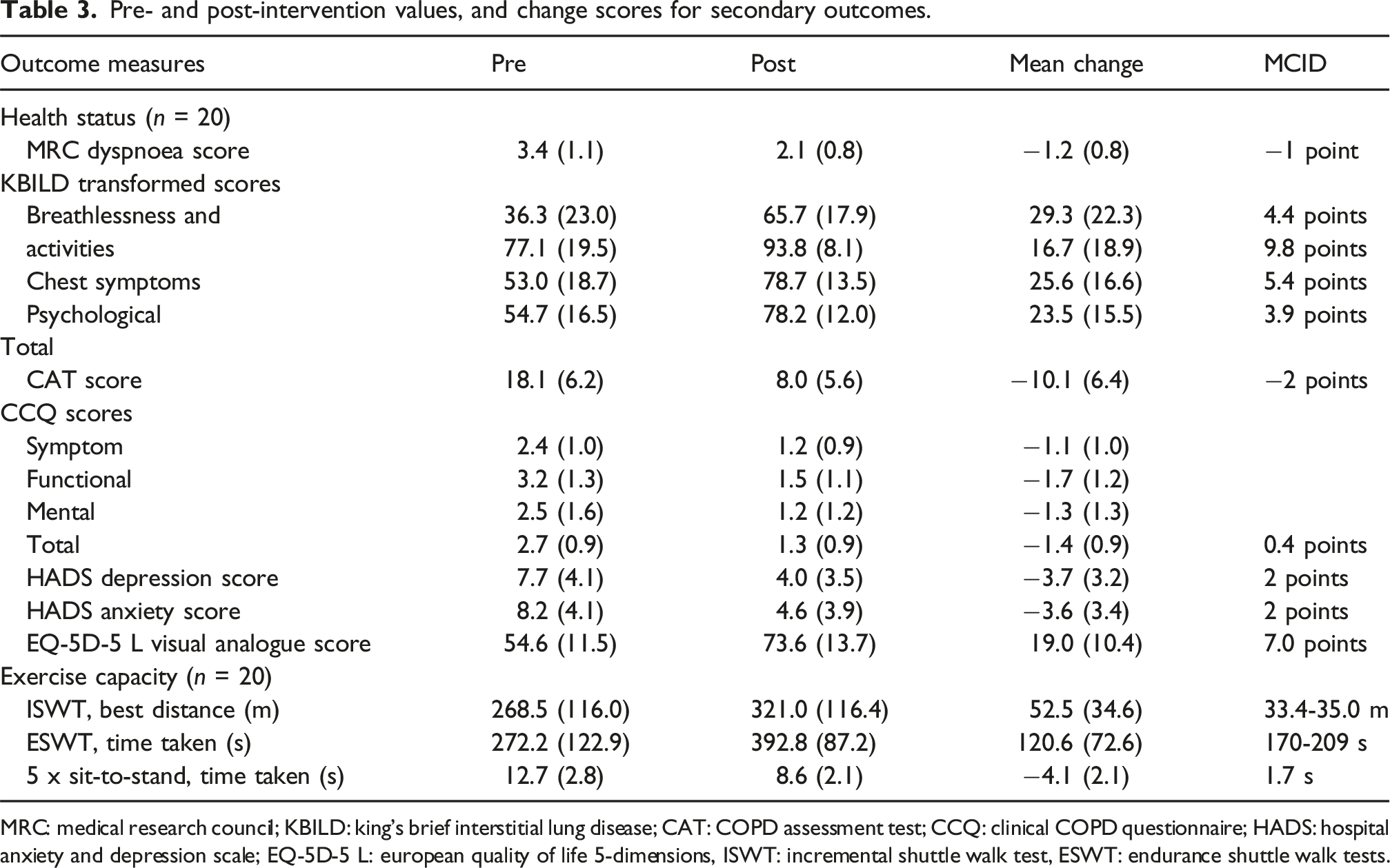
MRC: medical research council; KBILD: king’s brief interstitial lung disease; CAT: COPD assessment test; CCQ: clinical COPD questionnaire; HADS: hospital anxiety and depression scale; EQ-5D-5 L: european quality of life 5-dimensions, ISWT: incremental shuttle walk test, ESWT: endurance shuttle walk tests.
Discussion
Summary of the main findings
The aim of this trial was to assess the feasibility and acceptability of HBPR, via a culturally-tailored manual, among individuals with IPF in Delhi, India. The recruitment to and follow-up of the trial exceeded the criteria of feasibility, indicating that it is feasible to deliver the intervention. HBPR was acceptable to the majority of participants, suggesting that this is an appropriate mode of PR in this population and setting. Compliant participants reported perceived benefits, and those who were unable to fully adhere suggested improvements to the programme, including regular follow-ups from healthcare workers. Secondary outcome measures including exercise capacity, HRQoL, and mood showed potentially clinically meaningful improvements, aligning with the views of participants during interviews.
Interpretation of results
The 83% uptake of individuals with ILD observed in this trial was higher than those observed for traditional centre-based PR (∼63% of eligible) in this context and population. 41 A trial investigating the feasibility of HBPR in Malaysia reported a higher uptake rate (93%) but a lower completion rate (37%) due to COVID-19 restrictions. 42 The 6-week completion rate of 83% for our ‘SPACEforILD’ intervention mirrors the original 80% completion rate of ‘SPACEforCOPD’ trial in the UK, 15 and is greater than the completion rate reported in the UK national PR audit for COPD (67%). Significantly higher completion rates following HBPR compared to centre-based PR have also been reported in Australia. 43 The reported facilitators to our HBPR intervention, specifically low costs, minimal burden of travel, more flexibility regarding when to perform exercises, and family support, are consistent with literature from both high-44–46 and LMICs.10,13,42,47 These noted advantages of HBPR may help to overcome the barriers associated with more traditional, centre-based PR in LMICs. 48 The findings outlined above emphasize the benefit of having a menu of PR options to facilitate a patient choice approach to enhance the uptake and completion of PR in India.
Many of the female participants in this trial were not in work and identified as full-time homemakers, reflecting cultural norms in India, where women are primarily responsible for household duties. Such gendered expectations can limit women’s autonomy and confidence, often making them hesitant to travel alone, even for essential healthcare services like centre-based PR. Our HBPR intervention enabled women to care for their health without neglecting domestic responsibilities, which likely enhanced both its acceptability and adherence among these participants.
Participants in the present trial were provided with contact details for further support if needed, after the initial follow-up call. However, individuals reported the lack of direct supervision as the main barrier towards adhering to the programme. A previous trial investigating the acceptability of HBPR for individuals with COPD in Australia incorporated a home visit before weekly telephone calls to provide support, motivation, and education to participants. 46 This was reported to be a facilitator to the predominantly unsupervised programme for the majority of individuals, with one participant suggesting the need for more direct supervision. 46
Albeit not powered to detect significant improvements, we found promising changes in clinical outcomes, including dyspnoea, knowledge of ILD, anxiety, depression, exercise capacity, and lower limb physical function. A randomised controlled trial (RCT) assessing the effect of the ‘SPACEforCOPD’ manual found significant improvements in dyspnoea, 15 exercise capacity, and anxiety compared to usual care following the 6-weeks intervention comprising exercise and self-management strategies. Furthermore, ‘SPACEforCOPD’ is a cost-effective intervention within the UK-based healthcare system. 49 In India, centre-based PR for individuals with ILD has been shown to improve HRQoL and exercise capacity.41,50,51 A recent trial conducted in Manipal, India found similar improvements in these outcomes following a 4-weeks HBPR intervention. 16 Together, these findings demonstrate the potential for exercise and improved self-management through HBPR to increase PR provision whilst maintaining a high quality of service and patient care in India.
Strengths and limitations
This trial utilised a qualitative and quantitative approach to assess the feasibility and acceptability of a contextually adapted version of the evidence-based ‘SPACEforCOPD’ manual for individuals living with IPF in Delhi, India. The development of our manual involved individuals with IPF who provided important modifications and suggestions regarding cultural adaptations. The qualitative aspect of this trial allowed us to determine the acceptability of the intervention, and identify various facilitators, barriers, and future improvements to inform/improve future HBPR trials.
This was a single-centre trial conducted in Delhi, and therefore, findings are unlikely to be generalisable to India as a whole due to its large population and various cultures across different regions. According to the India Human Development Survey (IHDS), about 20% of Indian adults have some ability to speak English, with 4% reporting that they are fluent, while another 16% report having limited conversational skills. 52 English language access is also associated with urbanization, and India has a low urban ratio to rural settings, 53 thus, Hindi as the national language, is the most widely spoken across the country. Our trial participants were an affluent and highly educated population with the majority able to read and understand English but as well as not being in work. The reported suggestion for a more hybrid approach may reflect their employment status (70% not in employment) allowing them greater possibility to attend supervised centre-based PR sessions. The ‘SPACEforILD’ manual will be translated into Hindi and other local languages to facilitate implementation and testing in other areas and populations across India.
Due to the single-arm design of the trial, the interpretation of clinical results is limited, and details regarding exercise adherence were gathered subjectively, via interviews, only. For the original ‘SPACEforCOPD’ manual, between-group changes were only maintained for the ESWT at 6-months follow-up. 15 The long-term follow-up of ‘SPACEforILD’ remains unclear, emphasizing the need for a fully-powered RCT in this population.
Future recommendations
The finding that contextually-adapted HBPR via the ‘SPACEforILD’ manual is both feasible and acceptable from this trial justifies the need for a fully-powered RCT to assess the clinical- and cost-effectiveness of this intervention. Future work will need to address the lack of validated outcome measures in this population. Qualitative findings from the present trial emphasize the importance of implementing ‘SPACEforILD’ within a ‘patient choice’ model of PR, e.g., alongside secondary care and community options. Furthermore, there is a need to develop this manual in multiple languages to reach a broader range of individuals across India and beyond, into other LMICs and settings where digital PR options may not be suitable or easily accessible. Future work should aim to assess the feasibility of manual/HBPR for those with other chronic respiratory diseases, e.g., post-tuberculosis lung disease, and those with multiple long-term conditions.
Conclusion
Culturally-tailored, manual-based HBPR is feasible and acceptable in individuals with IPF in Delhi, India. Facilitators included less time restrictions to exercise, reduced need to travel, and family support. The main barrier reported was the lack of supervision resulting in reduced adherence, and recommendations suggested by participants included the need to translate the manual into a range of languages, and to implement it into a more hybrid approach. A fully-powered RCT is needed to assess the clinical- and cost-effectiveness of this intervention.
Supplemental Material
Supplemental Material - Feasibility and acceptability of home-based pulmonary rehabilitation for individuals with idiopathic pulmonary fibrosis in Delhi, India
Supplemental Material for Feasibility and acceptability of home-based pulmonary rehabilitation for individuals with idiopathic pulmonary fibrosis in Delhi, India by Humaira Hanif, Obaidullah Ahmed, James Manifield, Mohd Shibli, Amy Barradell, Zahira Ahmed, Dominic Malcolm, Andy Barton, Deepak Talwar, Mark W Orme and Sally J Singh in Chronic Respiratory Disease
Footnotes
Acknowledgments
For the purpose of open access, the author has applied a Creative Commons Attribution license (CC BY) to any Author Accepted Manuscript version arising from this submission. Professor Singh is a National Institute for Health and Care Research (NIHR) Senior Investigator.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Institute for Health and Care Research (NIHR) Global Health Research programme GHR 17/63/20 using UK aid from the UK Government to support global health research. The views expressed in this publication are those of the author(s) and not necessarily those of the NIHR or the UK government.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
