Abstract
Objective:
Pulmonary rehabilitation (PR) improves function, reduces symptoms and decreases healthcare usage in people with chronic obstructive pulmonary disease (COPD) following an acute exacerbation (AECOPD). However, rehabilitation uptake rates are low. This study aimed to address barriers to uptake and completion of PR following AECOPD.
Methods:
An action research approach was used to reflect on study feasibility, and to plan and implement an improved protocol. Phase I tested the feasibility of home-based PR started early after AECOPD. Phase II used qualitative interviews to identified potential barriers to program uptake. Phase III re-tested the program with changes to recruitment and assessment strategies.
Results:
Phase I: From 97 screened patients, 26 were eligible and 10 (38%) started home-based PR. Eight participants undertook ≥70% of PR sessions, achieving clinically meaningful improvement in 6-minute walk distance (mean (SD) change 76 (60) m) and chronic respiratory disease questionnaire total score (15 (21) units). Phase II: Potential barriers to uptake of home-based PR included access issues, confidence to exercise, and lack of information about PR benefits. Phase III: From 77 screened patients, 23 were eligible and 5 (22%) started the program.
Discussion:
Home-based PR improved clinical outcomes, but program eligibility and uptake remain challenging. Efforts should be made to ensure PR program eligibility criteria are broad enough to accommodate patient needs, and new ways of engaging patients are needed to improve PR uptake after AECOPD.
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive disease that is associated with reduced functional capacity, poor health-related quality of life and high mortality. 1 Acute exacerbations of COPD (AECOPD) contribute to disease progression and increasing symptoms. 2 AECOPD are commonly defined as a worsening of baseline symptoms 1 and are one of the leading causes of hospital admission and death in people with COPD. 3 This highlights the importance of identifying and providing interventions that reduce the negative consequences of exacerbations. 3,4
Robust evidence supports PR leading to improved exercise capacity and quality of life, and reduced symptoms and health care utilisation in people with stable COPD. 5 Additionally, a systematic review reported that PR started early after an AECOPD could decrease hospital admissions and mortality. 6 However participation rates for PR programs after AECOPD are exceedingly low. 7 In the United Kingdom, less than 10% of all people discharged from hospital following AECOPD complete an outpatient PR program. 8 Moreover, a multicentre study analysing PR participation in the US indicates less than 3% of people hospitalised with AECOPD received PR in the year following exacerbation, with fewer than 2% receiving PR within 6 months after hospitalisation. 7 Even fewer people (0.3%) commenced PR within the first month after hospital discharge. 7
Puhan et al. 6 undertook a systematic review of PR for people after AECOPD, with the programs included in the review varying according to length, location (inpatient or outpatient), timing of program commencement (before or after discharge), and degree of supervision. 6 While there were benefits of PR for the population, particularly with respect to risk of hospital readmission, program uptake rates were low. 6 Alternative PR models have been proposed to enhance program uptake in people with stable COPD. 9 The home-based model of PR is one such alternative, demonstrated to be safe and to deliver similar benefits to outpatient, centre-based, PR in people with stable COPD. 10 A nested qualitative study reported that participants felt well supported and the home-based program could fit in with their daily lives. 11 However, whether the same home-based model of PR, delivered early following hospitalisation for AECOPD is feasible and acceptable is unknown. The present study employed an action research design 12 to enable continuous learning regarding feasibility of home-based PR for people following AECOPD.
Methods
An action research methodology was chosen for this study because it allows improvements to be made in partnership with stakeholders, through cycles of planning, acting, observing and reflecting. 12 The cyclic approach, alternating between action and reflection, allows a greater understanding to be achieved by continuous refinement of methods. Three of the authors are health professionals at the study site (JB, MC, AEH) and were involved in design, data collection and reflection; the study arose because uptake of centre-based PR following a COPD exacerbation was acknowledged to be very low at our institution and had not been altered by previous improvement projects. Several of the authors had developed a home PR model for people with stable COPD 10 which had been adopted into practice at our institution, but this had not been trialled for patients following an exacerbation. This study builds on previous work using action research methods to build a home rehabilitation model in partnership with health professionals. 13 We considered both patients and health professionals to be stakeholders in this work, as the known barriers and facilitators to PR uptake impact on these groups in different ways. 14
This study comprised three phases (Figure 1): I) pilot study testing feasibility of the home-based PR program early following AECOPD; II) qualitative study to understand issues of feasibility from Phase I to support changes to the study protocol under investigation; and III) re-pilot study for feasibility and acceptance of the modified protocol. All phases included engagement with stakeholders, clinical reflection on the problem, planning, action (intervention or qualitative interviews) and evaluation, followed by further reflection. The study was approved by the Human Research Ethics Committees of Alfred Health (475/15), and written informed consent was obtained from all participants.
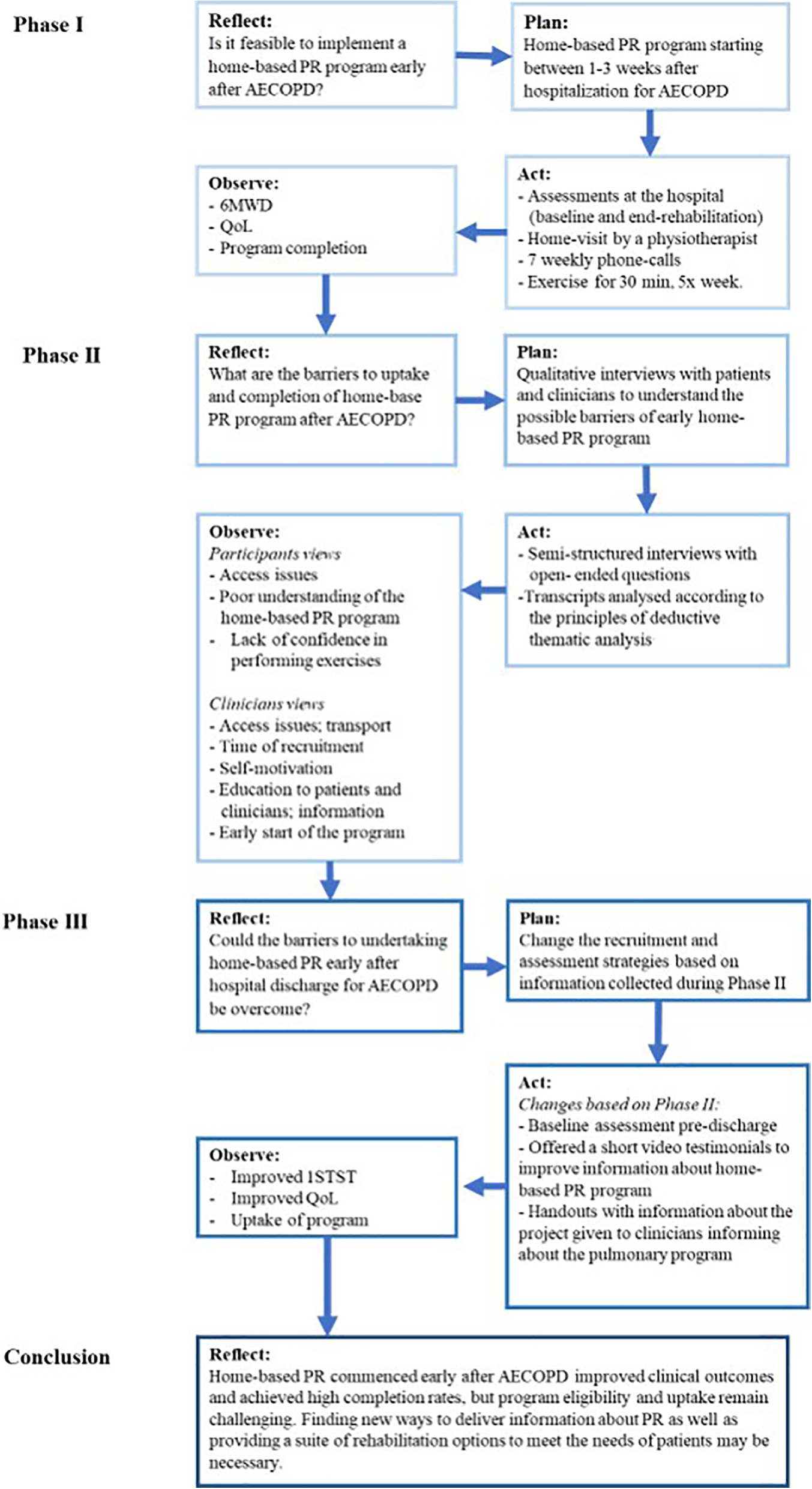
Flow-chart of the action research design including the clinical reflection, plan, action and evaluation of each phase of the action research. 12 PR: pulmonary rehabilitation; AECOPD: acute exacerbation of chronic obstructive respiratory disease; min: minutes; 6MWD: 6-minute walking distance; QoL: quality of life; 1STS: 1-minute sit-to-stand test.
Phase I – Feasibility study
Participants
Individuals with a confirmed diagnosis of COPD, smoking history of at least 10 pack years and aged over 40 years who were admitted to the respiratory or general medicine wards at the Alfred Hospital (Melbourne, Australia) for an AECOPD were eligible to be included. Participants were excluded if they had comorbidities which might prevent them from safely undertaking a home-based exercise program (e.g. balance deficits, cerebral or lower limb palsies, musculoskeletal impairment or cardiac conditions that would prevent independent exercise training), had attended a PR program in the previous 12 months, or could not consent to the study (cognitive impairment or non-English speaker).
Methods
The timing for approaching patients and the provision of information about home-based PR were not standardised. Although all patients were approached while on the ward, some participants approached on the second day of admission while others were approached close to discharge. The researcher explained the project, answered any questions about PR or the study, and provided information and consent forms to patients to read and decide if they would consent to participating. Individuals who consented to participate had a baseline assessment booked for the week after hospital discharge in the physiotherapy outpatient clinic (Alfred Hospital). After baseline assessment a home-visit was scheduled, to initiate the rehabilitation program. At the end of the program participants returned to the outpatient clinic for a final assessment.
Intervention
Participants undertook an early home-based PR program that commenced within 3 weeks of hospital discharge. The home-based rehabilitation program was individually prescribed and tailored to the patients’ needs, availability of any equipment and suitable home and community locations for exercise. The aerobic component of exercise training was prescribed based on 80% of the peak speed achieved during the 6-minute walk test (6MWT), converted to a distance walked over a prescribed time, and practiced with the physiotherapist (RM) at the initial visit. 10 The physiotherapist who conducted the home visit worked both clinically and with the research team. Where a 6MWT was unable to be performed exercise training was prescribed on the basis of symptoms (BORG 3-4). 10 Resistance training used free weights (with equipment accessible in the home environment) and functional activities (e.g. sit-to-stand from a dining chair, step ups on home stairs or in the neighbourhood, and water bottles for upper limb weights). 10 Participants were provided with a pedometer for monitoring speed and distance during walking training. Pedometer data was used by participants for their own motivation and to monitor progress, and individual pedometer data were not collected.
Participants were also provided with a home diary and instructed in its use. The diary allowed participants to document their exercise participation over the course of the program, as well as providing a template for weekly goal setting (exercise goals and other health goals). The diary was designed as a tool to assist patients to self-monitor and progress over the course of the program, and it was not used as an outcome measure. The initial home visit was followed by 7 weeks of phone calls following the principles of motivational interviewing. 10 Participants were encouraged to undertake exercise training (30 minutes aerobic training plus resistance training) five times per week and to document this in the diary. The structured weekly phone calls were delivered by a physiotherapist trained in motivational interviewing who reviewed the home diaries, assisted with goal setting and exercise progression, and delivered self-management training. During the calls participants were also provided with a menu of topics covering aspects of self-management (e.g. disease management, quit smoking, airway clearance, inhalers use, nutrition, exercises) 15 and were encouraged to choose one topic for discussion and goal setting each week.
Outcomes
Baseline demographics of age, gender, body mass index and lung function (spirometry) were collected from participant’s medical records. Feasibility outcomes for program implementation were eligibility, uptake and completion 16 (Table 1). Key clinical outcomes of interest were functional exercise capacity, 17 dyspnoea, 18 quality of life, 19 self efficacy, 20 and anxiety and depression 21 (Table 1). A researcher not involved in delivering the intervention assessed the participants pre- and post- the intervention period. Completion rates were collated at the end of the intervention period, with an a priori definition of completion as undertaking a minimum of 70% of planned PR sessions. 22
Phase I and III outcomes and instruments.
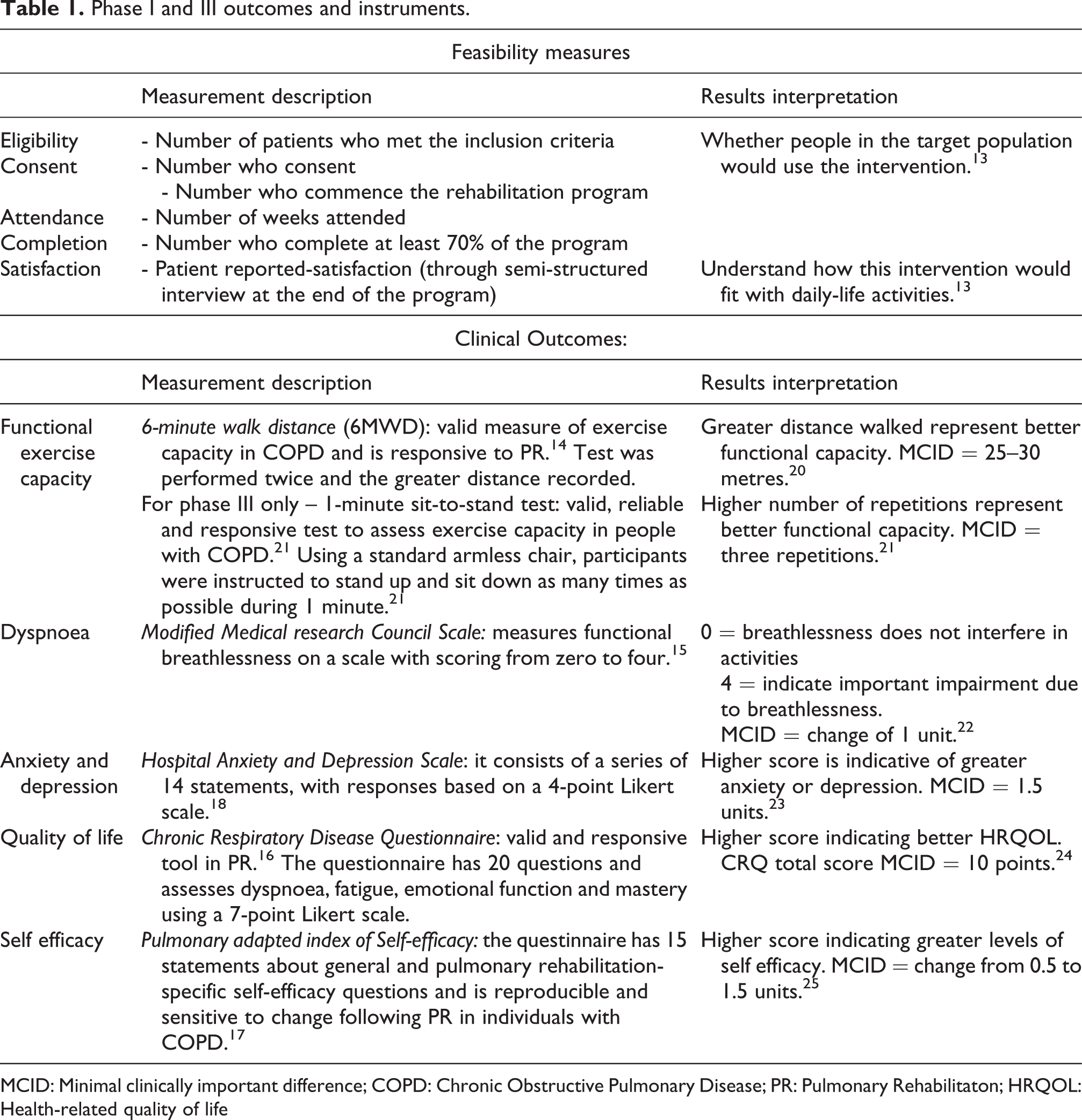
MCID: Minimal clinically important difference; COPD: Chronic Obstructive Pulmonary Disease; PR: Pulmonary Rehabilitaton; HRQOL: Health-related quality of life
Assessments
Clinical measures were recorded in the week after discharge (baseline), and following completion of the 8-week intervention period at the outpatient clinic at The Alfred Hospital. Following program completion, participants undertook a semi-structured interview to describe their experiences of home-based PR participation.
The outcomes were analysed using descriptive statistics, qualitative analysis, and compilation of basic data related to recruitiment. Paired t-tests were used to compare pre and post measurements.
Evaluation
During 6 months of recruitment, from 97 screened patients, 26 met the inclusion criteria (27%). Reasons for participant exclusion are detailed in Table 2. This included 26 patients (37% of those excluded) who were not eligible because they had participated in centred-based PR in the last 12 months. Of the 26 eligible participants, 15 (58%) consented to participate and 10 (38%) undertook the first assessment and started the program (Table 2). Reasons for not commencing the program were referral to palliative care (n = 1), feeling unwell (n = 1), new diagnosis of cancer (n = 1), unable to contact (n = 1) and failed to attend appointment (n = 1).
Feasibility outcomes from Phase I and Phase III.

n, number of participants; PR: Pulmonary rehabilitation; 6MWT: six-minute walk test.
Eight participants achieved >70% of the program (≥6 PR sessions); while two participants only completed two or three sessions of PR due to medical issues (n = 1) and starting an alternative home-based physiotherapy program (n = 1). All participants completed post-intervention questionnaires, and n = 8 attended the centre for post-rehabilitation physical assessment. At the conclusion of the PR program, participants demonstrated clinically important improvements in exercise capacity (mean (SD) change 6MWD 76 (60) m), and quality of life (CRQ total score 15 (21) units) (Table 3).
Participant characteristics and clinical outcomes from Phases I and III.
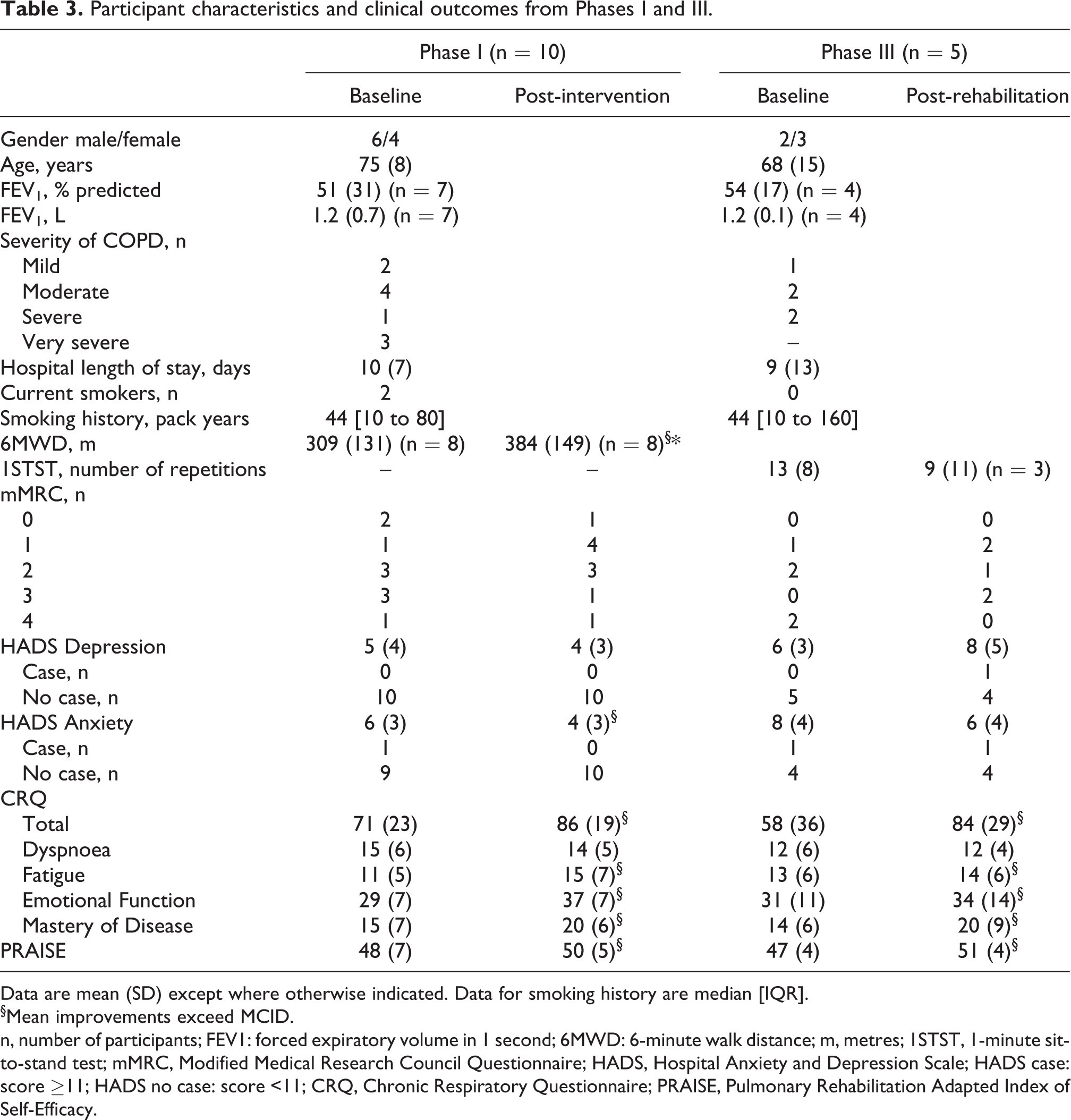
Data are mean (SD) except where otherwise indicated. Data for smoking history are median [IQR].
§Mean improvements exceed MCID.
n, number of participants; FEV1: forced expiratory volume in 1 second; 6MWD: 6-minute walk distance; m, metres; 1STST, 1-minute sit-to-stand test; mMRC, Modified Medical Research Council Questionnaire; HADS, Hospital Anxiety and Depression Scale; HADS case: score ≥11; HADS no case: score <11; CRQ, Chronic Respiratory Questionnaire; PRAISE, Pulmonary Rehabilitation Adapted Index of Self-Efficacy.
Qualitative interviews with participants (n = 9) after the program indicated they were satisfied with the structure and content of the home-based PR program (Box 1 and Supplement A).
Themes and subthemes identified during end rehabilitation interviews.
Motivation is a key to the program success
– Self-motivation is important to complete the program
– Phone-calls helped with motivation
– Increased confidence with the program
– The exercise diaries help to motivate and keep track of progress
Program was beneficial
– Could see improvement during the program
– Learned how to deal with disease and exercises
– Would recommend home-based pulmonary rehabilitation to people with COPD
Plans for the future
– Continue exercising after program ends
– Learned how to maintain a routine of exercises and that will be beneficial
Overcoming centre-based barriers
– Exercises are not scheduled, can be done during work and while travelling
– Preference for doing exercises alone
– Does not need to use public transportation
Clinical reflection
Data from Phase I revealed that 73% of patients were ineligible, with a significant number of exclusions due to participation in PR in the last 12 months. Many of these patients were debilitated due to their exacerbation, and some would have been willing to take part in a home-based PR program. There is evidence that benefits gained from completion of PR tend to be lost in the event of a respiratory exacerbation, particularly where hospitalisation is required, 7,8 suggesting this criterion may exclude people with the potential to benefit from home-based PR.
Only 38% of potential participants commenced home-based PR. Qualitative data indicated that these patients were satisfied with their experience. The reasons potential participants refused the offer of an early home-based PR program were similar to those previously reported, including not wanting to participate in a clinical trial; being unable to attend the hospital for assessment; not wanting a healthcare professional to visit the house; and preferring to attend centre-based PR. We were surprised at the very low uptake, as we had been offering home-based PR to stable patients at our centre for some time and it was well received. We did not feel we had enough information from Phase I to enable us to effectively redesign the manner in which patients following an exacerbation were offered the home-based PR program. Therefore, we concluded that our approach could be improved if it were informed by patient and clinician perspectives.
Phase II – Qualitative study
Plan
Phase II aimed to document the perspectives of both patients and clinicians regarding the reasons why people decline home-based PR following an AECOPD. This information would identify possible barriers to home-based PR.
Action
Participants
Clinicians: Clinicians who were currently working in PR (physiotherapists, exercise physiologists, respiratory medical staff), or on the general medical or respiratory wards at the Alfred Hospital were invited via email to participate.
Hospitalised Patients: Individuals with a confirmed diagnosis of COPD, smoking history of at least 10 pack years and aged over 40 years who were admitted to the respiratory or general medicine wards at the Alfred Hospital (Melbourne, Australia) for an AECOPD were recruited. No patient participants in Phase II had been participants in Phase I.
Methods
Clinicians and patients who agreed to participate in Phase II undertook semi-structured interviews (Supplement B). Interviews were undertaken over the phone or face-to-face (1:1), audio-recorded and transcribed verbatim for analysis. Clinicians were asked a series of open ended questions about their perspectives of home-based PR, timing of recruitment, and strategies for recruitment. Patients were asked their thoughts about possible constraints to taking part in a home-based PR program and suggestions for how to improve a program.
Analysis of qualitative data
The transcribed interviews were manually coded line-by-line and analysed according to the principles of deductive thematic analysis. 23 The data analysis was a four-step process that incorporated: a) immersing oneself in data, b) selecting meaningful units, c) condensing and labelling of data (coding), and d) clustering and formulation of themes. 23 All de-identified transcripts of the interviews were analysed by two researchers independently; both of whom had experience of analysing qualitative interviews and conducting PR (one with more than 10 years of experience, the other with 2 years). The researchers then compared major themes and any disagreements were solved by discussion. Data from clinician and participant interviews were analysed separately. Recruitment was ceased once saturation of themes was achieved.
Evaluation
Twelve clinicians and 14 hospitalised patients participated in Phase II. (Table 4).
Clinicians: Most clinicians felt that limited access to outpatient clinic for possible assessments, and a lack of information about PR and its benefits were potential barriers for patients to engage in home-based PR programs. The interviewed clinicians suggested it would be preferable to approach patients close to discharge when the patients are planning their return to daily life activities, meaning PR options could be presented as a part of the discharge plan. Clinicians interviewed also highlighted the need for them to have more information regarding ongoing research projects so they could recommend to patients the most suitable treatments available for each case.
Hospitalised Patients: Patients felt that potential barriers to uptake of home-based PR early after hospital discharge included access to outpatient clinic for assessments, and issues with and confidence in performing exercises. However, patient responses were more diverse compared to clinicians and indicated that most patients were not well informed about benefits of PR, nor what to expect from participating. This lack of knowledge impacted on their ability to suggest substantial protocol changes.
Results from Phase II – Qualitative interviews with patients and clinicians.

Clinical reflection
Phase II identified potential barriers including access to assessments issues, confidence in exercising after AECOPD, lack of information of PR benefits and structure. With that information, the protocol from Phase I was redesigned in an effort overcome these barriers. Because Phase I participants who undertook home-based PR were satisfied with its structure and content, the changes to the protocol for Phase III focused on strategies to improve program uptake. Specifically:
Time and location of the first assessment: Participants were recruited on the discharge day or the day before, and the first assessments were done at the time of the consent, in order to remove the requirement to travel back to the hospital for assessment. The 1-minute sit-to-stand test (1STST) was added to the assessment, as it is a valid, reliable and responsive tool to assess exercise capacity in people with COPD that requires minimal space to be performed 24 and may be more feasible post AECOPD (Table 1).
Improving information to patients about home-based PR benefits and structure: During recruitment, eligible participants were offered to watch short video testimonials from patients who had previously undertaken the home-based program; and,
Clinician information: When a potential participant was identified, a handout was given to the clinician to notify them of the patient’s eligibility and encouraging the clinician to open a discussion regarding PR options.
Phase III – Re-pilot the revised protocol
Action
Participants
Phase III adopted the same eligibility criteria from Phase I, with the exception that Phase III did not exclude participants based on their previous participation in PR programs, as reflection on Phase I had identified potential benefits for patients who were debilitated following an exacerbation. Participants were not included if they were currently enrolled in a PR program.
Intervention
There were no changes in the delivered intervention compared to Phase I.
Outcomes
Phase III maintained the same outcomes related to feasibility. 16 In Phase III the 1-minute sit-to-stand test was added to the measures from Phase I, all other measures remained unchanged and followed the same standards for assessment.
Assessments
After participants consented, clinical measures were recorded at baseline (prior to discharge from hospital) and immediately following completion of the 8-week intervention period. The outcomes of Phase III were measured with descriptive statistics regarding feasibiltiy and clinical outcomes, and qualitative analysis.
Evaluation
During 7 months of recruitment, from 77 screened patients, 19 were eligible (25%). Reasons for participant exclusion are detailed in Table 2. Of the 19 eligible participants, 10 (53%) consented to participate and 5 (26%) undertook the first assessment and started the program (Table 2). Reasons for not commencing the program were having a busy schedule (n = 2) and being unable to contact (n = 3).
In terms of recruitment strategies, information flyers were handed to clinicians on 10 occasions. Video testimonials were offered to 17 of 19 eligible participants, with n = 7 choosing to watch the full video (4 minutes) and n = 4 viewing part of it (Table 2). From the people who watched the video testimonials nine (82%) consented to participate in the trial. All four PR completers in Phase III had viewed the video.
Four participants achieved ≥70% of the program (≥6 PR sessions); while one participant only completed one session of PR due to working fulltime and having limited time to exercise (n = 1). None of the participants who started home-based PR were able to perform 6MWT at the baseline assessment as four were in isolation on the ward and one did not want to leave the room for the assessment. There was a clinically meaningful improvement in quality of life after home-based PR in Phase III with four participants exceeding the minimal clinically important difference for CRQ total score (Table 3). All participants that started the program were asked to perform the final assessments however, two participants could not return for assessments due to personal reasons and only responded to questionnaires; exercise capacity was not collected on these occasions.
Discussion
This feasibility study demonstrates the challenges in engaging patients in a PR program early after hospitalisation. Although home-based PR was well accepted by the participants who consented and engaged in the program, eligibility and uptake remained low. The present study demonstrated an overall uptake rate of 32% (38% phase I and 26% phase III). Despite concerted efforts to overcome possible barriers to home-based PR, uptake rates did not improve between Phase I and Phase III. Given the potential for PR to reduce hospital admissions, 6 the low eligibility for PR following AECOPD represents an unrealised opportunity to enhance patient and health system outcomes.
In this study we utilised a home-based PR program in an attempt to address well documented barriers to centre-based PR such as travel to the centre and competing demands on time. 25 As has been seen in people with stable COPD, 10 following hospitalisation for an AECOPD a home-based model of PR was acceptable to participants, had good retention in those who chose to commence the program, and produced clinically meaningful improvements in outcomes. Although the program was well received by those who undertook it, uptake in those who were eligible did not improve between Phase I and Phase III, and was less than that reported for centre-based post-exacerbation PR. 26 As the period following AECOPD may be both physically and emotionally taxing for people with COPD, this could impact on willingness to accept an offer of rehabilitation. In previous studies of centre-based post-exacerbation PR, uptake rates have ranged from 20% to 60% 8,27 –29 and use of a PR ‘taster’ during the inpatient stay was largely unacceptable to patients, 30 demonstrating the difficulty in engaging patients in any PR model at this time.
Despite a modest uptake rate for home-based PR in this trial, of those who commenced the program 80% completed at least 70% of the prescribed PR sessions (6 weeks). This completion rate is substantially higher than that seen in people with stable COPD attending centre-based PR, 26 however was not as great as that achieved by people with stable COPD undertaking home-based PR (91%). 10 Completing PR is crucial to achieving benefit, with people who complete PR being 56% less likely to be admitted to hospital in the following year than those who are unable to complete rehabilitation (HR 0.439, p = 0.02). 10 With recent data suggesting fewer than 3% of individuals complete PR within 12 months of a hospital admission for AECOPD, 7 in addition to overcoming practical obstacles to PR attendance, novel strategies are required to encourage patient engagement in the rehabilitation at this time.
During qualitative interviews in Phase II clinicians and patients indicated more information about the home-based PR program and its anticipated benefits would be useful. Limited patient and healthcare practitioner knowledge of PR and its expected outcomes can act as a barrier to PR uptake and completion. 14 To help overcome this potential barrier, video testimonials from people who have previously completed home-based PR were employed in Phase III. Providing health education using narrative communication in a video format is demonstrated to be more engaging and provides social role models, as compared to brochures or statistics. 31 Of the five participants who commenced home-based PR in Phase III all had viewed the video testimonials. While this appears positive, a recent study of video education to improve uptake of outpatient PR following AECOPD was found to have no effect. 32 Whether there is benefit of video testimonials for home-based PR is unclear. Given the period post hospitalisation is a difficult time to engage patients in rehabilitation, finding ways to engage patients with the rehabilitation process remains a challenge for clinicians and researchers alike.
Compounding modest uptake rates for home-based PR in this study, only 28% of all people admitted to hospital for AECOPD during the recruitment period were eligible for inclusion. This eligibility rate is lower than in other studies of post-exacerbation rehabilitation (29–60%) 27 –29,33 and for home-based PR in stable COPD (56%). 10 In the present study all individuals admitted to the hospital for AECOPD were screened for eligibility. Of people who did not meet inclusion criteria, nearly a quarter (24%) had a comorbid condition that impacted safety and ability to enrol in a home-based program, and a further 28% had issues associated with frailty. Multiple co-morbid conditions and frailty are common in people with COPD, with nearly all patients having at least one co-morbid condition and more than 50% having four or more comorbid conditions. 34 As many as one in four individuals hospitalised for an exacerbation of COPD are frail. 35 The presence of comorbidities and frailty in COPD is associated with increased likelihood of exacerbation and of hospitalisation, 34,35 confirming it is the most unwell and complex of patients with COPD who are the focus of post-exacerbation rehabilitation. Although the presence of co-morbidities and frailty explains a proportion of non-eligible patients, it also highlights that a home-based PR program while overcoming common barriers to program attendance may not be appropriate for everyone. Future studies might focus on new interventions suitable for complex patients with multi-morbidity and frailty.
Limitations of the study included a lack of data on adherence to the exercise component of the home program, which may have been useful to further explain the outcomes and reflect on program changes. Fidelity assessments were not conducted, so we cannot be certain that the intervention was delivered as intended, although we have conducted such assessments in our previous trial which used the same staff. 10 Although the early phases of work had identified increased provision of information to clinicians as a strategy to enhance uptake, we were only able to provide the clinician flier for 45% of eligible cases. This was usually because the clinician was not available on the ward, illustrating a weakness in this strategy. Although we used typical exclusion criteria for home-based rehabilitation programs (e.g. balance deficits, cerebral or lower limb palsies, musculoskeletal impairment or cardiac conditions that would prevent independent exercise training), the large number of excluded patients suggests that these criteria may limit participation in home-based PR. Interviews with patients who declined home-based PR in Phase I may have been informative, but ethics approval was not granted to interview these patients who had elected not to take part.
Conclusions
A home-based PR program commenced early after AECOPD achieved improvements in clinical outcomes and high completion rates, however program uptake remains challenging. Given the period of hospitalisation is a difficult time to engage patients in rehabilitation, it may be necessary to find new ways to deliver information about PR, as well as providing a suite of rehabilitation options to meet the needs of patients may be necessary. Future studies might focus on new interventions suitable for complex patients with multi-morbidity after exacerbations of COPD.
Supplemental material
Supplement_A_-_SemiStructured_interviews_and_quotes-PhaseI_R1 - Early home-based pulmonary rehabilitation following acute exacerbation of COPD: A feasibility study using an action research approach
Supplement_A_-_SemiStructured_interviews_and_quotes-PhaseI_R1 for Early home-based pulmonary rehabilitation following acute exacerbation of COPD: A feasibility study using an action research approach by Bruna Wageck, Narelle S Cox, Janet Bondarenko, Monique Corbett, Amanda Nichols, Rosemary Moore and Anne E Holland in Chronic Respiratory Disease
Supplemental material
Supplement_B_-_SemiStructured_interviews_R1 - Early home-based pulmonary rehabilitation following acute exacerbation of COPD: A feasibility study using an action research approach
Supplement_B_-_SemiStructured_interviews_R1 for Early home-based pulmonary rehabilitation following acute exacerbation of COPD: A feasibility study using an action research approach by Bruna Wageck, Narelle S Cox, Janet Bondarenko, Monique Corbett, Amanda Nichols, Rosemary Moore and Anne E Holland in Chronic Respiratory Disease
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B Wageck is the recipient of a full fee research scholarship (LTUFFRs) and postgraduate research scholarships (LTUPRS) from La Trobe University, Melbourne, Australia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
