Abstract
No disease-specific instruments exist in Dutch, French, Italian, and Swedish to measure health status in idiopathic pulmonary fibrosis (IPF) and other interstitial lung diseases (ILDs). The King’s Brief Interstitial Lung Disease (K-BILD) is a 15-item validated questionnaire assessing health status in patients with ILD. The aim of this study was to translate and validate the K-BILD to French, Italian, Swedish, and Dutch versions. The K-BILD was translated following a forward–backward multistep procedure and tested in structured patient interviews. Subsequently, 195 outpatients with ILD were asked to complete K-BILD, St. George’s Respiratory Questionnaire (SGRQ), and Euroqol EQ-5D-5L (EQ5D), twice, 2 weeks apart. Internal consistency, concurrent validity, and repeatability were determined. No major difficulties occurred in the translation processes. The K-BILD was considered comprehensible and relevant by patients. One hundred seventy-six patients (108 IPF and 68 other ILDs) completed the translated K-BILD. Internal consistency was good for all K-BILD modules (Cronbach’s α 0.70–0.93). Concurrent validity of K-BILD was strong compared with SGRQ (r = −0.86) and EQ5D (r = 0.68), low with transfer capacity of the lung for carbon monoxide corrected for hemoglobin (r = 0.33) and with forced vital capacity (r = 0.35). The K-BILD and its domains were repeatable over 2 weeks; intraclass correlation coefficients were 0.86–0.93 (n = 159). Known groups validity showed K-BILD was able to discriminate between patients based on severity of disease. K-BILD’s validity and reliability for patients with IPF was similar to that of other ILDs. The French, Italian, Swedish, and Dutch translated K-BILD questionnaires were well-received by patients and demonstrated excellent validity comparable to the original English K-BILD.
Keywords
Introduction
Health related quality of life (HRQL) is impaired in the majority of patients with interstitial lung disease (ILD) due to symptoms, such as dyspnoea and fatigue, limitations on physical activities, and social isolation. 1 –3 HRQL is quantified using disease-specific questionnaires on aspects of life that patients consider important. In clinical research, HRQL is an important endpoint to assess effectiveness of therapeutic interventions.
There are no disease-specific instruments to assess HRQL in idiopathic pulmonary fibrosis (IPF) and other ILD patients available in Dutch, French, Italian, and Swedish. Therefore, the St. George’s Respiratory Questionnaire (SGRQ), originally developed for chronic obstructive respiratory disease, is commonly used (50 items). 4 –7 In 2012, the King’s Brief Interstitial Lung Disease (K-BILD) health status questionnaire was made available. 8,9 The K-BILD questionnaire contains 15 questions and is much shorter than the SGRQ and easy to administer. It is well validated and can be used to assess HRQL in a wide range of ILDs. K-BILD also showed a stronger concurrent validity than the SGRQ with pulmonary function in patients with IPF. 8 The availability of the K-BILD in different languages could facilitate collaborative international research aiming to improve the quality of life in these rare diseases.
The aim of this study was to translate and validate the K-BILD to French, Italian, Swedish, and Dutch versions. The linguistic and psychometric validations of the Italian, French, Swedish, and Dutch K-BILD questionnaires are reported.
Methods
Linguistic validation: translation, patient interviews, and adaptation
The K-BILD is a 15-item validated, self-completed questionnaire on disease-specific health status with a seven point response scale. It has three domains: breathlessness and activities, psychological and chest symptoms, and one question on financial problems. The domain and total score ranges are 0–100, with the higher scores corresponding with better HRQL. 8
The translation and adaptation of the Dutch, French, Italian, and Swedish K-BILD questionnaires were conducted, respectively, at the pulmonary departments of Erasmus Medical Center in Rotterdam, the Netherlands, Louis Pradel Hospital, Lyon, France, the University of Catania, Italy, and the Karolinska University Hospital Solna, Stockholm, Sweden.
Permission to translate the K-BILD was asked from the copyright holders. 10 The K-BILD questionnaire was translated into Dutch, French, Italian, and Swedish, following a multistep procedure and in collaboration with the developers using their conceptual framework of items to ensure conceptual/semantic equivalence. 11 –13 The online supplement 1 provides details on all the 11 steps of the translational procedure. This included an external back translation and review by linguistic services of Mapi Language Services (Lyon, France).
For each country, the translated version was tested with structured interviews in at least five patients (interview questions are shown in the online supplement 2
The resulting final versions of the Dutch, French, Italian, and Swedish K-BILD are shown in the online supplements 3 to 6.
Psychometric validation of the Dutch K-BILD
Patients and measurements
All consecutive patients with ILD visiting the tertiary outpatient clinic of the pulmonary department of Erasmus Medical Center, between December 2013 and May 2014, were asked to participate. For Sweden, France, and Italy, patients were included between August 2015 and April 2016. Patients were excluded if they had sarcoidosis, emphysema (clinician’s judgment, based on lung function and computer tomography scan), or if there was a language or intellectual barrier. ILD was classified consistent with international guidelines. 14,15 Patients willing to participate were asked to complete two questionnaires: K-BILD and SGRQ, and two health status measurements: Punum Ladders and Euroqol EQ-5D-5L (EQ5D), at the day of the current visit and after 2 weeks. 16,17 The sequence of completing the questionnaires was: K-BILD, SGRQ, Punum Ladder, and EQ5D. Patients were instructed to fill in the questionnaires alone in a quiet place. Nonresponders received a phone call to remind them. Patients who did not complete > 85% of the questions were excluded from the study.
If performed in routine care, the results of pulmonary function tests (total lung capacity (TLC), forced vital capacity (FVC), and transfer capacity of the lung for carbon monoxide corrected for haemoglobin (TLCOc)) were recorded from the files. 18,19
The ethics committee of the Erasmus Medical Center, Rotterdam, the Netherlands, decided to exempt this study from review according to national and international regulations because of the noninterventional design (MEC-2013-498). All other hospitals approved of this decision. All patients gave written informed consent or approval by voluntarily returning the completed questionnaires.
Validation
For validation, we tested the following five different aspects:
Concurrent validity showing correlations between K-BILD scores and SGRQ scores, Punum Ladders, EQ5D, and lung function. Internal consistency reflecting the interrelatedness of items comprising the K-BILD. The test–retest reliability (repeatability) was determined by comparing the K-BILD scores at baseline and 2 weeks in patients whose condition was considered stable. Discriminative validity, reflecting the ability of an instrument to differentiate between groups of patients, was examined by comparing baseline health status scores of “known groups”. Effect size (ES) was calculated by determining partial η
2
in K-BILD scores between the groups.
20
Analysis
Data analysis was executed using SPSS version 21. Results are expressed as mean values ( ± standard deviation) unless otherwise stated. To determine concurrent validity between HRQL variables and clinical variables, we used Pearson correlation coefficient or Spearman’s rank correlation coefficients. Internal consistency was determined by calculating the Cronbach’s α coefficients for each domain and the total K-BILD. Cronbach’s α coefficient > 0.7 is considered a reliable internal validity. The test–retest reliability was assessed with intraclass correlation coefficient (ICC) and Bland–Altman plots. An ICC of 0.7 is considered the minimum standard for reliability. 21 Punum Ladders were used as a measure to assess if patients felt stable at 2 weeks. To assess discriminative validity and ES, students’ t-test or one-way analysis of variance was used.
Results
Permission to translate the K-BILD questionnaire was obtained by the copyright holders. Review by the developers of the cognitive interviews, comments, and back translations in each country resulted in minor changes to make sure the translated questionnaires reflected the intention of the original K-BILD. Demographics, translation comments, and changes per country per stage are shown in Table 1 and online supplement 7.
Characteristics of participants involved in linguistic validation per country.a

FVC: forced vital capacity; TLCOc: transfer capacity of the lung for carbon monoxide, corrected for hemoglobin concentration; IPF: idiopathic pulmonary fibrosis; NSIP: nonspecific interstitial pneumonia; CVD: collagen vascular disease.
aValues are numbers (percentages) or medians (range).
A total of 195 patients were recruited for the psychometric validation of the K-BILD. One hundred seventy-six patients (90%) completed and returned the questionnaire at week zero and 159 patients (82%) at week 2, with 0.2% missing items in the K-BILD questionnaire and 1.9% in the SGRQ. The diagnoses were: IPF (108), collagen vascular disease-associated ILD (19), chronic hypersensitivity pneumonitis (10), unclassifiable ILD (14), idiopathic nonspecific interstitial pneumonia (13), pulmonary alveolar proteinosis (2), obliterative bronchiolitis (3), organizing pneumonia (2), Langerhans cell histiocytosis (1), lymphangioleiomyomatosis (1), respiratory bronchiolitis-associated ILD (1), asbestosis (1), and desquamative interstitial pneumonia (1). Demographic information is shown in Table 2.
Demographics, HRQL, and clinical findings of participants involved in the psychometric validation of the K-BILD questionnaire: total for all countries, split by IPF and ILD (non-IPF), and split by individual country.

HRQL: health related quality of life; IPF: idiopathic pulmonary fibrosis; ILD: interstitial lung disease; K-BILD: King’s Brief Interstitial Lung Disease questionnaire; SGRQ: St. George’s Respiratory Questionnaire; QOL: quality of life; VAS: Visual Analogue scale; FVC: forced vital capacity; FEV1: forced expired volume in 1 second; TLC: total lung capacity; TLCOc: transfer capacity of the lung for carbon monoxide, corrected for hemoglobin concentration.
aIn Italy, the perceived health status questionnaire was not administered.
Lung function data were used when present; of 139 patients TLCOc data were available, 72 of the 139 patients had a TLCOc below 50% predicted. There were no floor or ceiling effects in the K-BILD total or domain scores; less than 15% of the participants achieved, respectively, the lowest or highest possible score. 21
Concurrent validity of the K-BILD domain and total scores with the validated SGRQ domain and total scores was strong for all domains. Correlation coefficients with other HRQL measures and lung function variables are shown in Table 3 for the total group and in online supplement 8 for the individual countries. The correlations between SGRQ total score and lung function parameters were comparable (FVC %predicted: r = −0.38, forced expired volume in 1 second %predicted: r = −0.30, TLC %predicted: r = −0.33, and TLCOc %predicted: r = −0.39).
Correlation coefficients between K-BILD scores and other HRQL scores and clinical variables, total for all countries.a,b

HRQL: health-related quality of life; K-BILD: King’s Brief Interstitial Lung Disease questionnaire; SGRQ: St. George’s Respiratory Questionnaire; VAS: Visual Analogue scale; FVC: forced vital capacity; FEV1: forced expired volume in 1 second; TLC: total lung capacity; TLCOc: transfer capacity of the lung for carbon monoxide, corrected for hemoglobin concentration.
aThe correlation coefficients for the corresponding domains are shown in bold.
bValues shown represent Pearson’s correlation coefficients, all p < 0.01 unless otherwise stated.
cp < 0.05.
dp > 0.05.
Internal consistency was good in the chest domain and excellent in the other domain and total scores (Table 4). Repeatability was tested in 159 patients; the average length of time between baseline and measurement at week 2 was 16 days. ICCs for consistency and Bland–Altman plot demonstrated good repeatability and thus reliability of the K-BILD (Table 4 and Figure 1 for the total group and online supplements 9 to 13 for the individual countries). Punum Ladders were completed by 156 patients, 99% had no change or minimal change in Punum scores quality of life between baseline and week 2, which confirmed their stable health status. Removing the two patients with major changes from test–retest analysis did not alter the results.
Internal consistency and reliability K-BILD (total all countries).a

K-BILD: King’s Brief Interstitial Lung Disease; ICC: intraclass correlation coefficient for K-BILD repeatability; 95%CI: 95% confidence interval.
aData shown are Cronbach’s α coefficient.
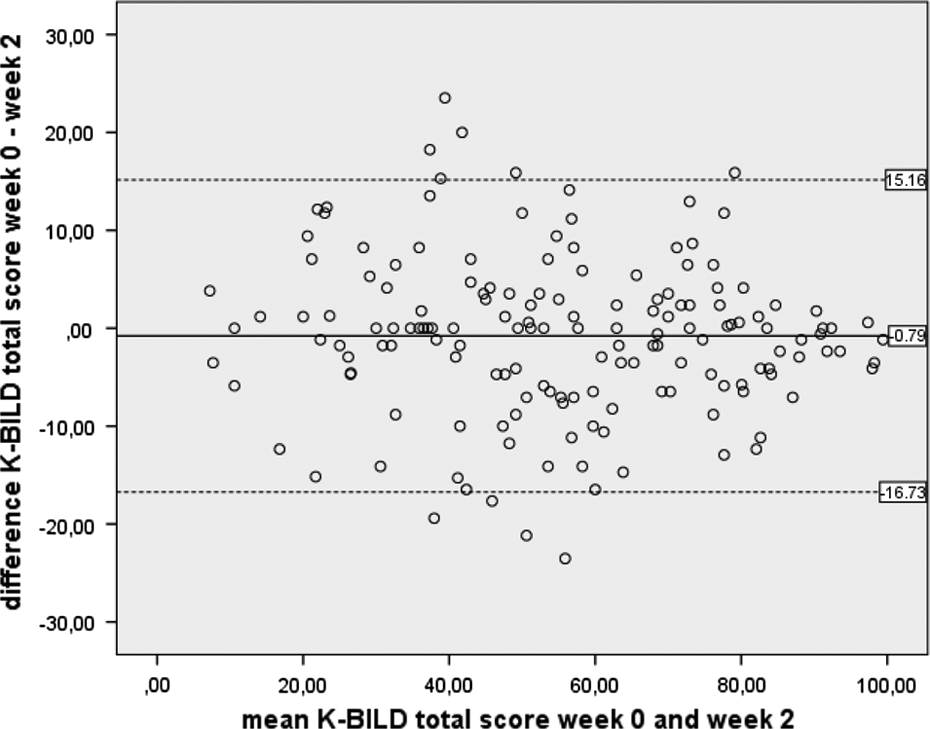
Bland Altman plot of repeatability of the K-BILD questionnaire of all countries. The solid line shows the mean difference and the dashed lines represent the 95% limits of agreement. K-BILD : King’s Brief Interstitial Lung Disease questionnaire.
Both K-BILD and SGRQ total scores were able to discriminate between patients based on severity of their disease (Table 5). The discriminative power of the K-BILD and SGRQ is expressed in ES between the known subgroups. The ES is the strongest (0.4) for symptom-based classification of groups and poor for those based on lung function and other non-symptom parameters, which is not surprising as they measure a different aspect of disease. The magnitude of the ES indicates both questionnaires having good discriminative power. Table 6 shows the concurrent validity, internal reliability, and repeatability of the K-BILD questionnaire in patients with IPF when comparable to patients with other ILDs (non-IPF).
K-BILD and SGRQ total scores in known groups.a
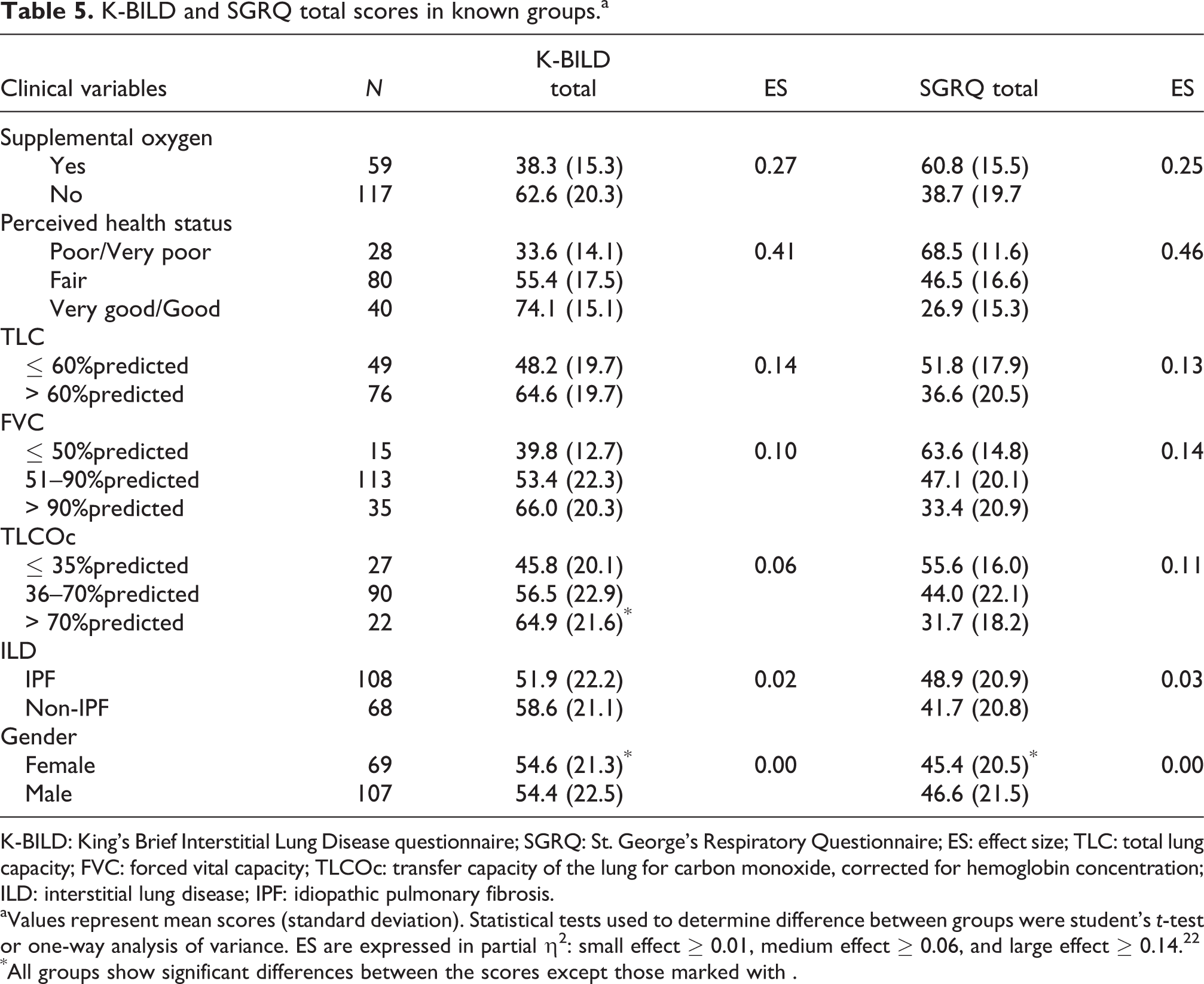
K-BILD: King’s Brief Interstitial Lung Disease questionnaire; SGRQ: St. George’s Respiratory Questionnaire; ES: effect size; TLC: total lung capacity; FVC: forced vital capacity; TLCOc: transfer capacity of the lung for carbon monoxide, corrected for hemoglobin concentration; ILD: interstitial lung disease; IPF: idiopathic pulmonary fibrosis.
aValues represent mean scores (standard deviation). Statistical tests used to determine difference between groups were student’s t-test or one-way analysis of variance. ES are expressed in partial η2: small effect ≥ 0.01, medium effect ≥ 0.06, and large effect ≥ 0.14.22
*All groups show significant differences between the scores except those marked with .
Concurrent validity, internal reliability, and repeatability of K-BILD in IPF in comparison with other ILDs, total for all countries.a

K-BILD: King’s Brief Interstitial Lung Disease questionnaire; IPF: idiopathic pulmonary fibrosis; ILD: interstitial lung disease; SGRQ: St. George’s Respiratory Questionnaire.
aData shown are Pearson’s correlation coefficients unless otherwise stated, p < 0.01.
Discussion
In this study, the K-BILD was translated into an Italian, French, Swedish, and Dutch version and psychometrically validated. It is the first health status questionnaire for IPF and other ILDs available in these languages. During the cultural adaptation process, only minor changes were necessary. The K-BILD was brief with only 15 items easy to administer, well-received by patients, and applicable to non-English speaking countries. The K-BILD was also validated for the first time in non-English speaking populations and showed good concurrent validity, internal consistency, repeatability, and discriminative performance, comparable with the original K-BILD. Also a strong correlation of the EQ5D index value with K-BILD was found. This had not been assessed before.
Instruments to measure HRQL have become increasingly important in trials and clinical care. However, major improvements are needed to develop and validate new or existing instruments. 23
The K-BILD questionnaire is the first disease-specific questionnaire to examine HRQL in patients with IPF and other ILDs. Other questionnaires were not specifically developed for ILDs; a-tool-to-assess-quality-of-life-in-IPF (ATAQ-IPF) and an IPF specific version of SGRQ-I were only validated in an IPF population. 22,24 The University of California San Diego Shortness of Breath Questionnaire only measures symptoms and was developed in a non-ILD population and tested for content and construct validity in IPF. 25 –27
In the absence of disease-specific measures for ILDs, clinically relevant patient-reported outcome measures for obstructive lung disease such as SGRQ have been used in trial assessing, for example, medication treatment in ILD/IPF. 28
The current patient population showed reduced HRQL in all domains of K-BILD and SGRQ, with the activity domain most impaired. This is in line with a review by Swigris of three studies that assessed HRQL in IPF and other ILD patients and also showed that HRQL was most impaired in the physical activity domains. 2 The mean (SD) K-BILD total score was 59 (22) in ILD patients and 52 (22) in IPF patients; in the original development study of the K-BILD, these scores were comparable with 59 (25) and 52 (26), respectively. 8
Concurrent validity and repeatability were comparable with the results of the original version. 8 In the current study, correlation of FVC and TLCOc with the breathlessness and activity domain was weaker than in the original study; FVC (0.42 vs. 0.51) and TLCOc (0.44 vs. 0.52). Correlations of SGRQ total score with FVC and TLCOc yielded comparable correlation coefficients to those of the K-BILD. The weak correlation of FVC with the HRQL questionnaires confirms that HRQL informs us about aspects of disease severity that are relevant to patients but cannot be measured with physiologic measures such as lung function. In other validation studies, the same results were found. In a study that assessed HRQL in 50 patients with ILD SGRQ total score correlated with FVC %predicted r = −0.45 and with TLCOc %predicted r = −0.55. 7 The SGRQ-I showed in IPF population correlations with FVC %predicted r = −0.33. 23 The ATAQ-IPF correlations revealed comparable results. 24
These findings confirm FVC contributes only partly to the impact ILD or IPF has on quality of life. TLCOc %predicted with a moderate correlation appears to be more related to quality of life in both our study and others. 7,23,24
It is interesting to note that in the current study, differences in between countries are seen in HRQL. In Italy, less impairment in HRQL is found both with the K-BILD and the SGRQ, while mean FVC values are comparable to the other countries. Also, correlations between FVC and K-BILD differed between countries. This could be due to small numbers; however, in Sweden and the Netherlands, correlations are similar to the original study from the United Kingdom. Although purely speculative, an alternative explanation could be that factors such as climate and diet influence disease burden or disease perception and consequently HRQL, with the Northern countries having more resemblance in these factors with the original study population from the United Kingdom and more similar outcomes. To the best of our knowledge, no studies have yet been performed in ILD looking at influences of diet and climate on disease and HRQL.
The K-BILD was developed for ILDs, including IPF. To assess more specifically its ability to measure HRQL in IPF, we compared the construct validity, internal reliability, and intraclass correlation between IPF and non-IPF ILD subgroups. These results show that the K-BILD is also a reliable and valid tool in IPF patients. Our study confirms HRQL is more affected in IPF than in other ILDs as has also been previously noted in studies using the generic measure Short Form-36. 2
The K-BILD questionnaire detected differences in disease severity. HRQL was more impaired in patients using supplemental oxygen (in line with the original study), with lower perceived health status and with lower lung function values (this was not tested in original study). In the original article of Patel et al., no ES are calculated. In our study, ES show that K-BILD discriminates better in the home oxygen and TLC subgroups, and the SGRQ discriminates better in the TLCOc and perceived health status subgroups (based on one question describing general health status). Both questionnaires had acceptable levels of missing items, K-BILD scored better with only 0.2% missing items versus 1.9% in SGRQ. The advantage of the K-BILD is that it is much shorter, 15 questions versus 50 questions.
With the economically challenging climate and new and expensive medications, governmental organizations increasingly investigate cost-effectiveness of treatment, with the benefit of interventions expressed in quality-adjusted life years (QALYs). A generally accepted tool for the calculation of QALYs is the EQ5D, a generic five questions measure of health. EQ5D was used in intervention studies in IPF to assess quality of life and to calculate cost-effectiveness of new treatment options. 29 In our study, K-BILD total score correlated well with EQ5D (0.68). The Dutch general population norm for the EQ5D index value is 0.91. 30 In our study, the mean EQ5D index value was 0.74 for ILD and 0.66 for the IPF subgroup.
A limitation of this study is that it did not assess responsiveness and minimal clinically important difference (MCID). The study of Patel et al. suggests that the K-BILD is a responsive health status outcome measure in ILD with an MCID of around eight; however, as they also state that this was only assessed in a small sample size and only four patients with large changes. 9 A larger study with longer follow up is needed. We therefore currently follow up a patient cohort prospectively, to gain information about responsiveness and MCID in a bigger multicultural cohort. Another limitation is that both in the original as well as in our study, only small numbers of patients with ultrarare ILD’s were included. Only larger international collaborative studies will be able to further validate the K-BILD in specific disease groups.
In conclusion, the current study developed a Dutch, Italian, French, and Swedish version of the K-BILD and demonstrated that the K-BILD is a reliable and valid instrument to measure HRQL in an international cohort of patients with ILD, consistent with the evidence of the original version. With only 15 items, it is easy to use in daily practice, and moreover, its use in different languages could facilitate collaborative international research aiming at improving quality of life in these rare diseases.
Footnotes
Acknowledgements
The authors like to thank Traclet J, Khouatra C, Durand M, Carlson L, Giot C, Puglisi S, Torrisi S, Klackenberg A, van den Toorn L, van den Blink B, Hoogsteden HC, van Manen MJG, and Muskens AM, for facilitating this study.
Author contributions
M Wapenaar (MSc) contributed to study conception and design; collection, analysis, and interpretation of data; and drafting the manuscript and revising it for important intellectual content and reading and approving the final version.
Dr Patel, Dr Birring, Dr van Domburg, and Dr Bakker contributed to the study conception and design; analysis and interpretation of data; and drafting and revising it for important intellectual content and reading and approving the final version.
Prof. Sköld, Prof. Cottin, Dr Vindigni, and Prof. Vancheri contributed to the study conception and design; collection, analysis, and interpretation of data; and drafting and critical review of the manuscript and reading and approving the final version.
Dr Wijsenbeek had full access to all the data in the study and takes final responsibility for the integrity of the data, the accuracy of the data analysis, and contributed to the study conception and design; collection, analysis, and interpretation of data; and drafting the manuscript and revising it for important intellectual content and reading and approval of the final version.
M Wapenaar (MSc) had full access to all the data in the study, takes final responsibility for the integrity of the data, the accuracy of the data analysis and for the decision to submit for publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Monique Wapenaar (MSc), Dr Patel, Dr Birring, Dr van Domburg, Dr Bakker, Dr Vindigni, and Prof. Vancheri have no conflicts of interest. Prof. Sköld has received honoraria for consulting, advisory boards, and lectures from AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Meda, Novartis, Mundipharma, Sandoz, Chiesi, Almirall, Intermune, and Roche, and research grants from Boehringer Ingelheim, Roche, and Sandoz. Prof. Cottin has received honoraria for consulting or participating to advisory board meetings, for speaking, and support for attending meetings from GSK, Intermune/Roche, Novartis, Sanofi, Biogen Idec., Actelion, Bayer, Boehringer Ingelheim, and Gilead. Dr Wijsenbeek has received honoraria for consulting or participating to advisory board meeting or speaking of Boehringer Ingelheim and Intermune/Roche, she received unrestricted research grants from Intermune, Hoffman la Roche, and Boehringer Ingelheim. All honoraria were paid to her institution.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigator initiated study was funded by a research grant from Intermune for the translation procedures.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
